Abstract
It has been widely reported that the incidence and severity of Clostridium difficile infection (CDI) have increased dramatically in North America and Europe. However, little is known about CDI in Mainland China. In this study, we aimed to investigate the incidence of CDI and the main epidemic and drug-resistant strains of C. difficile in Mainland China through meta-analysis of related studies published after the year 2010. A total of 51 eligible studies were included. The pooled incidence of toxigenic C. difficile among patients with diarrhoea was 14% (95% CI = 12–16%). In Mainland China, ST-37 and ST-3 were the most prevalent strains; fortunately, hypervirulent strains, such as ST-1 (BI/NAP1/027) and ST-11 (RT 078), have only occurred sporadically to date. The rates of C. difficile resistance to ciprofloxacin (98.3%; 95% CI = 96.9–99.7%), clindamycin (81.7%; 95% CI = 76.1–87.3%) and erythromycin (80.2%; 95% CI = 73.5–86.9%) are higher than in other counties; however, none of the C. difficile isolates reported in Mainland China were resistant to metronidazole (n/N = 0/960), vancomycin (n/N = 0/960), tigecycline (n/N = 0/41) or piperacillin/tazobactam(n/N = 0/288).
Similar content being viewed by others
Introduction
Clostridium difficile (C. difficile) is a Gram-positive, spore-forming, anaerobic bacterium that can colonize the human colon. Antibiotic therapy can have the adverse effect of disrupting the normal flora of the gut, in which case C. difficile may opportunistically dominate the gut and induce colitis. Not until J. G. Bartlett discovered the relationship between C. difficile and pseudomembranous colitis (PMC) in 19781 was attention focused on this anaerobic bacteria2. Indeed, Bartlett found that almost all PMC cases are caused by C. difficile3.
C. difficile produces toxins, including enterotoxin (toxin A, tcdA) and cytotoxin (toxin B, tcdB). Although the pathogenicity of toxin A is still debated, the function of toxin B in disease is well acknowledged in infected patients. Some strains of C. difficile, like RT 027, seem to be highly virulent. Some of the base-pairs of the negative regulatory gene tcdC are missing in this strain, which increases the production of toxin A and toxin B more than 16- and 23-fold, respectively. This increased pathogenicity is relevant to most outbreaks in Europe and North America4. According to several studies from the early 2000s, the incidence and severity of C. difficile infection (CDI) have increased dramatically in North America and Europe5,6,7. Another study found that C. difficile was the most commonly detected diarrhoeal pathogen in Western Australia8, and C. difficile RT 027 has been identified as the major strain responsible for outbreaks9,10,11,12. Moreover, the incidence and severity of hospital-acquired and community-acquired CDI are increasing worldwide13. One investigation published in The New England Journal of Medicine in 2014 showed that C. difficile was the most commonly reported pathogen (causing 12.1% of all health care–associated infections) in America14. Furthermore, C. difficile has become the primary pathogenic faecal bacteria associated with hospital infection, and the overall incidence of CDI has exceeded that of methicillin-resistant Staphylococcus aureus (MRSA) in parts of the United States15,16. Although there have been numerous studies of the epidemiology of CDI in North America and Europe, few studies have been undertaken elsewhere, particularly in Asia17.
There are many risk factors for CDI, including the use of antibiotics or proton pump inhibitors (PPIs), hospitalization, aging, gastrointestinal surgery, and conditions that may affect the colonic flora, among others18,19. However, exposure to antibiotics is the most important risk factor for the development of CDI5, and a high prevalence of indiscriminate and inappropriate use of antimicrobials exists in Asia17. In Mainland China, antibiotics are heavily consumed, and the abuse of antibiotics is fairly serious20. The rate of antibiotic usage by inpatients in Mainland China is as high as 80%, and the use of broad-spectrum antibiotics and joint use accounted for 58% of antibiotic treatments before 2004, according to an investigation by the WHO. This rate surpassed the international level (30%) to a large extent20,21. Another report found that the rate of antibiotic usage by inpatients in Mainland China was approximately 70% according to the Ministry of Health National Antimicrobial Resistance Investigation Net (Mohnarin). Moreover, almost every surgical patient receives antibiotics, and this usage rate is as high as 97%. In addition, the consumption of antibiotics per person each year in Mainland China is approximately 138 g on average, whereas only 13 g of antibiotics are consumed per person per year in the United States22. Although CDI is recognized as a major epidemic organism in North America and Europe, Chinese hospitals only sporadically report CDI. No large-scale studies have been performed to date, and therefore the status of CDI in Mainland China remains unknown. Moreover, the recognition of CDI among general healthcare workers and patients in Mainland China is very poor. This study intends to expand the effective research data and provide a more reliable meta-analysis based conclusion on the actual status of CDI and drug resistance in Mainland China. In particular, the following themes were focused and investigated: Topic 1: the incidence of CDI in Mainland China; Topic 2: the molecular epidemiology of C. difficile in Mainland China; and Topic 3: the antibiotic resistance of C. difficile in Mainland China. This information is critical for developing appropriate strategies to prevent CDI and the vast negative impact of such infections in Mainland China.
