Abstract
In tumor microenvironment, macrophages as a polarized M2 population promote tumor progression via releasing multiple cytokines and chemokines. A brown seaweed fucose-rich polysaccharide, fucoidan has antitumor activity and immune modulation through affecting tumor cells and lymphocytes. Here, we focused on the effect of fucoidan on macrophages especially M2 subtype. Our results demonstrated that fucoidan down-regulated partial cytokines and chemokines, especially a M2-type chemokine CCL22. Furthermore, fucoidan inhibited tumor cells migration and CD4+ T lymphocytes, especially Treg cells, recruitment induced by M2 macrophages conditioned medium through suppression of CCL22. Mechanismly, fucoidan inhibited CCL22 via suppressing p65-NF-κB phosphorylation and nuclear translocation. In addition, p38-MAPK and PI3K-AKT also affected the expression of CCL22 through differential modulation of NF-κB transcriptional activity. Taken together, we reveal an interesting result that fucoidan can inhibit tumor cell migration and lymphocytes recruitment by suppressing CCL22 in M2 macrophages via NF-κB-dependent transcription, which may be a novel and promising mechanism for tumor immunotherapy.
Similar content being viewed by others
Introduction
The complex tumor microenvironment plays a critical role in protecting the tumor cells from body repair mechanism and promoting tumor progression, invasion and metastasis, which is comprised of tumor cells, the stroma, blood and lymphatic vessels, and infiltrating immune cells1. Infiltrating immune cells largely response to tumor-derived molecular mechanisms, not only exhibit abortive activation, but are co-opted to promote tumor growth2. Macrophages are the most common cell population in tumor infiltrating immune cells, which account for 30–50%, known as tumor-associated macrophages (TAMs)3.
Macrophages are functionally plastic, and can alter their polarization state to accommodate different physiological conditions. M1 and M2 polarization states are extremes of phenotypic continuum4: “classically-activated” macrophages (M1) produce type I pro-inflammatory cytokines, participate in antigen presentation, and have tumoricidal activity. “alternatively-activated” macrophages (M2) produce type II cytokines, promote anti-inflammatory responses, and have pro-tumorigenic functions5. Under the influence of T and B cells or cancer cell-derived signals in tumor microenvironment, TAMs mainly exhibit an M2-like phenotype6, which promote the formation of blood vessels and lymphatic vessels, enhance tumor cell migration, and tumor proliferation and immune suppression7,8,9. M2 macrophages can be converted into M1, such activation is sufficient on its own to cause tumor rejection10. Therefore, the suitable formulation factors targeting immunosuppressive tumor microenvironment and modulating M2 macrophages repolarization or reprogramming may represent a breakthrough for macrophage-directed cancer immunotherapy11.
Fucoidan is a fucose-containing sulfated polysaccharide extracted from marine organisms, including brown algae species, which have been marketed as a dietary supplement or nutraceutical12. Fucoidan can target multiple receptors, such as scavenger receptors, or signaling molecules in various cell types, including tumor cells and immune cells13,14. Numerous studies have shown that fucoidan has antitumor effect which is mainly manifested as inhibition of tumor cell growth and migration, promoting lymphocyte proliferation and antitumor cytokine secretion15,16,17. Our previous studies have shown that fucoidan induce a functional maturation of human monocyte-derived dendritic cells18 and inhibit monocytes-induced angiogenesis19. However, the effect of fucoidan on the properties of macrophages subtypes is poorly understood.
Macrophages are versatile cells characterized by different functional transcriptional profiles in response to microenvironmental signals. Chemokines belong to a superfamily of small proteins with the role of cell chemoattractant in immune and inflammatory reactions, which has been a new dimension of transcriptional profiling to the characterization of different forms of macrophage activation20. CCL22 (also called macrophage derived chemokine, a CC-chemokine subfamily member) abundantly released by TAMs21, is commonly used as a marker of type M2 macrophages4,22. A recent study showed that M2 macrophages-derived CCL22 directly promote tumor migration capacities and correlate with venous infiltration23. CCL22 selectively recruits CCR4+ T lymphocytes (including: Th2 and regulatory T (Treg) cells) to tumor microenvironment through the chemotaxis of CCL22/CCR4 (the receptor of CCL22)24. Treg trafficking in tumor microenvironment play a critical role in the maintenance of immunosuppression.
The research adopted the THP-1 (human acute monocyte leukemia cell line) derived macrophages. We found that fucoidan significantly inhibited CCL22 expression in M2 macrophages via NF-κB pathway and further regulated tumor cells migration and lymphocytes recruitment which may represent a new mechanism for fucoidan in antitumor activity.
