Abstract
Field studies of wild vertebrates are frequently associated with extensive collections of banked fecal samples—unique resources for understanding ecological, behavioral and phylogenetic effects on the gut microbiome. However, we do not understand whether sample storage methods confound the ability to investigate interindividual variation in gut microbiome profiles. Here, we extend previous work on storage methods for gut microbiome samples by comparing immediate freezing, the gold standard of preservation, to three methods commonly used in vertebrate field studies: lyophilization, storage in ethanol and storage in RNAlater. We found that the signature of individual identity consistently outweighed storage effects: alpha diversity and beta diversity measures were significantly correlated across methods and while samples often clustered by donor, they never clustered by storage method. Provided that all analyzed samples are stored the same way, banked fecal samples therefore appear highly suitable for investigating variation in gut microbiota. Our results open the door to a much-expanded perspective on variation in the gut microbiome across species and ecological contexts.
Similar content being viewed by others
Introduction
Noninvasive collection is often the only feasible approach for obtaining samples from wild vertebrates, especially in threatened or endangered species1. Fecal samples are especially common, as they can be collected without disrupting study subjects, can often be unambiguously assigned to donors and can be longitudinally collected from the same animal over time. Such samples also contain abundant information about the genetics, endocrinology and parasite burden of the animals from which they are obtained. For these reasons, fecal samples may be the most extensively banked sample type available for wild vertebrates.
Such collections represent potentially invaluable resources for understanding interindividual, temporal and spatial variation in the gut microbiome in comparative or conservation contexts. However, sample storage methods vary widely across studies and in most cases, samples were not collected with microbiome analyses in mind. To assess the potential for mining existing sample banks, we investigated how three common field storage methods—storage in ethanol, storage in RNAlater, or storage in ethanol followed by lyophilization (typical for studies of steroid hormone levels)—affect gut microbiome diversity and composition estimates, compared to the gold standard of immediate freezing. We were particularly interested in comparing the roles of storage method versus individual identity, as the latter captures the biological variation generally of most interest to researchers (whereas storage-driven variance is purely technical). Although storage methods often explain substantial variation in microbiome composition when all other sources of variance are controlled2,3, the degree to which they confound other analyses depends on their importance relative to the effects of biologically interesting variation (interindividual, temporal and environmental). With one recent exception focused on humans and domestic dogs4, previous studies have focused on small numbers of study subjects (n ≤ 5), limiting their ability to evaluate this question2,3,5,6,7,8,9,10.
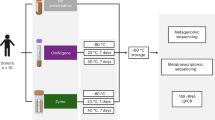
Here, we compared fecal samples collected from 13 captive adult rhesus macaques (Macaca mulatta), with each sample divided into four aliquots (one per storage condition we studied). We then evaluated individual identity versus technical storage effects on both alpha diversity and beta diversity estimates from high sequencing-depth 16S rRNA profiles. Notably, this study design provides a conservative estimate of the contribution of individual identity relative to technical variance, as all animals were sampled in standardized housing, dietary and social group conditions.
Results
For each individual in the study, we collected a fresh stool sample and divided it into four aliquots (n = 52 samples; Table S1). These were stored for 2–6 days prior to DNA extraction via: 1) immediate freezing at −20 °C; 2) immersion in absolute ethanol; 3) immersion in the preservative RNAlater; or 4) immersion in ethanol followed by lyophilization to powder (used for steroid hormone analysis and sometimes for genetic samples11,12; we subsequently refer to this condition simply as “lyophilized”). For each sample, we then generated amplicon libraries targeting the bacterial 16S rRNA V4 region13, multiplexed them for sequencing and performed quality filtering and OTU abundance estimation in QIIME14 (see Methods). We eliminated one ethanol sample because it generated very few reads; all remaining samples were rarefied to 54,633 reads for subsequent analyses. We identified 21,006 OTUs overall (mean per sample = 1,656 ± 237 s.d.; Table S1).
