Abstract
Allogeneic hematopoietic stem cell transplantation is a curative-intent treatment for patients with high-risk hematologic diseases. However, interstitial pneumonitis (IP) and other toxicities remain major concerns after total body irradiation (TBI). We have proposed using linear accelerators with rice-bag compensators for intensity modulation (IM-TBI), as an alternative to the traditional cobalt-60 teletherapy with lung-shielding technique (Co-TBI). Patients who received a TBI-based myeloablative conditioning regimen between 1995 and 2014 were recruited consecutively. Before March 2007, TBI was delivered using Co-TBI (n = 181); afterward, TBI was administered using IM-TBI (n = 126). Forty-four patients developed IP; of these cases, 19 were idiopathic. The IP-related mortality rate was 50% in the total IP cohort and 63% in the idiopathic subgroup. The 1-year cumulative incidences of IP and idiopathic IP were 16.5% and 7.4%, respectively; both rates were significantly higher in the Co-TBI group than in the IM-TBI group. Multivariate analysis revealed that Co-TBI was an independent prognostic factor for both total and idiopathic IP. In the acute myeloid leukemia subgroup, patients with different TBI techniques had similar outcomes for both overall and relapse-free survival. In conclusion, IM-TBI is an easy and effective TBI technique that could substantially reduce the complication rate of IP without compromising treatment efficacy.
Similar content being viewed by others
Introduction
Total body irradiation (TBI) has an established role in the conditioning regimens that are administered before allogeneic hematopoietic stem cell transplantation (HSCT)1,2,3,4. There are several rationales for administering TBI before HSCT: to lower the risk of engraftment failure through TBI’s immunosuppressive effects on the host; to eradicate residual malignant cells through its cytotoxic effects, especially in sanctuary sites like the central nervous system (CNS) and testes, which are difficult for chemotherapy to access; and to eradicate cells with genetic disorders, such as in cases of Fanconi’s anemia or Wiskott-Aldritch syndrome. Previous studies of conditioning regimens for acute leukemia have found that, as compared with chemotherapy alone, the combination of TBI and chemotherapy provided non-inferior outcomes5,6,7,8,9,10,11,12,13 and a lower CNS relapse rate9, even without cranial boost14. However, the addition of TBI would be expected to increase the risk of interstitial pneumonitis (IP), which has been the major dose-limiting toxicity. The incidence of IP has variously been documented as 10 to 85%9,15,16,17 and, strikingly, approximately half of these IP cases were fatal16,17. Many risk factors for IP were previously identified, including single-fraction TBI18, TBI with a higher total dose18, TBI with a higher dose rate15,19, and acute graft-versus-host disease (GvHD)15,16,17,18,20,21,22,23. To date, there have been relatively few studies of attempts to lower the IP rate by improving radiation techniques.
During the era of cobalt-60 teletherapy (Co-TBI) at our institute (1995 to March 2007), TBI was delivered anteroposteriorly using parallel opposed fields with lung blocks to lower the lung dose24. Since 2007, linear accelerators have become predominant, and we have developed a bilateral TBI technique that uses rice-bag compensators as intensity modulators (IM-TBI). Both techniques were delivered with a hyperfractionation schedule and a fixed dose of 12 Gray (Gy). The aim of this study is to evaluate the efficacy and complication rate of the newly developed TBI method.
Results
Patient characteristics
Among the 307 patients enrolled in this study, 181 patients received Co-TBI and 126 patients received IM-TBI. Data on forced vital capacity (FVC) and forced expiratory volume in 1 s (FEV1) were available for all patients. Information on total lung capacity (TLC) and carbon monoxide diffusing capacity of the lungs (DLCO) were available for 228 and 195 patients, respectively. The Co-TBI and IM-TBI groups were comparable in terms of age, sex, and the proportion of cases with complete remission (CR) at the time of HSCT (P = 0.375, 0.907, and 0.811, respectively). As compared with the Co-TBI group, more of the patients in the IM-TBI group had acute leukemia (86% vs. 75%, P = 0.022), initial CNS involvement (30% vs. 5%, P < 0.001), and GvHD of either the acute or chronic type (66% vs. 35%, P < 0.001 and 39% vs. 24%, P = 0.004, respectively). Regarding the pulmonary function test results, only TLC < 85% of predicted was more frequent in the IM-TBI group than in the Co-TBI group (19% vs. 8%, P = 0.012) (Table 1).
