Abstract
Several prenatal and perinatal factors have been found to be associated with developmental dyslexia (reading disorder) in alphabetic language. Given the absence of relevant studies of Chinese children, the present study tries to investigate these risk factors. A total of 45,850 students were recruited from grades three to six, from seven cities of Hubei province. Dyslexia in Chinese was diagnosed based on children’s clinical symptoms. The clinical symptoms of children’s reading performance were assessed by Dyslexia Checklist for Chinese Children (DCCC) and Pupil Rating Scale Revised Screening for Learning Disabilities (PRS) which were completed by parent/guardian and header teacher respectively. Chinese language exam was used to screen children with poor reading capacity. Questionnaires about prenatal and perinatal factors were completed by parent or guardian. Among the 34,748 eligible participants, 1,200 (3.45%) were diagnosed with dyslexia in Chinese. More boys suffered from dyslexia than the girls and the gender ratio was 3:1. Family history of neuropsychiatric diseases, maternal infectious diseases, difficult vaginal delivery, preterm birth, and neonatal asphyxia were found to increase the risk of developmental dyslexia in China. Closer longitudinal developmental monitoring and preventive measures should be taken for high risk children.
Similar content being viewed by others
Introduction
Developmental dyslexia is a disorder in children with normal intelligence and sensory abilities who show specific reading difficulties in accurate and/or fluent word recognition, spelling, and decoding abilities across different languages1. Children who suffer from dyslexia have more depressive moods, anxiety symptoms, somatic complaints, and behavioural problems than their peers who are normal readers2,3,4,5. A majority of children with dyslexia have persistent disorders in reading, which therefore impairs education attainment and income in adulthood6.
The prevalence of dyslexia varies across different languages and countries. For alphabetic languages, dyslexia is fairly pervasive with a prevalence ranging from 5% to 17.5%7. It was not until 1980s that the researchers realized the existence of Chinese dyslexia. A cross-culture study conducted in Taiwan, Japan and America in 1982 revealed that the prevalence of dyslexia in Chinese (7.5%) were similar with that in English (6.3%)8, and these bring more attention on Chinese dyslexia. A survey conducted in the academic year 1999/2000 in Hong Kong found that the rate of dyslexia was approximately 9.7% among students in grades one to four9. However, large-scale epidemiological study of dyslexia has been lacking in Mainland China in recent years.
Phonological processing is considered the core deficit of impaired reading in alphabetic and logographic language. A study in Chinese students found that children with impaired English reading have poorer Chinese reading performance than typical readers; functional magnetic resonance imaging results revealed that neural deficits involved for impaired phonological processing in English as the second language are similar with English as the first language10.
Evidences showed that dyslexia is associated with multiple genes and environmental risk factors. Nine risk loci (DYX1–DYX9) and six candidate genes (DYX1C1, DCDC2, KIAA0319, C2Orf3, MRPL19, ROBO1)1 are found to be linked with dyslexia. Besides, with the recent advances in genome-wide association studies11,12, more genes are found that may be involved in developmental dyslexia13,14. Parental education is an important environmental risk factor interacting with genes influence on reading disability15. Other possible environmental factors include the language and pre-literacy environments that parents provide to their children16. Our pervious screening study in a middle-size city found that mother’s education level, the time spend on electronic devices, and the literacy-related activity were associated with developmental dyslexia17,18. The purpose of this study was to explore potential environment risk factors for dyslexia from another aspect.
