Abstract
Estimating the abundance of pelagic fish species is a challenging task, due to their vast and remote habitat. Despite the development of satellite, archival and acoustic tagging techniques that allow the tracking of marine animals in their natural environments, these technologies have so far been underutilized in developing abundance estimations. We developed a new method for estimating the abundance of tropical tuna that employs these technologies and exploits the aggregative behavior of tuna around floating objects (FADs). We provided estimates of abundance indices based on a simulated set of tagged fish and studied the sensitivity of our method to different association dynamics, FAD numbers, population sizes and heterogeneities of the FAD-array. Taking the case study of yellowfin tuna (Thunnus albacares) acoustically-tagged in Hawaii, we implemented our approach on field data and derived for the first time the ratio between the associated and the total population. With more extensive and long-term monitoring of FAD-associated tunas and good estimates of the numbers of fish at FADs, our method could provide fisheries-independent estimates of populations of tropical tuna. The same approach can be applied to obtain population assessments for any marine and terrestrial species that display associative behavior and from which behavioral data have been acquired using acoustic, archival or satellite tags.
Similar content being viewed by others
Introduction
Estimating the abundance of animal populations is central to modern ecology and conservation, both for terrestrial and marine species. This field continues to grow, not only because of the introduction of new statistical approaches and tools, but also due to the parallel technological advances that underpin new ways to conduct animal censuses through remote detection and telemetry1,2,3,4,5,6,7. Many survey methods used in terrestrial population ecology are based on the so-called distance-sampling approaches, where individuals are counted (or their signs, like animal footprints, droppings or sounds) over random points, quadrants or line transects1,8,9,10,11. From these measurements, absolute or relative abundance indices are derived by integrating the measured density of organisms over a certain area. Analogous methods have been employed in marine population ecology. Line transects have been widely used for small pelagic fish species using active acoustic techniques12,13,14. Equivalently, line transects based on visual inspections are commonly employed in abundance and diversity assessments of benthic species15,16. Additionally, aerial surveys allowed estimating the abundance for those species that are visible from the sea surface, like the Atlantic bluefin tuna17,18,19,20, whales21 and dolphins22. However, for the majority of large pelagic fish species, which are sparsely and patchily distributed in very large three-dimensional habitats, these types of surveys are difficult to conduct. An alternative approach for abundance estimates is the use of mark-recapture experiments. In this approach, which is widely employed in both terrestrial and marine ecology, animals collected in a series of samples are tagged and then released back into the population, where the marked animals are assumed to mix uniformly with the unmarked population. The total population is then estimated according to the ratio of marked to unmarked individuals that are recaptured9. However, conventional tagging data alone are rarely used to estimate the abundance of large pelagic fish and are generally employed in integrated assessment models along with other fisheries-dependent datasets (i.e. catch data) to obtain abundance estimates (see e.g. ref. 23). Besides, conventional tagging data are mainly exploited to estimate mortality and movement rates24,25,26. It is noteworthy that these conventional tagging projects also provide key information on the biology of large pelagic fish, such as growth rates and migration patterns27,28,29,30,31. However, although they allowed unprecedented sampling of subpopulations of animals, these approaches suffer possible bias when applied to pelagic fish species. Among other sources of error, these methods are affected by the disparate ways in which the fish are recaptured and require dedicated approaches to account for the erroneous reported recapture locations32. Additionally, there also may be problems with the way the marked individuals are distributed within the population that is being estimated. Finally, specific to marine ecology are those methods based on fisheries data, relying on the concept of catch-per-unit-of-effort (CPUE) indices33,34. These approaches are based on the idea that knowing how much effort is put into catching and removing fish from the population can provide a relative index of abundance, with the assumption that the same amount of effort will always remove the same proportion of the population that is present. However, rapid technological shifts in fisheries (and concomitant changes in harvest efficiency) make it difficult to analyze the time series of historical data and require dedicated standardization methods to account for this variability34. These CPUE indices are used in integrated assessment models in combination with conventional tagging data23.
The recent introduction of animal remote tracking technologies through satellite, archival and acoustic tagging allows unprecedented opportunities for gaining knowledge of the spatial and behavioral ecology of different species in their natural habitats. Thanks to this technology, marine scientists can now gain more insights about movements and behavior of large pelagic fish (e.g. refs 35, 36, 37, 38). However, despite these technological developments, few methods based on the knowledge of animal behavior have been proposed for estimating their abundance3,39,40. Here we propose a way to integrate telemetered behavioral data (specifically, the association of tropical tuna with floating objects, see below) into a new method of obtaining indices of population abundance. We argue that it is possible to use components of this associative behavior (namely, the residence and absence times at different aggregation sites), to estimate the size of local populations from which the groups of associated animals are drawn. This method is independent of understanding the causative factors that underpin the associative behavior. Specifically, we considered the issue of estimating tropical tuna abundance, taking advantage of their associative behavior with floating objects. Considering their ecological and economic importance and that currently no method exists for obtaining direct, fisheries-independent estimates of their populations, the development of new approaches for evaluating the abundance of tropical tuna is crucial. Different species of tropical tunas, such as skipjack (Katsuwonus pelamis), yellowfin (Thunnus albacares) and bigeye (Thunnus obesus) tuna are known to associate with natural or man-made floating objects, usually called Fish Aggregating Devices (FADs) and fishers have been exploiting this associative behavior for years41. In recent decades, this natural phenomenon has been exploited by purse seine tuna fisheries, which deploy a large number of drifting FADs to increase their chances to locate and catch tropical tuna42. In the following, we demonstrate, through modeling and data analysis, that knowledge of tropical tuna behavior around FADs and quantification of individual residence times around floating objects can provide a new path for direct estimates of populations of tropical tuna.
Methods
Model definition
We considered a system of N fish individuals in an array of p FADs43,44,45. A fish can be in one of the two following states: it can either be associated with one of the FADs, or be unassociated, i.e., occupy a portion of the sea outside of the zone of influence of any FAD. Considering that the total number of fish N is a conserved quantity (assume no recruitment and mortality of fish and balanced exit/entry fluxes of fish within the area and timescale of interest), the fish population at time t is a constant that can be expressed as:

where Xi(t) is the number of fish individuals associated to FAD i at time t,  is the total number of fish associated with all FADs and Xu(t) is the number of unassociated fish. The time evolution of the number of associated fish is described through a system of p differential equations of the form43:
is the total number of fish associated with all FADs and Xu(t) is the number of unassociated fish. The time evolution of the number of associated fish is described through a system of p differential equations of the form43:

where μi denotes the probability for unassociated fish to associate with FAD i (with the index i = 1, … p running over all FADs) and θi denotes the probability for an associated fish to depart FAD i and become unassociated. Similarly, the time evolution of the number of unassociated fish reads:

Considering equation (2) at equilibrium, the ratio between the number of associated fish at a given FAD i and the unassociated population can be expressed as:

which implies  . Taking into account this relation and equation (1), we can write:
. Taking into account this relation and equation (1), we can write:

which provides the number of associated fish relative to the total fish population in terms of the parameters μi and θi that set the system’s dynamics (equation (2)).
Similarly, considering equation (5) for a specific FAD (denoted as FAD 1 in the following) leads:

which provides the ratio between the population of fish associated with FAD 1 and the total population. Equation (6) implies that it is possible to relate the total population to the overall association dynamics and the population associated at one FAD only:

where X1 is the population associated at FAD 1.
Derivation of abundance indices from continuous residence and absence times
Following the recent literature on FADs, the continuous bouts of time that individuals spend at the FADs or out of them are herein referred to as continuous residence times (CRT) and continuous absence times (CAT), respectively (see e.g. refs 45, 46, 47). By exploiting the methods of survival analysis, the model parameters in equation (2) can be inferred from the survival curves of CRTs and CATs45,46. The association dynamics defined in equation (2), where the probabilities μi and θi are two time-independent constants, implies a memoryless process with an exponential distribution of CRTs and CATs45. The survival curve of CRTs can be written as:

where Ci represent the proportion of CRTs recorded at FAD i and the arguments of the exponentials θi correspond to the probabilities to depart from FAD i. From the above relation, it is possible to infer the probabilities  by fitting the survival curve of CRTs with a multiple exponential model. The coefficients Ci are related to the probability to associate with FAD i relative to the overall probability to associate with one of the p FADs and can be expressed in terms of the model parameters μi as follows:
by fitting the survival curve of CRTs with a multiple exponential model. The coefficients Ci are related to the probability to associate with FAD i relative to the overall probability to associate with one of the p FADs and can be expressed in terms of the model parameters μi as follows:

Similarly, the survival curve of CATs for a time-independent process follows:

where  is the probability to associate with one of the p FADs. Combining equations (9) and (10) allows to infer the probability
is the probability to associate with one of the p FADs. Combining equations (9) and (10) allows to infer the probability  to reach FAD i as:
to reach FAD i as:

where  and
and  are estimated from the fits of the survival curves of CRTs and CATs with equations (8) and (10), respectively.
are estimated from the fits of the survival curves of CRTs and CATs with equations (8) and (10), respectively.
Equations (8) and (10) imply that the average CRTs and CATs can be related to  and
and  as follows:
as follows:

and

where  is the average continuous residence time recorded at FAD i and
is the average continuous residence time recorded at FAD i and  is the average continuous absence time spent off the FADs. Substituting equations (11, 12, 13) into equations (5) and (7) leads, respectively:
is the average continuous absence time spent off the FADs. Substituting equations (11, 12, 13) into equations (5) and (7) leads, respectively:

and

where  is the average association time estimated over all FADs,
is the average association time estimated over all FADs,  is the proportion of CRT recorded at FAD 1 and
is the proportion of CRT recorded at FAD 1 and  is the estimated population at FAD 1. Equation (14) provides the estimated ratio between the associated and total population form knowledge of the average residence and absence times only and the index Φ is thereafter referred to as association index. Similarly, the index Ω in equation (15) is thereafter referred to as the abundance index.
is the estimated population at FAD 1. Equation (14) provides the estimated ratio between the associated and total population form knowledge of the average residence and absence times only and the index Φ is thereafter referred to as association index. Similarly, the index Ω in equation (15) is thereafter referred to as the abundance index.
Stochastic simulations: Algorithm description
The association dynamics described in equations (2, 3) was simulated by considering a system of N fish in an array of p FADs. Each fish individual was assigned to one of the p + 1 following states: either a fish was associated to one of the p FADs, or it was unassociated. At each time step t (with t = 1, . . . , Tend), each of the unassociated fish Xu(t) could move to FAD i (with i = 1, . . . , p) according to the probability μi. Equivalently, each of the associated fish Xi at FAD i could depart from that FAD according to the probability θi. The acceptance/rejection of the trial moves were implemented through comparison of μi and θi with a pseudo-random number ξ sampled from a uniform distribution in the interval (0, 1]. The trial move of departing a FAD i was accepted when ξ ≤ θi. Equivalently, an unassociated individual moved to FAD i when  . In the following, the choice in the model parameters ensured the positive-definiteness of the probabilities and the normalization conditions (
. In the following, the choice in the model parameters ensured the positive-definiteness of the probabilities and the normalization conditions ( and θi ≤ 1). The initial position of all fish was assigned to the unassociated state and the system was let evolving in time following the above procedure, up to the end of the simulation at t = Tend. For each time step we recorded the observables of interest: number fish in each of the p + 1 states and position of the fish individuals.
and θi ≤ 1). The initial position of all fish was assigned to the unassociated state and the system was let evolving in time following the above procedure, up to the end of the simulation at t = Tend. For each time step we recorded the observables of interest: number fish in each of the p + 1 states and position of the fish individuals.
The simulations were run for 1000 replica. For each replica, the system’s properties were studied at equilibrium, when the average number of fish per FAD and outside of the FADs was constant in time. To this purpose, we excluded from the analysis a time lapse Tstart located at the beginning of the simulation. At Tstart a number of fish NT (the so-called tagged fish) were sampled at FAD 1 (the FAD of tagging) among the X1 fish present at this FAD. The choice of following only a subset of individuals mimics electronic tagging experiments, where the number of tagged fish is generally much smaller than the total population present in a FAD array. For each of the NT individuals we calculated the CRTs (CATs), as the continuous bouts of time spent at each FAD (outside of the FADs) without any interruption. For each tagged fish, each CRT was followed by a CAT (by construction) and the algorithm kept track of the series of CRTs and CATs sequentially recorded for each fish during the simulation. To this purpose, each tagged individual i was associated to a vector vi = ( ), where
), where  (
( ) corresponds to the jth CRT (CAT) recorded for fish i and
) corresponds to the jth CRT (CAT) recorded for fish i and  is the last CRT recorded during the simulation for individual i (notice that ni can vary among individuals depending on the lengths of their CRTs/CATs). For each replica, the time-averaged number of associated fish
is the last CRT recorded during the simulation for individual i (notice that ni can vary among individuals depending on the lengths of their CRTs/CATs). For each replica, the time-averaged number of associated fish  at FAD 1 (see equation (15)) was estimated from Tstart up to the end of the simulation Tend. In order to reproduce tagging experiments of different lengths, the average residence times and absence times (
at FAD 1 (see equation (15)) was estimated from Tstart up to the end of the simulation Tend. In order to reproduce tagging experiments of different lengths, the average residence times and absence times ( ,
,  and
and  ), as well as the proportion of CRTs recorded at FAD 1 (
), as well as the proportion of CRTs recorded at FAD 1 ( ) (see equations (14) and (15)) were estimated for variable numbers of CRTs and CATs. To this purpose, we considered a subset of CRTs/CATs
) (see equations (14) and (15)) were estimated for variable numbers of CRTs and CATs. To this purpose, we considered a subset of CRTs/CATs  = (
= ( ) obtained from the individual vectors vi defined above. Variable numbers of CRTs/CATs were obtained by pooling the vectors
) obtained from the individual vectors vi defined above. Variable numbers of CRTs/CATs were obtained by pooling the vectors  for increasing values of k (k being the same for all tagged fish and k < min(ni)). The association and abundance indices were then estimated for each replica following equations (14) and (15) and the average and standard deviation of each index calculated over the replica. The sensitivity of our results with respect to the tagging strategy was analyzed by plotting the association and abundance indices as a function of the total number of CRT considered. Also, the sensitivity of the indices relative to the inclusion of the first residence times recorded at the FAD of tagging (FAD 1) was analyzed, by including or excluding the first CRT recorded at FAD 1 (denoted below as CRT1). In the case where CRT1 was excluded, divergences in equation (15) due to the fact that no CRT were recorded at FAD 1 after the tagging, could lead to an undefined abundance index for some of the replica. This generally occurred when the total number of CRTs considered for each tagged individual was small relative to the probability to reach FAD 1. In order to avoid any bias, we considered values of the total number of CRTs where the abundance index was defined for all replica. For all case studies, the model was run for Tstart = 1.0e4, Tend = 1.0e5 and NT = 10 and a sensitivity analysis was conducted on the other model parameters, see paragraph below.
for increasing values of k (k being the same for all tagged fish and k < min(ni)). The association and abundance indices were then estimated for each replica following equations (14) and (15) and the average and standard deviation of each index calculated over the replica. The sensitivity of our results with respect to the tagging strategy was analyzed by plotting the association and abundance indices as a function of the total number of CRT considered. Also, the sensitivity of the indices relative to the inclusion of the first residence times recorded at the FAD of tagging (FAD 1) was analyzed, by including or excluding the first CRT recorded at FAD 1 (denoted below as CRT1). In the case where CRT1 was excluded, divergences in equation (15) due to the fact that no CRT were recorded at FAD 1 after the tagging, could lead to an undefined abundance index for some of the replica. This generally occurred when the total number of CRTs considered for each tagged individual was small relative to the probability to reach FAD 1. In order to avoid any bias, we considered values of the total number of CRTs where the abundance index was defined for all replica. For all case studies, the model was run for Tstart = 1.0e4, Tend = 1.0e5 and NT = 10 and a sensitivity analysis was conducted on the other model parameters, see paragraph below.
Sensitivity analysis of the abundance indices
First, the model was run for a homogeneous system, where all the FADs had the same arrival and departure probabilities μi = μ and θi = θ. Here, the following case studies were considered: (i) variable association dynamics μ/θ, (ii) population sizes N and (iii) numbers of FADs p. For case study (i), the departure probability θ was fixed to a constant value and only the dependence on the association probabilities μ was studied, since the properties of the homogeneous system at equilibrium only depend on the ratio μ/θ43. Secondly, we studied a heterogeneous system with two different FAD classes, denoted as FAD-class A and B (with model parameters μA, θA and μB, θB respectively). Here, the FAD of tagging (FAD 1) was assigned to FAD-class A and the following case studies were considered: (iv) variable association probabilities μB, (v) departure probabilities θB and (vi) numbers of FADs belonging to FAD-class A relative to class B, keeping constant all the other parameters. The model parameters considered for the homogeneous and heterogeneous case studies are reported in Tables 1 and 2, respectively.
Experimental data analysis
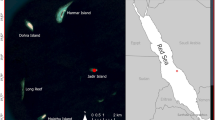
We considered a passive acoustic telemetry dataset collected from tagged yellowfin tuna monitored in an array of 13 instrumented FADs around the island of Oahu, Hawaii (USA), see Fig. 1. Details on the tagging procedure, tag specification and FAD array instrumentation can be found in refs 46 and 47 and in Appendix 1 of the Supplementary Material. We considered 28 yellowfin tuna with fork length larger than 50 cm tagged in 2003, see Supplementary Table S1. The choice of these individuals was based on previous studies that unveiled a homogeneous associative behavior for tagged individuals of this size class48. The choice of the time period was due to the fact that in 2003 the largest number of individuals of this size class was tagged and detected over a large portion of the FAD array. The association and absence times at/off the FADs were estimated using the definition of CRT and CAT, respectively, employed in ref. 46. Supplementary Table S2 resumes the CRTs recorded over the FAD array for the dataset.