Characteristics of and risk factors for interstitial pneumonitis
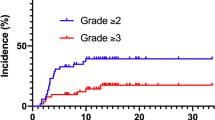
During the follow-up period (median duration, 138.8 months), 44 patients developed IP, 19 cases of which were idiopathic (Tables 1 and 2). The median interval from the date of HSCT to the development of IP was 2.5 months (range, 0.2 to 34.4 months). Eighty percent of the patients with IP were diagnosed within 6 months after HSCT. The overall 1-year cumulative incidences of IP and idiopathic IP were 16.5% and 7.4%, respectively.
Regarding treatments, 41% (18/44) of patients in the total IP group and 53% (10/19) of those in the idiopathic IP group received corticosteroid. Concomitant infection was the main obstacle for the patients who were unable to receive corticosteroid. The IP-related respiratory failure rates were comparable between patients in the total IP cohort and idiopathic IP subgroup (55% [24/44] vs. 53% [10/19], respectively, P > 0.999). Among the survivors with follow-up lung function tests, 57% (4/7) had moderate to severe restrictive ventilator defect. The total IP-related mortality rate was 50% (22/44), which was similar to the idiopathic IP-related mortality rate (63% [12/19], P = 0.414).
The risk factors for both total IP and idiopathic IP included non-CR at HSCT (19% vs. 12%, P = 0.020 and 9% vs. 5%, P = 0.050, respectively) and abnormal FVC before HSCT (24% vs. 12%, P = 0.010 and 13% vs. 5%, P = 0.012, respectively) (Tables 3 and 4). Although Co-TBI was a significant predictive factor for total IP (19% vs. 8%, P = 0.012, Table 3 and Fig. 1A), it only had borderline significance as a predictive factor for idiopathic IP (8% vs. 3%, P = 0.071, Table 4 and Fig. 1B).
In a multivariate Cox proportional hazards regression analysis for total IP, non-CR at HSCT, Co-TBI, and pre-HSCT abnormal FVC were independent predictors of higher incidence (Table 3); however, only Co-TBI was an independent prognostic factor for a higher incidence of idiopathic IP (Table 4).
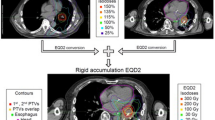
Comparisons of the lung doses for Co-TBI and IM-TBI are provided in Fig. 2A,B. The dose volume histogram showed that the lung dose was comparable between these two groups, but the lung dose distribution was much more homogenous for the IM-TBI plan than for the Co-TBI plan (Fig. 2A). The iso-dose curve also showed more hot spots in the entrance and exit regions in the Co-TBI plan (Fig. 2B).
(A) The dose volume histogram showed the median dose was similar between these different techniques, but the lung dose distribution was much more homogenous for the IM-TBI plan. Solid line: Co-TBI; Dashed line: IM-TBI. (B) The iso-dose curve of lung revealed that there were more hot spots in the entrance and exit regions in the Co-TBI plan.
Prognostic impact in acute myeloid leukemia (AML) patients
In order to elucidate the prognostic impacts of different TBI techniques, we analyzed survival in the relative homogeneous group of patients with AML (n = 111). Univariate analyses of overall survival (OS) showed that age greater than 30 years (12.4 vs. 51.5 months, P = 0.037) and non-CR at HSCT (5.6 vs. 92.9 months, P < 0.001) were significant unfavorable prognostic factors. Other clinical parameters, including initial CNS involvement, unfavorable-risk cytogenetics, different TBI techniques, and GvHD (either acute or chronic), did not show any significant associations with survival in the univariate analyses. In a multivariate analysis of OS, only non-CR at HSCT was an unfavorable prognostic factor (relative risk [RR] 4.0, 95% confidence interval [CI] 2.4–6.7, P < 0.001, Table 5).