As is known, developmental dyslexia is a kind of neurodevelopmental disorder. Risk factors such as maternal malnutrition, drugs and alcohol, maternal diseases, and unfavourable events during the perinatal period could disrupt normal brain development and lead to dysfunction. Several factors have been reported to have adverse effects on reading or language ability. Preterm birth was one of the most frequently reported risk factors on children’s reading disabilities. A recent meta-analysis found that school-aged children born preterm had poorer decoding and reading comprehension performance than their full-term peers19. A population-based cohort study indicated that former late preterm may not increase the risk of learning disabilities20. Study on school-aged children from Avon Longitudinal Study of Parents and Children (ALSPAC) found that prenatal nicotine exposure was associated with poor performance in specific reading skill outcomes, especially in decoding single words21. Mascheretti et al.22 reported that maternal smoking during pregnancy interacted with dyslexia candidate gene DYX1C1 and affected dyslexia-related reading, spelling and memory phenotypes. Maternal hypertension diseases were found reduced the verbal ability, not non-verbal ability, in the 10 aged children from Western Australian Pregnancy Cohort (Raine) Study23. van Handel et al.24 reviewed that mild neonatal encephalopathy following perinatal asphyxia increased the risk of reading and spelling difficulties in children with norm intelligence quotient. Children who affected neonatal hyperbilirubinemia had persisting problems with reading, writing and mathematics in a prospective birth cohort25.
The long-term effects of prenatal and perinatal risk factors on children’s neurodevelopment could be alleviated by effective intervention policies and therefore these differ in different countries and cultures. Conclusions draw from populations in alphabetic language environment may not be as applicable to Chinese children because of the different socioeconomic level and language backgrounds. Due to the lack of related studies in China, we conducted this descriptive epidemiological study of primary school students. Our aim was to screen prenatal and perinatal factors and elucidate the distribution of dyslexia in Chinese.
Results
The Prevalence and Distribution of Dyslexic Children
Of all the 34,748 students, 1,200 (3.45%) were diagnosed with dyslexia in Chinese. The prevalence of dyslexia varies in different cities (P = 0.02). The differences between the grades were significant (P < 0.001). There was a downward trend between dyslexia and grades (P for trend < 0.05), with the lowest prevalence (2.86%) among sixth grade students. More boys suffered from dyslexia than girls and the gender ratio was 3:1. The dyslexic children and non-dyslexic children were balanced on age (P = 0.316). See details in Table 1.
Association between Prenatal and Perinatal Factors and Dyslexia
Table 2 presents the association between potential prenatal and perinatal risk factors and dyslexia, adjusted for maternal occupation, maternal education, and family income. The distribution of dyslexic children was different from non-dyslexic children with respect to maternal occupation, maternal education, and family income (P < 0.001). Dyslexic children had significant higher proportion of family history of neuropsychiatric diseases than non-dyslexic children (2.3% vs. 0.8%). We observed that the dyslexic children were significantly more likely than non-dyslexic children to have maternal infectious diseases (9.17% vs. 5.83%), maternal anti-infectious medication (2.18% vs. 1.39%), maternal antipsychotics (0.91% vs. 0.34%), and maternal smoking (0.53% vs. 0.20%) during prenatal period. The distribution of dyslexic children was different from non-dyslexic children on gestational weeks (P < 0.001). With regard to difficult vaginal delivery and neonatal asphyxia, the proportions of dyslexic children were higher than non-dyslexic children (3.12% vs. 1.75%, 3.76% vs. 1.42%).
After adjustment for possible confounding variables, the risk of dyslexia remained significantly associated with family history of neuropsychiatric diseases (OR = 2.15, 95% CI = 1.30–3.55), maternal infectious diseases (OR = 1.59, 95% CI = 1.25–2.03), preterm birth (OR = 1.30, 95% CI = 1.01–1.66), difficult vaginal delivery (OR = 1.58, 95% CI = 1.03–2.42), and neonatal asphyxia (OR = 2.38, 95% CI = 1.61–3.52).
Discussion
In the present study, we observed that the prevalence of Chinese language developmental dyslexia was 3.45%. More boys suffered from dyslexia than girls. Family history of neuropsychiatric diseases, infectious diseases, difficult vaginal delivery, preterm birth, and neonatal asphyxia increased the risk of developmental dyslexia. As one of the rare large-scale surveys on Chinese dyslexia, our results not only demonstrate the prevalence and relative risk factors of dyslexia in Chinese, but also provide a data source for cross-culture study.