Results
Search results
We reviewed 7 electronic databases and identified 1,440 articles published from 2010 to 2016 (Fig. 1). After initial evaluation of the titles and abstracts, 1,351 articles were excluded because of their irrelevance and duplication. The full text of the remaining articles was reviewed. Among the 89 articles, 38 were excluded again for specific reasons: 8 were reviews, 4 studies did not include data from 2010–2016, 1 study had fewer than 5 samples, 12 studies used the same samples, 5 studies didn’t originate from Mainland China, 2 studies were repeated in different languages, and incomplete information was provided in 6 studies. Finally, 51 studies were included in this systematic review and meta-analysis. Of these, 39 articles focused on Topic 1, 16 focused on Topic 2 and 10 focused on Topic 3.
Characteristics of the eligible studies
All features of the 51 eligible studies are listed in Supplementary Tables 1, 2 and 3. Regarding Topic 1, Beijing and Guangdong each produced 6 articles; 4 articles were from Hunan; Hubei and Shanghai each produced 3 articles; Jiangsu, Hebei and Sichuan each produced 2 articles; 7 articles concerned Zhejiang; and 1 article each originated from Anhui, Gansu, Henan, and Ningxia. A total of 15,313 samples were collected to detect the existence of C. difficile and the associated toxins. Some 16 of the included studies were related to Topic 2, and multi-locus sequence typing (MLST) and PCR ribotyping (RT) were used to detect the molecular type of C. difficile. Ten articles were relevant to Topic 3. Antibiotic sensitivity testing (AST) were carried out with 3 methods differently; 6 used the agar dilution method; 3 used the E-test; and 1 study combined the agar dilution, disk diffusion and E-test methods.
The incidence of CDI in Mainland China
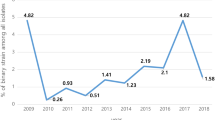
In total, 39 studies, including 13 provinces and 15,313 samples, reported the positive rate of toxigenic C. difficile (Table 1). We estimated the mean positive rate of toxigenic C. difficile in diarrhoea patients in Mainland China to be 14% (95% CI = 12–16%) (n/N = 2,132/15,313), with a high level of heterogeneity between the estimated rates (I-squared = 92.9%, p < 0.001) (Fig. 2). Figure 3 shows the incidence of CDI in mainland China over the past 6 years. In particular, the positive rates reported in Hubei (23%; 95% CI = 20–26%), Hebei (19%; 95% CI = 15–24%), Anhui (19%; 95% CI = 12–26%) and Sichuan (17%; 95% CI = 13–22%) were very high, whereas Ningxia (4%; 95% CI = 1–6%) and Henan (3%; 95% CI = −1–8%) showed lower positive rates.
The incidence of CDI in Mainland China (Created by an online Chinese website.http://c.dituhui.com/apps/range).
The molecular epidemiology of C. difficile
We next reviewed the molecular classification methods, including MLST and RT, for C. difficile in the published articles. As shown in Table 2, ST-37 (17.2%; 95% CI = 12.2–22.1%) (n/N = 152/913) and ST-3 (18.1%; 95% CI = 8.3–27.8%) (n/N = 67/295) were the dominant strains in Mainland China. Focusing on studies that used the RT method, RT 006 (55%; 95% CI = 39.6–70.4%) (n/N = 22/40) was the predominant strain in Mainland China, followed by RT 017 (17.2%; 95% CI = 12.2%1–22.1%) (n/N = 152/913) as the second epidemic strain. Fortunately, hypervirulent strains, such as ST-1 (BI/NAP1/027) and ST-11 (RT 078), were only reported sporadically, and no outbreaks have been reported to date (Table 3).
The antibiotic resistance of C. difficile
A total of 24 drugs were reported in the included studies. However, 14 drugs were investigated by several studies (Table 4), while the others were only examined in single studies. The results shown in Table 4 are more credible because they include several studies and a large number of samples. None of these strains were resistant to metronidazole (n/N = 0/960), vancomycin (n/N = 0/960), piperacillin/tazobactam (n/N = 0/288), or tigecycline (n/N = 0/41). However, these strains were highly resistant to erythromycin (80.2%; 95% CI = 73.5–86.9%) (n/N = 340/433), clindamycin (81.7%; 95% CI = 76.1–87.3%) (n/N = 476/581) and ciprofloxacin (98.3%; 95% CI = 96.9–99.7%) (n/N = 688/694). The results of the other 10 single-drug studies showed that the rate of resistance to teicoplanin was 0% (n/N = 0/20), amoxicillin/clavulanic acid was 0% (n/N = 0/22), ampicillin was 0% (n/N = 0/22), ceftriaxone was 100% (n/N = 20/20), piperacillin was 0% (n/N = 0/22), penicillin was 27.3% (n/N = 6/22), fidaxomicin was 0% (n/N = 0/45), chloramphenicol was 2% (n/N = 7/334), cefoxitin was 92.6% (n/N = 87/94) and imipenem was 3% (n/N = 4/138). None of the strains were resistant to fidaxomicin, amoxicillin/clavulanic acid, ampicillin, piperacillin, or teicoplanin, and nearly all of the strains were sensitive to chloramphenicol and imipenem according to the published articles23,24,25,26,27,28. The strains reported in one article were resistant to ceftriaxone and cefoxitin25,29.
