Abstract
Using a large longitudinal data set spanning 4 years, we examined whether a change in self-reported sleep duration is associated with metabolic syndrome (MetS). Current analysis included 15,753 participants who were free of MetS during both 2006–2007 and 2010–2011. Sleep duration was categorized into seven groups: ≤5.5 h, 6.0–6.5 h, 7.0 h, 7.5–8.0 h, ≥8.5 h, decrease ≥2 h, and increase ≥2 h. Cox proportional hazards models were used to calculate hazard ratios (HRs) and their confidence intervals (CI) for MetS, according to sleep duration. Compared to the reference group of persistent 7-h sleepers, a decrease of ≥2 h sleep per night was associated with a higher risk of incident MetS (HR = 1.23, 95% CI = 1.05–1.44) in analyses adjusted for age, sex, sleep duration at baseline, marital status, monthly income per family member, education level, smoking status, drinking status, physical activity, body mass index, snoring status and resting heart rate. An increased risk of MetS incidence was also observed in persistent short sleepers (average ≤5.5 h/night; HR = 1.22, 95% CI = 1.01–1.50). This study suggests individuals whose sleep duration decreases ≥2 h per night are at an increased risk of MetS.
Similar content being viewed by others
Introduction
Metabolic syndrome (MetS) is closely linked to lifestyle and clustering of risk factors, including abdominal adiposity, hypertension, hypertriglyceridemia, low high-density lipoprotein (HDL) cholesterol and hyperglycemia. Given that MetS is becoming a pandemic, with prevalence rates between 20 and 30% among the adult population1, identification of modifiable risk factors associated with the development of MetS is important to public health. Prior cross-sectional studies suggest short sleep duration is significantly associated with increased risk of MetS2,3,4,5. Recent prospective analyses have also provided robust evidence of a similar association between short sleep duration and a higher incidence of MetS6,7,8. Conversely, other studies have shown an association between long sleep duration and MetS9,10. Thus, it is necessary to assess the temporal relationship between sleep duration and MetS.
Increasing concerns about reductions in night time sleep duration caused by our new 24/7 society11,12 cannot be addressed by studies based on a one-off measurement of sleep duration. Thus, it is impossible to determine from such studies6,7,8 whether associations observed between short or long sleep duration and MetS are generated by persistent exposure, or whether decreases or increases from a normal average of 7 h per night also confer risk. A significant decrease or increase in sleep duration has also been associated with increased all-cause mortality13. Cross-sectional data from the 2005–2006 National Health and Nutrition Examination Survey looked at the change in objective sleep duration and individual cardiometabolic risk factors14. Data from the Whitehall II study suggests that individuals whose sleep duration increases are at an increased risk of type 2 diabetes, although we are unaware of any population level investigations of change in sleep duration and incidence of MetS15.
In this study, we examined potential longitudinal associations between changes in sleep duration over a 4-year exposure period (the reference group of persistent 7-h sleepers ref. 16) and incidence of MetS in the subsequent 4-year period. Using a large prospective study from Kailuan, we took account of sleep duration and snoring status at baseline examination, as a potential confounder and mediator of these associations.
Results
Table 1 shows the general characteristics of study participants according to the incidence of MetS. Baseline SBP, DBP, TC, BMI, TG, FBG, and RHR were significantly higher, and HDL-C was significantly lower in individuals who developed MetS compared with non-MetS (p < 0.001).
Table 2 shows baseline characteristics according to sleep duration. Significant association was found between sleep duration and age, sex, education level, income level, smoking status, drinking status, physical activity, BMI, SBP, DBP, FBG, TG, HDL-C, RHR, snoring status, history of stroke, myocardial infarction, and cancer (p < 0.001).
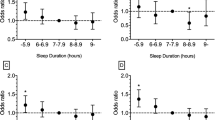
During an average 3.4-year follow-up, 6,302 participants (40.0%) developed MetS. Age, sex, sleep duration at baseline, sex, marital status, monthly income per family member, education level, smoking status, drinking status, physical activity, body mass index, snoring status and resting heart rate were designated as confounding factors in Model 2. After adjusting for these confounding factors, participants who slept ≤5.5 h per night had an increased risk of developing MetS compared with participants who persistently slept 7 h (HR = 1.22, 95% CI = 1.01–1.50; all p-values <0.05). Compared with the reference group of persistent 7-h sleepers, a decrease of ≥2 h sleep per night was associated with a higher risk of MetS incidence (HR = 1.23, 95% CI = 1.05–1.44; all p-values <0.05) (Table 3). Moreover, the association between decreased sleep duration and risk of MetS remained significant upon repeating our analysis and excluding individuals with stroke, myocardial infarction and cancer, respectively (Table 4).