Regarding univariate analyses of relapse-free survival (RFS), age greater than 30 years (8.2 vs. 28.7, P = 0.034) and non-CR at HSCT (3.7 vs. 59.5 months, P < 0.001) were significant unfavorable prognostic factors. In resemblance with our multivariate analysis of OS, only non-CR at HSCT was an unfavorable prognostic factor in the multivariate analysis of RFS (RR 4.4, 95% CI 2.6–7.3, P < 0.001, Table 5).
Discussion
Heterogeneous definitions of TBI-related IP have been used in the literature. In some studies, the definition of TBI-related IP included all causes of IP, including both infectious and idiopathic cases19, while other studies included idiopathic cases alone16. In addition, several confounding factors, such as underlying pulmonary disease with abnormal lung function, regimens of conditioning radiochemotherapy, GvHD, and concurrent infection, make it challenging to determine an accurate diagnosis for IP17. In this study, we analyzed both total IP and idiopathic IP to minimize the impact of the infectious causes.
The large majority of the patients who developed IP did so within 1 year after HSCT (97% for the IM-TBI group and 90% for the Co-TBI group, P = 0.407). Furthermore, 99% (65/66) of the survivors in the IM-TBI group and 97% of the survivors in the Co-TBI group (56/58, P = 0.599) were observed for more than 1 year, making it possible to detect most of the IP development after HSCT.
The possibility of developing IP is always a major concern for conditioning regimens that include TBI. Several patient-specific risk factors for IP were reported previously, including old age15,20, poor performance status15, and acute leukemia or myelodysplastic syndrome15,16. Each of these risk factors is non-modifiable. Post-HSCT events, such as GvHD, play important roles in IP development15,16,17,18,20,21,22,23, but they are closely associated with post-HSCT disease status and the primary care physicians’ treatment policies. The most important of the remaining modifiable risk factors for IP development are probably related to TBI itself19. The toxicity of TBI mainly depends on the total dose18, dose rates15,19, fractionation18, and application of lung shielding17,18. To the best of our knowledge, the present study included one of the largest patient cohorts. Furthermore, all patients received homogenous myeloablative conditioning regimens that consisted of TBI with a total dose of 12 Gy. In this study, we have provided the first report of a radiation dose-modulation method that was simple, cheap, effective, and easy to apply. The incidences of IP and idiopathic IP were lower in the IM-TBI group than in the Co-TBI group, even though patients in the IM-TBI group tended to have more risk factors for IP, including a higher prevalence of acute leukemia, more frequent abnormal pulmonary function test results before HSCT, and more frequent acute or chronic GvHD. In both univariate and multivariate analyses, IM-TBI was a favorable risk factor for IP development (Tables 3 and 4). Schneider and colleagues also commented that the reduced incidence of IP in patients receiving IM-TBI might be explained by the more homogenous lung dose distribution and the lower number of focal hot spots that were generated by the rice-bag compensators23,25.
The present investigation is one of the first studies to show that non-CR at HSCT is an unfavorable prognostic factor for IP, using both univariate and multivariate analyses. This might be explained by physicians’ attempts to induce more GvHD in patients without CR at HSCT, who were believed to have dismal outcomes. Previous studies concerning the association of TBI and IP were mainly based on mixed and various hematologic malignancies1,12,15,16,17,18,19,20,21,22,24,25,26,27,28. Thomas et al. were the first to describe TBI in transplantation for patients with AML, but their reports were limited to patients in first complete remission2,3. Although discussions about the side effects of TBI may have been included in other clinical trials that have compared TBI with various conditioning regimens, the patients in these trials may not have been representative of the general AML population29. In contrast, we enrolled consecutive patients—including those who were not in CR or who even suffered from relapse—so we could investigate the impact of CR on IP development.
Regarding the validity of our comparison of the two TBI techniques, a major concern is the different periods during which the techniques were performed. To eliminate the potential biases that might arise from these different periods, such as improvements in post-HSCT care, we additionally compared the IP rates of patients who received TBI-free conditioning chemotherapy composed of cyclophosphamide (60 mg · kg−1 · day−1 for 2 days) and busulfan (intravenous 3.2 mg · kg−1 · day−1 or oral 4 mg · kg−1 · day−1 for 4 days) (BuCy)30,31,32 during these two different eras. We divided the patients who were treated with BuCy during 1995–2014 into two groups, using March 1, 2007 as the splitting date. The 1-year cumulative incidence of IP in patients who received standard BuCy did not differ significantly between the two periods (before March 2007 vs. after March 2007, 8.6% vs. 12.2%, P = 0.420). These results suggest that the risk of IP was not reduced by changes in factors unrelated to IM-TBI, such as developments in post-HSCT care.