The prevalence obtained in our study is consistent with our previous result of 3.9% in a middle sample size research17. The result of our study is different from those obtained from Hong Kong (9.7%)9 and Taiwan (7.5%)8. The difference in prevalence between our study and the above studies may result from a variety of definition criteria. The diagnostic criteria of dyslexia applied in Hong Kong and Taiwan using the cut-off score of 1 SD below the mean. Our study used the cut-off of 2 SD from the mean. Types of script may play a role in determining the phenotype of dyslexia in Chinese students. As is known, simplified characters used in Mainland China are much easier to learn than traditional characters used in Hong Kong and Taiwan. An alphabetic phonetic system called pinyin used by Mainland Chinese students to read Chinese characters may also affect the prevalence of dyslexia.
There is a significant male predominance of dyslexia in our study (3:1) which is in the range of previously reported results (from 1.5:1 to 3.1:1)26. Compared with clinical data, our results to some extent avoid the bias result from high comorbidities in boys. Structural neuroimaging studies found less grey matter volume in the left inferior parietal cortex of dyslexic boys, while anatomical variants exist in early sensory and motor cortices of dyslexic girls27. Slower neural responses to speech of male adults compared with females helps to explain the higher incidence of reading impairment in males28. The mechanisms of sex differences in the structure and function of the brain and their role in dyslexia still needs more research.
It is known that dyslexia are familial and moderately heritable. Our results showed an association between family history of neuropsychiatric diseases and dyslexia. Shared genetic risk factors that influence the diseases may be the biologic mechanism underlying the link. Nucleotide changes in the ASD candidate gene, AUTS2, were reported to be associated with ADHD, epilepsy, and dyslexia29. However, conclusions about each specific neuropsychiatric disease could not be drawn from the present results and further investigation is needed.
We found an association between maternal infectious diseases and dyslexia. Epidemiological studies have shown that maternal infection could increase the risk of mental illness in offspring30. The mechanism for this probably involves the maternal immune response which may have an effect on the foetus. Maternal immune activation (MIA) resulting from prenatal exposure to infectious pathogens or inflammatory stimuli is increasingly recognised as playing an important etiological role in neuropsychiatric disorders with neurodevelopmental features31. The Interleukin-17a pathway, which is involved in the MIA of the virally infected mother, has been reported to induce behavioural abnormalities in offspring in rodent testing32.
There have been many improvements in perinatal care, resulting in the survival of a large number of infants with poor birth conditions, such as prematurity, birth disease, and abnormal delivery. Infants with poor birth conditions have greater risk of medical disabilities and behavioural and psychological problems in childhood than their peers. Our study showed increasing prevalence of dyslexia in students between grade three and grade six who had preterm births. Conclusions about the long-term effect of late preterm birth (34 to 37 weeks) on neurodevelopment, learning, and behavioural problems are inconsistent20,33. In our study, we did not have precise gestational weeks. Since most preterm births occur at moderate rather than extreme prematurity, it can be deduced that our results reflect the adverse effects of moderate preterm birth on reading ability. A magnetic resonance imaging study found that moderate and late preterm infants exhibit widespread brain white matter microstructural alterations compared with full-term controls34. Reduced organisation or volume of brain white matter in the left superior longitudinal fasciculus, which connects frontal and temporal language regions, may underpin developmental dyslexia35.
Neonatal asphyxia is the leading specific cause of neonatal mortality in low- and low-middle-income countries and is also the main cause of long-term morbidity including mental retardation, cerebral palsy, and other neurodevelopmental disorders36. Hypoxic ischemic brain injury is the main cause of neurodevelopmental sequelae in birth asphyxia survivors. The rodent model showed moderate hypoxia ischemia could reduce cortical activity, dendrite development, and impaired visual cortical plasticity37. Most studies about the long-term outcomes of infants with neonatal asphyxia are concentrated on the first two years; however, our study showed an association between asphyxia at birth and dyslexia at school age. The students we investigated are eligible to study in normal primary school without mental retardation and severe medical disabilities. It could be concluded that infants who survive neonatal asphyxia, even if they have no symptoms of neurological disorders, have a substantially increased risk of dyslexia.
