Abstract
Enterohemorrhagic Escherichia coli O157:H7 (EHEC) has caused foodborne outbreaks worldwide and the bacterium forms antimicrobial-tolerant biofilms. We investigated the abilities of various plant essential oils and their components to inhibit biofilm formation by EHEC. Bay, clove, pimento berry oils and their major common constituent eugenol at 0.005% (v/v) were found to markedly inhibit EHEC biofilm formation without affecting planktonic cell growth. In addition, three other eugenol derivatives isoeugenol, 2-methoxy-4-propylphenol, and 4-ethylguaiacol had antibiofilm activity, indicating that the C-1 hydroxyl unit, the C-2 methoxy unit, and C-4 alkyl or alkane chain on the benzene ring of eugenol play important roles in antibiofilm activity. Interestingly, these essential oils and eugenol did not inhibit biofilm formation by three laboratory E. coli K-12 strains that reduced curli fimbriae production. Transcriptional analysis showed that eugenol down-regulated 17 of 28 genes analysed, including curli genes (csgABDFG), type I fimbriae genes (fimCDH) and ler-controlled toxin genes (espD, escJ, escR, and tir), which are required for biofilm formation and the attachment and effacement phenotype. In addition, biocompatible poly(lactic-co-glycolic acid) coatings containing clove oil or eugenol exhibited efficient biofilm inhibition on solid surfaces. In a Caenorhabditis elegans nematode model, clove oil and eugenol attenuated the virulence of EHEC.
Similar content being viewed by others
Introduction
Enterohemorrhagic Escherichia coli O157:H7 (EHEC) is responsible for outbreaks of hemorrhagic colitis and associated bloody diarrhea1. EHEC forms attaching and effacing (AE) lesions on human epithelial cells and produces Shiga-like toxins, which are responsible for the development of hemolytic-uremic syndrome2. Unfortunately, no effective therapy is available because antimicrobial agents increase the risk of developing hemolytic-uremic syndrome, a major cause of acute renal failure in children1.
The first stage of EHEC infection involves the adhesion of bacterial cells to host cells and the formation of microcolonies leading to colonization of the large intestine2. EHEC is also able to form biofilms on various biotic and abiotic surfaces, such as, on plants, stainless steel, glass, and polymers3,4. These biofilms are resistant to conventional antimicrobial agents, host defenses, and external stresses. Accordingly, in clinical and industrial environments EHEC biofilms pose a substantial challenge, and methods of controlling these biofilms are urgently required. The mechanism of EHEC biofilm formation is complex, which has been the subject of research. The importance of fimbriae, including curli and pili, for EHEC biofilm formation has been well-documented4,5,6. Swimming and swarming motilities influence the biofilm development of E. coli7,8,9. Also, several signaling molecules, such as cyclic di-GMP, autoinducer-2, and indole, are known to be involved in the biofilm formation of E. coli7,10.
Essential oils derived from plants have been widely used as antimicrobial and flavoring agents due to their variable terpenoid and phenolic contents11, and as a result, the anti-bacterial and anti-fungal activities of several essential oils have been well-studied11,12,13. However, traditional antimicrobial agents primarily designed to inhibit cell growth often result in bacterial drug resistance, and thus, essential oils have been studied using other developmental approaches, such as, the inhibition of biofilm formation, toxin production, bacterial quorum sensing, and of adhesive factors. For example, carvacrol and eugenol14, grapefruit limonoids15, β-sitosterol glucoside from clementine peel16, ginkgolic acids from Ginkgo biloba17, and cinnamaldehyde and eugenol from cinnamon bark oil18, which are found in essential oils, have been reported to inhibit EHEC biofilm formation. However, few studies have been undertaken to compare the antibiofilm characteristics of large numbers of essential oils.
In this study, 83 essential oils were initially screened for their ability to inhibit EHEC biofilm formation. Three oils, namely, bay, clove, and pimento berry oils demonstrated strong anti-biofilm activity against EHEC at sub-inhibitory concentrations, but not against laboratory E. coli strains. Chemical structure-activity assays revealed that eugenol and three other eugenol derivatives had anti-biofilm activity. In order to understand their action mechanisms, transcriptional analysis, motility analysis, and electron microscopy were utilized. In addition, a biocompatible poly(lactic-co-glycolic acid) surface coatings containing biofilm inhibitors were prepared and their antibiofilm effects were examined. Finally, an in vivo Caenorhabditis elegans model was used to study the effects of eugenol and of clove oil to confirm their antivirulence effects on EHEC.
Results
Anti-biofilm effects of essential oils against EHEC
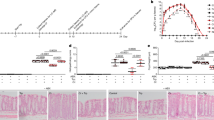
To identify new anti-biofilm agents, 83 essential oils were initially screened in 96-well plates at a concentration of 0.005% (v/v) to minimize antimicrobial effects. Several essential oils were found to inhibit EHEC biofilm formation, but with widely different efficiencies. Detailed information on EHEC growth and biofilm formation in the presence of the 83 essential oils is provided in Supplementary Table S1. Notably, four essential oils, namely bay, cinnamon bark, clove, and pimento berry oil inhibited EHEC biofilm formation by more than 75%. No growth reduction of EHEC cells above 30% at OD620 was observed at 0.005% (v/v) as compared with untreated controls. Kim et al. found that cinnamon bark oil18 had antibiofilm activity against EHEC, but this is the first time that bay, clove, and pimento berry oils have been reported to have antibiofilm activity. In the present study, more detailed study showed bay, clove, and pimento berry oil all dose-dependently inhibited EHEC biofilm formation in 96-well polystyrene plates (Fig. 1a–c). Since bacteria form biofilms on the bottoms and sides of these plates, confocal laser microscopy and EHEC expressing green fluorescent protein were used to observe biofilm formation on glass, and our microscopic observations confirmed that all three essential oils dramatically inhibited biofilm formation on the bottom of glass (Fig. 1d). Biofilm inhibition was further confirmed by COMSTAT analysis. More specifically, bay, clove, and pimento berry oils reduced all three measured parameters (biomass, mean thickness, and substratum coverage) of EHEC (Table 1), and biomass (volume/area) and mean thickness were reduced by >80% by all three oils at 0.005% (v/v).