Results
The cytokines transcription of human THP-1-derived macrophages are alerted by fucoidan during polarization process
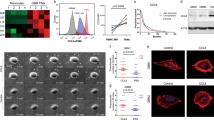
The traditional concepts of M1 and M2 subtypes are seen as two extremes of a continuum of intermediate forms4. The effects of fucoidan were detected during macrophages polarization. Fucoidan was added before THP-1-derived macrophages polarization, which is illustrated in the schematic diagram in Fig. 1a. After 48 h polarization, cells were adherent and had a fusiform/fibroblastic appearance. There were no significant differences of morphologic characteristics between fucoidan treated and untreated M0, M1 or M2 macrophage-like populations (denoted as “M0”, “M1” and “M2”, Fig. 1b). mRNA expression of some cytokines and surface marker of M1 and M2 was performed by quantitative real-time PCR. Our results showed that the mRNA expression of TNF-α, IL-1β and IL-6 described as M1 markers tended to be higher in M1 than in M0 and/or M2-like macrophages. The mRNA expression of TGF-β, MRC-1 and IL-10 described as M2 markers were significantly higher in M2 than in M0 and/or M1 macrophages. Fucoidan significantly affected the cytokines transcription and down-regulate TNF-α, IL-1β, IL-6, TGF-β and IL-10 in M0, M1 and/or M2-like macrophages (Fig. 1c).
Fucoidan modulates specific gene transcription of human THP-1-derived macrophages during the polarization process.
(a) A diagrammatic illustration for the macrophage polarization and fucoidan treatment of THP-1 cells. Under PMA (100 ng/ml) stimulation, THP-1 cells differentiated into macrophage-like cells. LPS (100 ng/ml) and IFN-γ (20 ng/ml) stimulated PMA-activated macrophages into M1-phenotype. IL-4 (20 ng/ml) and IL-13 (20 ng/ml) stimulated PMA-activated macrophages into M2-phenotype. (b) Morphology of macrophages after 48 h polarization with or without fucoidan treatment. The scale bar represents 50 μm. (c) The expression of typical M1/M2 type cytokines and surface marker in non-treated and fucoidan-treated M0/M1/M2-like macrophages were assessed by quantitative real-time PCR and expressed as a fold change compared with M0-like macrophages. M1 biomarkers including TNF-α, IL-1β and IL-6; M2 biomarkers including TGF-β, IL-10 and MRC-1. The values represent means ± SD, n = 3: *P < 0.05, **P < 0.01.
Fucoidan regulates the CC chemokines profiles and particularly down-regulate the CCL22 expression and secretion level in M2 macrophages
Here we analyzed the mRNA expression of six CC-chemokines including CCL2, CCL3, CCL4, CCL5, CCL18 and CCL22, which primarily are expressed in macrophages. As shown in Fig. 2a, CCL2 and CCL22, which were respectively described as M1 and M2 markers, were significantly higher in M1 or M2-like macrophages. Fucoidan down-regulated the mRNA expression of CCL2, CCL4, CCL5 and CCL22 in M0, M1 and/or M2-like macrophages. It is notable the mRNA level of CCL22 was approximate 50-fold decrease in fucoidan-treated THP-1-derived M2 macrophages (Fig. 2a). The secretion level of CCL22 in the supernatant after 48 h polarization was also decreased by fucoidan in M2-like macrophages (Fig. 2b). The down-regulation of CCL22 by fucoidan was further verified in the primary cells isolated from peripheral blood. After M-CSF treatment, monocytes were differentiated into M0 macrophages in vitro. Then stimulated with IL-4 and IL-13, the M0 polarized into M2 subtype. The expression and secretion of CCL22 were rose significantly. Fucoidan notably reduced the expression and extracellular concentrations of CCL22 (Fig. 2c,d). By taking the polarized M2-like macrophages as the research object, the expression of CCL22 was up-regulated by IL-4 and IL-13, significantly. Among them, IL-4 played the major role in the up-regulation of CCL22 expression and secretion. And IL-13 may play a synergic effect (Fig. 2e,f).
Inhibitory effect of fucoidan on CCL22 production in macrophages.
(a) Fucoidan modulates CC chemokines transcription of THP-1-derived macrophages during the polarization process. The expression of selected CC chemokines in non-treated and fucoidan-treated M0/M1/M2-like macrophages were assessed by quantitative real-time PCR and expressed as a fold change compared with M0 macrophages. (b) Fucoidan modulates CCL22 secretion level of THP-1-derived macrophages during the polarization process. After 48 h polarization, the conditioned media created by non-treated and fucoidan-treated M0/M1/M2-like macrophages were collected. ELISA was then used on the various conditioned media to measure the levels of secreted CCL22. (c,d) CCL22 transcription (c) and concentration in culture media (d) of peripheral blood monocyte-derived macrophages. (e,f) CCL22 transcription (e) and concentration (f) in non-treated and fucoidan-treated polarized M2 macrophages exposure to IL-4 and/or IL-13. The quantitative real-time PCR data expressed as relative fold change compared with control (polarized M2 macrophages). The value represent means ± SD, n = 3: *P < 0.05, **P < 0.01.
Fucoidan inhibits the effect of M2 supernatant on tumor cell migration and CD4+ T lymphocytes recruitment via the down-regulation of CCL22
In the above study, we found the strong inhibition of CCL22 by fucoidan in polarizing and polarized M2 macrophages. In order to explore the influence of fucoidan on M2 macrophages functions, the indirect co-culture models based on transwell chambers (Fig. 3a) were established to analyze the tumor cell migration and T lymphocytes recruitment, which were promoted by macrophages-derived CCL22, as described in previous studies.
Fucoidan inhibited the migration of tumor cells and affected T lymphocytes recruitment via suppressing CCL22 production.