Discussion
Because only a limited number of studies have investigated C. difficile in Mainland China, some provinces were only investigated in one study and others were not included in the analyses. Although the data were not sufficient to provide an accurate conclusion, we still obtained valuable information through meta-analysis of related published data. In this study, we found that the incidence of toxigenic C. difficile in Mainland China was 14% (95% CI = 12–16%) (n/N = 2,132/15,313). Fortunately, highly virulent strains were rarely reported, although CDI outbreaks have been reported more than 100 times in North America and Europe since 200330. From 2000 to 2009, the confirmed CDI cases were more than doubled from 139,000 to 336,600 in US hospitals, and the number of patients with a primary confirmed CDI cases tripled from 33,000 to 111,00031. Moreover, the incidence, mortality, and medical care costs of CDIs have reached historic highs in America31. However, Asian countries seem to have a low incidence of CDI. One survey conducted in Japan found only a small number of patients with serious CDI and recurrent CDI. Meanwhile, regarding the annual number of CDI patients at all of the hospitals that participated in this survey, the largest group of hospitals reported treating “1 to 5 patients a year,” representing 17.8% of the sample, and the second largest group of hospitals reported treated “no patients a year,” representing 13.1%32. In addition, the CDI incidence in Korea was estimated to be 5.06 per 100,000 population in 2011, whereas the estimated incidence in America per 100,000 population ranged from 30 to 120 cases of community-associated infection and from 50 to 160 cases of health care-associated infection across the Emerging Infections Program (EIP) sites in the same year33,34. In Mainland China, the positive rate of toxigenic C. difficile infection in some provinces, such as Ningxia and Henan, was close to zero. This result was different with Jin K et al. reviewed in 2010, in which no significant geographical variation of CDI was found in Mainland China35. Therefore, we re-analysed the articles concerning Ningxia and Henan. The CDI study in Henan only included 60 samples, and the authors detected 9 cases of C. difficile, but only 2 produced toxins. The investigation in Ningxia included 233 diarrhoea patients; of these, 162 cases were due to non-infectious diarrhoea and 71 cases were caused by pathogenic bacteria, where 9 C. difficile strains were assayed with the VIDAS CDAB kit. On the other hand, Anhui, Hebei, Hubei and Sichuan reported a much higher positive rate, which attracted our attention. These studies were reviewed, which showed that some risk factors were associated with these results. First, the diarrhoea patients included in these studies represented special groups; some diarrhoea patients came from the intensive care unit (ICU), some suffered from hematologic diseases, and some patients had used antibiotics for a long time. Moreover, some diarrhoea patients were diagnosed with antibiotic-associated diarrhoea (AAD). Thus, these results may increase the positive rates of CDI to a high level. In addition, 2,570 patients from 9 articles were diagnosed with AAD or confirmed to have used antibiotics, and this feature undoubtedly increased the rate of CDI in our survey. Notably, studies from Guangdong province included one-third of all the pooled nationwide cases approximately, which may have impacted the overall results of this study. Thus, reanalysis was necessary. We deleted the data from Guangdong and recalculated the mean incidence of CDI based on the data from the other 12 provinces. Finally, the recalculated incidence of CDI was 14% (95% CI = 11–16%) (n/N = 1,343/10,024). The same phenomenon was found from the studies of Zhejiang province. Therefore, we recalculated without the data from Zhejiang and got the same result of 14% (95% CI = 12–17%) (n/N = 1,732/11,583). Therefore, these changes did not produce a significant difference in the overall rate of CDI, in other words, the data from Guangdong and Zhejiang had a small impact on the nationwide average value of CDI incidence despite their big data weight.
Based on the risk factors mentioned above, some researchers are worried about the incidence of CDI in Mainland China due to the growing elderly population and the well-recognized problem of over-prescribing broad-spectrum antibiotics in Mainland China35. The lifestyles of Chinese people, particularly those of young Chinese, have changed tremendously over the last 20 years, partially because of the rapid growth of the Chinese economy. These lifestyle changes, which were not as common in Mainland China in the past, may represent a major contributor to the increasing emergence of CDI19.
Some 16 articles included in this meta-analysis did not detect ST-1 (BI/NAP1/027) or ST-11 (RT 078) in Mainland China. According to our data, ST-37 (RT 017) strains were predominant in Mainland China. This result is in agreement with previous research results showing that ST-37 (RT 017) was the dominant type in Mainland China36,37. In contrast, one study showed that ST-37 (RT 017) strains were rare among healthy individuals38. However, we also learned from scattered reports that highly virulent strains, such as RT 078 and RT 027, have been isolated in Mainland China. In 2008, Cheng, V. C. et al. detected the RT 027 strain of C. difficile for the first time, and Huang, H. H. et al. first reported the discovery of RT 078 in Mainland China in 201139,40. However, the epidemic strains in Mainland China are not the highly virulent type of C. difficile, which may be the reason why CDI has not had an outbreak in our country in recent years. Additionally, the number of reports about toxin A-negative and toxin B-positive strains has gradually increased in recent years41. Many studies in Mainland China have found the same situation42. In Asia, the numbers of toxin A-negative and toxin B-positive strains are significantly higher than that in European and American countries, which may also explain why the prevalent strain in Asia is RT 01726,43 and not RT 027. Nevertheless, rational tests and multi-centre or national-level surveillance for CDI, particularly for RT 027, should be introduced to provide essential data and guide future clinical practice44. RT 027 C. difficile isolates were detected in Beijing in 2012 and 2013, following the first discovery in Hong Kong39,44. Thus, it is time to stop neglecting CDI in Mainland China, and this problem should receive greater awareness.