Effects of bay, clove, and pimento berry oils on EHEC biofilm formation.
Biofilm formation (OD570) by EHEC was quantified in the presence of each of the three essential oils selected for further study after culture for 24 h in 96-well plates (a–c). Biofilm formation by EHEC/pCM18 tagged with green fluorescent protein in the presence of essential oils (0.005%) was confirmed by confocal laser microscopy (d). Scale bar = 50 μm.
Identification of the active anti-biofilm components in essential oils
To identify the active anti-biofilm components in the above three essential oils, GC-MS analysis was performed and as a result 33 different compounds were identified (Table 2). Eugenol was the predominant component and accounted for more than 62% of all three oils. In addition, myrcene, chavicol, methyleugenol, and β-caryophyllene were found to be present at >7.8%. This result concurs with previous reports, in which bay oil19, clove oil20, and pimento berry oil21 were found to contain about 60% eugenol. As was expected, eugenol was also found to dose-dependently inhibit EHEC biofilm formation (Fig. 2b), whereas other compounds, such as, myrcene and β-caryophyllene, did not show anti-biofilm activity at concentration under 0.005% (data not shown). These results suggest that eugenol is largely responsible for the anti-biofilm activity of these oils.
Inhibition of biofilm formation by eugenol-like compounds.
Chemical structures are shown (a). Biofilm formation by EHEC was quantified in the presence of selected chemicals after incubation for 24 h in 96-well polystyrene plates without shaking (b). At least two independent experiments were conducted (total 12 wells). Error bars indicate standard deviations. Biofilms formed in the presence of 4-ethylguaiacol or eugenol (0.005%) were observed under a confocal laser microscope (c). Scale bar = 50 μm. *P < 0.05 versus non-treated controls.
The antimicrobial activities of essential oils and eugenol were investigated by measuring minimum inhibitory concentrations (MICs) and EHEC planktonic cell growth. The MICs of the three oils and eugenol against EHEC were >0.1%, which is consistent with previously reported values12,22. Notably, MICs against EHEC were 20-times higher than the concentrations (~0.005%) required for antibiofilm activity. Furthermore, the three oils and eugenol at concentrations ≤0.005% did not retard planktonic cell growth, whereas at 0.01%, they reduced final cell density by about 20% (Supplementary Fig. S1). These findings indicate reduced biofilm formation by the three oils or eugenol was due to antibiofilm activity and not to antimicrobial activity.
Anti-biofilm activities of eugenol derivatives on EHEC
To identify the structural motif present in eugenol responsible for antibiofilm activity against EHEC, we investigated the antibiofilm activities of eleven eugenol-related compounds (Fig. 2a). We found eugenol, 4-ethylguaiacol, isoeugenol, and 2-methoxy-4-propylphenol at 0.005% markedly inhibited EHEC biofilm formation by ≥50% versus untreated controls. Specifically, eugenol at 0.005% decreased EHEC biofilm formation by 87%, whereas the other seven compounds (guaiacol, 4-ethylcatechol, thymol, vanillin, carvacrol, methyl eugenol, and 2-methoxyhydroquinone) at concentrations of up to 0.005% did not significantly inhibit EHEC biofilm formation. Although, it has been previously reported that vanillin inhibits biofilm formation by Aeromonas hydrophila23, it did not inhibit EHEC biofilm formation in the present study.
Interestingly, antibiofilm activity against EHEC was found to be closely related to the presence and size of a C-4 alkane chain, and the presence of a hydroxyl group and a methoxy group at the C-1 and C-2 positions of the benzene ring, because eugenol, 4-ethylguaiacol, isoeugenol, and 2-methoxy-4-propylphenol all possess a C-4 alkane or alkyl chain and C-1 hydroxyl and C-2 methoxy groups (Fig. 2a). However, methyl eugenol, which has an alkyl chain and a methoxy unit at C-1 and C-2 lacked inhibitory activity, indicating the eugenol backbone and the C-1 hydroxyl and C-2 methoxy groups are required for antibiofilm activity. Because eugenol reduced biofilm formation most, we focused on it and eugenol-rich clove oil for further study.
Microscopic observations confirmed that eugenol and 4-ethylguaiacol at 0.005% (v/v) markedly inhibited EHEC biofilm formation (Fig. 2c). In addition, COMSTAT analysis confirmed that eugenol and 4-ethylguaiacol reduced biofilm biomass, mean thickness, and substratum coverage (Table 1).
Effects of the three essential oils and eugenol on biofilm formation by other E. coli strains
It is important that we develop therapeutic compounds that inhibit pathogenic biofilm formation but leave beneficial commensal biofilms unharmed24. Thus, the effects of the three essential oils and eugenol were investigated on three laboratory E. coli strains: BW25113, MG1655, and TG1. Unlike that observed for EHEC, neither eugenol nor the three oils had any biofilm inhibitory effects on these three E. coli K-12 strains (Fig. 3a–c). The three laboratory E. coli strains developed poor biofilms in comparison to the EHEC strain as judged by crystal violet staining. The biofilm formation of the untreated laboratory strains was similar to that of the EHEC strain treated with 0.005% eugenol. It is intriguing how eugenol specifically inhibits biofilm formation by EHEC. Because curli fimbriae are critical for E. coli biofilm development25, we investigated fimbriae productions by EHEC and the three laboratory strains. Interestingly, EHEC produced more fimbriae than the three laboratory E. coli strains on Congo red plates (Fig. 3d), which suggests eugenol inhibits the development of fimbriated E. coli like EHEC.