For both of the TBI techniques that were investigated in the present study, the incidences of IP and idiopathic IP were lower than has been recorded in some previous data19. These lower IP rates might be explained by the use of lung shielding, either with lung blocks (in Co-TBI) or with rice-bag compensators (in IM-TBI). Sampath et al. reported a reduction in the incidence of IP from 11 to 2.3% with lung shielding33. This result supports our finding that a reduced incidence of IP could be achieved by using shielding accessories to tailor the dose to the lung.
In the past 10 years, BuCy has increasingly been used as the first choice option. BuCy has been applied instead of a TBI-based conditioning regimen in consideration of the late toxicities of TBI and evidence that BuCy and TBI have comparable treatment outcomes5,6,7,8,9,10,11,12,13. However, TBI offers some irreplaceable benefits. For example, it does not spare sanctuary regions; it provides homogenous high doses, regardless of blood supply; it offers less cross-resistance with other antineoplastic agents; and it makes it possible to tailor the dose distribution by shielding or boosting sites of interest. In our practice, the TBI-containing conditioning regimen was often reserved for refractory disease, relapsed disease, or patients with CNS involvement (Table 1).
In univariate and multivariate analyses of the patients with AML, the use of different TBI techniques was not associated with any significant differences in OS or RFS. These findings indicate that the modified TBI technique, IM-TBI, was able to effectively reduce the risk of IP without compromising the treatment outcomes of patients with AML. However, because the study cohort included limited numbers of patients with acute lymphoblastic leukemia and lymphoma, survival analyses were not carried out in these two patient subgroups.
One of the limitations to this study was that we did not have complete performance status data. Weiner et al. stated that a pre-transplantation Karnofsky performance status score of less than 100% was a risk factor for IP in patients who received HSCT (RR 2.1, P < 0.0001)15. Unfortunately, as a consequence of the retrospective nature of this study, we were unable to analyze the associations between Karnofsky performance status score and IP development.
In conclusion, the rice-bag compensator-containing TBI technique (IM-TBI) may be capable of providing a substantially lower IP rate than the conventional lung-shielding TBI technique (Co-TBI). Additionally, in the subgroup of patients with AML, the IM-TBI technique may be able to provide OS and RFS rates that are similar to those associated with Co-TBI. Further efforts should be invested in developing new and modified techniques of tailoring dose distributions to sites of interest, with the ultimate goal of reducing the rates of late toxicities.
Methods and Materials
Patients
This study recruited a total of 307 consecutive patients who received HSCT with a TBI-based myeloablative conditioning regimen from 1995 to 2014 at the National Taiwan University Hospital, including both children and adults. All patients received a pre-HSCT baseline pulmonary function test, routine chest radiograph, and high resolution computed tomography (CT) of the lungs. The patients who received the two different techniques of TBI (IM-TBI and Co-TBI) were compared in terms of the following characteristics: age, definite diagnosis, pre-HSCT disease status, conditioning regimens, and pre-HSCT pulmonary function test results, including FEV1, FVC, TLC, and DLCO. This study was approved by the Research Ethics Committee of the National Taiwan University Hospital and was performed in accordance with the Declaration of Helsinki, including all relevant details. Every patient provided written informed consent.
Conditioning radiochemotherapy and post-HSCT care
The majority of the patients (243/307, 79.1%) received conditioning radiochemotherapy that consisted of cyclophosphamide and TBI. Other conditioning regimens were composed of TBI and melphalan, fludarabine, etoposide, and/or cytarabine. The medications for GvHD prophylaxis included anti-thymocyte globulin, cyclosporine, methotrexate, and/or mycophenolate mofetil34. Trimethoprim/sulfamethoxazole was used for pneumocystis jirovecii pneumonia prophylaxis. After 2007, regular blood sampling tests for serum cytomegalovirus viral load were initiated after conditioning chemotherapy, and lasted for at least 6 months after HSCT. Pre-emptive anti-cytomegalovirus therapy was delivered if the serum cytomegalovirus viral load became elevated.