CDI shows a close relationship with AAD, as described above. A number of international studies reported on the wide use of broad-spectrum antimicrobials, particularly the third generation of cephalosporin, the fluoroquinolone drugs and the clindamycin drugs, which subsequently increased the resistance to these drugs. Meanwhile, the balance of intestinal flora is destroyed following antibiotic treatment, which serves as a risk factor for C. difficile-associated diarrhoea (CDAD)45,46,47,48. The resistance rates to ciprofloxacin and cefoxitin were almost 100% in this study, which is consistent with these previous reports. According to the study by Pituch H et al., the rate of resistance to moxifloxacin was 38.5–40.1%, which is similar to that identified in this study (39.0%; 95% CI = 27.9–50.1%)49. Additionally, Ilchmann, C. and Oka, K. et al. found that the rates of resistance to clindamycin and erythromycin were more than 80%50,51, which is also similar to the results of our study ((81.7%; 95% CI = 76.1–87.3%); (80.2%; 95% CI = 73.5–86.9%), respectively). In recent years, a foreign survey reported the appearance of metronidazole- and/or vancomycin-resistant strains52. For example, Martin H et al. discovered that the rate of drug resistance to metronidazole and vancomycin was 1.8% and 0.4% respectively53. However, the articles included in this study did not identify metronidazole- and/or vancomycin-resistant strains. Therefore, the resistance profiles of C. difficile to these two drugs differ between Mainland China and other countries. In Mainland China, only heteroresistance to metronidazole was reported in Shanghai54, and this type of resistance only appears in the first generation of cultured strains and will disappear after several generations55. However, the epidemic ST-37 strain in Mainland China presented the highest resistance rate to many antibiotics, particularly clindamycin, tetracycline, and moxifloxacin, and all ST-37 isolates exhibited multi-drug resistance56. Some studies also indicated that the recurrence rate has increased in recent years following the use of metronidazole and/or vancomycin57. For example, the recurrence rates following metronidazole and vancomycin were reported at 25% and 60%, respectively46.
We realize that there are additional limitations of this study. First, we included few articles about C. difficile in Mainland China, particularly those reporting on the incidence and antibiotic resistance. Second, there is no standard test for detecting toxigenic C. difficile. Enzyme immunoassays and PCR were widely used to detect toxigenic C. difficile, even though the positive detection rate of toxigenic C. difficile was not statistically significant58. Third, the methods used to detect antibiotic resistance differed between studies. The agar dilution method and E-test were most commonly adopted in these articles. Moreover, the selection criteria for Topic 1 have disadvantages as well, as these criteria do not separate diarrhoeal patients into high-risk groups and low-risk groups, such as AAD and non-AAD. Moreover, studies published in local journals, which were not indexed in the electronic databases, might have been missed in this meta-analysis. Last, significant heterogeneity was observed between studies. For example, the incidence of toxigenic C. difficile among diarrhoea patients varied in different subgroups, such as outpatients and inpatients, ordinary inpatients and ICU patients. However, we did not obtain sufficient information about these subgroups for further analysis. Therefore, the prevalence and antibiotic resistance of C. difficile in Mainland China requires further study.
Despite the limitations listed above, there are some noteworthy findings of this study. First, the incidence of toxigenic C. difficile among diarrhoea patients in Mainland China was 14% (95% CI = 12–16%) (n/N = 2,132/15,313), and this rate may be slightly higher than the actual status because some patients included in Topic 1 had additional risk factors. Second, although RT 014/020 were the most common RT strains in Western Australia8, RT 017 (ST-37) was the major epidemic strain in Mainland China. The existence of RT 027 (ST-1) and RT 078 (ST-11) in Mainland China was only reported in a few studies, whereas these are the dominant strains in outbreaks in Europe and North America. Finally, the rates of resistance to erythromycin and clindamycin were greater than 80%, and the rates of resistance to ciprofloxacin and cefoxitin almost reached 100%. These results in Mainland China are consistent with other reports on C. difficile drug resistance in other countries. Most importantly, Mainland China has not detected the metronidazole- and vancomycin-resistant strains of C. difficile to date. Together, the information provided in this study will help guide rational drug use in the clinic and enhance the awareness of C. difficile infection and the epidemiological characteristics of these infections in Mainland China.
Methods
Search strategy
We searched the NCBI PubMed, Web of Science, Science Director, OVID, China National Knowledge Infrastructure (CNKI), Wanfang (Chinese) and Weipu (Chinese) databases to identify research studies that described C. difficile infections in Mainland China. The following keywords were used in the searches: “C. difficile”, “prevalence”, “incidence”, “molecular epidemiology”, “in Mainland China” and “antibiotic resistance.” The search was limited to publications from 2010 to 2016. The same strategies were used for each database. We placed no language restrictions on the searches or search results.
Selection criteria
Two investigators (Chenjie Tang and Lunbiao Cui) independently reviewed the potentially appropriate studies to determine whether they met the predetermined eligibility criteria. Disagreements between the reviewers were resolved by consulting the other authors. Studies obtained from the literature search were checked by titles and abstracts. The titles and abstracts of the potential references were carefully scanned to exclude irrelevant articles. The remaining articles were evaluated to identify research that contained the relevant information, and the full texts were then reviewed in depth. In addition, the inclusion and exclusion criteria were established by the investigators prior to the literature review, and we also estimated and scored the relevance and quality of the references according to JBI (Joanna Briggs Institute). The inclusion criteria were as follows: Topic 1: C. difficile and its toxins were detected in stool samples from diarrhoea patients; Topic 2: C. difficile was detected by MLST or RT methods; and Topic 3: AST was performed on C. difficile. Moreover, studies were only included if they constituted original works and if the samples in these studies came from Mainland China. Only studies published within the last 6 years were included. Studies were excluded if they met the following conditions: (1) review or case report; (2) the data were not from 2010–2016; (3) studies with less than 5 samples; (4) samples from different studies were repeated; (5) the samples did not come from Mainland China; (6) the studies were repeated in different languages; (7) the information was not complete; and (8) the samples did not come from the diarrhoea patients included in Topic 1. Figure 1 shows the flowchart of the procedure used to select the articles.