Effects of bay, clove, pimento berry oils and eugenol on biofilm formation by three laboratory E. coli strains and fimbriae production. Biofilm formation (OD570) by E. coli BW25113 (a), MG1655 (b), and TG1 (c) was quantified in the presence of essential oils or eugenol after culture for 24 h in 96-well plates without shaking. Fimbriae production was assessed on Congo red plates after 24 h (d). At least three independent experiments were conducted.
Effect of the three essential oils and eugenol on EHEC motility
The impacts of three essential oils and eugenol on the swarming and swimming motilities of EHEC were also investigated because motility plays an important role in E. coli biofilm formation9,26. While the three oils clearly reduced swarming motility, eugenol did not (Supplementary Fig. S2a), and neither the three oils nor eugenol changed EHEC swimming motility (Supplementary Fig. S2b). Additionally, the presence of elongated swarming and swimming was measured as the diameter (in millimeters) of the zone of expansion (Supplementary Fig. S2c,d). The result suggests that the anti-biofilm effect of eugenol on EHEC is not closely related to motility.
Transcriptional changes in EHEC cells induced by clove oil or eugenol
To investigate the genetic bases of EHEC biofilm inhibition by clove oil and eugenol, qRT-PCR was performed to examine the differential expressions of biofilm- and virulence-related genes in treated and non-treated EHEC cells. Most noticeably, both clove oil and eugenol most markedly inhibited the expression of curli genes (csgABDFG) by 8-fold to 155-fold, while they less appreciably changed the expression of type I fimbrial and other fimbrial genes (Supplementary Table S2), whereas they did not affect the expression of rrsG (a housekeeping gene) (Fig. 4). Also, clove oil down-regulated several motility genes (swarming genes fimA and fimH and swimming genes flhD, fliA, and motB), and LEE transcriptional regulator ler gene (Fig. 4a), which concur with its inhibition of motility (Supplementary Fig. S2). On the other hand, eugenol altered the expression of motility genes less but decreased the expressions of ler and ler-controlled toxin genes (espD, escJ, escR, and tir) (Fig. 4b). Neither clove oil nor eugenol appreciably affected the expressions of quorum sensing genes (luxS, luxR and tnaA) or Shiga-like toxin genes (stx1 and stx2). Previously, it has been reported that curli fimbriae production is crucial for biofilm formation by EHEC strains, particularly ATCC 43895 strain or called EDL933 strain used in this study4,5,6. This qRT-PCR result indicates that clove oil and eugenol strongly reduce the transcription of curli genes, as well as reducing the transcription of several other EHEC genes, including type I fimbriae genes.
Transcriptional profiles of EHEC cells treated with or without clove oil or eugenol.
EHEC was cultivated in LB broth with or without clove oil (a) or eugenol (b) at 0.005% (v/v) for 2 h with agitation (250 rpm) at 37 °C. Transcriptional profiles were measured by qRT-PCR. Relative gene expressions represent transcriptional levels after treatment with clove oil or eugenol versus untreated controls (value 1.0). The experiment was performed in duplicate.
Eugenol reduced fimbriae formation by EHEC
Because fimbriae are important for EHEC biofilm formation4,6,27, and the gene expression of the csg operon producing curli fimbriae production was found to be markedly down-regulated by clove oil and eugenol (Fig. 4), we investigated curli fimbriae production by SEM and Congo red that specifically binds curli fimbriae. In-line with our gene expression data, both clove oil and eugenol at 0.005% clearly reduced fimbriae production (Fig. 5a) and specifically curli production (Fig. 5b), which indicates down-regulation of curli genes and of curli fimbriae inhibition underlie the antibiofilm activities of clove oil and of eugenol.
Effects of clove oil and eugenol on curli fimbriae production by EHEC.
Fimbriae productions by EHEC cells grown with or without clove oil or eugenol (0.005%) were observed by SEM (a). For SEM analysis, cells were cultured in LB broth with or without clove oil or eugenol at 37 °C for 2 h. The scale bar represents 3 μm. Congo red binding assay for the detection of curli fimbriae production (b). EHEC cells were grown in LB broth in the presence of Congo red and Commassie brilliant blue at 37 °C for 24 h. Culture supernatants were measured at 490 nm and cell pellets were photographed. Dark red color indicates curli production stained by Congo red and Commassie brilliant blue.
Biofilm inhibition using a biocompatible polymer coating
To prevent biofilm formation on a solid surface, biofilm inhibitors were incorporated into a biocompatible poly(D,L-lactide-coglycolide) (PLGA; a polymer with many medical, pharmaceutical, and industrial uses). As was expected, PLGA coatings containing clove oil or eugenol (0.005%) markedly inhibited EHEC biofilm formation (Fig. 6). COMSTAT analysis showed that both clove oil and eugenol reduced biofilm biomass, mean thickness, and substratum coverage by ≥90% (Table 3). These results complement observed biofilm reductions observed on polystyrene plates (Figs 1a–c and 2b) and glass (Figs 1d and 2c). Also, the result demonstrates that biocompatible polymer coatings could be potentially used to prevent pathogenic biofilm formation on biomedical and food processing surfaces.
Inhibition of biofilm formation by PLGA coatings containing clove oil or eugenol.
Biofilms formed by EHEC/pCM18-GFP were investigated by confocal laser microscopy. Clove oil or eugenol (0.005%, v/v) was incorporated into PLGA and biofilms were grown at 37 °C for 24 h without shaking. Scale bars represent 50 μm. At least three independent experiments were conducted.