Discussion
Our findings suggest persistent short sleep (average <5.5 h) increases the risk of MetS incidence. In contrast, increased sleep duration was not associated with higher incidence of MetS compared with individuals persistently sleeping 7 h per night. Compared to these individuals, we also found that a decrease of ≥2 h in sleep duration over a four-year exposure period was associated with an increased risk of developing MetS in analyses adjusted for age, sex, sleep duration at baseline, sex, marital status, monthly income per family member, education level, smoking status, drinking status, physical activity, body mass index, snoring status and resting heart rate. A set of sensitivity analyses further confirmed these findings.
After 3.4 years of follow-up, the overall incidence of MetS in this study was 40.0%, which was significantly higher than a previously published report about midlife adults comprising a sub-cohort of ARIRANG and Quebec Family Study patients6,7,8. A recent meta-analysis and several prospective studies have suggested that short rather than long sleep duration is significantly associated with increased risk of MetS2,6,7,8. Our study results were quite similar to the above-published study. However, some studies have shown that longer sleep duration may also be a risk factor in participants9,10. Prior evidence connecting sleep duration to cardiometabolic risk is varied17, with a reported U-shaped relationship between sleep duration and MetS18, type 2 diabetes19,20, and mortality13,21. Various factors may contribute to this difference, such as geographic and ethnic variations, varying clinical definitions of MetS, limited confounding factors, and major diseases affecting sleep duration. Thus, in our study, a greater quantity of important influencing factors has been analyzed than in previous research, including RHR and snoring status. First, taking into consideration prior evidence showing RHR is an independent risk factor for existing MetS and a powerful predictor for future incidence of MetS22,23,24,25, we adjusted for RHR in our full model. Second, poor sleep efficiency, often relating to snoring status, has a significant correlation with over-activity of the sympathetic nervous system, which could result in insulin resistance26 and increased blood pressure27. In light of this, snoring was considered as a confounding factor in our assessment of the relative risks for MetS. Moreover, age, sex, socioeconomic status may contribute to the association between the altered sleep duration and incident MetS, as advanced age is associated with changes in sleep architecture with increased difficulties in sleep initiation and maintenance28. And, sleep problems are particularly common in people with anxiety, depression, bipolar disorder, and poor socioeconomic status. Compared to men, women are more likely to develop depression and sleep problems. Therefore, we adjusted for age, sex, income level and education level in the full model. Finally, subjects with stroke, myocardial infarction, and cancer may experience sleep deprivation, which could lead to biased results. Therefore, we further confirmed our finding that short sleep duration increases the risk of MetS incidence by repeating the same analysis and excluding individuals with stroke, myocardial infarction and cancer, respectively.
An inherent limitation of previous studies has been the reliance on a single time point by which to assess sleep duration, which may have occurred several decades prior to the event, and is therefore likely to yield biased estimates of the association. Moreover, there has been no consideration of how sleep duration varies within individuals over time, and the subsequent impact this could have on changes in sleep duration and future risk of MetS. As the current study appears to be the first to demonstrate an association between decreased sleep duration and incidence of MetS in a large cohort study from China, our findings thus provide the first evidence that change in sleep duration is not only strongly associated with future risk of MetS, it is also likely to be a more accurate indicator of the true magnitude of risk as compared with a single measure of sleep duration self-reported several years before the onset of MetS.
It is an interesting and new observation that a decrease in sleep duration over a 4-year period is more deleterious than persistent short sleep. This may be related to the shortened sleep duration (≤5.5 h) among sleepers with a significant decrease in sleep duration. There are a number of biological mechanisms through which reduced sleep duration may lead to MetS. Experiments have demonstrated short-term sleep deprivation among healthy subjects results in adverse physiological changes, including decreased glucose tolerance and increased insulin resistance, sympathetic tone and blood pressure29.