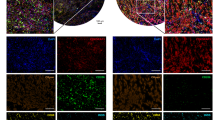
(a) Diagram showing the protocol for MHCC-97 migration assay and the PBMC/CD4+ lymphocytes recruitment assay. Upper: Supernatants of THP-1-derived M2 macrophages under non- or fucoidan pretreatment were collected and add into lower chamber of transwell with or without neutralizing anti-CCL22 antibody or recombine human CCL22, respectively. Down left: MHCC-97H were seeded in upper chamber. Transwelled cells were stained after 24 h and counted. Down right: PBMC (I) from healthy human peripheral blood separated by lymphocyte separation liquid or microbeads selected CD4+ lymphocytes (II) were used as upper chamber components of transwell. Transwelled cells were collected after 6h, and flow cytometry was used to detect the cellular components. FCM: flow cytometry. (b) Migration assays were carried out in MHCC-97H cells by using transwell chamber assay. Migrating cells located on the lower surface were fixed in methanol and stained with eosin. The scale bar represents 50 μm. The graph represents fold change. (c) Flow cytometry settings used to detect human CD3+CD4−/+ cells among PBMC recruited to lower chamber contained non-treated and fucoidan-treated THP-1-derived M2 macrophages supernatants from donor human PBMC seeded in the upper chambers (left). %CD3+CD4+ and %CD3+CD4− cells are the percentage of CD3+CD4+ or CD3+CD4− cells among recruited PBMC (right). (d,e) Flow cytometry settings used to detect human CD25+FoxP3+ (Treg) cells among CD4+ lymphocytes recruited to the bottom chamber filled with conditioned M2 supernatants as tumor cell migration assay. Graphs showed the total numbers of CD4+ lymphocytes (d) and the flow cytometry (left) and percentage (right) of CD25+FoxP3+ cells among recruited CD4+ lymphocytes (e). Control group: the bottom chamber filled with M2 supernatants. The value represent means ± SD, n = 3: *P < 0.05, **P < 0.01 versus the control.
As shown in Fig. 3b, the migration of MHCC-97H cells (Human hepatoma cells) toward M2 supernatants was significantly reduced when the THP-1-derived M2 macrophages were pretreated with fucoidan for 24 h or CCL22 neutralizing Abs were added to the coculture system. Recombinant human CCL22 (rhCCL22) induced the recovery of MHCC-97H migration ability to some extent in fucoidan pretreated THP-1-derived M2 macrophages supernatants.
Then we identified the population of CD3+CD4− and CD3+CD4+ T cells in PBMC recruited by M2 macrophages supernatants using flow cytometry. The CD3+CD4− T cells were more abundant in the cells recruited by fucoidan-preteated-M2 macrophages supernatants. Relatively, the proportion of CD3+CD4+ T lymphocyte was lower under the chemiotaxis by supernatants from fucoidan stimulated THP-1-derived M2 macrophages (Fig. 3c). To explore whether fucoidan pretreatment of M2 macrophages can inhibit CD4+ T lymphocyte migration, we isolated the CD4+ T cells from PBMC by immunomagnetic selection using anti-CD4 microbeads for transwell assay. The fucoidan pretreatment of THP-1-derived M2 macrophages reduced the number of CD4+ T lymphocytes recruited by M2 conditioned medium (Fig. 3d). As CCL22 is a potent chemokine contributing to Treg cells recruitment, we tried to explore whether fucoidan regulated Treg cells migration mediated by M2 macrophages supernatant. Medium from the fucoidan pretreated THP-1-derived M2 macrophages recruited significantly less CD4+CD25+FoxP3+ (Treg) cells than control medium. The addition of CCL22 neutralizing Abs reduced the preferential recruitment of CD4+CD25+FoxP3+ cells by control medium. And rhCCL22 preferentially recruited CD4+CD25+FoxP3+ cells in fucoidan-pretreated THP-1-derived M2 macrophages supernatant (Fig. 3e), which supporting the idea that CCL22 selectively recruit Tregs.
CCL22 down-regulation in THP-1-derived M2 macrophages by fucoidan stimulation via NF-κB pathway
As p38-MAPK, PI3K-AKT and NF-κB signal pathways are affected by fucoidan or engage in expression regulation of CCL22 in other cell types. Phosphorylation of p38-MAPK, AKT and p65-NF-κB were detected to evaluate the mechanism of CCL22 down-regulation by fucoidan in THP-1-derived macrophages. Results from western blot analysis indicated that the phosphorylation of p38-MAPK were up-regulated by fucoidan stimulation in macrophages. At the same time, the phosphorylation of AKT and p65-NF-κB were inhibited (Fig. 4a). Furthermore, we confirmed the inhibition of NF-κB activity by immunofluorescence microscopy assay. As shown in Fig. 4b, treatment of fucoidan induced faint immunostaining of p65-NF-κB translocated into the nucleus. Then we used the inhibitors of PI3K, p38-MAPK and NF-κB to analyze the effect on CCL22 expression and secretion. However, we accidentally found that Wortmannin (PI3K-AKT pathway inhibitor) can significantly increase the expression and secretion of CCL22, p38-MAPK pathway inhibitor SB203580 and NF-κB pathway inhibitor BAY 11-7082 can significantly down-regulate the expression and secretion of CCL22 (Fig. 5a,b). In western blot analysis, Wortmannin which inhibited the PI3K-AKT pathway, promoted the phosphorylation of p65-NF-κB and p38-MAPK. SB203580 which inhibited the p38-MAPK pathway, also inhibited the phosphorylation of p65-NF-κB and AKT at the same time (Fig. 5c). Similar results of NF-κB activation were observed by immunofluorescence (Fig. 5d).