Clove oil and eugenol reduced EHEC virulence in the nematode model
Since EHEC colonizes and replicates in the digestive tract of C. elegans and has the ability to kill the nematode28, the effects of clove oil and eugenol on EHEC virulence were investigated by assessing C. elegans survival. In the agreement with previous results28, EHEC significantly reduced the lifespan of C. elegans as compared with E. coli OP50, which is a common food source for the nematode. Moreover, it was found that clove oil or eugenol at 0.005% prolonged C. elegans survival in the presence of EHEC. In fact, the effects of clove oil and eugenol were similar (Fig. 7). This result is in-line with the observed down-regulation of biofilm formation and of the LEE transcriptional regulator ler by eugenol (Fig. 4). In addition, to examine the toxicity of eugenol, C. elegans survival was investigated using E. coli OP50. E. coli OP50 fed eugenol at a concentration of 0.005% was found to have no effect on C. elegans survival (Fig. 7).
Discussion
EHEC infection is problematic worldwide because of the lack of an effective therapy and the formation of antibiotic-resistant biofilms by EHEC serotypes. The present study demonstrates that three essential oils (bay, clove, and pimento berry oils) exhibit high antibiofilm activity against EHEC without affecting its planktonic cell growth. Hence, unlike antibiotics that aim to inhibit cell growth, biofilm inhibitors that do not inhibit bacterial growth could reduce the risk of drug resistance. In the present study, identification of the active compound and a chemical structure-activity relationship investigation revealed the antibiofilm activities of eugenol and its derivatives. Furthermore, transcriptional and phenotypic assays provided clues regarding the mechanism of biofilm inhibition and virulence attenuation by eugenol and eugenol-rich oils.
Our investigation of fimbriae production, motility, and laboratory E. coli biofilm formation, and qRT-PCR analysis showed that the antibiofilm activities of eugenol and the three oils were due to the suppression of fimbriae production (Figs 3 and 5) and other genes (Fig. 4) in EHEC. It is well known that fimbriae or pili play important role in the biofilm formation of laboratory and pathogenic E. coli such as EHEC9,27,29. EHEC strains contain a series of fimbriae such as curli fimbriae (Csg), type I fimbriae (Fim), E. coli common pilus (Ecp), F9 fimbriae (Z2200), and other fimbrial proteins29. It has been also reported that several phytochemicals, such as, 3-indolylacetonitrile30, phloretin31, resveratrol (and its dimer viniferin)32,33, cinnamaldehyde18, coumarin34, and ginkgolic acids17 inhibit EHEC biofilm formation primarily by inhibiting curli fimbriae production. Hence, it appears that fimbriae-reducing ability is not rare in the plant kingdom and that the fimbriae inhibition could be viewed a practical target for suppressing EHEC biofilm formation.
To date, a dozen essential oils have been shown to possess antibacterial and antibiofilm activities against wide range of pathogenic bacteria13,35,36. Previously, eugenol was found to inhibit biofilm formation by Staphylococcus aureus37, Candida albicans38, Listeria monocytogenes14, and EHEC14,18, and to inhibit quorum sensing of Pseudomonas aeruginosa39. The present study shows for the first time that eugenol-rich oils (bay, clove, and pimento berry oils) at 0.005% (about 50 μg/ml) have antibiofilm activity against EHEC (Fig. 1), and provides detail of chemical structure-activity relationships and action mechanism involved. Eugenol is used in perfumes, flavorings, and as a local antiseptic and anaesthetic40, and is abundant found in clove oil, nutmeg, cinnamon, basil, and bay leaves. This study suggests another application for inexpensive eugenol-rich oils to control EHEC infection.
Our chemical structure-activity relationship study revealed that the C-1 hydroxyl, the C-2 methoxy, and a C-4 alkyl or alkane chain on the aromatic ring of eugenol are required for antibiofilm against EHEC (Fig. 2). Many eugenol derivatives can be synthesized41,42, and several phytochemicals, such as, zingerones, gingerols, shogaols, and paradols bearing the eugenol motif are found in plant extracts. Thus, further investigations of eugenol derivatives are probably worthwhile to identify more potent antibiofilm and anti-virulence compounds.
EHEC produces potent Shiga-like toxins and encodes the LEE operon genes, which are both required for forming attaching and effacing lesions to host epithelial cells2. Furthermore, it has been shown the majority of LEE genes are positively regulated by the ler gene (LEE-encoded regulator)43. In the present study, eugenol was found to down-regulate the expression of ler and ler-controlled toxin genes (espD, escJ, escR, and tir) and curli-producing genes (csgABDFG) (Fig. 4). The pathogenicity of EHEC was attenuated by eugenol and clove oil (Fig. 7), but they did not harm laboratory E. coli biofilms (Fig. 3). Therefore, it appears eugenol could be used as a lead compound for the development of biofilm inhibitors and toxin producing inhibitors for fimbriae-rich and LEE-encoding E. coli strains, such as, enteropathogenic Escherichia coli44, uropathogenic Escherichia coli45, and enteroaggregative Escherichia coli46.
In the present study, eugenol, eugenol derivatives, and essential oils were found to reduce biofilm formation by and the virulence of EHEC. Furthermore, PLGA coatings containing eugenol or eugenol-rich clove oil effectively prevented EHEC biofilm formation on solid surfaces (Fig. 6). These results suggest that eugenol and its derivatives have potential uses in antivirulence strategies against persistent EHEC infection. In addition, it would be interesting to explore the uses of inexpensive eugenol-rich oils in the food, cosmetics, fishery, agricultural, and environmental industries and in the medical field.