Total body irradiation
Two techniques of TBI were used during different time periods in this study. After March 2007, the radiotherapy technique for TBI was switched from cobalt-60 teletherapy machines (Co-TBI) to linear accelerators with rice-bag compensators for intensity modulation (IM-TBI). Both techniques included two parallel opposed diagonal fields (40 × 40 cm), which were accomplished through rotation of the fully opened collimator by 45 degrees, to cover the entire body.
In the Co-TBI group, patients were treated with a cobalt-60 1.25 MeV teletherapy machine at a source-surface distance equaling 300 cm. Patients received 1.5 Gy per fraction for 3 to 4 days, to a total dose of 12 Gy, and were irradiated anteroposteriorly with a dose rate of 4.4–5.8 cGy/min. Adults were treated in a standing upright position, while children were treated in a reclining position24. To lower the exposure of the lungs, 1-cm Cerrobend blocks were placed on top of an acrylic box tray as a beam spoiler. The region of the chest wall under the shielding lung blocks would be boosted with electron beams of appropriate energy ranging from 6 to 9 MeV. The final lung dose would be reduced to 70 to 80% of the prescribed dose.
In the IM-TBI group, TBIs were delivered with linear accelerators using 10 megavolt photons at source-axis distance equaling 380–470 cm. The total dose was 12 Gy, and was delivered in 8 fractions within 4 consecutive days. The dose rate was 5–10 cGy/min, depending on the patients’ body thickness. Patients lay in a supine position on a 32 cm-wide headboard with both arms positioned on the chest and the legs bent. We used the lateral diameter of the body at the level of the umbilicus as the reference thickness. If the reference thickness was more than 32 cm, additional bolus was applied on the bilateral outer sides of the headboard; on the other hand, polystyrene foam would be placed on the inner side of the headboard if the reference thickness was less than 32 cm. The rice-bag compensators were placed around the head, neck, shoulders, and chest to fill the interspace of these regions, and to decrease the variation of lateral body thickness along the patient axis (Fig. 3). The lung shielding was established using the rice bag and the patients’ arms as lateral compensators (Fig. 3C). A large spoiler screen made of 2-cm thick acrylic was placed within 10 cm of the patients’ surface to bring the surface dose to at least 90% of the prescribed dose. CT-based simulation and treatment planning were performed to ensure dose uniformity within ± 10%, as measured by thermoluminescent detectors that were placed at the bilateral eyes, neck, bilateral waist, umbilicus, lower back, and groin. The lungs were attenuated to a median dose of 10 Gy (range 9.6 to 10.8 Gy).
To compare the homogeneity of IM-TBI and Co-TBI, we simulated a three-dimensional plan for Co-TBI by inputting the Co-60 beam data into the Pinnacle Treatment Planning System, version 9.2.0.60009 (Philips Healthcare, USA). This allowed us to compare the dose volume histogram and iso-dose curve of the lung between the Co-TBI and IM-TBI groups.
Interstitial pneumonitis
All patients with respiratory symptoms and abnormal chest radiograph findings received high resolution CT of the lungs. IP was defined as multilobar infiltrates on routine chest radiographs and CT in the absence of congestive heart failure, renal failure, or iatrogenic fluid overload17,35. The etiology of IP was investigated through a detailed examination of microbiological stains and cultures, as well as polymerase chain reaction tests for cytomegalovirus, pneumocystis jiroveci pneumonia, tuberculosis36,37, and other diseases. Around 25% of the patients received bronchoalveolar lavage or open lung biopsy38. If there was no clear pathogenic micro-organism, the IP was classified as idiopathic IP16.
Cytogenetics
Bone marrow cells were harvested directly or after 1 to 3 days of unstimulated culture, as described previously39. Metaphase chromosomes were banded using the trypsin-Giemsa technique and karyotyped according to the International System for Human Cytogenetic Nomenclature. Cytogenetic findings were risk-stratified according to the modified Medical Research Council classification40.