Map of the FAD array around the island of Oahu, Hawaii.
Source: http://www.hawaii.edu/HIMB/FADS. Original Map was modified by P. Lopez (IRD) using Adobe Illustrator CS 2.
The survival curves of CRTs and CATs were compared through the Cox proportional hazards regression model49, using the survival library50. Such comparison was performed for two purposes: i) assessing the classes of homogeneous FADs present in the array and ii) verifying that the equilibrium condition assumed in equation (4) was fulfilled. In order to assess the classes of homogeneous FADs, only the FADs with more than 10 CRTs recorded during the study period where analyzed individually. Reversely, for the FADs that were rarely visited, all the CRTs were pooled in a unique survival curve. The equilibrium condition was tested for each class of FADs by comparing survival curves of CRTs recorded over consecutive months that presented sufficient numbers of CRTs. The same test was run for CATs recorded over consecutive months. The hypothesis of time-independence in the association and departure probabilities (see equation (2)) was tested by fitting the survival curves of both CRTs and CATs with three models: exponential, double exponential and power law, using the nls function50. The first two models correspond to a memoryless, time-independent dynamics whereas the latter implies time-dependent probabilities to depart from and/or reach a FAD45,46, see Appendix 2 in the Supplementary material for more details. Goodness of fits was compared through the Akaike information criterion (AIC)51. When the AIC values of two models were close, the inspection of the standard errors of the model parameters and the principle of model parsimony drove the choice of the best fit. The probabilities of departing from FADs were inferred from the model fits of the survival curves of CRTs following equation (8) for each class of homogeneous FADs. The probabilities of reaching the FADs were estimated following equation (11). In this case, the sensitivity to the inclusion/exclusion of the first CRTs recorded after the tagging for each tagged individual (CRT1) was considered by including/excluding them in the proportion of CRTs ( in equation (11)) recorded at the FAD-class of tagging (see Table S1). The association and abundance index were then estimated from equations (14) and (15) and the stochastic simulations described above were run using the inferred model parameters, in order to assess the sensitivity of the indices to the number of CRTs and the inclusion of CRT1.
in equation (11)) recorded at the FAD-class of tagging (see Table S1). The association and abundance index were then estimated from equations (14) and (15) and the stochastic simulations described above were run using the inferred model parameters, in order to assess the sensitivity of the indices to the number of CRTs and the inclusion of CRT1.
Ethic statement
The methods for handling and tagging yellowfin tuna were carried out in accordance with the relevant guidelines on animal experimentation and experimental surgery on fish. All experimental protocols were approved by the University of Hawaii Institutional Animal Care and Use Committee (IACUC).
Results
Results from stochastic simulations
Homogeneous system
Globally, for a homogeneous system with equal probabilities of joining or departing from a FAD (Table 1), our model results indicate that the association index (equation (14)) is robust and shows little sensitivity to the tagging strategy and the model parameters (see Fig. 2A,C,E). On the other hand, the abundance index (equation (15)) was more sensitive to the number of CRTs (Fig. 2B,D,F). The inclusion of the first CRT recorded at the FAD of tagging (CRT1) in equation (15) led to an underestimated population for all case studies. When CRT1 was excluded from equation (15), the abundance index showed higher accuracies but larger variabilities. For all case studies, the asymptotic limit was reached for both indices for high numbers of CRTs. Case study (i) (Fig. 2A,B) demonstrated that both indices converged to the asymptotic values for any value of the association probability μ (Fig. 2A,B), the convergence being independent on the value of μ. Case study (ii) (Fig. 2C,D) demonstrate that both indices were not sensitive to the size of the population considered within the FAD array. Finally, changes in the total number of FADs (case study (iii)) resulted to be equivalent to varying μ for the association index (Fig. 2E). For the abundance index (Fig. 2F), including CRT1 in equation (15) produced higher losses in the accuracy for higher numbers of FADs. For example, the estimated population that showed high accuracies for 1000 CRTs and 10 FADs was lowered to nearly 50% of the true population in the presence of 100 FADs. This loss in accuracy was lower when excluding CRT1, but the variability of the abundance index increased when increasing the number of FADs.
Homogeneous system.
Association (left column) and abundance (right column) indices as a function of the total number of CRT. (A,B) Case study (i), with different probabilities to reach the FADs. (C,D) Case study (ii), with different population sizes. (E,F) Case study (iii) with different numbers of FADs. Empty/filled points correspond to the estimated indices with/without the first CRT recorded for each fish at the FAD of tagging (CRT1). The horizontal lines denote the asymptotic limits.
Heterogeneous system
All case studies converged to the asymptotic limit for large numbers of CRTs, see Fig. 3. Globally, the convergence of the association index showed little dependence on the properties of an heterogeneous array of FADs (Fig. 3A,D). The only exception was found for case study (v), where variable probabilities θB to depart from FAD-class B were considered (Fig. 3C). Here, when CRT1 was included in equation (14), smaller θB led to an underestimated association index. The abundance index showed an opposite trend. Higher dependencies on the model parameters and the tagging strategies were found for case studies (iv) and (vi) (Fig. 3B,F). When increasing the probability μB to reach FAD-class B (case study (iv) and Fig. 3B), the inclusion of CRT1 lead to an underestimated population. If CRT1 was excluded, the abundance index showed higher variabilities over the replica for larger values of μB. Case study (vi) showed a similar trend to case study (iv), where larger proportions of FAD-class B induced lower accuracies and higher variabilities in the abundance index (Fig. 3F).
Heterogeneous system.
Association (left column) and abundance (right column) indices as a function of the total number of CRT. (A,B) Case study (iv), with different probabilities to reach the FAD-class B. (C,D) Case study (ii), with different probabilities to depart from FAD-class B. (E,F) Case study (iii) with different proportions of FADs in FAD-class B relative to FAD-class A. Empty/filled points correspond to the estimated indices with/without the first CRT recorded for each fish at the FAD of tagging (CRT1). The black points denote the homogeneous system with parameters of FAD-class A. The horizontal lines denote the asymptotic limits.
Applications to tropical tuna in an array of FADs
The Cox proportional hazard model run over the survival curves of CRTs revealed two classes of FADs: FAD HH (denoted below as FAD-class 1) and FADs CO and V (FAD-class 2), see Supplementary Table S3. The CRTs recorded at the remaining FADs (FADs II, J, LL, R, T, X, MM, U, S and BO) were pooled and compared to these two classes of FADs. This comparison revealed that these FADs were not statistically different from FAD-class 2 (Supplementary Table S3). The Cox proportional hazard model run over the CRTs of FAD-classes 1 and 2 recorded at subsequent months demonstrated a temporal homogeneity for both FAD classes (Supplementary Table S4). The same result was obtained for CATs recorded at subsequent months, thus confirming that the equilibrium condition was attained (Supplementary Table S4). The comparison of goodness of fits between single exponential, double exponential and power law demonstrated that the survival curves of both CRTs recorded under FAD-class 1 and FAD-class 2 were exponentially distributed. FAD-class 1 was well described by a single exponential model and characterized by a mean CRT of 21.3 days. FAD-class 2 was best fitted by a double exponential model corresponding to two behavioral modes. Values of the AIC of exponential and power-law models were very close for FAD-class 1. Therefore, the exponential model was chosen for model parsimony, see Supplementary Table S5 and Appendix 4). The first behavioral mode represented 33% of the CRTs of FAD-class 2 with a mean duration of 0.07 days (1.7 hours), while the second behavioral mode represented 67% of the CRTs of FAD-class 2 with a mean duration of 3.7 days. Finally, the survival curves of CATs were best fitted by a single exponential model, see Supplementary Table S5, with a mean duration of 2.5 days for absence times. Table 3 resumes the optimal fitting parameters for both the CRTs and the CATs. Given the exponential survival curves of CRTs and CATs related to time-independent probabilities to depart from/reach the FADs and the fulfillment of the equilibrium condition, the model defined in equations (2, 3, 4, 5) was suitable to reproduce the observed association dynamics. In the presence of two FAD classes and a double exponential model for FAD-class 2 (Appendix 3 in the Supplementary Information), the association index can be written as:

with:

and in the limit  :
:

Estimates of the departure probabilities  ,
,  and
and  could directly be obtained from the fits of the survival curves of CRTs (Table 3). Moreover, the arrival probabilities
could directly be obtained from the fits of the survival curves of CRTs (Table 3). Moreover, the arrival probabilities  ,
,  and
and  could be estimated from the fitted parameters in Table 3 through the following equations:
could be estimated from the fitted parameters in Table 3 through the following equations:

where  (
( ) is the number of CRTs recorded at FAD-class 1 (FAD-class 2),
) is the number of CRTs recorded at FAD-class 1 (FAD-class 2),  is the total number of CRTs,
is the total number of CRTs,  is the fraction of CRTs associated to short residence times for FAD-class 2 and
is the fraction of CRTs associated to short residence times for FAD-class 2 and  is the estimated probability to associate with one of the FADs of the array related to the survival curve of CAT, see equation (10). Application of the analytical formula (17) and (18) led to an association index of 0.721 (±0.087) and 0.720 (±0.086), respectively, which implies that the shortest timescales related to
is the estimated probability to associate with one of the FADs of the array related to the survival curve of CAT, see equation (10). Application of the analytical formula (17) and (18) led to an association index of 0.721 (±0.087) and 0.720 (±0.086), respectively, which implies that the shortest timescales related to  and
and  can be neglected and the asymptotic limit in equation (18) holds. Application of the analytical formula (18) with the exclusion of CRT1 in (19) led to an association index of 0.688 (±0.083), which is close to the previous values. Details for the derivation of the association index can be found in Appendix 5 of the Supplementary Material. The stochastic model was run with parameters obtained from the fitted values of Table 3 for 1000 replica, in the limit
can be neglected and the asymptotic limit in equation (18) holds. Application of the analytical formula (18) with the exclusion of CRT1 in (19) led to an association index of 0.688 (±0.083), which is close to the previous values. Details for the derivation of the association index can be found in Appendix 5 of the Supplementary Material. The stochastic model was run with parameters obtained from the fitted values of Table 3 for 1000 replica, in the limit  , with and without taking into account CRT1 in the estimate of the model parameters (see Supplementary Table S6). The estimated association index showed little sensitivity relative to the number of CRTs recorded, see Fig. 4A. Secondly, the model could provide an estimate of the abundance index, in the hypothetical case where we could measure the population at FAD-class 1 (Fig. 4B). Table 4 reports the estimated association index and the estimated abundance index for a simulated population of 10 000 individuals. Relative errors of 1–3% and 5–10% were obtained for the association and abundance index, respectively, when the total number of CRT considered in the model was equal to 100 (i.e., of the same order of magnitude of the field experiment) thus providing indications that this approach can consistently be applied to realistic systems.
, with and without taking into account CRT1 in the estimate of the model parameters (see Supplementary Table S6). The estimated association index showed little sensitivity relative to the number of CRTs recorded, see Fig. 4A. Secondly, the model could provide an estimate of the abundance index, in the hypothetical case where we could measure the population at FAD-class 1 (Fig. 4B). Table 4 reports the estimated association index and the estimated abundance index for a simulated population of 10 000 individuals. Relative errors of 1–3% and 5–10% were obtained for the association and abundance index, respectively, when the total number of CRT considered in the model was equal to 100 (i.e., of the same order of magnitude of the field experiment) thus providing indications that this approach can consistently be applied to realistic systems.
Discussion
We propose a new way of deriving animal abundance (tuna in our case study) that relies on their associative behaviour around aggregating points and on the possibility of monitoring the dynamics of this behaviour through electronic tagging technologies. Our approach is based on knowing the amount of time spent by individuals at or away from aggregation points – in this case, using acoustic telemetry. These two measurable quantities can now be accessible through satellite, acoustic or archival tagging technologies in instances where it is possible to tag a subset of individuals of the species of interest and detect their characteristic associative behaviour. From knowledge of residence and absence times only, our approach provides the so-called association index, which informs on the fraction of the population that can be found at the aggregation points relative to the local population. Interestingly, when the total number of associated individuals at one of the aggregation points is known, our approach provides absolute abundance indices, linking directly the number of associated individuals to the local population present within the sampled region.
The sensitivity analysis conducted on the association index revealed that high accuracies can already be obtained for small number of CRTs (<100), both for homogeneous and heterogeneous FAD arrays. Reversely, the abundance index converged less rapidly and provided underestimated (overestimated) abundances at small number of CRTs when including (excluding) the first CRT recorded at the FAD of tagging (CRT1). Such bias is due to the error in the estimates of the proportion of residence times ( in equation (15)) spent at the FAD of tagging, which is overestimated (underestimated) when including (excluding) the first CRT recorded at the FAD of tagging. Despite the overall population is at equilibrium, the tagging of NT fish is conducted at a single FAD (FAD1) so the tagged sub-population is out of equilibrium (being concentrated only at FAD1). The sensitivity analysis conducted herein on CRT1 allows to evaluate the effect of this out-of-equilibrium state of the tagged fish. Fishing and tagging operations are generally conducted on a subportion of the FAD array (for example, in our dataset of Hawaii the yellowfin tuna were tagged at only two FADs, CO and HH, see Table S1) and all data are generally included in the analyses to maximise the use of the information collected in the field. Ideally, one should wait for the system of tagged individuals to be at equilibrium. In practice, this equilibrium condition is difficult to assess because the number of tagged individuals in a FAD array is, unfortunately, relatively small and each of the tagged individuals spends a different amount of time in the FAD array. Here, we demonstrated that the abundance index may show a strong sensitivity to this tagging strategy and that the effect of including the first CRT should be considered in the estimated index.
in equation (15)) spent at the FAD of tagging, which is overestimated (underestimated) when including (excluding) the first CRT recorded at the FAD of tagging. Despite the overall population is at equilibrium, the tagging of NT fish is conducted at a single FAD (FAD1) so the tagged sub-population is out of equilibrium (being concentrated only at FAD1). The sensitivity analysis conducted herein on CRT1 allows to evaluate the effect of this out-of-equilibrium state of the tagged fish. Fishing and tagging operations are generally conducted on a subportion of the FAD array (for example, in our dataset of Hawaii the yellowfin tuna were tagged at only two FADs, CO and HH, see Table S1) and all data are generally included in the analyses to maximise the use of the information collected in the field. Ideally, one should wait for the system of tagged individuals to be at equilibrium. In practice, this equilibrium condition is difficult to assess because the number of tagged individuals in a FAD array is, unfortunately, relatively small and each of the tagged individuals spends a different amount of time in the FAD array. Here, we demonstrated that the abundance index may show a strong sensitivity to this tagging strategy and that the effect of including the first CRT should be considered in the estimated index.