Materials and Methods
Bacterial strain, essential oils, and growth conditions
All experiments were conducted at 37 °C in Luria-Bertani (LB) medium, which was used to culture E. coli O157:H7 (ATCC 43895, EDL933 strain) and three laboratory E. coli K-12 strains (MG1655, BW25113, and TG1). To culture E. coli O157:H7/pCM18 tagged with green fluorescent protein, LB broth containing 300 μg/ml of erythromycin was used to maintain the pCM18-GFP plasmid. Bacterial cells were initially streaked from −80 °C glycerol stock on LB plates, grown on plates, and then were cultured from a fresh single colony in LB broth. For phenotypic assays, overnight cultures (stationary phase cells) were inoculated again in LB broth at an initial turbidity of 0.05 at 600 nm. The 83 essential oils (Supplementary Table S1) were obtained from Berjé (Bloomfield, NJ, USA) or Jin Aromatics (Anyang, Gyeonggi Province, Korea). Other chemicals (guaiacol, 4-ethylcatechol, thymol, 2-methoxyhydroquinone, vanillin, carvacrol, methyl eugenol, 2-methoxy-4-propylphenol, isoeugenol, 4-ethylguaiacol, and eugenol) were purchased from Sigma-Aldrich (St. Louis, USA). For cell growth measurements, turbidity was measured at 600 nm using a spectrophotometer (Optizen 2120UV, Mecasys, Korea).
Crystal-violet static biofilm formation assay
A static biofilm formation assay was performed in 96-well polystyrene plates (SPL Life Sciences, Korea), as previously described30. Briefly, overnight cultures were inoculated in LB broth (total volume 300 μl) at an initial turbidity of 0.05 at 600 nm and cultured with or without bay, clove, or pimento berry oils or components for 24 h without shaking at 37 °C. To quantify biofilm formation, cell cultures were washed three times with H2O to remove all non-adherent cells. Biofilms were stained with crystal violet for 20 min, rinsed three times with H2O, extracted with 95% ethanol, and absorbances were measured at 570 nm. Results are the averages of at least twelve replicate wells.
Gas chromatograph/mass spectroscopy (GC-MS) analysis
Detailed chemical compositions of the three active essential oils were obtained by GC/MS using an Agilent 6890N GC DB-5 MS fused silica capillary column (30 m × 0.25 m i.d., film thickness 0.25 μm) and a Jeol JMS 700 mass spectrometer. Capillary column details and temperature conditions for the analysis were as previously described47. GC-MS was performed using an electron ionization system at 70 eV. Helium was used as the carrier gas at a flow rate of 1 ml/min. The temperatures of the GC injector and MS transfer line were 280 °C and 250 °C, respectively. An initial oven temperature of 50 °C was maintained for 2 min and then increased to 250 °C at a rate of 10 °C/min and this was followed with a holding period at 250 °C for 10 min. Diluted samples (1.0 ml, 1/100 (v/v) in methanol) were injected manually in split-less mode. The relative percentages of oil components were calculated by normalizing peak areas and are expressed as percentages. Components were identified using GC retention times and by matching mass spectra with entries in the Wiley and NIST libraries.
Confocal laser scanning microscopy and COMSTAT analysis
Bacterial cells (E. coli O157:H7/pCM18 tagged with green fluorescent protein) were cultured in glass-bottomed dishes (SPL Life Sciences, Korea) without shaking with or without essential oils or eugenol. Planktonic cells were removed by washing with PBS three times, and biofilms were visualized by excitation using an Ar laser 488 nm (emission wavelengths 500 to 550 nm) under a confocal laser microscope (Nikon eclipse Ti, Tokyo) using a 20 × objective48. Color confocal images were constructed using NIS-Elements C version 3.2 (Nikon eclipse). For each experiment, at least 10 random positions in two independent cultures were chosen for microscopic analysis.
To quantify biofilm formation, color confocal images (20 image stacks) were converted to gray scale using ImageJ. COMSTAT biofilm software49 was used to determine biomasses (μm3 per μm2), mean thicknesses (μm), and substratum coverages (%). Thresholding value was fixed for all image stacks, and at least 4 positions and 20 planar images per position were analyzed.
Fimbriae assay using a Congo red and scanning electron microscopy (SEM)
To measure curli fimbriae production, LB agar medium containing 20 μg/ml Congo red (Sigma), 10 μg/ml Coomassie brilliant blue (Sigma), and 15 g L–1 agar was used, as previously described50. Curli fimbriae production was visualized after 24 h of incubation at 37 °C on Congo red plates. In addition, a Congo red binding assay was performed, as previously described51. Briefly, overnight culture of E. coli O157:H7 EDL933 was reinoculated (100:1) with 0.01% eugenol or clove oil in LB medium with 20 μg/ml Congo red and 10 μg/ml Coomassie brilliant blue in 14-ml round bottom tubes and incubated at 37 °C for 24 h with 250 rpm shaking. Incubated cells were collected by centrifugation at 16,600 × g for 15 min. Optical density of the supernatants was measured at 490 nm. Also, SEM was used to observe fimbriae production, as previously described30. Briefly, EHEC cells were incubated for 2 h at 37 °C with agitation at 250 rpm, and then re-incubated for 3 h more with or without clove oil or eugenol (0.005%) at 37 °C with shaking. After prompt fixation with glutaraldehyde and formaldehyde, cells were collected by filtering through a 0.45 μm nylon filter under vacuum. The filter was then cut into 0.5 × 0.5 mm squares and post-fixed in sodium phosphate buffer, osmium, ethanol, and isoamyl acetate, and critical-point dried. Specimens were examined using an SEM (S-4100; Hitachi, Japan) at 15 kV and magnifications ranging from 2,000X to 10,000X.
Swimming and swarming motility
Swimming motility was assayed using 0.3% agar plates containing 1% tryptone and 0.25% NaCl, and swarming motility was assayed using LB broth supplemented with 0.8% glucose and 0.5% agar, as previously described32. Essential oils, eugenol or DMSO (the control) were added to motility agar. EHEC was grown to an OD600 of 1.0 and then ~0.2 μl aliquots of cultures were placed in motility plates using a sterilized pipette tip. Sizes of swimming halos were measured 16 h later. Each experiment was performed using at least three independent cultures.