Data extraction
All data were extracted by two independent investigators. Disagreements in data extraction were resolved by reaching a consensus in accordance with the original study and the other authors’ opinions. The following relevant data were extracted in three predefined tables. Details about the data extraction can be found in the Supplementary Tables 1, 2 and 3.
Statistical analysis
Microsoft Excel (version 12.0) and Stata (version 12.0) were used in this meta-analysis. Based on the possibility of significant heterogeneity, we used the Q-statistic (p < 0.05 was considered to indicate statistically significant heterogeneity) to estimate the heterogeneity between studies. A random effect model (REM) or fixed effect model (FEM) was chosen for the meta-analysis according to the p value. We calculated the incidence of toxigenic C. difficile among diarrhoea patients in the provinces of Mainland China, as well as the molecular epidemiology and antibiotic resistance using 95% confidence intervals (CIs) and a suitable model. Data manipulation and statistical analyses were performed using Stata 12.0(StataCorp. 2011. Stata Statistical Software: Release 12. College Station, TX: StataCorp LP.).
Additional Information
How to cite this article: Tang, C. et al. The incidence and drug resistance of Clostridium difficile infection in Mainland China: a systematic review and meta-analysis. Sci. Rep. 6, 37865; doi: 10.1038/srep37865 (2016).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Bartlett, J. G., Chang, T. W., Gurwith, M., Gorbach, S. L. & Onderdonk, A. B. Antibiotic-associated pseudomembranous colitis due to toxin-producing clostridia. The New England journal of medicine. 298, 531–534 (1978).
Moreno, M. A., Furtner, F. & Rivara, F. P. Clostridium difficile: a cause of diarrhea in children. JAMA pediatrics. 167, 592 (2013).
Bartlett, J. G. Clinical practice. Antibiotic-associated diarrhea. The New England journal of medicine. 346, 334–339 (2002).
O’Connor, J. R., Johnson, S. & Gerding, D. N. Clostridium difficile infection caused by the epidemic BI/NAP1/027 strain. Gastroenterology. 136, 1913–1924 (2009).
Cohen, S. H. et al. Clinical practice guidelines for Clostridium difficile infection in adults: 2010 update by the society for healthcare epidemiology of America (SHEA) and the infectious diseases society of America (IDSA). Infection control and hospital epidemiology. 31, 431–455 (2010).
Kuijper, E. J. et al. Update of Clostridium difficile-associated disease due to PCR ribotype 027 in Europe. Euro surveillance: bulletin Europeen sur les maladies transmissibles = European communicable disease bulletin. 12, E1–2 (2007).
McDonald, L. C. et al. Recommendations for surveillance of Clostridium difficile-associated disease. Infection control and hospital epidemiology. 28, 140–145 (2007).
Collins, D. A. & Riley, T. V. Routine detection of Clostridium difficile in Western Australia. Anaerobe. 37, 34–37 (2016).
Baldoni, D., Gutierrez, M., Timmer, W. & Dingemanse, J. Cadazolid, a novel antibiotic with potent activity against Clostridium difficile: safety, tolerability and pharmacokinetics in healthy subjects following single and multiple oral doses. The Journal of antimicrobial chemotherapy. 69, 706–714 (2014).
McDonald, L. C. et al. An epidemic, toxin gene-variant strain of Clostridium difficile. The New England journal of medicine. 353, 2433–2441 (2005).
McFarland, L. V. Update on the changing epidemiology of Clostridium difficile-associated disease. Nature clinical practice. Gastroenterology & hepatology. 5, 40–48 (2008).
Warny, M. et al. Toxin production by an emerging strain of Clostridium difficile associated with outbreaks of severe disease in North America and Europe. Lancet. 366, 1079–1084 (2005).
Rea, M. C. et al. Effect of broad- and narrow-spectrum antimicrobials on Clostridium difficile and microbial diversity in a model of the distal colon. Proceedings of the National Academy of Sciences of the United States of America. 108 Suppl 1, 4639–4644 (2011).
Magill, S. S. et al. Multistate point-prevalence survey of health care-associated infections. The New England journal of medicine. 370, 1198–1208 (2014).
Kardas, P., Devine, S., Golembesky, A. & Roberts, C. A systematic review and meta-analysis of misuse of antibiotic therapies in the community. International journal of antimicrobial agents. 26, 106–113 (2005).
Harvey, N., Dennison, E. & Cooper, C. Osteoporosis: impact on health and economics. Nature reviews. Rheumatology. 6, 99–105 (2010).
Putsathit, P., Kiratisin, P., Ngamwongsatit, P. & Riley, T. V. Clostridium difficile infection in Thailand. International journal of antimicrobial agents. 45, 1–7 (2015).