After adjusting for neonatal asphyxia, difficult vaginal delivery is an independent risk factor for dyslexia. Prolonged duration of the second stage of labour could increase the risk of perinatal adverse outcomes38. An early follow-up study got contrary results, abnormal labour were not found associated with neurologic abnormalities (including speech delay and learning disabilities)39. Our data showed a mild but significant association between difficult vaginal delivery and child dyslexia, which suggested subtle neuronal damage. Since the cause, manifestation, and treatment of difficult vaginal delivery are complicated, consequently interpretation of its long-term effect on neurodevelopment should be done with caution.
Our study is an observational study about the prenatal and perinatal risk factors on dyslexia in Chinese, and prevalence and distribution were also described. We screened some risk factors, such as family history of neuropsychiatric diseases, maternal infectious disease, difficult vaginal delivery, preterm birth, and neonatal asphyxia.
There are several potential limitations to this study. Fist, this is a retrospective observational study and recall bias was unavoidable. We did not get objective information of prenatal and perinatal risk factors from medical records. Some detailed information such as specific medication, the severity of maternal disease, and the degrees of prematurity or asphyxia could not be obtained. However, we did receive relatively correct answers about some critical events, for example, whether the mother or infants had or had not suffered from particular diseases, since pregnancy and childbirth are special experiences for a family. Second, the participants in our investigation are from Hubei province, which may limit generalization to other Chinese population. This study provided clues for further study and data source for comparison with similar studies. Third, the present study had restricted power to detect the association between maternal smoking and antipsychotics and dyslexia. One possible cause is that the rates of maternal smoking and antipsychotics among pregnancy women are rare in China. This does not mean that children being exposed to smoking and antipsychotics in utero are risk-free of dyslexia, as it has been shown that maternal smoking and antipsychotics had adverse effect on offspring’s reading ability21,40. Finally, we did not obtain the family history of dyslexia and other language disorders, because our team was the first one studying dyslexia in Chinese in Hubei province. In summary, our study is a screen study and give a profile of prenatal and perinatal risk factors of dyslexia. Further investigation is needed to understand whether the observed associations are causal.
The findings from our study suggest that infants who are born with prenatal and perinatal risk factors, even if they are eligible to study in normal primary schools, would have higher risk of reading difficulty than their peers. It is necessary to undertake closer longitudinal developmental monitoring and preventive measures for high risk children.
Methods
Sources of Data and Ethics Statement
The data in this study were collected from an on-going project of the Tongji Reading Environment and Dyslexia Study (READ). The READ project launched in 2011 and aims to explore environmental and genetic factors affecting dyslexia. The READ was approved by the Ethical Committee of the Medical Association of Tongji Medical College, Huazhong University of Science and Technology. The methods of the present study were carried out in accordance with the approved guidelines. Written informed consent was obtained from the parents or guardians of the participant children after the nature of the study were explained.
Participants
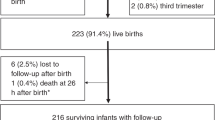
The present study was conducted on primary school students in seven cities of the Hubei province, including Qianjiang, Tianmen, Shayang, Suizhou, Jingshan, Yidu, and Yiling. The children enrolled in our investigation had passed the entrance examination and were eligible to study in normal primary school. Children with abnormal intelligence would recorded in school and were excluded from our study. In total, 45,850 students from grade three to grade six in selected schools participated in the investigation.
Measuring tools
Questionnaires about prenatal and perinatal factors were filled out by parents or guardians. Instruction for filling and the contact information of our team were presented on the questionnaires, in case the fillers have any problems. The questionnaires also contain the variables of family socioeconomic status and home literacy environment. The Dyslexia Checklist for Chinese Children (DCCC) and Pupil Rating Scale Revised Screening for Learning Disabilities (PRS) were used to assess children’s daily reading behaviours and were completed by parent/guardian and header teacher respectively. Score of Chinese language exam in school was used to screen children with poor reading capacity.