The strengths of our study include a prospective cohort design, large sample size, the Asian ethnicity of our participants, and a broad spectrum of potential confounding parameters. However, there are several potential limitations of our study. First, we only collected information on sleep duration by self-reported questionnaires, without 24-h polysomnography information. However, self-reported measures showed good agreement when compared to quantitative sleep assessment with monitoring30,31. Second, our study did not exclude participants with obstructive sleep apnea syndrome, and there is some evidence that sleep apnea is associated with increased risk of MetS32. Furthermore, we did not collect sufficient information on the pre- or post-menopause status of women, which appears to be an important determinant of MetS risk in women8. Previous studies have reported gender-specific differences in sleep patterns may be influenced by differences in social or household roles, or sex hormones33. Finally, most of the participants from the Kailuan coal mine were male; thus, the sex distribution of participants was unbalanced and cannot be viewed as a representative sample of the general Chinese population.
In conclusion, our findings demonstrate an association between reduced sleep duration and increased future risk of MetS. This study also highlights the need to take into consideration change of sleep duration when estimating risk, rather than relying on a single measure of exposure that often precedes the outcome by several decades. Our results should encourage and support individuals to maintain or adopt a 7-h sleep duration each night, as this could have significant beneficial effects in stemming the growing prevalence of MetS.
Methods
Ethics Statement
In compliance with the Declaration of Helsinki, the protocol for this study was approved by the Ethics Committee of Kailuan General Hospital and all participants provided informed written consent with signatures34,35.
Study Design and Participants
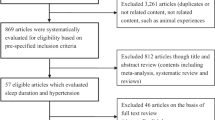
The Kailuan study was a prospective cohort study designed to investigate the association of risk factors and chronic disease. The Kailuan community, located at the center of the Kailuan Coal Industry in Hebei Province, China, has approximately 7.2 million inhabitants with 11 hospitals responsible for their healthcare. From June 2006 to October 2007, a total of 155,418 employees (including retired individuals) in the community were invited to participate and 65.31% of them agreed to be participants. A total of 101,510 participants (81,110 men and 20,400 women, aged 18–98 years old) were recruited into the Kailuan study. During baseline analysis, a total of 85,757 participants were excluded from the recruited population, including 33,651 participants lacking face-to-face follow-up data during 2010–2011 survey, 43,154 participants diagnosed with MetS prior to the 2010–2011 survey, and 6,176 participants lacking complete data regarding sleep duration and other indicators. In addition, 2,776 individuals who did not participate in the 2012–2013 and 2014–2015 surveys were excluded. The remaining 15,753 participants were included in the final analysis (Fig. 1). Follow-up evaluations included biennial measurement of laboratory parameters and recording of adverse events. All physicians and nurses had rigorous, unified training before conducting this study.
Assessment of Potential Covariates
All participants underwent a clinical examination and standardized interview. Physical activity was evaluated based on individual responses to questions regarding the types and frequencies of physical activity at work and during leisure time. Physical activity was classified as “≥4 times per week and ≥20 min at a time”, “<80 min per week”, or “none”. Smoking and drinking statuses were classified as “never”, “former”, or “current” according to self-reported information. Monthly income per family member (at baseline) was categorized as “<¥600”, “¥600–799”, “¥800–999” and “≥¥1,000”.
Anthropomorphic parameters such as height, weight, and waist circumference were measured. Body mass index (BMI) was calculated as weight/height (kg/m2). Systolic blood pressure (SBP) and diastolic blood pressure (DBP) were measured thrice in the seated position using a mercury sphygmomanometer, and the average of three readings was used for analyses.
Blood samples were collected from the antecubital vein after an overnight fast. Venous blood was obtained for determination of routine chemistry, including fasting blood glucose (FBG), high-density lipoprotein-cholesterol (HDL-C), total cholesterol (TC), and triglycerides (TG). Resting heart rate (RHR) was measured and calculated from electrocardiogram recordings after subjects acclimated to the hospital setting for ≥30 min and were in the supine position for ≥5 min.