Effect of Fucoidan on the signaling pathways and NF-κB nuclear translocation in THP-1-derived M0 and M2 polarized macrophages.
(a) M0 and M2 macrophages derived from THP-1 cells were treated with PBS or fucoidan (100 μg/ml) for 1 h. Whole-cell lysates were analyzed by western blot using the appropriate antibodies. The original blot is presented in Supplementary Fig. S1. Densitometry ratios of phospho-p65-NF-κB, AKT and p38 were normalized to the total p65-NF-κB, AKT and p38. The value represent means ± SD, n = 3; *P < 0.05, **P < 0.01 versus the PBS treated group, respectively. (b) p65-NF-κB immunofluorescence of THP-1-derived M0 and M2 polarized macrophages, which were treated as described in (a). Green (anti-p65-NF-κB) indicates p65-NF-κB distribution, and blue indicates the location of nucleus. The scale bar represents 10 μm.
Suppression of CCL22 expression by fucoidan is mediated by a p38- and PI3K-AKT- independent inhibition of the NF-κB pathway.
(a,b) THP-1-derived M2 macrophages were treated with the NF-κB inhibitor BAY11-7082, the PI3K inhibitor Wortmannin or the p38-MAPK inhibitor SB203580 for 24 h. The cells and culture supernatants were collected. CCL22 transcription (a) and concentration (b) were measured by quantitative real-time PCR and ELISA, respectively. (c) M2 macrophages derived from THP-1 cells were pretreated with the Wortmannin or the SB203580 for 30 min, followed by incubation with or without Fucoidan for 1 h. Whole-cell lysates were analyzed by western blot using the appropriate antibodies. The original blots are presented in Supplementary Fig. S2. Densitometry ratios of phospho-p65-NF-κB, AKT and p38-MAPK were normalized to β-actin levels. The cells treated with DMSO were used as control. The value represent means ± SD, n = 3. *P < 0.05, **P < 0.01 versus the control. (d) p65-NF-κB immunofluorescence of THP-1-derived M0 and M2 polarized macrophages, which were treated as described in (c). Green (anti-p65-NF-κB) indicates p65-NF-κB distribution, and blue indicates the location of nucleus. The scale bar represents 10 μm.
Discussion
Specific cytokines and chemokines produced by TAMs maintain a suitable microenvironment for tumor growth and progression21. Recently, the strategies including blocking macrophages pro-tumor polarization and effector function have all been used successfully in preclinical tumor models25. The reprogramming of cytokines and chemokines will result in a progression of functional changes of M1 and M2 macrophages26. In this study, we found that fucoidan inhibited partial cytokines and chemokines belonging to either M1 or M2 phenotypes, which suggested that the macrophage repolarization induced by fucoidan was not significant. The down-regulation of cytokines and chemokines probably restrain the effector function of macrophages and maybe even lead to modify the tumor microenvironment. As a chemotactic factor, CCL22 was mainly generated by M2 macrophages27, which induced by Th2 cytokines IL-4 and IL-1328. We discovered that the transcription of CCL22 was markedly inhibited in fucoidan-treated M2 macrophages during and after polarization, which lead to the low secretion level of CCL22.
CCL22 is highly expressed in a variety of malignant tumors including ovarian tumor, colorectal tumor, breast tumor, hepatocellular carcinoma and follicular lymphoma, and is related to poor prognosis29,30,31,32,33. It has been recently shown that CCL22 directly induce tumor cells migration34. The secretion of CCL22 by macrophages has a chemotaxis effect on Treg cells trafficking, which are suppressive and able to block tumor-specific immunity35. We discovered that the tumor cells and Treg cells migration were all inhibited in the conditioned medium from the fucoidan-pretreated M2 macrophages due to the lower concentration of CCL22. These result suggest that the antitumor effect of fucoidan may be potentially via inhibiting CCL22 production by macrophages in tumor microenvironment, which need further in vivo investigation.
We next investigated the molecular mechanism of fucoidan-induced CCL22 down-regulation. Previous studies have shown that CCL22 production is dependent on p38-MAPK and NF-κB in various cell types36,37,38. P38-MAPK, PI3K-AKT and NF-κB pathways are influenced by fucoidan39,40,41. In macrophages, p38-MAPK signaling pathway enhances the NF-κB transactivation42. PI3K-AKT inactivates the glycogen synthase kinase-β (GSK-β) by inducing its phosphorylation, then inhibit the transactivational activity of NF-κB43. Surprisingly, we found that fucoidan significantly up-regulated the phosphorylation of p38-MAPK, inhibited the phosphorylation of AKT and the activity of p65-NF-κB in THP-1-derived macrophages. Further experiments using the signal pathway inhibitors showed the contrary effects of p38-MAPK and PI3K-AKT pathway on NF-κB activation, which induced or inhibited the expression and secretion of CCL22. Analysis of the promoter region of CCL22 showed that the NF-κB binding site in the CCL22 promoter region is essential for the induction of CCL22 expression37. Therefore, although fucoidan activated p38-MAPK and inhibited the PI3K-AKT pathway, the production of CCL22 was finally inhibited via the suppression of NF-κB activity (Fig. 6). NF-κB plays an essential role in modulating antitumor immunity44. NF-κB activation accounts for increased transcription of several hundred genes. A reduction of myeloid NF-κB activity is associated with tumor surveillance and a reduction in tumor growth, which is likely due to decreased expression of several genes, whose products enhance tumor development, rather than a single one45. So we speculate the antitumor activity of fucoidan targeting macrophages may depend primarily on the suppression of NF-κB activity.