Globally, for a homogeneous system, our analysis revealed that both indices showed little sensitivity in relation to the association dynamics and the total population. Reversely, the abundance index was highly sensitive to the number of FADs, with lower accuracies for higher numbers of FADs. Similarly to the trend of the index relative to the inclusion of CRT1, such bias can be explained by the increasing error in the estimates of ( ) for larger numbers of FADs. This statement, however, should be considered with attention. The sensitivity analysis conducted here considers populations at equilibrium and with the same association dynamics. The approach does not intend to be predictive on how the association dynamics changes with respect to changes in the population size. It rather relies on the fact that, for any population size, if we can measure the association dynamics from field data, we can obtain accurate abundance estimates. Such equilibrium hypothesis relies on the fact that the behavioral timescales are much shorter than those related to the population dynamics. The same consideration should be taken for the other case studies considered in this paper, for example changes in the numbers of FADs (case study (iii)). The sensitivity analysis did not consider possible changes in the associative dynamics of tuna when the number of FADs is modified. The assumption here is that we can, in any case, measure the association dynamics for a given population size, number of FADs or FAD-array heterogeneities, and that the accuracies of the estimated association and abundance indices may depend on such variables.
) for larger numbers of FADs. This statement, however, should be considered with attention. The sensitivity analysis conducted here considers populations at equilibrium and with the same association dynamics. The approach does not intend to be predictive on how the association dynamics changes with respect to changes in the population size. It rather relies on the fact that, for any population size, if we can measure the association dynamics from field data, we can obtain accurate abundance estimates. Such equilibrium hypothesis relies on the fact that the behavioral timescales are much shorter than those related to the population dynamics. The same consideration should be taken for the other case studies considered in this paper, for example changes in the numbers of FADs (case study (iii)). The sensitivity analysis did not consider possible changes in the associative dynamics of tuna when the number of FADs is modified. The assumption here is that we can, in any case, measure the association dynamics for a given population size, number of FADs or FAD-array heterogeneities, and that the accuracies of the estimated association and abundance indices may depend on such variables.
The heterogeneous model allowed taking into account possible FAD-array heterogeneities, either induced by the characteristics of the FADs themselves, or by social interactions43. Our analysis was independent on the causative factors that induced such heterogeneities and the possible role of social interactions was indirectly taken into account by considering heterogeneous values of μi and θi. For all case studies, the association index confirmed a high robustness also for a heterogeneous system. The only exception was case study (v) (heterogeneous probabilities of departing from the FADs), where, for θB ≪ θA, the inclusion of CRT1 led to an underestimated association index. This happens because the proportion of CRTs associated to FAD-class B is underestimated relative to FAD-class A (where tagging occurs) and thus the association index is closer to the homogeneous case (θB = θA) for small numbers of CRT. Reversely, the abundance index revealed a sensitivity to heterogeneous probabilities of joining the FADs, as well as on the relative number of FADs of each class. The best FAD for tagging fish and measuring the local biomass resulted to be the more attractive one (i.e., the one with higher μi, thus with the best estimates of Ci) and higher accuracies could be obtained when the FAD-class of tagging constitutes the majority of the array.
The application of this approach to field data was key for validating the main assumptions upon which the model is built: (i) time-independent dynamics and (ii) equilibrium condition satisfied. The hypothesis of time-independence in the probabilities of joining or departing from a FAD could be tested through the approaches of survival analysis. The survival curves of both CRTs and CATs could be best fitted by exponential models, which imply time-independent probabilities of joining or departing from a FAD. In some cases, power-law models also performed well. The distinction between multiple exponentials and power laws is not always straightforward, particularly for reduced number of points52. We therefore adopted the principle of parsimony and considered the simplest model that fitted well the experimental curves, thus choosing the exponentials models that best fitted the data. The equilibrium hypothesis found support from the comparison of survival curves of CRTs and CATs obtained at subsequent months, which demonstrate that the associative behavior of tuna did not change in time. Unfortunately, we did not dispose of measurements of the number of associated tuna, nor of its time evolution, to the purpose of assessing that equilibrium was attained at the level of the aggregation. Validating the equilibrium condition on the associated biomass (for example, through the echosounder buoys53) will undoubtedly be a key component of future studies of this type. Also, we could not verify the stationarity of the overall population. The fact that the tagged fish left the FAD array during the tagging experiment indicates that the possibility that yellowfin tuna leaves the FAD array certainly exists. However, verifying that other yellowfin tuna reached the FAD array in the meantime was not possible. Another important outcome of the method application to a realistic dataset was the assessment of the sensitivity of the association and abundance indices for an experimentally-observed association dynamics. To this purpose, the model parameters were derived from the fits of the survival curves of CRTs and CATs. In this respect, our results are promising and open the route to future applications of this method on tropical tuna and species showing the same associative behavior. We could demonstrate that for values of the model parameters inferred from field data, the accuracy of the association index is very high, already for a reduced numbers of residence times. Collecting field data of this kind is challenging: the number of fish that can be tagged is bounded by the high costs of the electronic tags and the issues of acoustic collisions that occur when too many individuals are tagged45,54. Our study demonstrates that such limits do not constitute a major issue for population assessments of tropical tuna. With the theoretical model we have demonstrated the possible bias induced by CRT1. In the analysis of experimental data, this translates in a biased estimate of μi, the probability to reach the FADs where tagging operations have been conducted (equation 11). For this reason, we considered two possible values of μi, one estimated with the inclusion of CRT1 and the other without CRT1. Reversely, all CRTs were included in the survival analysis of CRTs, assuming that there the effect of tagging on the residence times was negligible. This choice was dictated by the fact that we would reduce too much our database to have good survival curves when excluding CRT1. Also, in survival analysis, it is well known that left censoring does not introduce a bias, since the starting point (the beginning of CRT1) is defined by an event (fish tagging) which can be considered randomized relative to the time the tagged fish have already spent at the FAD and thus does not affect the properties of the survival curve.
Our model provides for the first time an estimate of the relative abundance of tropical tuna that can be considered to be within the general vicinity of an array of FADs. The estimated association index indicates high values for the ratio between the number of associated and total individuals around Oahu (Hawaii), of the order of 70%. One possible explanation for this high value could be that the individuals captured and tagged at the FADs are drawn from a subpopulation with a high “associative character”47. This possibility could be addressed by new tagging experiments where individuals would be tagged both at the aggregative points (FADs in the case of tuna) and away from them. Furthermore, based on the outcomes of the sensitivity analysis conducted theoretically (Fig. 2C,D), where the accuracy of the association and abundance indices was demonstrated to be independent on the size of the population, we could also test the robustness of the abundance index for realistic association dynamics. Assuming a population of 10 000 individuals and knowledge of the aggregated population at one FAD, we could provide an order of magnitude of the accuracy of our method. Remarkably, we found relative errors on the total population estimates ranging between of 3–10% and relative standard deviations of the same order of magnitude already for 100 CRTs, which indicate the applicability of this approach for abundance estimates of yellowfin tuna in a realistic experimental setting. Combined with estimates of the actual number of yellowfin tuna at Oahu FADs our method could provide an estimate of the local population around Oahu. In this respect, it is important to stress that the abundance index derived through this approach concerns only the fraction of the population of tropical tuna that can be found in association with FADs, i.e. skipjack tuna and small yellowfin and bigeye tuna.