Statistical analysis
We compared the characteristics of the Co-TBI and IM-TBI groups using chi-square tests. OS was measured from the date of donor stem cell infusion (described as the date of HSCT elsewhere in this article) to the date of last follow-up or death from any cause, whereas relapse was defined as the reappearance of at least 5% leukemic blasts in bone marrow aspiration smears or new extramedullary leukemia in patients with a previously documented CR41. RFS was measured from the date of HSCT until relapse or death from any cause, whichever occurred first. OS, RFS, and cumulative incidence of IP were estimated using the Kaplan-Meier method. Multivariate Cox proportional hazards regression analyses were used to investigate independent prognostic factors for OS and RFS. The proportional hazards assumption (i.e., the constant hazards assumption) was examined by using time-dependent covariate Cox regression before conducting the multivariate Cox proportional hazards regression. A P-value < 0.05 was considered statistically significant. All statistical analyses were performed using SPSS software, version 20 (SPSS Inc., Chicago, IL, USA).
Additional Information
How to cite this article: Chiang, Y. et al. Reduced incidence of interstitial pneumonitis after allogeneic hematopoietic stem cell transplantation using a modified technique of total body irradiation. Sci. Rep. 6, 36730; doi: 10.1038/srep36730 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Thomas, E. D. et al. One hundred patients with acute leukemia treated by chemotherapy, total body irradiation, and allogeneic marrow transplantation. Blood 49, 511–533 (1977).
Thomas, E. D. et al. Marrow transplantation for acute nonlymphoblastic leukemia in first remission. N. Engl. J. Med. 301, 597–599 (1979).
Thomas, E. D. et al. Marrow transplantation for acute nonlymphoblastic leukemic in first remission using fractionated or single-dose irradiation. Int. J. Radiat. Oncol. Biol. Phys. 8, 817–821 (1982).
Brochstein, J. A. et al. Allogeneic bone marrow transplantation after hyperfractionated total-body irradiation and cyclophosphamide in children with acute leukemia. N. Engl. J. Med. 317, 1618–1624 (1987).
Uberti, J. P. et al. Comparative analysis of BU and CY versus CY and TBI in full intensity unrelated marrow donor transplantation for AML, CML and myelodysplasia. Bone Marrow Transplant. 46, 34–43 (2011).
Blume, K. G. et al. A prospective randomized comparison of total body irradiation-etoposide versus busulfan-cyclophosphamide as preparatory regimens for bone marrow transplantation in patients with leukemia who were not in first remission: a Southwest Oncology Group study. Blood 81, 2187–2193 (1993).
Devergie, A. et al. Allogeneic bone marrow transplantation for chronic myeloid leukemia in first chronic phase: a randomized trial of busulfan-cytoxan versus cytoxan-total body irradiation as preparative regimen: a report from the French Society of Bone Marrow Graft (SFGM). Blood 85, 2263–2268 (1995).
Hartman, A. R., Williams, S. F. & Dillon, J. J. Survival, disease-free survival and adverse effects of conditioning for allogeneic bone marrow transplantation with busulfan/cyclophosphamide vs total body irradiation: a meta-analysis. Bone Marrow Transplant. 22, 439–443 (1998).
Litzow, M. R. et al. Comparison of outcome following allogeneic bone marrow transplantation with cyclophosphamide-total body irradiation versus busulphan-cyclophosphamide conditioning regimens for acute myelogenous leukaemia in first remission. Br. J. Haematol. 119, 1115–1124 (2002).
Nagler, A. et al. Allogeneic hematopoietic stem-cell transplantation for acute myeloid leukemia in remission: comparison of intravenous busulfan plus cyclophosphamide (Cy) versus total-body irradiation plus Cy as conditioning regimen–a report from the acute leukemia working party of the European group for blood and marrow transplantation. J. Clin. Oncol. 31, 3549–3556 (2013).
Blaise, D. et al. Allogeneic bone marrow transplantation for acute myeloid leukemia in first remission: a randomized trial of a busulfan-Cytoxan versus Cytoxan-total body irradiation as preparative regimen: a report from the Group d’Etudes de la Greffe de Moelle Osseuse. Blood 79, 2578–2582 (1992).