The resulting data recapitulated previous observations showing storage condition effects on mean alpha diversity2,3,5,7. In our case, lyophilized samples exhibited lower Shannon’s Diversity Index (SDI) values relative to other conditions (Tukey’s HSD: p between 7.6 × 10−5 and 0.063) and samples stored in RNAlater exhibited somewhat higher values, although this comparison was only significant in comparison to the lyophilized condition (p = 7.6 × 10−5; Fig. 1a; Table S2). Among the OTUs that were present at an abundance ≥0.1% in any sample (n = 696 OTUs), none were identified as present in one condition but absent in the other three storage conditions and only 11—primarily of the order Clostridiales—were absent in one storage condition but present in the others (always missing in the lyophilized condition: Table S3). Of these 11 OTUs, 8 were very low abundance (<0.05% relative abundance) in all but one female.
Storage effects on alpha diversity.
(a) Shannon’s Diversity Index (SDI) values (y-axis) shown as a function of storage method (x-axis), with each individual plotted in a different color. Lyophilized samples have lower SDI values than other storage methods (Tukey’s HSD, lyophilized-frozen: p = 0.004; lyophilized-RNAlater: p = 7.6 × 10−5; lyophilized-ethanol: p = 0.063; Table S2). (b) SDI values are significantly correlated within individuals, between all storage methods (Pearson’s correlation, p < 0.05). Each dot represents an individual and each panel shows the correlation between SDI values obtained from two different storage methods.
Overall, SDI values retained a strong signature of individual identity. Specifically, SDI values were significantly correlated across samples from the same individual across storage methods (Fig. 1b). Further, although we observed several changes in the rank order of SDI values by individual, across conditions (Fig. 1a), individual identity explained a larger proportion of variance in SDI across samples than storage condition (ANOVA: 50% versus 36%). We obtained qualitatively similar but weaker results for the number of OTUs identified in each sample (Figure S1), suggesting that the combination of species richness and evenness captured by SDI is more stable than richness alone.
Beta diversity measures of community similarity (Tables S4–S6) were also more influenced by individual identity than sample storage condition. 66.3% of variation in taxonomic abundance could be explained by individual identity, compared to 14.3% by storage method (PERMANOVA on a Bray-Curtis dissimilarity matrix; p < 0.001 for both predictors; similar results were obtained for weighted UniFrac: 53.1% versus 26.2%; and for unweighted UniFrac: 43.2% versus 8.2%). Bray-Curtis dissimilarities were much higher for pairs of samples collected from different individuals (mean = 0.51 ± 0.11 s.d. within condition; 0.56 ± 0.11 s.d. between conditions) than for samples collected from the same individual using different storage conditions (mean = 0.35 ± 0.11 s.d., Fig. 2a; see also Figure S2). Samples from the same individual, but not storage condition, also clustered together in a hierarchical clustering analysis using either Bray-Curtis or unweighted UniFrac measures (Fig. 2b; Figure S3a; no clustering, either by individual or storage condition, was observable using weighted UniFrac: Figure S3b). Most importantly, relative distances between individuals remained consistent across storage conditions. For example, pairwise correlations between Bray-Curtis dissimilarity matrices calculated separately for each condition were highly correlated (r = 0.59 to 0.88, all p < 0.005; Table S7), with similar patterns observed using weighted or unweighted UniFrac (Tables S8 and S9).
Storage effects on beta diversity.
(a) Bray-Curtis dissimilarity values (y-axis) comparing the same individual from samples collected under different storage conditions (red), different individuals with samples collected under the same storage conditions (blue) and different individuals with samples collected under different storage conditions (green). Median Bray-Curtis dissimilarity calculated from subsampling reads from the same sample (i.e., the minimum dissimilarity due to read resampling alone) is indicated by the gray dashed line. Because of the large number of data points, all pairwise comparisons are highly significant (Wilcoxon Rank Sum test, p < 1 × 10−9). However, the dissimilarity values for same individual/different storage are much lower on average (mean = 0.35 ± 0.11 s.d.) than dissimilarity values measured between individuals in either the same (mean = 0.51 ± 0.11 s.d.) or different (0.56 ± 0.11 s.d.) storage conditions. (b) Bray-Curtis dissimilarities cluster more strongly by individual (colors along the left-hand sidebar, with one color per individual) than by storage method (colors shown on the top, next to the dendrogram and in the boxed legend).