A great advantage of this model is that it is simple and contains very few parameters: residence times at aggregation sites, residence times off aggregation sites and the population at one of the aggregating points are all that is required. In particular, equation (5) provides estimate of the relative abundance of associated individuals for a generalized aggregative system, independent of the mechanism that leads to the aggregation. Within our approach the effects of social interactions and the spatial heterogeneity induced by the environment can be naturally taken into account. The only assumption here is that the probability of joining or leaving the aggregation points is constant in time and that the population is stationary during the period of observation. For these reasons, this method is applicable to different species and case studies, not only to the one presented here. Each time animals show an associative behaviour, whatever the reason, it is possible to derive the relative abundance of associated animals for that region. This opens the possibility of using natural sites but also artificial sites that are designed specifically to aggregate animals and derive indices of abundance. This simplicity also implies that there is room for improvement, for example considering a spatialized model where the connectivity between FADs depends on their locations in the array.
Within the population-ecology literature, mark-recapture methods are generally employed for species that gather at specific locations, whereas distance-sampling methods are considered more effective for dispersed populations11. Our method can be considered as a combination of these two approaches. On one side, it is based on a subset of tagged individuals and exploits the aggregation of animals at particular points in space, increasing the detectability of those species that are otherwise dispersed over large areas. On the other side, it relies on detecting the presence/absence of these animals at the aggregating points. Its novelty and strength rely on the additional information obtained from the measurement of association and absence times. This information allows integrating the association dynamics within the sampling theory, thus accounting for the probability of observing (or not) the animal at the sampled aggregative points due to their behaviour.
More generally, our approach opens the route towards fisheries-independent abundance estimates of tropical tuna over wider regions, a crucial issue in fisheries management since no method currently exists for obtaining direct estimates of their populations. Using current generation archival tags, with light sensors, it is now possible to reconstruct the most probable tracks of individual fish and determine, for species exhibiting distinct diel vertical patterns depending on their type of association (e.g. bigeye tuna55), their associative history over several months and years. Equivalently, the long lifetime of acoustic tags now allows covering long periods of time. Furthermore, fishers currently use Global Positioning Systems (GPS) buoys with echosounders to obtain information on the presence and abundance of associated tuna (with no information on the species) around drifting FADs56. Access of these data would provide scientists with unprecedented large datasets regarding the amount of tuna at floating objects, which, when combined with tagging data, would allow first direct estimates of tuna populations57. The geographical extent of this population could be estimated by assessing the range of movements of fish caught and released at FADs in the region (as is currently occurring around anchored FADs in Hawaii using satellite telemetry techniques - Holland, unpublished data). Applying our approach would complement the current estimates obtained through more classical fisheries-dependent methods. In the hypothetical case where the number of fish at FADs remained high even if the wider population declined drastically, our method could really be innovative with respect to abundance estimates based on catch data. With our approach, we can estimate the proportion of associated population relative to the total population. Therefore, our abundance and association indices would be complementary to the fisheries-dependent abundance indices, and alert in the presence of “apparent” constant catches but an increasing ratio of associated population. Finally, our approach would allow the estimation of populations of other species that associate with floating objects, which represent the bycatch of tropical tuna purse seine fisheries. Populations of these species are rarely assessed due to lack of methods and data. Tagging these species would allow estimating their relative abundance and better evaluate the impacts of FAD fishing on pelagic ecosystems42.
In the same way that oceanographers have historically deployed different kinds of instrumented probes to study the physical dynamics of the oceans, it is now time for fisheries scientists to adopt the same strategy. By deploying networks of instruments that can observe the biological components of the oceans, both through FAD instrumentation and fish tagging, fisheries biologists can significantly improve their knowledge of the pelagic ecosystem. This new technology could be used to feed appropriate models with fishery-independent data that are vital for improved resource management strategies.
Additional Information
How to cite this article: Capello, M. et al. Population assessment of tropical tuna based on their associative behavior around floating objects. Sci. Rep. 6, 36415; doi: 10.1038/srep36415 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Schwarz, C. J. & Seber, G. A. F. Estimating animal abundance: Review iii. Stat. Sci. 14, 427–456 (1999).
Turner, W. et al. Remote sensing for biodiversity science and conservation. Trends Ecol. Evol. 18, 306–314 (2003).
Morales, J. M. et al. Building the bridge between animal movement and population dynamics. Philos. T. Roy. Soc. B: Biol. Sci. 365, 2289–2301 (2010).
OĆonnell, A. F., Nichols, J. D. & Karanth, K. U. Camera traps in animal ecology (Springer, 280 pp, 2011).
Blumstein, D. T. et al. Acoustic monitoring in terrestrial environments using microphone arrays: applications, technological considerations and prospectus. J. Appl. Ecol. 48, 758–767 (2011).
Marques, T. A. et al. Estimating animal population density using passive acoustics. Biol. Rev. (2012).
Burton, A. C. et al. Wildlife camera trapping: a review and recommendations for linking surveys to ecological processes. J. Appl. Ecol. 52, 675–685 (2015).
Wilson, K. R. & Anderson, D. R. Evaluation of a density estimator based on a trapping web and distance sampling theory. Ecology 66, 1185–1194 (1985).
Buckland, S. T., Goudie, I. B. J. & Borchers, D. L. Wildlife population assessment: past developments and future directions. Biometrics 65, 1–12 (2000).
Wilson, G. J. & Delahay, R. J. A review of methods to estimate the abundance of terrestrial carnivores using field signs and observation. Wildlife Res. 28, 151–164 (2001).
Thomas, L. et al. Distance sampling. In Encyclopedia of Environmetrics, 544–552 ( A. H. El-Shaarawi & W. W. Piegorsch . John Wiley and Sons, Chichester, 2002).
Petitgas, P. Geostatistics for fish stock assessments: a review and an acoustic application. ICES J. Mar. Sci. 50, 285–298 (1993).
Misund, O. A. Underwater acoustics in marine fisheries and fisheries research. Rev. Fish. Biol. Fish. 7, 1–34 (1997).
Barange, M., Hampton, I. & Roel, B. A. Trends in the abundance and distribution of anchovy and sardine on the south african continental shelf in the 1990s, deduced from acoustic surveys. S. Afr. J. Mar. Sci. 21, 367–391 (1999).
Ensign, W. E., Angermeier, P. L. & Dolloff, C. A. Use of line transect methods to estimate abundance of benthic stream fishes. Can. J. Fish. Aquat. Sci. 52, 213–222 (1995).
Gray, J. S. The measurement of marine species diversity, with an application to the benthic fauna of the norwegian continental shelf. J. Exp. Mar. Biol. Ecol. 250, 23–49 (2000).
Lutcavage, M., Kraus, S. & Hoggard, W. Aerial survey of giant bluefin tuna, thunnus thynnus, in the great bahama bank, straits of florida. Fish. Bull. 95, 300–310 (1997).
Royer, F., Fromentin, J. M. & Gaspar, P. Association between bluefin tuna schools and oceanic features in the western mediterranean. Mar. Ecol. Prog. Ser. 269, 249–263 (2004).
Bonhommeau, S., Farrugio, H., Poisson, F. & Fromentin, J.-M. Aerial surveys of bluefin tuna in the western mediterranean sea: retrospective, prospective, perspective. Collective Volume of Scientific Papers 65, 801–811 (2010).