Ringden, O. et al. A randomized trial comparing busulfan with total body irradiation as conditioning in allogeneic marrow transplant recipients with leukemia: a report from the Nordic Bone Marrow Transplantation Group. Blood 83, 2723–2730 (1994).
Kroger, N. et al. Comparison of total body irradiation vs busulfan in combination with cyclophosphamide as conditioning for unrelated stem cell transplantation in CML patients. Bone Marrow Transplant. 27, 349–354 (2001).
Alexander, B. M. et al. Utility of cranial boost in addition to total body irradiation in the treatment of high risk acute lymphoblastic leukemia. Int. J. Radiat. Oncol. Biol. Phys. 63, 1191–1196 (2005).
Weiner, R. S. et al. Interstitial pneumonitis after bone marrow transplantation. Assessment of risk factors. Ann. Intern. Med. 104, 168–175 (1986).
Keane, T. J., Van Dyk, J. & Rider, W. D. Idiopathic interstitial pneumonia following bone marrow transplantation: the relationship with total body irradiation. Int. J. Radiat. Oncol. Biol. Phys. 7, 1365–1370 (1981).
Chen, C. I. et al. Radiation-associated pneumonitis following autologous stem cell transplantation: predictive factors, disease characteristics and treatment outcomes. Bone Marrow Transplant. 27, 177–182 (2001).
Pino y Torres, J. L. et al. Risk factors in interstitial pneumonitis following allogenic bone marrow transplantation. Int. J. Radiat. Oncol. Biol. Phys. 8, 1301–1307 (1982).
Carruthers, S. A. & Wallington, M. M. Total body irradiation and pneumonitis risk: a review of outcomes. Br. J. Cancer 90, 2080–2084 (2004).
Fukuda, T. et al. Risks and outcomes of idiopathic pneumonia syndrome after nonmyeloablative and conventional conditioning regimens for allogeneic hematopoietic stem cell transplantation. Blood 102, 2777–2785 (2003).
Bortin, M. M., Kay, H. E., Gale, R. P. & Rimm, A. A. Factors associated with interstitial pneumonitis after bone-marrow transplantation for acute leukaemia. Lancet 1, 437–439 (1982).
Deeg, H. J. Acute and delayed toxicities of total body irradiation. Seattle Marrow Transplant Team. Int. J. Radiat. Oncol. Biol. Phys. 9, 1933–1939 (1983).
Schneider, R. A., Schultze, J., Jensen, J. M., Hebbinghaus, D. & Galalae, R. M. Long-term outcome after static intensity-modulated total body radiotherapy using compensators stratified by pediatric and adult cohorts. Int. J. Radiat. Oncol. Biol. Phys. 70, 194–202 (2008).
Shank, B. et al. Hyperfractionated total body irradiation for bone marrow transplantation. Results in seventy leukemia patients with allogeneic transplants. Int. J. Radiat. Oncol. Biol. Phys. 9, 1607–1611 (1983).
Schneider, R. A. et al. 20 years of experience in static intensity-modulated total-body irradiation and lung toxicity. Results in 257 consecutive patients. Strahlenther. Onkol. 183, 545–551 (2007).
Girinsky, T. et al. Consequences of two different doses to the lungs during a single dose of total body irradiation: results of a randomized study on 85 patients. Int. J. Radiat. Oncol. Biol. Phys. 30, 821–824 (1994).
Oya, N. et al. Influence of radiation dose rate and lung dose on interstitial pneumonitis after fractionated total body irradiation: acute parotitis may predict interstitial pneumonitis. Int. J. Hematol. 83, 86–91 (2006).
Kelsey, C. R. et al. Severe pulmonary toxicity after myeloablative conditioning using total body irradiation: an assessment of risk factors. Int. J. Radiat. Oncol. Biol. Phys. 81, 812–818 (2011).
Mengis, C., Aebi, S., Tobler, A., Dahler, W. & Fey, M. F. Assessment of differences in patient populations selected for excluded from participation in clinical phase III acute myelogenous leukemia trials. J. Clin. Oncol. 21, 3933–3939 (2003).