Discussion
Together, our results indicate that, while mean alpha and beta diversity values are sometimes altered by storage condition, biologically relevant signatures of individual identity tend to be retained, especially for measures of beta diversity. Our findings agree with previous studies using fewer individuals6,7,8,10 and extend them to three of the most commonly used storage methods in vertebrate field studies. For many types of studies, storage condition per se may therefore be less important than maintaining consistency in storage methods within a data set (suggesting caution when performing comparisons across data sets that use different methods, unless the variable of interest has a considerably larger effect size than storage condition). Notably, in natural populations, interindividual differences in diet, social contacts and habitat will likely amplify the signature of individual identity even further.
Thus, while immediate freezing will always be preferred when possible, when it is not possible—as is often the case in field studies—other methods provide reasonable alternatives. Although the differences are small, alpha diversity tended to be more highly correlated between frozen and ethanol-stored samples than frozen and RNAlater-stored samples, suggesting that ethanol may be as good or better of a storage medium than more expensive options (consistent with previous work3,4,15,16).
Together, our findings support the utility of using banked fecal sample collections from field studies for analyses of gut microbiome variation. These collections are not only substantial (ranging up to tens of thousands of samples), but are also often longitudinal, complemented by extensive demographic and behavioral metadata and focused on species of particular conservation concern. As a note of caution, our study examined only short-term storage, so the long-term effects of storage in alternative media remain important to examine (but see4, where storage up to 8 weeks produced minimal sample preservation effects). However, given appropriate controls for sample age and DNA quality, our results suggest that sample banks from field studies may represent extraordinary, largely untapped resources for understanding the causes, consequences and diversity of gut microbial structure.
Methods
Study subjects, sample collection and sequencing
Study subjects were 13 adult female rhesus macaques (Macaca mulatta), members of 7 different social groups housed at the Yerkes National Primate Research Center (YNPRC). These groups were formed as part of a separate study on the relationship between dominance rank and gene regulation. All groups were maintained in standardized indoor-outdoor housing runs (25 m × 25 m per run), under standardized demographic (5 adult females per group), dietary and observational conditions.
Fecal samples were collected within 10–15 minutes after deposition and subdivided into four equal subsamples. The first subsample was frozen immediately at −20 °C; the second subsample immersed in the commercial preservative RNAlater (Life Technologies, Carlsbad, CA); and the third and fourth subsamples immersed in absolute ethanol. After one day of room temperature storage, one of the ethanol-stored subsamples was lyophilized following standard methods used to process fecal samples for steroid hormone analysis in primate field studies (ethanol evaporation followed by 0.1 millibar of vacuum pressure at −50 °C11). DNA from all samples was extracted using MO BIO’s PowerSoil DNA Isolation kit (MO BIO Laboratories, Inc., Carlsbad, CA). For lyophilized samples, extractions were obtained from 0.05 g of sample instead of 0.25 g to avoid complete absorption of liquid in the first steps of the DNA extraction. We note that the lyophilization procedure involves additional sample handling relative to the other conditions we tested, which could introduce additional opportunity for sample contamination. However, the general agreement between lyophilized samples and the other samples suggests this is not a major concern if proper sterile technique is employed.
16S rRNA library preparation (targeting the V4 region) and sequencing were conducted at the University of Minnesota Genomics Center, using forward and reverse primers 515F and 806R13 followed by amplification with indexing primers and 300 bp paired-end sequencing on a MiSeq flowcell (see Table S1 for sample-specific details).
All animal work was carried out in accordance with the relevant guidelines and regulations established by AALAC International (YNPRC is a fully accredited animal housing facility). This research was conducted under Institutional Animal Care and Use Committee protocols approved by Emory University (IACUC YER-2001677-040715GA) and Duke University (IACUC A079-12-03).
Data analysis
Following sample demultiplexing, primer sequences were removed from the raw reads using CutAdapt v.1.7.117. Because CutAdapt does not always detect reverse primers effectively, the first 29 base pairs (theoretically the primer and linker sequences) were removed from reverse reads. Reads were truncated at the first base pair with a PHRED quality score ≤3 and forward and reverse reads were then merged using USEARCH v6.118. Read pairs that failed to merge were discarded. We used QIIME v1.8 to conduct further quality control filtering14, using default parameters except for the minimum acceptable per-base Phred score parameter, which we increased from 4 to 20. Putative chimeric sequences were identified using UCHIME (implemented in USEARCH v6.119) and sequences were discarded from the sample when both reference-based (against the RDP Gold training database v920) and de novo abundance-based methods flagged them as likely chimeras.