Bauer, R., Bonhommeau, S., Brisset, B. & Fromentin, J. Aerial surveys to monitor bluefin tuna abundance and track efficiency of management measures. Mar. Ecol. Prog. Series 534, 221–234 (2015).
George, J. C., Zeh, J., Suydam, R. & Clark, C. Abundance and population trend (1978–2001) of western arctic bowhead whales surveyed near barrow. Alaska Marine Mammal Science 20, 755–773 (2004).
Slooten, E., Dawson, S. M. & Rayment, W. J. Aerial surveys for coastal dolphins: Abundance of hector’s dolphins off the south island west coast, new zealand. Marine Mammal Science 20, 477–490 (2004).
Harley, L., Davies, N., Hampton, J. & McKechnie, S. Stock Assessment of Bigeye Tuna in the Western and Central Pacific Ocean. Western and Central Pacific Fisheries Commission, Tenth Regular Session, Majuro, Republic of the Marshall Islands. WCPFC-SC10-2014/SA-WP-01 (2014).
Bertignac, M., Hampton, J. & Coan, Jr. A. L. Estimates of exploitation rates for north pacific albacore, thunnus alalunga, from tagging data. Fish. Bull. 97, 421–433 (1999).
Sibert, J. R. & Hampton, J. Mobility of tropical tunas and the implications for fisheries management. Mar. Policy 27, 87–95 (2003).
Gaertner, D., Hallier, J. P. & Maunder, M. N. A tag attrition model as a means to estimate the efficiency of two types of tags used in tropical tuna fisheries. Fish. Res. 69, 171–180 (2004).
Sibert, J. R., Hampton, J., Fournier, D. A. & Bills, P. J. An advection-diffusion-reaction model for the estimation of fish movement parameters from tagging data, with application to skipjack tuna (Katsuwonus pelamis). Can. J. Fish. Aquat. Sci. 56, 925–938 (1999).
Hampton, J. Natural mortality rates in tropical tunas-size really does matter. Can. J. Fish. Aquat. Sci. 57, 1002–1010 (2000).
Shiham, A. M. & Sibert, J. R. Population dynamics and movements of skipjack tuna (katsuwonus pelamis) in the maldivian fishery: analysis of tagging data from an advection-diffusion-reaction model. Aquat. Liv. Res. 15, 13–23 (2002).
Eveson, J. P., Laslett, G. M. & Polacheck, T. An integrated model for growth incorporating tag-recapture, length-frequency, and direct aging data. Can. J. Fish. Aquat. Sci. 61, 292–306 (2004).
Gaertner, D., Delgado de Molina, A., Ariz, J., Pianet, R. & Hallier, J. Variability of the growth parameters of the skipjack tuna (katsuwonus pelamis) among areas in the eastern atlantic: analysis from tagging data within a meta-analysis approach. Aquat. Living Resour. 21, 349–356 (2008).
Leroy, B. et al. Lessons learned from implementing three, large-scale tuna tagging programs in the western and central pacific ocean. Fish. Res. 163, 23–33 (2015).
Quinn, T. J. & Deriso, R. B. Quantitative Fish Dynamics (New York, Oxford University Press, 1999).
Maunder, M. N. et al. Interpreting catch per unit effort data to assess the status of individual stocks and communities. ICES J. Mar. Sci. 63, 1373–1385 (2006).
Jorgensen, S. J. et al. Philopatry and migration of pacific white sharks. Proc. R. Soc. B. 277, 679–688 (2009).
Humphries, N. E., Queiroz, N., Jrm, D., Pade, N. G. & Musyl, M. K. Environmental context explains levy and brownian movement patterns of marine predators. Nature 465, 1066–1069 (2010).
Schaefer, K. M., Fuller, D. W. & Block, B. A. Movements, behavior, and habitat utilization of yellowfin tuna (thunnus albacares) in the pacific ocean off baja california, mexico, determined from archival tag data analyses, including unscented kalman filtering. Fish. Res. 112, 22–37 (2011).
Block, B. A. et al. Tracking apex marine predator movements in a dynamic ocean. Nature 475, 86–90 (2011).
Davis, T. L. & Stanley, a. C. Vertical and horizontal movements of southern bluefin tuna (thunnus maccoyii) in the great australian bight observed with ultrasonic telemetry. Fish. Bull. 100, 448–465 (2002).
Sippel, T. et al. Using movement data from electronic tags in fisheries stock assessment: a review of models, technology and experimental design. Fish. Res. 163, 152–160 (2015).
Uda, M. Types of skipjack schools and their fishing qualities: the shoals of katuwo and their angling. Bull. Jap. Soc. Sci. Fish. 2, 107–111 (1933).
Dagorn, L., Holland, K., Restrepo, V. & Moreno, G. Is it good or bad to fish with fads? what are the real impacts of the use of drifting fads on pelagic marine ecosystems? Fish Fish. 14, 391–415 (2013).
Sempo, G., Dagorn, L., Robert, M. & Deneubourg, J.-l. Impact of increasing deployment of artificial floating objects on the spatial distribution of social fish species. J. Appl. Ecol. 50, 1081–1092 (2013).
Robert, M., Dagorn, L. & Deneubourg, J. The aggregation of tuna around floating objects: What could be the underlying social mechanisms? J. Theor. Biol. 359, 161–170 (2014).
Capello, M. et al. A methodological framework to estimate the site fidelity of tagged animals using passive acoustic telemetry. PLoS ONE 10(8), e0134002 (2015).
Robert, M. et al. Intra-individual behavioral variability displayed by tuna at fish aggregating devices (fads). Mar. Ecol. Prog. Ser. 484, 239–247 (2013).
Dagorn, L., Holland, K. & Itano, D. Behavior of yellowfin (thunnus albacares) and bigeye (thunnus obesus) tuna in a network of fish aggregating devices (fads). Mar. Biol. 151, 595–606 (2007).
Robert, M., Dagorn, L., Deneubourg, J., Itano, D. & Holland, K. Size-dependent behavior of tuna in an array of fish aggregating devices (fads). Mar. Biol. 159, 907–914 (2012).
Cox, D. Regression models and life tables (with discussion). J. R. Stat. Soc. Series B, 187–220 (1972).
R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria, https://www.R-project.org/ (2015).
Akaike, H. Information measures and model selection. Bull. Int. Stat. Inst. 50, 277–290 (1973).
Chu-Shore, J., Westover, M. B. & Bianchi, M. T. Power law versus exponential state transition dynamics: Application to sleep-wake architecture. PLoS ONE 5(12), e14204 (2010).
Lopez, J., Moreno, G., Boyra, G. & Dagorn, L. A model based on data from echosounder buoys to estimate biomass of fish species associated with fish aggregating devices. Fish. Bull. 114, 166–178 (2016).
Forget, F. G. et al. Behaviour and vulnerability of target and non-target species at drifting fish aggregating devices (fads) in the tropical tuna purse seine fishery determined by acoustic telemetry. Can. J. Fish. Aquat. Sci. 72, 1398–1405 (2015).
Schaefer, K. M. & Fuller, D. W. Vertical movements, behavior, and habitat of bigeye tuna (thunnus obesus) in the equatorial eastern pacific ocean, ascertained from archival tag data. Mar. Biol. 157, 2625–2642 (2010).
Lopez, J., Moreno, G., Sancristobal, I. & Murua, J. Evolution and current state of the technology of echo-sounder buoys used by spanish tropical tuna purse seiners in the atlantic, indian and pacific oceans. Fish. Res. 155, 127–137 (2014).
Moreno, G. et al. Fish aggregating devices (fads) as scientific platforms. Fish. Res. (2015).
Acknowledgements
This research was supported by a Marie Curie Intra European Fellowship within the 7th European Community Framework Programme, project QUAESITUM (Grant Agreement Number PIEF-GA-2011-299519) and the ANR project BLUEMED (ANR-14-ACHN-0002-01). Tagging in Hawaii was funded by Cooperative Agreement number NA17RS1230 between the Joint Institute for Marine and Atmospheric Research (JIMAR) and the National Oceanographic and Atmospheric Administration (NOAA). The views expressed herein are those of the authors and do not necessarily reflect the views of NOAA or any of its sub-agencies. A part of the analysis was carried out with financial support from the Commission of the European Communities, specifically the RTD programme of Framework Programme 7, “Theme 2-Food, Agriculture, Fisheries and Biotechnology”, through the research project MADE (mitigating adverse ecological impacts of open ocean fisheries). It does not necessarily reflect its views and in no way anticipates the Commission’s future policy in this area. This work was inspired by (1) the research project FADIO (Fish Aggregating Devices as Instrumented Observatories of pelagic ecosystems), funded by the European Union, DG Research, 5th Framework Package, contract QLRI-CT-2001-02773 as well as by (2) the 2012 international Stock Assessment workshop: understanding purse seine CPUE, funded by the International Seafood Sustainability Foundation (ISSF).
Author information
Authors and Affiliations
Contributions
M.C., J.-L.D. and M.R. conceived the model. L.D. and K.H. conducted the experiments. M.C. and M.R. analyzed the results. K.S., L.D. and K.H. evaluated the method applicability. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Capello, M., Deneubourg, J., Robert, M. et al. Population assessment of tropical tuna based on their associative behavior around floating objects. Sci Rep 6, 36415 (2016). https://doi.org/10.1038/srep36415
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep36415
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.