Assessment of Sleep Duration
Sleep duration was elicited by the question “How many hours of sleep have you gotten on an average night in the preceding 3 months?” Response categories were ≤5, 6, 7, 8, and ≥9 h. Sleep duration in 2006–2007 and 2010–2011 was used to determine changes in sleep duration over the two exposure periods. To calculate change, baseline sleep duration (2006–2007) was subtracted from the sleep duration reported at follow-up (2010–2011). Decreased sleep was defined as a decrease of ≥2 h in sleep duration; whereas, increased sleep was defined as an increase of ≥2 h in sleep duration. Changes in sleep duration with the range of 0–1 h between 2006 and 2010 surveys was not considered to be different and classified as ‘no change in sleep duration’. For these stable sleepers, average sleep duration was calculated and categorized into five levels: ≤5.5, 6.0–6.5, 7.0, 7.5–8.0 and ≥8.5 h.
In addition, participants were asked, “Do you generally snore when you sleep?” Response alternatives were “yes” and “no.”
Follow-Up and Diagnosis of Metabolic Syndrome (MetS)
Participants were followed up by face-to-face interviews at every 2-year routine medical examination until December 31, 2015, or to the event of interest or death. Follow-ups were performed by trained physicians who were blinded to baseline data. MetS was diagnosed when a participant had three or more of the following components: 1) Waist circumference ≥90 cm for men or ≥80 cm for women; 2) TG ≥1.7 mmol/l; 3) HDL-C <1.03 mmol/l for men or <1.30 mmol/l for women; 4) SBP/DBP ≥130/85 mmHg or current use of antihypertensive medications; 5) FBG ≥5.6 mmol/l, previous diagnosis of type 2 diabetes, or current use of oral hypoglycemic agents or insulin36.
Statistical analysis
Continuous variables were expressed as means ± standard deviations; whereas, categorical variables were expressed as percentages. We compared parameters according to each sleep duration group. One-way analysis of variance (ANOVA) was used for non-paired samples of normally distributed parameters and the Kruskal-Waillis test was applied for non-parametric variables. A Chi-squared test was applied to compare categorical variables. A multivariate analysis was performed using two models: Model 1 was adjusted for age, sex, and sleep duration at baseline; Model 2 included Model 1 parameters plus monthly income per family member, education level, marital status, smoking status, drinking status, physical activity, BMI, snoring status and RHR; We used Cox proportional hazards modeling to calculate the hazard ratio (HR) and 95% confidence interval (CI) of MetS, using the group with persistent 7-h sleep duration as a reference category. Person-years were calculated from the date of the 2010 survey was conducted to the date when MetS was detected (depending on the analysis in question), date of death or date of participating in the last interview in this analysis, whichever came first. Further, as individuals with major fatal diseases could impact our assessment of sleep duration and future MetS risk, we conducted three sensitivity analyses to test the robustness of our findings by repeating our aforementioned analysis and excluding individuals with stroke, myocardial infarction and cancer, respectively. Statistical analysis was performed using SAS 9.3 statistical software (SAS Institute, Cary, NC).
Additional Information
How to cite this article: Song, Q. et al. Changes in sleep duration and risk of metabolic syndrome: the Kailuan prospective study. Sci. Rep. 6, 36861; doi: 10.1038/srep36861 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Grundy, S. M. Metabolic syndrome pandemic. Arteriosclerosis, thrombosis, and vascular biology 28, 629–636, doi: 10.1161/ATVBAHA.107.151092 (2008).
Xi, B., He, D., Zhang, M., Xue, J. & Zhou, D. Short sleep duration predicts risk of metabolic syndrome: a systematic review and meta-analysis. Sleep medicine reviews 18, 293–297, doi: 10.1016/j.smrv.2013.06.001 (2014).
Chaput, J. P., McNeil, J., Despres, J. P., Bouchard, C. & Tremblay, A. Seven to eight hours of sleep a night is associated with a lower prevalence of the metabolic syndrome and reduced overall cardiometabolic risk in adults. PloS one 8, e72832, doi: 10.1371/journal.pone.0072832 (2013).
Wu, M. C. et al. Short sleep duration associated with a higher prevalence of metabolic syndrome in an apparently healthy population. Preventive medicine 55, 305–309, doi: 10.1016/j.ypmed.2012.07.013 (2012).