The DCCC is a specialized rating scale for dyslexia in Chinese. The scale contains 58 items which were drawn from the clinical symptoms described in ICD-10, DSM-IV, and relative references. The responses are in a 5-point Likert-scale format ranging from ‘never do’ to ‘always do’ (1 = never do, 5 = always do). Higher score means more serious reading difficulty. The reliability coefficient is 0.724 (Cronbach’s a)17. The PRS is a convenient tool widely used to undertake a diagnose of learning disability in China, containing a total of 24 items representing 5 functional regions (i.e., listening comprehension, time and spatial judgments, social behaviour, motion ability, memory and language ability). The reliability coefficients (Cronbach’s a) were higher than 0. 90 for four functional regions and 0.84 for one region41. The score of each question ranges from 1 to 5. Higher score means better learning ability.
For the purpose of this study, dyslexia in Chinese was diagnosed using the following criteria: (a) the score of DCCC was 2 standard deviations higher than the mean score of all the students in same grade; (b) the score of PRS was lower than 65 points; (c) the Chinese language exam was below the tenth percentile of all children in the same grade; and (d) children who had suffered from intellectual disability, brain injury, visual and auditory disorders, epilepsy, and other neurological disorders were excluded.
Data Collection
The number of questionnaires obtained from parents or guardians was 37,309 and 1,736 were excluded from analysis because of poor quality (missing rate more than 50%). In total, there were 35,573 valid parent/guardian questionnaires retained for further analysis. Among all the valid data, 5063 participants were from Qianjiang, which were reported in our pervious article17,18.
After excluding the data of children who had suffered from brain injury, visual and auditory disorders, epilepsy, and other neurological disorders, 34,748 were included in the final analysis.
Statistical Analysis
Continuous variables were described as mean ± standard deviation (SD) and tested with t-test; Categorical variables were described as proportion (%) and tested with Chi-square test or Fisher’s exact probability. Logistic regression analysis was performed to calculate the odds ratio (OR) and 95% confidence intervals (95% CI) of the association between dyslexia in Chinese and prenatal and perinatal risk factors. All P values were two-tailed with a significant level of 0.05. Statistical analyses were carried out using SPSS 13.0 software.
Additional Information
How to cite this article: Liu, L. et al. Descriptive epidemiology of prenatal and perinatal risk factors in a Chinese population with reading disorder. Sci. Rep. 6, 36697; doi: 10.1038/srep36697 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Peterson, R. L. & Pennington, B. F. Developmental dyslexia. Lancet 379, 1997–2007 (2012).
Casey, R., Levy, S. E., Brown, K. & Brooks-Gunn, J. Impaired emotional health in children with mild reading disability. J. Dev. Behav. Pediatr. 13, 256–260 (1992).
Heiervang, E., Stevenson, J., Lund, A. & Hugdahl, K. Behaviour problems in children with dyslexia. Nord. J. Psychiatry 55, 251–256 (2001).
Maughan, B., Rowe, R., Loeber, R. & Stouthamer-Loeber, M. Reading problems and depressed mood. J. Abnorm. Child Psychol. 31, 219–229 (2003).
Willcutt, E. G. & Pennington, B. F. Psychiatric comorbidity in children and adolescents with reading disability. J. Child Psychol. Psychiatry 41, 1039–1048 (2000).
McLaughlin, M. J., Speirs, K. E. & Shenassa, E. D. Reading disability and adult attained education and income: evidence from a 30-year longitudinal study of a population-based sample. J. Learn. Disabil. 47, 374–386 (2014).
Demonet, J. F., Taylor, M. J. & Chaix, Y. Developmental dyslexia. Lancet 363, 1451–1460 (2004).
Stevenson, H. W. et al. Reading disabilities: the case of Chinese, Japanese, and English. Child Dev. 53, 1164–1181 (1982).
Chan, D. W., Ho, C. S. h., Tsang, S. m., Lee, S. h. & Chung, K. K. H. Prevalence, gender ratio and gender differences in reading-related cognitive abilities among Chinese children with dyslexia in Hong Kong. Educ. Stud. 33, 249–265 (2007).