Lessa, F. C., Gould, C. V. & McDonald, L. C. Current status of Clostridium difficile infection epidemiology. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America. 55 Suppl 2, S65–70 (2012).
Fang, W. J. et al. Clostridium difficile carriage in hospitalized cancer patients: a prospective investigation in eastern China. BMC infectious diseases. 14, 523 (2014).
HU, Y. et al. Exploring on the Status, Reasons and Countermeasures of Antibiotics Abuse. Chinese Journal of Social Medicine. 128–130 (2013).
Li, Z. et al. Analysis of the antibiotic use in our country. International medicine & health guidance news. 83–84 (2005).
Li, F. C. & Liping Liu . The status and strategy of antibiotics abuse in our country. Chin J of Clinical Rational Drug Use. 175–177 (2014).
Wang, J. A Prospective Study on Risk Factors for Toxigenic Clostridium difficile Colonization and Infection among Inpatients with Hematologic Disease and Analysis of Drug Resistance of Clinical Isolates [Master]: Hebei Medical University; 2014.
Tang, H. X. Detection and analysis of Clostridium difficile in diarrhea patients [Master]: GuangZhou Medical University; 2014.
li, Y. Q., Nie, Y. Q. & Yang, Y. M. Analysis of drug resistance in Clostridium difficile isolation. Chin J Gastroenteroal Hepatol. 922–925 (2010).
Zhou, F. et al. Ribotyping and antimicrobial resistance patterns of Clostridium difficile during two periods within an interval of 5 years. Chin J Infect Chemother. 116–120 (2014).
Yang, J. Study on the molecular epidemiology and antibiotic resistance of Clostridium difficile isolates [Master]: Hebei Medical University; 2012.
Tian, T. T. Molecular epidemiology and drug resistance analysis of Clostridium difficile carried by the healthy population [Master]: Hebei Medical University; 2015.
Dong, D. F. et al. Clinical and microbiological characterization of Clostridium difficile infection in a tertiary care hospital in Shanghai, China. Chinese medical journal. 127, 1601–1607 (2014).
Kelly, C. P. & LaMont, J. T. Clostridium difficile–more difficult than ever. The New England journal of medicine. 359, 1932–1940 (2008).
Lucado, J., Gould, C. & Elixhauser, A. Clostridium Difficile Infections (CDI). In Hospital Stays, 2009: Statistical Brief #124 Healthcare Cost and Utilization Project (HCUP). Statistical Briefs. Rockville (MD).: Agency for Health Care Policy and Research (US); 2006.
Yamagishi, Y. & Mikamo, H. Recent epidemiology of Clostridium difficile infection in Japan. The Japanese journal of antibiotics. 68, 345–358 (2015).
Lessa, F. C. et al. Burden of Clostridium difficile infection in the United States. The New England journal of medicine. 372, 825–834 (2015).
Choi, H. Y. et al. The epidemiology and economic burden of Clostridium difficile infection in Korea. BioMed research international. 2015, 510386 (2015).
Jin, K., Wang, S., Huang, Z. & Lu, S. Clostridium difficile infections in China. Journal of biomedical research. 24, 411–416 (2010).
Hawkey, P. M. et al. Molecular epidemiology of Clostridium difficile infection in a major chinese hospital: an underrecognized problem in Asia? Journal of clinical microbiology. 51, 3308–3313 (2013).
Yan, Q. et al. Multilocus sequence typing (MLST). analysis of 104 Clostridium difficile strains isolated from China. Epidemiology and infection. 141, 195–199 (2013).
Tian, T.-t. et al. Molecular Characterization of Clostridium difficile Isolates from Human Subjects and the Environment. PloS one. 11 (2016).
Cheng, V. C. et al. Clostridium difficile ribotype 027 arrives in Hong Kong. International journal of antimicrobial agents. 34, 492–493 (2009).
Huang, H. H. et al. Antimicrobial resistance in 188 clinical isolates of Clostridium difficile. Chin J Infect Chemother. 1–5 (2011).
Rupnik, M. How to detect Clostridium difficile variant strains in a routine laboratory. Clinical microbiology and infection: the official publication of the European Society of Clinical Microbiology and Infectious Diseases. 7, 417–420 (2001).
Wang, J. et al. PCR ribotyping and toxin gene polymorphism of Clostridium difficile in China. Chinese J Infection Control. 1–7 (2014).
Kim, H. et al. Increasing prevalence of toxin A-negative, toxin B-positive isolates of Clostridium difficile in Korea: impact on laboratory diagnosis. Journal of clinical microbiology. 46, 1116–1117 (2008).
Cheng, J. W. et al. The First Two Clostridium difficile Ribotype 027/ST1 Isolates Identified in Beijing, China-an Emerging Problem or a Neglected Threat? Scientific reports. 6, 18834 (2016).
van der Kooi, T. I. et al. Antibiotic use and other risk factors at hospital level for outbreaks with Clostridium difficile PCR ribotype 027. Journal of medical microbiology. 57, 709–716 (2008).
Surawicz, C. M. & Alexander, J. Treatment of refractory and recurrent Clostridium difficile infection. Nature reviews. Gastroenterology & hepatology. 8, 330–339 (2011).
Owens, R. C.,, Jr., Donskey, C. J., Gaynes, R. P., Loo, V. G. & Muto, C. A. Antimicrobial-associated risk factors for Clostridium difficile infection. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America. 46 Suppl 1, S19–31 (2008).