Kobayashi, D., Takahashi, O., Deshpande, G. A., Shimbo, T. & Fukui, T. Relation between metabolic syndrome and sleep duration in Japan: a large scale cross-sectional study. Intern Med 50, 103–107 (2011).
Kim, J. Y. et al. A prospective study of total sleep duration and incident metabolic syndrome: the ARIRANG study. Sleep medicine 16, 1511–1515, doi: 10.1016/j.sleep.2015.06.024 (2015).
Chaput, J. P., McNeil, J., Despres, J. P., Bouchard, C. & Tremblay, A. Short sleep duration as a risk factor for the development of the metabolic syndrome in adults. Preventive medicine 57, 872–877, doi: 10.1016/j.ypmed.2013.09.022 (2013).
Choi, J. K. et al. Association between short sleep duration and high incidence of metabolic syndrome in midlife women. The Tohoku journal of experimental medicine 225, 187–193 (2011).
Arora, T. et al. Self-reported long total sleep duration is associated with metabolic syndrome: the Guangzhou Biobank Cohort Study. Diabetes care 34, 2317–2319, doi: 10.2337/dc11-0647 (2011).
Santos, A. C., Ebrahim, S. & Barros, H. Alcohol intake, smoking, sleeping hours, physical activity and the metabolic syndrome. Preventive medicine 44, 328–334, doi: 10.1016/j.ypmed.2006.11.016 (2007).
Matricciani, L., Olds, T. & Petkov, J. In search of lost sleep: secular trends in the sleep time of school-aged children and adolescents. Sleep medicine reviews 16, 203–211, doi: 10.1016/j.smrv.2011.03.005 (2012).
Bin, Y. S., Marshall, N. S. & Glozier, N. Secular trends in adult sleep duration: a systematic review. Sleep medicine reviews 16, 223–230, doi: 10.1016/j.smrv.2011.07.003 (2012).
Ferrie, J. E. et al. A prospective study of change in sleep duration: associations with mortality in the Whitehall II cohort. Sleep 30, 1659–1666 (2007).
Kanagasabai, T., Riddell, M. C., Macpherson, A. K. & Ardern, C. I. Changes in polysomnography-based sleep duration and sleep efficiency and the risk of developing hypertension,diabetes,dyslipidemia and obesity:a follow-up study. Sleep 39, abstract supplement (2016).
Ferrie, J. E. et al. Change in Sleep Duration and Type 2 Diabetes: The Whitehall II Study. Diabetes care 38, 1467–1472, doi: 10.2337/dc15-0186 (2015).
Watson, N. F. et al. Recommended Amount of Sleep for a Healthy Adult: A Joint Consensus Statement of the American Academy of Sleep Medicine and Sleep Research Society. Sleep 38, 843–844, doi: 10.5665/sleep.4716 (2015).
Hall, M. H. et al. Self-reported sleep duration is associated with the metabolic syndrome in midlife adults. Sleep 31, 635–643 (2008).
Li, X. et al. U-shaped relationships between sleep duration and metabolic syndrome and metabolic syndrome components in males: a prospective cohort study. Sleep medicine 16, 949–954, doi: 10.1016/j.sleep.2015.03.024 (2015).
Jackson, C. L., Redline, S., Kawachi, I. & Hu, F. B. Association between sleep duration and diabetes in black and white adults. Diabetes care 36, 3557–3565, doi: 10.2337/dc13-0777 (2013).
Yaggi, H. K., Araujo, A. B. & McKinlay, J. B. Sleep duration as a risk factor for the development of type 2 diabetes. Diabetes care 29, 657–661 (2006).
Patel, S. R. et al. A prospective study of sleep duration and mortality risk in women. Sleep 27, 440–444 (2004).
Jiang, X. et al. Metabolic syndrome is associated with and predicted by resting heart rate: a cross-sectional and longitudinal study. Heart 101, 44–49, doi: 10.1136/heartjnl-2014-305685 (2015).
O'Hartaigh, B. et al. Influence of heart rate at rest for predicting the metabolic syndrome in older Chinese adults. Acta diabetologica 50, 325–331, doi: 10.1007/s00592-012-0396-z (2013).