To identify operational taxonomic units (OTUs) in our data set, we used the open-reference OTU picking pipeline in QIIME. Specifically, the set of chimera-filtered reads was first clustered using the UCLUST v1.2.22 algorithm and the GreenGenes database (May 2013 release21), with a 97% identity threshold. Sequences that failed to cluster against the reference database were then clustered de novo, with sequences that failed both clustering attempts discarded. A representative sequence for each cluster was selected based on the most abundant sequence and then aligned using PyNAST v1.2.222. Sequences that failed to align were discarded. Taxonomic identity was assigned to aligned OTUs using the RDP classifier v2.223, retrained to the May 2013 release of the GreenGenes database21. Singleton OTUs were removed from the OTU table as they tend to be enriched for sequencing errors. We also removed one sample due to low read count.
For all subsequent analyses, we rarefied the OTU table to 54,633 reads per sample using the QIIME v 1.8.0 script single_rarefaction.py. Subsampling reads from individuals with uniformly high coverage across storage conditions supported the stability of our summary statistics at this level of rarefaction (Figure S4). We calculated alpha diversity measures using the QIIME v 1.8.0 script alpha_diversity.py and beta diversity measures using the corresponding QIIME v 1.8.0 script beta_diversity.py14. To estimate the minimum beta diversity dissimilarity due to random resampling error, we used high sequencing depth samples for which at least five times the number of rarefied reads were available (273,165 reads; n = 12). We drew five random subsamples from the total quality-filtered read count for each of these samples. We then calculated all pairwise Bray-Curtis dissimilarity values between subsamples from the same original sample. The median of these dissimilarity values is shown as the dashed line in Fig. 2a (and for weighted and unweighted UniFrac analyses in Figure S2).
All statistical analyses on alpha and beta diversity values were conducted in R v 3.1.124 using either the R base packages or, for PERMANOVA, the R package vegan25 and the R package ade4 v 1.7–226.
Data Availability
All raw read data and associated sample metadata are available in the NCBI Short Read Archive (SRP072517).
Additional Information
How to cite this article: Blekhman, R. et al. Common methods for fecal sample storage in field studies yield consistent signatures of individual identity in microbiome sequencing data. Sci. Rep. 6, 31519; doi: 10.1038/srep31519 (2016).
References
Höss, M., Kohn, M., Pääbo, S., Knauer, F. & Schröder, W. Excrement analysis by PCR. Nature 359, 199–199 (1992).
Choo, J. M., Leong, L. E. & Rogers, G. B. Sample storage conditions significantly influence faecal microbiome profiles. Scientific Reports 5 (2015).
Hale, V. L., Tan, C. L., Knight, R. & Amato, K. R. Effect of preservation method on spider monkey (Ateles geoffroyi) fecal microbiota over 8weeks. Journal of Microbiological Methods 113, 16–26 (2015).
Song, S. J. et al. Preservation methods differ in fecal microbiome stability, affecting suitability for field studies. mSystems 1, e00021–00016 (2016).
Cardona, S. et al. Storage conditions of intestinal microbiota matter in metagenomic analysis. BMC Microbiology 12, 158 (2012).
Carroll, I. M., Ringel-Kulka, T., Siddle, J. P., Klaenhammer, T. R. & Ringel, Y. Characterization of the fecal microbiota using high-throughput sequencing reveals a stable microbial community during storage. PLoS One 7, e46953 (2012).
Dominianni, C., Wu, J., Hayes, R. B. & Ahn, J. Comparison of methods for fecal microbiome biospecimen collection. BMC Microbiology 14, 103 (2014).
Fouhy, F. et al. The Effects of Freezing on Faecal Microbiota as Determined Using MiSeq Sequencing and Culture-Based Investigations. PloS One 10, e0119355 (2015).
Lauber, C. L., Zhou, N., Gordon, J. I., Knight, R. & Fierer, N. Effect of storage conditions on the assessment of bacterial community structure in soil and human-associated samples. FEMS Microbiology Letters 307, 80–86 (2010).
Voigt, A. Y. et al. Temporal and technical variability of human gut metagenomes. Genome Biology 16, 73 (2015).
