Abstract
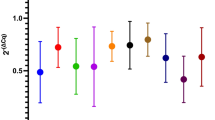
Evaluation of gene expression levels by reverse transcription quantitative real-time PCR (RT-qPCR) has for many years been the favourite approach for discovering disease-associated alterations. Normalization of results to stably expressed reference genes (RGs) is pivotal to obtain reliable results. This is especially important in relation to neurodegenerative diseases where disease-related structural changes may affect the most commonly used RGs. We analysed 15 candidate RGs in 98 brain samples from two brain regions from Alzheimer’s disease (AD), Parkinson’s disease (PD), Multiple System Atrophy, and Progressive Supranuclear Palsy patients. Using RefFinder, a web-based tool for evaluating RG stability, we identified the most stable RGs to be UBE2D2, CYC1, and RPL13 which we recommend for future RT-qPCR studies on human brain tissue from these patients. None of the investigated genes were affected by experimental variables such as RIN, PMI, or age. Findings were further validated by expression analyses of a target gene GSK3B, known to be affected by AD and PD. We obtained high variations in GSK3B levels when contrasting the results using different sets of common RG underlining the importance of a priori validation of RGs for RT-qPCR studies.
Similar content being viewed by others
Article PDF
Change history
23 July 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
Author information
Authors and Affiliations
Additional information
Electronic supplementary material
The online version of this article (doi:10.1038/srep37116) contains supplementary material, which is available to authorized users.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Rydbirk, R., Folke, J., Winge, K. et al. Assessment of brain reference genes for RT-qPCR studies in neurodegenerative diseases. Sci Rep 6, 37116 (2016). https://doi.org/10.1038/srep37116
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep37116
This article is cited by
-
Novel Somatostatin Receptor-4 Agonist SM-I-26 Mitigates Lipopolysaccharide-Induced Inflammatory Gene Expression in Microglia
Neurochemical Research (2022)
-
Assessment of common housekeeping genes as reference for gene expression studies using RT-qPCR in mouse choroid plexus
Scientific Reports (2021)
-
Validation of reference genes for use in untreated bovine fibroblasts
Scientific Reports (2021)
-
Epigenetic modulation of AREL1 and increased HLA expression in brains of multiple system atrophy patients
Acta Neuropathologica Communications (2020)
-
Genome-wide DNA methylation meta-analysis in the brains of suicide completers
Translational Psychiatry (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.