RNA isolation and quantitative real-time RT-PCR
For transcriptional analysis, EHEC was inoculated into 25 ml of LB broth in 250 ml shake flasks at a starting OD600 of 0.05, and then cultured at 37 °C for 3 h with agitation (250 rpm) in the presence or absence of clove oil or eugenol (0.005%) for another 2 hours. RNase inhibitor (RNAlater, Ambion, TX, USA) was added to prevent RNA degradation. Total RNA was isolated using a Qiagen RNeasy mini Kit (Valencia, CA, USA).
qRT-PCR was used to investigate the transcription levels of curli genes (csgA, csgB, csgD, csgF and csgG), type I fimbriae genes (fimA, fimC, fimD, and fimH), other fimbriae genes (ecpA, ecpR, and Z2200), cellulose gene (bcsA), motility genes (flhD, fliA, motB, and qseB), AI-2 quorum sensing genes (luxS and luxR), indole-synthesis gene (tnaA), shiga-like toxin genes (stx1 and stx2), and LEE-encoded regulator genes (ler, espD, escJ, escR, and tir) in EHEC treated with or without clove oil or eugenol (0.005%). Gene specific primers were used and rrsG was used as a housekeeping control (Supplementary Table S3). The qRT-PCR method used was an adaptation of a previously described method30. qRT-PCR was performed using a SYBR Green master mix (Applied Biosystems, Foster City, USA) and an ABI StepOne Real-Time PCR System (Applied Biosystems) on two independent cultures.
Surface coating with biofilm inhibitors
To fabricate a coating containing biofilm inhibitor, we used biodegradable poly(D,L-lactide-coglycolide) (PLGA) as previously described52. Briefly, essential oils or eugenol (final concentration, 0.005% v/v) were mixed into 2% PLGA dissolved in chloroform and 25 μl of these mixtures were applied to slide-glass bottomed dishes to produce a 0.7~0.8 cm diameter coatings, which were then air-dried for 1 h and sterilized by UV exposure for 4 h. To induce biofilm formation on coated glass surfaces, cells (E. coli O157:H7/pCM18 tagged with green fluorescent protein; 4 × 107 CFU/ml) were re-inoculated into LB. Samples were then incubated at 37 °C for 24 h. Planktonic cells were removed by washing with PBS twice, and biofilm cells in PBS buffer were visualized by confocal laser microscopy.
The Caenorhabditis elegans nematode model
The C. elegans killing assay was performed as described previously53. In brief, E. coli O157:H7 was cultured with or without clove oil or eugenol (0.005%) at 37 °C for 18 h, and then 10 μl of E. coli O157:H7 was spread onto NGM plates. E. coli OP50 was used as a control strain. L4/young adult fer-15;fem-154 worms (n = 60) were infected by placing them on the lawns. Nematodes were then incubated at 20 °C and scored as alive or dead on a daily basis by gently touching them with a platinum wire. Three independent experiments were conducted.
Statistical analysis
Sample sizes of all experiments are indicated in ‘Material and Methods’. Results are expressed as means ± standard deviations. The Student’s t-test was used to determine the significances of differences between samples and non-treated controls. Statistical significance was accepted for p values <0.05, and significant changes are indicated by asterisks in figures.
Additional Information
How to cite this article: Kim, Y.-G. et al. Essential Oils and Eugenols Inhibit Biofilm Formation and the Virulence of Escherichia coli O157:H7. Sci. Rep. 6, 36377; doi: 10.1038/srep36377 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Tarr, P. I., Gordon, C. A. & Chandler, W. L. Shiga-toxin-producing Escherichia coli and haemolytic uraemic syndrome. Lancet 365, 1073–1086 (2005).
Nataro, J. P. & Kaper, J. B. Diarrheagenic Escherichia coli. Clin Microbiol Rev 11, 142–201 (1998).
Patel, J., Sharma, M. & Ravishakar, S. Effect of curli expression and hydrophobicity of Escherichia coli O157:H7 on attachment to fresh produce surfaces. J Appl Microbiol 110, 737–745 (2011).
Ryu, J. H. & Beuchat, L. R. Biofilm formation by Escherichia coli O157:H7 on stainless steel: effect of exopolysaccharide and curli production on its resistance to chlorine. Appl Environ Microbiol 71, 247–254 (2005).
Lee, J.-H., Kim, Y.-G., Cho, M. H., Wood, T. K. & Lee, J. Transcriptomic analysis for genetic mechanisms of the factors related to biofilm formation in Escherichia coli O157:H7. Curr Microbiol 62, 1321–1330 (2011).
Uhlich, G. A., Cooke, P. H. & Solomon, E. B. Analyses of the red-dry-rough phenotype of an Escherichia coli O157:H7 strain and its role in biofilm formation and resistance to antibacterial agents. Appl Environ Microbiol 72, 2564–2572 (2006).
Wood, T. K. Insights on Escherichia coli biofilm formation and inhibition from whole-transcriptome profiling. Environ Microbiol 11, 1–15 (2009).
Beloin, C., Roux, A. & Ghigo, J. M. Escherichia coli biofilms. Curr Top Microbiol Immunol 322, 249–289 (2008).
Pratt, L. A. & Kolter, R. Genetic analysis of Escherichia coli biofilm formation: roles of flagella, motility, chemotaxis and type I pili. Mol Microbiol 30, 285–293 (1998).
Worthington, R. J., Richards, J. J. & Melander, C. Small molecule control of bacterial biofilms. Org Biomol Chem 10, 7457–7474 (2012).
Helander, I. M. et al. Characterization of the action of selected essential oil components on gram-negative bacteria. J Agric Food Chem 46, 3590–3595 (1998).