Loo, V. G. et al. A predominantly clonal multi-institutional outbreak of Clostridium difficile-associated diarrhea with high morbidity and mortality. The New England journal of medicine. 353, 2442–2449 (2005).
Pituch, H. et al. Characterization and antimicrobial susceptibility of Clostridium difficile strains isolated from adult patients with diarrhoea hospitalized in two university hospitals in Poland, 2004–2006. Journal of medical microbiology. 60, 1200–1205 (2011).
Ilchmann, C. et al. Comparison of resistance against erythromycin and moxifloxacin, presence of binary toxin gene and PCR ribotypes in Clostridium difficile isolates from 1990 and 2008. European journal of clinical microbiology & infectious diseases: official publication of the European Society of Clinical Microbiology. 29, 1571–1573 (2010).
Oka, K. et al. Molecular and microbiological characterization of Clostridium difficile isolates from single, relapse, and reinfection cases. Journal of clinical microbiology. 50, 915–921 (2012).
Johnson, S., Sanchez, J. L. & Gerding, D. N. Metronidazole resistance in Clostridium difficile. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America. 31, 625–626 (2000).
Martin, H. et al. Characterization of Clostridium difficile strains isolated from patients in Ontario, Canada, from 2004 to 2006. Journal of clinical microbiology. 46, 2999–3004 (2008).
Huang, H. H. et al. Antimicrobial susceptibility and heteroresistance in Chinese Clostridium difficile strains. Anaerobe. 16, 633–635 (2010).
Pelaez, T. et al. Metronidazole resistance in Clostridium difficile is heterogeneous. Journal of clinical microbiology. 46, 3028–3032 (2008).
Dong, D. et al. Clinical and microbiological characterization of Clostridium difficile infection in a tertiary care hospital in Shanghai, China. Chinese medical journal. 127, 1601–1607 (2014).
Pelaez, T. et al. In vitro activity of ramoplanin against Clostridium difficile, including strains with reduced susceptibility to vancomycin or with resistance to metronidazole. Antimicrobial agents and chemotherapy. 49, 1157–1159 (2005).
Wu, J. S. The fluorescent PCR applied in the detection of Clostridium difficile Zhejiang JITCWM. 346–347 (2011).
Kong, J. & Cai, X. The comparison and analysis of two Clostridium difficile detection methods. Chin J Clin Lab Sci. 632–633 (2014).
Yu, S., Ai, X. & Hu, Y. Analysis of pathogenic bacteria in antibiotic - associated diarrhea in the hospital elderly. The Chinese Journal of Clinical Pharmacology. 27, 976–979 (2011).
Jia, H. B. Study on the Rapid Identification and Toxin Detection of Clinical Clostridium difficile Strains [Master]: Peking Union Medical College; 2011.
Guo, S., Xu, X. W. & Dong, F. Risk factors of Clostridium difficile-associated diarrhea in children. Zhonghua yi xue za zhi. 92, 1836–1840 (2012).
Han, X. H. et al. Clostridium difficile infection in hospitalized cancer patients in Beijing, China is facilitated by receipt of cancer chemotherapy. Anaerobe. 24, 82–84 (2013).
Zhao, C. & Xu, X. Virulence genes and clinical features of Clostridium difficile-associated diarrhea in children. Zhonghua er ke za zhi. Chinese journal of pediatrics. 53, 522–527 (2015).
Feng, W. et al. The application and epidemiological research of xTAG GPP multiplex PCR in the diagnosis of infectious diarrhea. Zhonghua yi xue za zhi. 95, 435–439 (2015).
Song, W. B. et al. Investigation on Clostridium difficile infection among diarrhea patients in Lanzhou area. Chinese Journal of Microecology. 26, 1138–1139, 1145 (2014).
Yang, F. Y., Li, P. & Li, Y. Q. PCR ribotyping profiles of Clostridium difficile and risk factors of Clostridium difficile-associated diarrhea. Journal of Nursing Science. 1–3 (2012).
Wang, P. The Epidemiological Study of Clostridium difficile Infection in South China and Evaluation of a New Molecular Biological Detection Technology [Dr.]: Southern Medical University; 2014.
Deng, J. K. Clinical evaluation of xTAG GPP for simultaneous enteropathogens detection and of MALDI-TOF MS for rapid identification of enteropathogenic bacteria [Master]: Southern Medical University; 2014.
Mu, X. P. et al. Detection and analysis of Clostridium difficile toxin in feces of hospitalized children with diarrhea. China Tropical Medicine. 1124–1126 (2014).
Wang, R. L. et al. The investigation of Clostridium difficile infection in patients with diarrhea. The Journal of Practical Medicine. 144–146 (2015).
Lu, Y. L. & Jin, H. J. Clostridium difficile diarrhea and its laboratory diagnosis. Henan J Prev Med. 391–392+415 (2013).
Xu, Y. Q. & Deng, M. Epidemiological characteristics of Clostridium difficile infections and current status of infection control. Chin J Nosocomiol. 23, 5073–5075 (2013).
Galaydick, J. et al. Seek and you shall find: prevalence of Clostridium difficile in Wuhan, China. American journal of infection control. 43, 301–302 (2015).
Cheng, G. P. et al. Analysis of Clostridium difficile associated diarrhea in pediatric patients with antibiotic-associated diarrhea. Chin J Pediatr. 53, 220–224 (2015).