You, H. et al. Neural deficits in second language reading: fMRI evidence from Chinese children with English reading impairment. NeuroImage 57, 760–770 (2011).
Gong, J. et al. A functional polymorphism in lnc-LAMC2-1:1 confers risk of colorectal cancer by affecting miRNA binding. Carcinogenesis 37, 443–451 (2016).
The Wellcome Trust Case Control Consortium. Genome-wide association study of 14,000 cases of seven common diseases and 3,000 shared controls. Nature 447, 661–678 (2007).
Roeske, D. et al. First genome-wide association scan on neurophysiological endophenotypes points to trans-regulation effects on SLC2A3 in dyslexic children. Mol. Psychiatry 16, 97–107 (2011).
Shao, S. et al. The Roles of Genes in the Neuronal Migration and Neurite Outgrowth Network in Developmental Dyslexia: Single- and Multiple-Risk Genetic Variants. Mol. Neurobiol. 53, 3967–3975 (2015).
Friend, A., Defries, J. C. & Olson, R. K. Parental education moderates genetic influences on reading disability. Psychol. Sci. 19, 1124–1130 (2008).
Korat, O., Arafat, S. H., Aram, D. & Klein, P. Book Reading Mediation, SES, Home Literacy Environment, and Children’s Literacy: Evidence from Arabic-Speaking Families. First Lang. 33, 132–154 (2013).
Sun, Z. et al. Prevalence and associated risk factors of dyslexic children in a middle-sized city of China: a cross-sectional study. PloS one 8, doi: 10.1371/journal.pone.0056688 (2013).
He, Z. et al. Does long time spending on the electronic devices affect the reading abilities? A cross-sectional study among Chinese school-aged children. Res. Dev. Disabil. 35, 3645–3654 (2014).
Kovachy, V. N., Adams, J. N., Tamaresis, J. S. & Feldman, H. M. Reading abilities in school-aged preterm children: a review and meta-analysis. Dev. Med. Child Neurol. 57, 410–419 (2015).
Harris, M. N. et al. ADHD and learning disabilities in former late preterm infants: a population-based birth cohort. Pediatrics 132, doi: 10.1542/peds.2012-3588 (2013).
Cho, K., Frijters, J. C., Zhang, H., Miller, L. L. & Gruen, J. R. Prenatal exposure to nicotine and impaired reading performance. J. Pediatr. 162, 713–718 (2013).
Mascheretti, S. et al. An assessment of gene-by-environment interactions in developmental dyslexia-related phenotypes. Genes Brain Behav. 12, 47–55 (2013).
Whitehouse, A. J., Robinson, M., Newnham, J. P. & Pennell, C. E. Do hypertensive diseases of pregnancy disrupt neurocognitive development in offspring? Paediatr. Perinat Epidemiol. 26, 101–108 (2012).
van Handel, M., Swaab, H., de Vries, L. S. & Jongmans, M. J. Long-term cognitive and behavioral consequences of neonatal encephalopathy following perinatal asphyxia: a review. Eur. J. Pediatr. 166, 645–654 (2007).
Hokkanen, L., Launes, J. & Michelsson, K. Adult neurobehavioral outcome of hyperbilirubinemia in full term neonates-a 30 year prospective follow-up study. PeerJ. 2, doi: 10.7717/peerj.294 (2014).
Rutter, M. et al. Sex differences in developmental reading disability: new findings from 4 epidemiological studies. Jama 291, 2007–2012 (2004).
Evans, T. M., Flowers, D. L., Napoliello, E. M. & Eden, G. F. Sex-specific gray matter volume differences in females with developmental dyslexia. Brain Struct. Funct. 219, 1041–1054 (2014).
Krizman, J., Skoe, E. & Kraus, N. Sex differences in auditory subcortical function. Clin. Neurophysiol. 123, 590–597 (2012).
Oksenberg, N., Stevison, L., Wall, J. D. & Ahituv, N. Function and regulation of AUTS2, a gene implicated in autism and human evolution. PLoS Genet. 9, doi: 10.1371/journal.pgen.1003221 (2013).