Hammer, K. A., Carson, C. F. & Riley, T. V. Antimicrobial activity of essential oils and other plant extracts. J Appl Microbiol 86, 985–990 (1999).
Kavanaugh, N. L. & Ribbeck, K. Selected antimicrobial essential oils eradicate Pseudomonas spp. and Staphylococcus aureus biofilms. Appl Environ Microbiol 78, 4057–4061 (2012).
Pérez-Conesa, D., McLandsborough, L. & Weiss, J. Inhibition and inactivation of Listeria monocytogenes and Escherichia coli O157:H7 colony biofilms by micellar-encapsulated eugenol and carvacrol. J Food Protect 69, 2947–2954 (2006).
Vikram, A., Jesudhasan, P. R., Jayaprakasha, G. K., Pillai, B. S. & Patil, B. S. Grapefruit bioactive limonoids modulate E. coli O157:H7 TTSS and biofilm. Int J Food Microbiol 140, 109–116 (2010).
Vikram, A., Jayaprakasha, G. K., Uckoo, R. M. & Patil, B. S. Inhibition of Escherichia coli O157:H7 motility and biofilm by β-sitosterol glucoside. BBA-Gen Subjects 1830, 5219–5228 (2013).
Lee, J.-H., Kim, Y.-G., Ryu, S. Y., Cho, M. H. & Lee, J. Ginkgolic acids and Ginkgo biloba extract inhibit Escherichia coli O157:H7 and Staphylococcus aureus biofilm formation. Int J Food Microbiol 174, 47–55 (2014).
Kim, Y.-G., Lee, J.-H., Kim, S.-I., Baek, K.-H. & Lee, J. Cinnamon bark oil and its components inhibit biofilm formation and toxin production. Int J Food Microbiol 195, 30–39 (2015).
Hoferl, M. et al. Correlation of antimicrobial activities of various essential oils and their main aromatic volatile constituents. J Essent Oil Res 21, 459–463 (2009).
Bhuiyan, M. N. I. Constituents of the essential oil from leaves and buds of clove (Syzigium caryophyllatum (L.) Alston). Afr J Pharm Pharmacol 6, 1260–1263 (2012).
Seo, S. M. et al. Fumigant antitermitic activity of plant essential oils and components from ajowan (Trachyspermum ammi), allspice (Pimenta dioica), caraway (Carum carvi), dill (Anethum graveolens), geranium (Pelargonium graveolens), and litsea (Litsea cubeba) oils against japanese termite (Reticulitermes speratus Kolbe). J Agric Food Chem 57, 6596–6602 (2009).
Pei, R. S., Zhou, F., Ji, B. P. & Xu, J. Evaluation of combined antibacterial effects of eugenol, cinnamaldehyde, thymol, and carvacrol against E. coli with an improved method. J Food Sci 74, M379–M383 (2009).
Ponnusamy, K., Paul, D. & Kweon, J. H. Inhibition of quorum sensing mechanism and Aeromonas hydrophila biofilm formation by vanillin. Environ Eng Sci 26, 1359–1363 (2009).
Kolter, R. & Greenberg, E. P. Microbial sciences: the superficial life of microbes. Nature 441, 300–302 (2006).
Rodrigues, D. F. & Elimelech, M. Role of type 1 fimbriae and mannose in the development of Escherichia coli K12 biofilm: from initial cell adhesion to biofilm formation. Biofouling 25, 401–411 (2009).
Ren, D., Sims, J. J. & Wood, T. K. Inhibition of biofilm formation and swarming of Escherichia coli by (5Z)-4-bromo-5-(bromomethylene)-3-butyl-2(5H)-furanone. Environ Microbiol 3, 731–736 (2001).
Rendón, M. A. et al. Commensal and pathogenic Escherichia coli use a common pilus adherence factor for epithelial cell colonization. Proc Natl Acad Sci USA 104, 10637–10642 (2007).
Chou, T. C. et al. Enterohaemorrhagic Escherichia coli O157:H7 Shiga-like toxin 1 is required for full pathogenicity and activation of the p38 mitogen-activated protein kinase pathway in Caenorhabditis elegans. Cell Microbiol 15, 82–97 (2013).
McWilliams, B. D. & Torres, A. G. Enterohemorrhagic Escherichia coli adhesins. Microbiol Spectr 2 (2014).
Lee, J.-H., Cho, M. H. & Lee, J. 3-Indolylacetonitrile decreases Escherichia coli O157:H7 biofilm formation and Pseudomonas aeruginosa virulence. Environ Microbiol 13, 62–73 (2011).
Lee, J.-H. et al. Apple flavonoid phloretin inhibits Escherichia coli O157:H7 biofilm formation and ameliorates colon inflammation in rats. Infect Immun 79, 4819–4827 (2011).
Lee, J.-H. et al. Diverse plant extracts and trans-resveratrol inhibit biofilm formation and swarming of Escherichia coli O157:H7. Biofouling 29, 1189–1203 (2013).
Cho, H. S. et al. Inhibition of Pseudomonas aeruginosa and Escherichia coli O157:H7 biofilm formation by plant metabolite ε-viniferin. J Agric Food Chem 61, 7120–7126 (2013).
Lee, J.-H. et al. Coumarins reduce biofilm formation and the virulence of Escherichia coli O157:H7. Phytomedicine 21, 1037–1042 (2014).
Niu, C. & Gilbert, E. S. Colorimetric method for identifying plant essential oil components that affect biofilm formation and structure. Appl Environ Microbiol 70, 6951–6956 (2004).
Kerekes, E. B. et al. Anti-biofilm forming and anti-quorum sensing activity of selected essential oils and their main components on food-related micro-organisms. J Appl Microbiol 115, 933–942 (2013).
Yadav, M. K., Chae, S. W., Im, G. J., Chung, J. W. & Song, J. J. Eugenol: a phyto-compound effective against methicillin-resistant and methicillin-sensitive Staphylococcus aureus clinical strain biofilms. PLoS One 10, e0119564 (2015).