Based on the chemotherapy-induced immune suppression, the combination of immunotherapy, which mainly depend on the action of T cells and natural killer (NK) cells, and chemotherapy would be not advantageous for improving therapeutic effect. Macrophages might be more resistant to chemotherapy than other immune cells, and TAMs even promote tumor chemoresistance46. Immunotherapy target the activation of macrophages might therefore be synergistic with chemotherapy47. Thus, as a marine natural drug, fucoidan could be used as a promising antitumor agent targeting macrophages for tumor immunotherapies.
Materials and Methods
Abs and reagents
Recombinant human CCL22, M-CSF, IFN-γ, IL-4 and IL-13 were purchased from R&D Systems (Minneapolis, MN, USA). Fucoidan (from Fucus vesiculosus), PMA, LPS, Wortmannin, SB203580 and BAY 11-7082 were purchased from Sigma-Aldrich (St Louis, MO, USA). Monoclonal antibody to CCL22 and the isotype control were purchased from R&D Systems.
Cell culture
The human monocytes cell line THP-1 was purchased from American Type Culture Collection (ATCC, Rockville, MD, USA). Human hepatoma cell line MHCC-97H was purchased from Liver Cancer Institute of Fudan University (Shanghai, China). Cells were grown in RPMI 1640 medium (HyClone, Logan, UT, USA) containing 10% fetal bovine serum (FBS) (Gibco, Grand Island, NY, USA), 100 U/ml penicillin and 100 μg/ml streptomycin (Invitrogen, Carlsbad, CA, USA) at 37 °C and 5% CO2 in a humidified incubator.
Differentiation and polarization of THP-1 and monocyte-derived macrophages
THP-1 cells were differentiated into M0 by PMA (100 ng/ml) treatment for 48 h in RPMI 1640 medium supplemented with 10% FBS. For M1 subtype polarization experiments, after 6 h PMA treatment, 100 ng/ml LPS and 20 ng/ml IFN-γ were add for another 42 h. For M2 subtype polarization experiments, after 6 h PMA treatment, 20 ng/ml IL-4 and IL-13 were add for another 42 h48. Monocytes from PBMC were differentiated into unpolarized macrophages by M-CSF (50 ng/ml) stimulation for 6 days in RPMI 1640 medium with 10% FBS, and then polarized to M1 with LPS and IFN-γ or M2 with IL-4 and IL-13 for another 2 days.
RNA extraction, reverse transcriptase and quantitative Real-time polymerase chain reaction
Total RNA was extracted by Trizol reagent (Sigma-Aldrich) and reverse transcribed to cDNA, as described previously49. Quantitative real-time PCR was performed using SYBR Green real-time PCR Master Mix (Toyobo, Osaka, Japan) with 1 μg cDNA as the template and the primers (shown in Supplementary Table S1) on a LightCycler® 480 qPCR machine (Roche Diagnostics, Penzberg, Germany). Gene expression data were analyzed with LightCycler® software vision 4.0.
Enzyme-linked immunosorbent assay
The CCL22 concentration in the culture supernatant was measured by ELISA kit (Catalog No: DMD00, R&D Systems) and followed the protocol by the manufacturer’s introduction.
Preparation and isolation of PBMC and cell subtypes
Human peripheral blood mononuclear cells (PBMCs) were isolated from leukocyte-enriched buffy coats of healthy donors by Ficoll (Sigma-Aldrich) density gradient centrifugation. The collection and use of blood complied with relevant guidelines and institutional practices from the Ethics Committees of Qilu Hospital of Shandong University. Written informed consent was obtained from all subjects. Our study was specially approved by Ethics Committees of Qilu Hospital of Shandong University (Ethical approval No. KYLL-2013-069). For monocytes isolation, monocytes were isolated by magnetic separation using the Miltenyi Monocyte Isolation kit (Miltenyi Biotec, Bergisch-Gladbach, Germany) according to the manufacturer’s instructions, and cultured in 6-well plates at a density of 2 × 106 cells/well in RPMI 1640 medium supplemented with 10% FBS. CD4+ T lymphocytes were purified from PBMC by CD4 Microbeads (Miltenyi Biotec), as described by the manufacturer. More than 98% of the isolated cells were positive for CD3 and CD4, as measured by flow cytometry using APC- or FITC-conjugated Abs against CD3 (OKT3) and CD4 (OKT4) (Biolegend, San Diego, CA, USA).