Khan, M. Z., Altmann, J., Isani, S. S. & Yu, J. A matter of time: evaluating the storage of fecal samples for steroid analysis. General and Comparative Endocrinology 128, 57–64 (2002).
Nsubuga, A. M. et al. Factors affecting the amount of genomic DNA extracted from ape faeces and the identification of an improved sample storage method. Molecular Ecology 13, 2089–2094 (2004).
Caporaso, J. G. et al. Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. The ISME journal 6, 1621–1624 (2012).
Caporaso, J. G. et al. QIIME allows analysis of high-throughput community sequencing data. Nature Methods 7, 335–336 (2010).
Sinha, R. et al. Collecting fecal samples for microbiome analyses in epidemiology studies. Cancer Epidemiology Biomarkers & Prevention 25, 407–416 (2016).
Vlčková, K., Mrázek, J., Kopečný, J. & Petrželková, K. J. Evaluation of different storage methods to characterize the fecal bacterial communities of captive western lowland gorillas (Gorilla gorilla gorilla). Journal of Microbiological Methods 91, 45–51 (2012).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet. journal 17, 10–12 (2011).
Edgar, R. C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26, 2460–2461 (2010).
Edgar, R. C., Haas, B. J., Clemente, J. C., Quince, C. & Knight, R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27, 2194–2200 (2011).
Cole, J. R. et al. The Ribosomal Database Project (RDP-II): sequences and tools for high-throughput rRNA analysis. Nucleic Acids Research 33, D294–D296 (2005).
DeSantis, T. Z. et al. Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Applied and Environmental Microbiology 72, 5069–5072 (2006).
Caporaso, J. G. et al. PyNAST: a flexible tool for aligning sequences to a template alignment. Bioinformatics 26, 266–267 (2010).
Wang, Q., Garrity, G. M., Tiedje, J. M. & Cole, J. R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Applied and Environmental Microbiology 73, 5261–5267 (2007).
R Development Core Team. R: a language and environment for statistical computing (R Foundation for Statistical Computing, Vienna, Austria, 2014).
Oksanen, J. et al. The vegan package. Community Ecology Package, 631–637 (2007).
Dray, S. & Dufour, A.-B. The ade4 package: implementing the duality diagram for ecologists. Journal of Statistical Software 22, 1–20 (2007).
Acknowledgements
We gratefully acknowledge support from the National Science Foundation (IOS 1053461 to E.A.A.), National Institutes of Health (R01-GM102562 to J.T., L.B.B. and M.E.W. and 2-P2CHD065563-06 to Seth Sanders) and the Clare Boothe Luce Foundation (E.A.A.). We thank A. Tripp, J. Whitley and J. Johnson for their support in collecting samples, M. Gearhart for assistance with computational analysis, members of the Blekhman lab for helpful discussions and K. Amato for constructive comments on a previous version of this manuscript. This work was carried out using computing resources at the Minnesota Supercomputing Institute.
Author information
Authors and Affiliations
Contributions
Conceptualization: R.B., E.A.A., L.B.B. and J.T.; Investigation: R.B., K.T., M.L.Y., L.G., L.E.G. and J.T.; Writing–Original Draft: R.B., K.T. and J.T.; Writing–Review & Editing: E.A.A., L.B.B., Z.P.J., M.E.W., J.K., M.L.Y., L.G. and L.E.G.; Funding Acquisition: E.A.A., L.B.B., M.E.W. and J.T.; Resources: Z.P.J., M.E.W. and J.K.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Blekhman, R., Tang, K., Archie, E. et al. Common methods for fecal sample storage in field studies yield consistent signatures of individual identity in microbiome sequencing data. Sci Rep 6, 31519 (2016). https://doi.org/10.1038/srep31519
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep31519
This article is cited by
-
Functional host-specific adaptation of the intestinal microbiome in hominids
Nature Communications (2024)
-
Seasonal shifts in the gut microbiome indicate plastic responses to diet in wild geladas
Microbiome (2021)
-
Clinical health issues, reproductive hormones, and metabolic hormones associated with gut microbiome structure in African and Asian elephants
Animal Microbiome (2021)
-
A Recent Overview of Microbes and Microbiome Preservation
Indian Journal of Microbiology (2020)
-
Improving the standards for gut microbiome analysis of fecal samples: insights from the field biology of Japanese macaques on Yakushima Island
Primates (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.