He, M., Du, M., Fan, M. & Bian, Z. In vitro activity of eugenol against Candida albicans biofilms. Mycopathologia 163, 137–143 (2007).
Zhou, L., Zheng, H., Tang, Y., Yu, W. & Gong, Q. Eugenol inhibits quorum sensing at sub-inhibitory concentrations. Biotechnol Lett 35, 631–637 (2013).
Jadhav, B. K., Khandelwal, K. R., Ketkar, A. R. & Pisal, S. S. Formulation and evaluation of mucoadhesive tablets containing eugenol for the treatment of periodontal diseases. Drug Dev Ind Pharm 30, 195–203 (2004).
Mastelic, J. et al. Comparative study on the antioxidant and biological activities of carvacrol, thymol, and eugenol derivatives. J Agric Food Chem 56, 3989–3996 (2008).
Sadeghian, H., Seyedi, S. M., Saberi, M. R., Arghiani, Z. & Riazi, M. Design and synthesis of eugenol derivatives, as potent 15-lipoxygenase inhibitors. Bioorg Med Chem 16, 890–901 (2008).
Mellies, J. L., Elliott, S. J., Sperandio, V., Donnenberg, M. S. & Kaper, J. B. The Per regulon of enteropathogenic Escherichia coli: identification of a regulatory cascade and a novel transcriptional activator, the locus of enterocyte effacement (LEE)-encoded regulator (Ler). Mol Microbiol 33, 296–306 (1999).
Saldaña, Z. et al. Synergistic role of curli and cellulose in cell adherence and biofilm formation of attaching and effacing Escherichia coli and identification of Fis as a negative regulator of curli. Environ Microbiol 11, 992–1006 (2009).
Cegelski, L. et al. Small-molecule inhibitors target Escherichia coli amyloid biogenesis and biofilm formation. Nat Chem Biol 5, 913–919 (2009).
Richter, A. M., Povolotsky, T. L., Wieler, L. H. & Hengge, R. Cyclic-di-GMP signalling and biofilm-related properties of the Shiga toxin-producing 2011 German outbreak Escherichia coli O104:H4. EMBO Mol Med 6, 1622–1637 (2014).
Bajpai, V. K., Sharma, A. & Baek, K. H. Antibacterial mode of action of Cudrania tricuspidata fruit essential oil, affecting membrane permeability and surface characteristics of food-borne pathogens. Food Control 32, 582–590 (2013).
Kim, Y.-G. et al. Antibiofilm activity of Streptomyces sp. BFI 230 and Kribbella sp. BFI 1562 against Pseudomonas aeruginosa. Appl Microbiol Biotechnol 96, 1607–1617 (2012).
Heydorn, A. et al. Quantification of biofilm structures by the novel computer program COMSTAT. Microbiology 146, 2395–2407 (2000).
Reisner, A., Krogfelt, K. A., Klein, B. M., Zechner, E. L. & Molin, S. In vitro biofilm formation of commensal and pathogenic Escherichia coli strains: impact of environmental and genetic factors. J Bacteriol 188, 3572–3581 (2006).
Lee, V. T. et al. A cyclic-di-GMP receptor required for bacterial exopolysaccharide production. Mol Microbiol 65, 1474–1484 (2007).
Zodrow, K. R., Schiffman, J. D. & Elimelech, M. Biodegradable polymer (PLGA) coatings featuring cinnamaldehyde and carvacrol mitigate biofilm formation. Langmuir 28, 13993–13999 (2012).
Lee, J.-H. et al. Indole and 7-benzyloxyindole attenuate the virulence of Staphylococcus aureus. Appl Microbiol Biotechnol 97, 4543–4552 (2013).
Murphy, C. T. et al. Genes that act downstream of DAF-16 to influence the lifespan of Caenorhabditis elegans. Nature 424, 277–283 (2003).
Acknowledgements
The 83 essential oils used in this study were kindly provided by Prof. Young-Joon Ahn at Seoul National University. This research was supported by National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIP) (#2015R1A2A2A01004542 to J. Lee) and by the Basic Science Research Program through the NRF funded by the Ministry of Education (#215C000232 to J-H. Lee) and by Priority Research Centers Program through the NRF funded by the Ministry of Education (2014R1A6A1031189).
Author information
Authors and Affiliations
Contributions
J.L. and J.-H.L. conceived the study. Y.-G.K., J.-H.L., G.G. and J.G.P. designed research, performed experiments, analyzed the data, and helped draft the manuscript. S.-I.K. provided a library of essential oils. J.-H.L. and J.L. wrote much of the manuscript. All authors have read and approved the final manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Kim, YG., Lee, JH., Gwon, G. et al. Essential Oils and Eugenols Inhibit Biofilm Formation and the Virulence of Escherichia coli O157:H7. Sci Rep 6, 36377 (2016). https://doi.org/10.1038/srep36377
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep36377
This article is cited by
-
A Review of Challenges and Solutions of Biofilm Formation of Escherichia coli: Conventional and Novel Methods of Prevention and Control
Food and Bioprocess Technology (2024)
-
Repurposing carvacrol, cinnamaldehyde, and eugenol as potential anti-quorum sensing agents against uropathogenic Escherichia coli isolates in Alexandria, Egypt
BMC Microbiology (2023)
-
In-vitro antibacterial activity and mechanism of Monarda didyma essential oils against Carbapenem-resistant Klebsiella pneumoniae
BMC Microbiology (2023)
-
Determination of antibacterial and anti-biofilm potential of Kewda essential oil against Staphylococcus aureus and Klebsiella pneumoniae
Biologia (2023)
-
Activity of Essential Oils Against Multidrug-Resistant Salmonella enteritidis
Current Microbiology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.