In vitro migration assay
Supernatants from THP-1 derived M2 macrophages with or without fucoidan pretreatment (24 h), recombination human CCL22 (100 ng/ml) or blocking mAbs (1 μg/ml) were plated into the bottom of migration chambers of 24-well plate. For tumor cell migration assay, 5 × 104 MHCC-97H cells in 200 μl RPMI 1640 medium containing 0.1% FBS were added to the top of 8 μm-pore Costar Transwell chambers (Corning Life Sciences, Corning, NY, USA). After incubated at 37 °C for 24 h in an atmosphere containing 5% CO2, the cells on the top side of filter were wiped off with cotton swab, and migrating cells located on the lower surface were fixed in methanol and stained with eosin. For lymphocyte migration assay, 4 × 106 fresh human PBMCs or 2 × 106 isolated CD4+ T cells in 200 μl RPMI 1640 medium containing 0.1% FBS were seeded in the top of the 5 μm-pore Costar Transwell chambers (Corning Life Sciences). Following 6 h incubation at 37 °C, cells migrating to the lower chambers were collected and used for flow cytometry.
Flow cytometry
Migrated cells were collected and stained with a mixed solution of FITC-conjugated mAbs against CD4, PerCP-conjugated mAbs against CD25 (M-A251) and APC-conjugated mAbs against CD3 (Biolegend) for 20 min. Then cells were pretreated with FoxP3 staining buffer set (Catalog No: 00-5523-00, eBioscience, San Diego, CA, USA) according to the manufacturer’s instructions and stained with PE-conjugated mAbs against Foxp3 (206D, Biolegend). Cells were analyzed with a FACS Calibur flow cytometer (Becton-Dickinson (BD), San Jose, CA, USA).
Western blot analysis
Cells in six-well plates were washed with PBS and lysed with equal volumes of RIPA buffer containing 1 mM PMSF on ice and then centrifuged for 10 min at 12000 rpm under 4 °C. Lysates with equal amounts of protein were separated by 10% SDS-PAGE and then transferred onto PVDF membrane (Millipore, Bedford, MA, USA). The membranes were blocked for 1 h at room temperature with 5% BSA in TBS containing 0.1% Tween20 and then incubated overnight at 4 °C with the following primary antibodies: phospho-Ser536-NF-κB p65, NF-κB p65, phospho-p38-MAPK, MAPK, phospho-AKT, AKT (Cell Signaling Technology, Danvers, Mass, USA); β-actin (Santa Cruz Biotechnology, Santa Cruz, CA, USA). The membranes were exposed to horseradish peroxidase-labeled secondary antibodies (1:3000) for 1 h at room temperature and detected by enhanced chemiluminescence detection system (Amersham Imager 600, GE Healthcare Life Sciences, Little Chalfont, UK and ChemiDoc™ Touch Imaging System, Bio-Rad, Hercules, CA).
Immunofluorescence staining
For immunofluorescence analysis of nuclear translocation of NF-κB p65, macrophages were fixed in 4% paraformaldehyde for 30 min and incubated with anti-p65 NF-κB antibody (1:400, Catalog No: 8242, Cell Signaling Technology) overnight at 4 °C. The next day, cells were washed three times in PBS, and then incubated with anti-rabbit Alexa Fluor 488-conjugated secondary antibody for 1 h at room temperature. Nuclear was stained by DAPI for 5 min at room temperature. The location of NF-κB was detected by immunofluorescence microscopy (Olympus IX81).
Statistics analysis
The software SPSS 19.0 was used for statistical analysis. All data were presented as means ± SD. Two-tailed Student’s t-test or one-way ANOVA analysis was used to determine significance. P < 0.05 was considered statistically significant.
Additional Information
How to cite this article: Sun, J. et al. Fucoidan inhibits CCL22 production through NF-κB pathway in M2 macrophages: a potential therapeutic strategy for cancer. Sci. Rep. 6, 35855; doi: 10.1038/srep35855 (2016).
References
Quail, D. F. & Joyce, J. A. Microenvironmental regulation of tumor progression and metastasis. Nature medicine. 19, 1423–1437 (2013).
Whiteside, T. L. The tumor microenvironment and its role in promoting tumor growth. Oncogene. 27, 5904–5912 (2008).
Balkwill, F., Charles, K. A. & Mantovani, A. Smoldering and polarized inflammation in the initiation and promotion of malignant disease. Cancer cell. 7, 211–217 (2005).
Mantovani, A., Sozzani, S., Locati, M., Allavena, P. & Sica, A. Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 23, 549–555 (2002).
Biswas, S. K. & Mantovani, A. Macrophage plasticity and interaction with lymphocyte subsets: cancer as a paradigm. Nature immunology. 11, 889–896 (2010).
Mantovani, A. & Sica, A. Macrophages, innate immunity and cancer: balance, tolerance, and diversity. Curr Opin Immunol. 22, 231–237 (2010).
Han, Q., Shi, H. & Liu, F. CD163 M2-type tumor-associated macrophage support the suppression of tumor-infiltrating T cells in osteosarcoma. International immunopharmacology. 34, 101–106 (2016).
Kimura, Y., Sumiyoshi, M. & Baba, K. Antitumor and Antimetastatic Activity of Synthetic Hydroxystilbenes Through Inhibition of Lymphangiogenesis and M2 Macrophage Differentiation of Tumor-associated Macrophages. Anticancer research. 36, 137–148 (2016).