Bai, J., Lei, Y. T. & Zhang, J. B. Clinical Characteristics of Clostridium Difficile-associated Diarrhea and Prevention Strategy. Chinese and Foreign Medical Research. 1–3 (2013).
Chen, W. et al. The etiology, molecular epidemiology and clinical characteristics of Clostridium difficile infections in Xiangya Hospital. Chin J Lab Med. 855–860 (2014).
Li, C. H. Epidemiological studies of C. difficile multilocus sequence typing and risk factors [Dr.]: Central South University; 2014.
Wu, L. Real-time PCR Method for the Detection of Clostridium difficile and Cryptosporidium parvum in biological samples [Master]: Suzhou University; 2012.
Li, Y. J. et al. Etiological study of infectious diarrhea among some hospitalized children in Suzhou City. Occupation and Health. 358–361 (2015).
Yang, Y. The analysis of antibiotic associated diarrhea clinical laboratory test results. Journal of Ningxia Medical University. 86–88 (2014).
Zhou, F. F., Wu, S., Klena, J. D. & Huang, H. H. Clinical characteristics of Clostridium difficile infection in hospitalized patients with antibiotic-associated diarrhea in a university hospital in China. European journal of clinical microbiology & infectious diseases: official publication of the European Society of Clinical Microbiology. 33, 1773–1779 (2014).
Chen, X. et al. Clinical evaluation of GeneXpert real-time fluorescence quantitation PCR in rapid detection of Clostridium difficile. Laboratory Medicine. 590–592+580 (2014).
Gao, Y. T. et al. Analysis of clinical characteristics of Clostridium difficile infection for hospitalized patients with diarrhea Journal of Shanghai Jiaotong University Medical Science. 1481–1484+1496 (2014).
Zhang, C. Y. et al. Clinical Analysis and Laboratory Diagnosis of Clostridium (C.). Difficile- associated Diarrhea. Parasitoses and Infectious Diseases. 68–72 (2012).
Wang, X., Cai, L., Yu, R., Huang, W. & Zong, Z. ICU-Onset Clostridium difficile infection in a university hospital in China: a prospective cohort study. PloS one. 9, e111735 (2014).
Chen, Y. B. et al. Molecular epidemiology of Clostridium difficile in a tertiary hospital of China. Journal of medical microbiology. 63, 562–569 (2014).
Lu, X. F., Wu, J., Xu, S., Cai, J. & Wu, X. A sensitive method for detection of Clostridium difficile in diarrheal patients. Chin Crit Care Med. 27, 525–526 (2015).
Pan, J. S. et al. Incidence of Clostridium difficile-associated diarrhea in elderly patients. Chin J of Nosocomiol. 5176–5177+5180 (2013).
Zhu, S. S. et al. Comparison of polymerase chain reaction ribotyping, toxinotyping and nutritional aspects of toxin production of Clostridium difficile strains. Biomedical reports. 2, 477–480 (2014).
Xu, W. et al. The Clostridium difficiletest detection analysis of elderly hospitalized patients with diarrhea in Shaoxing. Shanghai Journal of Preventive Medicine. 28, 200–201 (2016).
Li, W. G. et al. MLST typing of Clostridium difficile isolated from rural and urban China: a comparative study. Chin J Nosocomiol. 22, 2699–2701 (2012).
Wang, Z. et al. Virulence and genotype distribution of Clostridium difficile isolated from infants and childen. Chin J Clin Lab Sci. 27–30 (2016).
Lidan, C. et al. Molecular characterization and antimicrobial susceptibility of tcdA-negative Clostridium difficile isolates from Guangzhou, China. Diagnostic microbiology and infectious disease. 84, 361–365 (2016).
Acknowledgements
This study was supported by a grant from the Key Laboratory of Enteric Pathogenic Microbiology of the Ministry Health (EM201401) and the Project of the Key Laboratory for Laboratory Medicine of Jiangsu Province (No. XK201114).
Author information
Authors and Affiliations
Contributions
C.T. and L.C. reviewed the potentially appropriate studies and prepared the manuscript; Y.X., L.X.1, and P.S.1 reviewed the papers according to the three topics, respectively. C.L. and W.X. conducted the statistical analyses, interpreted the data and critically revised the manuscript. G.L. supervised all phases of the study and critically revised the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Tang, C., Cui, L., Xu, Y. et al. The incidence and drug resistance of Clostridium difficile infection in Mainland China: a systematic review and meta-analysis. Sci Rep 6, 37865 (2016). https://doi.org/10.1038/srep37865
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep37865
This article is cited by
-
An epidemiological surveillance study (2021–2022): detection of a high diversity of Clostridioides difficile isolates in one tertiary hospital in Chongqing, Southwest China
BMC Infectious Diseases (2023)
-
Prevalence and molecular subtyping of Blastocystis in patients with Clostridium difficile infection, Singapore
Parasites & Vectors (2021)
-
A 2-step algorithm combining glutamate dehydrogenase and nucleic acid amplification tests for the detection of Clostridioides difficile in stool specimens
European Journal of Clinical Microbiology & Infectious Diseases (2021)
-
Molecular characteristics of Clostridium difficile in children with acute gastroenteritis from Zhejiang
BMC Infectious Diseases (2020)
-
Epidemiology of Clostridium difficile infection in hospitalized adults and the first isolation of C. difficile PCR ribotype 027 in central China
BMC Infectious Diseases (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.