Tutschka, P. J., Copelan, E. A. & Klein, J. P. Bone marrow transplantation for leukemia following a new busulfan and cyclophosphamide regimen. Blood 70, 1382–1388 (1987).
Santos, G. W. et al. Marrow transplantation for acute nonlymphocytic leukemia after treatment with busulfan and cyclophosphamide. N. Engl. J. Med. 309, 1347–1353 (1983).
Bertz, H., Potthoff, K., Mertelsmann, R. & Finke, J. Busulfan/cyclophosphamide in volunteer unrelated donor (VUD) BMT: excellent feasibility and low incidence of treatment-related toxicity. Bone Marrow Transplant. 19, 1169–1173 (1997).
Sampath, S., Schultheiss, T. E. & Wong, J. Dose response and factors related to interstitial pneumonitis after bone marrow transplant. Int. J. Radiat. Oncol. Biol. Phys. 63, 876–884 (2005).
Chou, S. C. et al. Prognostic implication of gene mutations on overall survival in the adult acute myeloid leukemia patients receiving or not receiving allogeneic hematopoietic stem cell transplantations. Leuk. Res. 38, 1278–1284 (2014).
Panoskaltsis-Mortari, A. et al. An official American Thoracic Society research statement: noninfectious lung injury after hematopoietic stem cell transplantation: idiopathic pneumonia syndrome. Am. J. Respir. Crit. Care Med. 183, 1262–1279 (2011).
Ku, S. C. et al. Pulmonary tuberculosis in allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant. 27, 1293–1297 (2001).
Chen, C. Y. et al. Mycobacterial infections in adult patients with hematological malignancy. Eur. J. Clin. Microbiol. Infect. Dis. 31, 1059–1066 (2012).
Wang, J. Y. et al. Diffuse pulmonary infiltrates after bone marrow transplantation: the role of open lung biopsy. Ann. Thorac. Surg. 78, 267–272 (2004).
Tien, H. F. et al. Correlation of cytogenetic results with immunophenotype, genotype, clinical features, and ras mutation in acute myeloid leukemia. A study of 235 Chinese patients in Taiwan. Cancer genetics and cytogenetics 84, 60–68 (1995).
Grimwade, D. et al. Refinement of cytogenetic classification in acute myeloid leukemia: determination of prognostic significance of rare recurring chromosomal abnormalities among 5876 younger adult patients treated in the United Kingdom Medical Research Council trials. Blood 116, 354–365 (2010).
Cheson, B. D. et al. Revised recommendations of the International Working Group for Diagnosis, Standardization of Response Criteria, Treatment Outcomes, and Reporting Standards for Therapeutic Trials in Acute Myeloid Leukemia. J. Clin. Oncol. 21, 4642–4649 (2003).
Author information
Authors and Affiliations
Contributions
Contribution: Y.C. and C.-H.T. were responsible for study design and plan, literature collection, data management and interpretation, statistical analysis and manuscript writing; C.-Y.L. was responsible for statistical analysis and interpretation of the statistical findings; S.-H.K., Y.-H.C., M.Y., C.-C.L., S.-Y.H., B.-S.K., C.-T.L., H.-A.H., W.-C.C., J.-H.L., C.-C.L., S.-J.W., S.-C.H., Y.-C.C., K.-H.L., D.-T.L., H.-T.C., M.-Y.L., Y.-L.Y., H.-H.C., S.-C.C., C.-L.C., C.-Y.C., W.T., J.-L.T. and H.-F.T. contributed patient cohort and clinical data; M.-C.L. performed the chromosomal studies; X.-W.L. was responsible for the data management; J.-K.W. prepared the dose volume histogram and iso-dose curve; J.-L.T. and Y.-H.C. planned, designed, coordinated the study over the entire period and wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Chiang, Y., Tsai, CH., Kuo, SH. et al. Reduced incidence of interstitial pneumonitis after allogeneic hematopoietic stem cell transplantation using a modified technique of total body irradiation. Sci Rep 6, 36730 (2016). https://doi.org/10.1038/srep36730
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep36730
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.