Jeong, S. K. et al. Tumor associated macrophages provide the survival resistance of tumor cells to hypoxic microenvironmental condition through IL-6 receptor-mediated signals. Immunobiology. 10.1016/j.imbio.2015.11.010 (2015).
O’Sullivan, T. et al. Cancer immunoediting by the innate immune system in the absence of adaptive immunity. The Journal of experimental medicine. 209, 1869–1882 (2012).
Mills, C. D., Lenz, L. L. & Harris, R. A. A Breakthrough: Macrophage-Directed Cancer Immunotherapy. Cancer research. 76, 513–516 (2016).
Atashrazm, F., Lowenthal, R. M., Woods, G. M., Holloway, A. F. & Dickinson, J. L. Fucoidan and cancer: a multifunctional molecule with anti-tumor potential. Marine drugs. 13, 2327–2346 (2015).
Kwak, J. Y. Fucoidan as a marine anticancer agent in preclinical development. Marine drugs. 12, 851–870 (2014).
Ben, J. et al. Major vault protein regulates class A scavenger receptor-mediated tumor necrosis factor-alpha synthesis and apoptosis in macrophages. The Journal of biological chemistry. 288, 20076–20084 (2013).
Chen, M. C., Hsu, W. L., Hwang, P. A. & Chou, T. C. Low Molecular Weight Fucoidan Inhibits Tumor Angiogenesis through Downregulation of HIF-1/VEGF Signaling under Hypoxia. Marine drugs. 13, 4436–4451 (2015).
Shimizu, J. et al. Proportion of murine cytotoxic T cells is increased by high molecular-weight fucoidan extracted from Okinawa mozuku (Cladosiphon okamuranus). Journal of health science. 51, 394–397 (2005).
Takai, M., Miyazaki, Y., Tachibana, H. & Yamada, K. The enhancing effect of fucoidan derived from Undaria pinnatifida on immunoglobulin production by mouse spleen lymphocytes. Bioscience, biotechnology, and biochemistry. 78, 1743–1747 (2014).
Yang, M. et al. Fucoidan stimulation induces a functional maturation of human monocyte-derived dendritic cells. International immunopharmacology. 8, 1754–1760 (2008).
Lv, Y. et al. Comparison of the effects of marchantin C and fucoidan on sFlt-1 and angiogenesis in glioma microenvironment. The Journal of pharmacy and pharmacology. 64, 604–609 (2012).
Mantovani, A. et al. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol. 25, 677–686 (2004).
Solinas, G., Germano, G., Mantovani, A. & Allavena, P. Tumor-associated macrophages (TAM) as major players of the cancer-related inflammation. Journal of leukocyte biology. 86, 1065–1073 (2009).
Jaguin, M., Houlbert, N., Fardel, O. & Lecureur, V. Polarization profiles of human M-CSF-generated macrophages and comparison of M1-markers in classically activated macrophages from GM-CSF and M-CSF origin. Cell Immunol. 281, 51–61 (2013).
Yeung, O. W. et al. Alternatively activated (M2) macrophages promote tumour growth and invasiveness in hepatocellular carcinoma. Journal of hepatology. 62, 607–616 (2015).
Gobert, M. et al. Regulatory T cells recruited through CCL22/CCR4 are selectively activated in lymphoid infiltrates surrounding primary breast tumors and lead to an adverse clinical outcome. Cancer research. 69, 2000–2009 (2009).
Ruffell, B. & Coussens, L. M. Macrophages and therapeutic resistance in cancer. Cancer cell. 27, 462–472 (2015).
Stout, R. D., Watkins, S. K. & Suttles, J. Functional plasticity of macrophages: in situ reprogramming of tumor-associated macrophages. Journal of leukocyte biology. 86, 1105–1109 (2009).
Godiska, R. et al. Human macrophage-derived chemokine (MDC), a novel chemoattractant for monocytes, monocyte-derived dendritic cells, and natural killer cells. The Journal of experimental medicine. 185, 1595–1604 (1997).
Andrew, D. P. et al. STCP-1 (MDC) CC chemokine acts specifically on chronically activated Th2 lymphocytes and is produced by monocytes on stimulation with Th2 cytokines IL-4 and IL-13. J Immunol. 161, 5027–5038 (1998).
Anz, D. et al. Suppression of intratumoral CCL22 by type i interferon inhibits migration of regulatory T cells and blocks cancer progression. Cancer research. 75, 4483–4493 (2015).
Wertel, I. et al. Macrophage-derived chemokine CCL22 and regulatory T cells in ovarian cancer patients. Tumour biology : the journal of the International Society for Oncodevelopmental Biology and Medicine. 36, 4811–4817 (2015).
Li, Y. Q. et al. Tumor secretion of CCL22 activates intratumoral Treg infiltration and is independent prognostic predictor of breast cancer. PloS one. 8, e76379 (2013).
Yang, P. et al. TGF-beta-miR-34a-CCL22 signaling-induced Treg cell recruitment promotes venous metastases of HBV-positive hepatocellular carcinoma. Cancer cell. 22, 291–303 (2012).