Labouesse, M. A., Langhans, W. & Meyer, U. Long-term pathological consequences of prenatal infection: beyond brain disorders. Am. J. Physiol. Regul. Integr. Comp. Physiol. 309, doi: 10.1152/ajpregu.00087.2015 (2015).
Patrich, E., Piontkewitz, Y., Peretz, A., Weiner, I. & Attali, B. Maternal immune activation produces neonatal excitability defects in offspring hippocampal neurons from pregnant rats treated with poly I:C. Sci. Rep. 6, doi: 10.1038/srep19106 (2016).
Choi, G. B. et al. The maternal interleukin-17a pathway in mice promotes autism-like phenotypes in offspring. Science 351, 933–939 (2016).
Moster, D., Lie, R. T. & Markestad, T. Long-term medical and social consequences of preterm birth. N. Engl. J. Med. 359, 262–273 (2008).
Kelly, C. E. et al. Moderate and late preterm infants exhibit widespread brain white matter microstructure alterations at term-equivalent age relative to term-born controls. Brain Imaging Behav. 10, 41–49 (2016).
Vandermosten, M. et al. A tractography study in dyslexia: neuroanatomic correlates of orthographic, phonological and speech processing. Brain 135, 935–948 (2012).
Wallander, J. L. et al. Brain research to ameliorate impaired neurodevelopment–home-based intervention trial (BRAIN-HIT). BMC Pediatr. 10, doi: 10.1186/1471-2431-10-27 (2010).
Ranasinghe, S. et al. Reduced Cortical Activity Impairs Development and Plasticity after Neonatal Hypoxia Ischemia. J. Neurosci. 35, 11946–11959 (2015).
Allen, V. M., Baskett, T. F., O’Connell, C. M., McKeen, D. & Allen, A. C. Maternal and perinatal outcomes with increasing duration of the second stage of labor. Obstet. Gynecol. 113, 1248–1258 (2009).
Rosen, M. G., Debanne, S. M., Thompson, K. & Dickinson, J. C. Abnormal labor and infant brain damage. Obstet. Gynecol. 80, 961–965 (1992).
El Marroun, H., White, T., Verhulst, F. C. & Tiemeier, H. Maternal use of antidepressant or anxiolytic medication during pregnancy and childhood neurodevelopmental outcomes: a systematic review. Eur. Child Adolesc. Psychiatry 23, 973–992 (2014).
Wang Zhong et al. Reliability and validity of revised pupil rating scale in regional implementation of learning disability screening. Chin. Prev. Med. 11, 682–685 (2010).
Acknowledgements
This research was supported by National Natural Science Foundation of China (81273092, 81673194) to Song Ranran. Project from Health and Family Planning Commission of Hubei Province (WJ2015MB019). The Fundamental Research Funds for the Central Universities (HUST: 2014TS053).
Author information
Authors and Affiliations
Contributions
L.L.F. designed and wrote the manuscript. W.J., S.S.S., L.X., K.R., and Z.X.H. performed the investigation and analysed data. S.R.R. supervised the project and wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Liu, L., Wang, J., Shao, S. et al. Descriptive epidemiology of prenatal and perinatal risk factors in a Chinese population with reading disorder. Sci Rep 6, 36697 (2016). https://doi.org/10.1038/srep36697
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep36697
This article is cited by
-
Association between urinary 2-hydroxyethyl mercapturic acid and dyslexia among school-aged children
Environmental Science and Pollution Research (2023)
-
Prevalence of developmental dyslexia in primary school children: a protocol for systematic review and meta-analysis
World Journal of Pediatrics (2022)
-
Orthographic Processing of Developmental Dyslexic Children in China: Evidence from an Event-Related Potential Study
Current Medical Science (2021)
-
The effect of pre-pregnancy hair dye exposure on infant birth weight: a nested case-control study
BMC Pregnancy and Childbirth (2018)
-
A gradient relationship between low birth weight and IQ: A meta-analysis
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.