Rogowski, O. et al. Elevated resting heart rate is associated with the metabolic syndrome. Cardiovascular diabetology 8, 55, doi: 10.1186/1475-2840-8-55 (2009).
Oda, E. & Aizawa, Y. Resting heart rate predicts metabolic syndrome in apparently healthy non-obese Japanese men. Acta diabetologica 51, 85–90, doi: 10.1007/s00592-013-0503-9 (2014).
Hall, M. H. et al. Sleep is associated with the metabolic syndrome in a multi-ethnic cohort of midlife women: the SWAN Sleep Study. Sleep 35, 783–790, doi: 10.5665/sleep.1874 (2012).
Kario, K. Obstructive sleep apnea syndrome and hypertension: mechanism of the linkage and 24-h blood pressure control. Hypertension research: official journal of the Japanese Society of Hypertension 32, 537–541, doi: 10.1038/hr.2009.73 (2009).
Prinz, P. N. Age impairments in sleep, metabolic and immune functions. Experimental gerontology 39, 1739–1743, doi: 10.1016/j.exger.2004.06.023 (2004).
Spiegel, K., Leproult, R. & Van Cauter, E. Impact of sleep debt on metabolic and endocrine function. Lancet 354, 1435–1439, doi: 10.1016/S0140-6736(99)01376-8 (1999).
Gangwisch, J. E. et al. Short sleep duration as a risk factor for hypertension: analyses of the first National Health and Nutrition Examination Survey. Hypertension 47, 833–839, doi: 10.1161/01.HYP.0000217362.34748.e0 (2006).
Lockley, S. W., Skene, D. J. & Arendt, J. Comparison between subjective and actigraphic measurement of sleep and sleep rhythms. Journal of sleep research 8, 175–183 (1999).
Xu, S. et al. The association between obstructive sleep apnea and metabolic syndrome: a systematic review and meta-analysis. BMC pulmonary medicine 15, 105, doi: 10.1186/s12890-015-0102-3 (2015).
Antonijevic, I. A., Murck, H., Frieboes, R., Holsboer, F. & Steiger, A. On the gender differences in sleep-endocrine regulation in young normal humans. Neuroendocrinology 70, 280–287, doi: 54487 (1999).
Wang, A. et al. Measures of adiposity and risk of stroke in China: a result from the Kailuan study. PloS one 8, e61665, doi: 10.1371/journal.pone.0061665 (2013).
Zhang, Q. et al. Ideal cardiovascular health metrics and the risks of ischemic and intracerebral hemorrhagic stroke. Stroke; a journal of cerebral circulation 44, 2451–2456, doi: 10.1161/STROKEAHA.113.678839 (2013).
Grundy, S. M. et al. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation 112, 2735–2752, doi: 10.1161/CIRCULATIONAHA.105.169404 (2005).
Acknowledgements
We thank all study participants, their relatives, the members of the survey teams at the 11 regional hospitals of the Kailuan Medical Group, and the project development and management teams at the Beijing Tiantan hospital and the Kailuan Group.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: X.W. and S.W. Performed the experiments and collected data: X.L. and W.Z. Analyzed the data and wrote the paper: Q.S. and X.L. Reviewed manuscript: Q.S., X.L., W.Z., X.W. and S.W. Edited manuscript: X.L.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Song, Q., Liu, X., Zhou, W. et al. Changes in sleep duration and risk of metabolic syndrome: the Kailuan prospective study. Sci Rep 6, 36861 (2016). https://doi.org/10.1038/srep36861
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep36861
This article is cited by
-
The effect of nutrition and physical activity on sleep quality among adults: a scoping review
Sleep Science and Practice (2023)
-
Association of sleep duration and insomnia with metabolic syndrome and its components in the Women’s Health Initiative
BMC Endocrine Disorders (2022)
-
Association between high-density lipoprotein cholesterol and all-cause mortality in the general population of northern China
Scientific Reports (2019)
-
Night Sleep Duration and Risk of Incident Anemia in a Chinese Population: A Prospective Cohort Study
Scientific Reports (2018)
-
Long Sleep Duration Is an Independent Risk Factor for Incident Atrial Fibrillation in a Chinese Population: A Prospective Cohort Study
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.