Rawal, S. et al. Cross talk between follicular Th cells and tumor cells in human follicular lymphoma promotes immune evasion in the tumor microenvironment. J Immunol. 190, 6681–6693 (2013).
Cao, L., Hu, X., Zhang, J., Huang, G. & Zhang, Y. The role of the CCL22-CCR4 axis in the metastasis of gastric cancer cells into omental milky spots. J Transl Med. 12, 267 (2014).
Curiel, T. J. et al. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nature medicine. 10, 942–949 (2004).
Yano, C. et al. Mechanism of Macrophage-Derived Chemokine/CCL22 Production by HaCaT Keratinocytes. Ann Dermatol. 27, 152–156 (2015).
Nakayama, T. et al. Selective induction of Th2-attracting chemokines CCL17 and CCL22 in human B cells by latent membrane protein 1 of Epstein-Barr virus. Journal of virology. 78, 1665–1674 (2004).
Shoji, N., Asano, K., Furuta, A., Hirano, K. & Suzaki, H. Effect of histamine H1 receptor antagonists on TARC/CCL17 and MDC/CCL22 production from CD14+ cells induced by antigenic stimulation in vitro. International archives of allergy and immunology. 155, 38–51 (2011).
Lee, H., Kim, J. S. & Kim, E. Fucoidan from seaweed Fucus vesiculosus inhibits migration and invasion of human lung cancer cell via PI3K-Akt-mTOR pathways. PloS one. 7, e50624 (2012).
Nakamura, T., Suzuki, H., Wada, Y., Kodama, T. & Doi, T. Fucoidan induces nitric oxide production via p38 mitogen-activated protein kinase and NF-kappaB-dependent signaling pathways through macrophage scavenger receptors. Biochemical and biophysical research communications. 343, 286–294 (2006).
Sapharikas, E., Lokajczyk, A., Fischer, A. M. & Boisson-Vidal, C. Fucoidan Stimulates Monocyte Migration via ERK/p38 Signaling Pathways and MMP9 Secretion. Marine drugs. 13, 4156–4170 (2015).
Sun, J., Ramnath, R. D., Zhi, L., Tamizhselvi, R. & Bhatia, M. Substance P enhances NF-kappaB transactivation and chemokine response in murine macrophages via ERK1/2 and p38 MAPK signaling pathways. American journal of physiology. Cell physiology 294, C1586–C1596 (2008).
Guha, M. & Mackman, N. The phosphatidylinositol 3-kinase-Akt pathway limits lipopolysaccharide activation of signaling pathways and expression of inflammatory mediators in human monocytic cells. The Journal of biological chemistry. 277, 32124–32132 (2002).
Richmond, A. & Yang, J. The role of NF-kB in modulating antitumor immunity. Oncoimmunology. 5, e1005522 (2016).
Greten, F. R. et al. IKKbeta links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell. 118, 285–296 (2004).
De Palma, M. & Lewis, C. E. Cancer: Macrophages limit chemotherapy. Nature. 472, 303–304 (2011).
Buhtoiarov, I. N. et al. Anti-tumour synergy of cytotoxic chemotherapy and anti-CD40 plus CpG-ODN immunotherapy through repolarization of tumour-associated macrophages. Immunology. 132, 226–239 (2011).
Zhao, P. et al. Response gene to complement 32 (RGC-32) expression on M2-polarized and tumor-associated macrophages is M-CSF-dependent and enhanced by tumor-derived IL-4. Cell Mol Immunol. 12, 692–699 (2015).
Sun, J. et al. Hypoxia induces T-cell apoptosis by inhibiting chemokine C receptor 7 expression: the role of adenosine receptor A(2). Cell Mol Immunol. 7, 77–82 (2010).
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (No. 31470885, No. 31300752, No. 31270971, No. 81300510, No. 81502615).
Author information
Authors and Affiliations
Contributions
J.S., J.T.S. and X.Q. conceived and designed the experiments. J.S., J.T.S. and L.Z. preformed the experiments. B.F.S. collected samples. Q.S. and Y.L. acquared and analyzed the data. J.S. and J.T.S. wrote the manuscript text and prepared Figures. J.S., J.T.S., D.Y., Y.Z. and X.Q. contributed to the discussion and reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Sun, J., Sun, J., Song, B. et al. Fucoidan inhibits CCL22 production through NF-κB pathway in M2 macrophages: a potential therapeutic strategy for cancer. Sci Rep 6, 35855 (2016). https://doi.org/10.1038/srep35855
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep35855
This article is cited by
-
Immune modulation in malignant pleural effusion: from microenvironment to therapeutic implications
Cancer Cell International (2024)
-
Downstream STING pathways IRF3 and NF-κB differentially regulate CCL22 in response to cytosolic dsDNA
Cancer Gene Therapy (2024)
-
Anticancer potential of algae-derived metabolites: recent updates and breakthroughs
Future Journal of Pharmaceutical Sciences (2023)
-
Oligo-Fucoidan supplementation enhances the effect of Olaparib on preventing metastasis and recurrence of triple-negative breast cancer in mice
Journal of Biomedical Science (2022)
-
Tumor cell-derived asymmetric dimethylarginine regulates macrophage functions and polarization
Cancer Cell International (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.