Abstract
Directed self-assembly of block copolymers is a scalable method to fabricate well-ordered patterns over the wafer scale with feature sizes below the resolution of conventional lithography. Typically, lithographically-defined prepatterns with varying chemical contrast are used to rationally guide the assembly of block copolymers. The directed self-assembly to obtain accurate registration and alignment is largely influenced by the assembly kinetics. Furthermore, a considerably broad processing window is favored for industrial manufacturing. Using an atomically-thin layer of graphene on germanium, after two simple processing steps, we create a novel chemical pattern to direct the assembly of polystyrene-block-poly(methyl methacrylate). Faster assembly kinetics are observed on graphene/germanium chemical patterns than on conventional chemical patterns based on polymer mats and brushes. This new chemical pattern allows for assembly on a wide range of guiding periods and along designed 90° bending structures. We also achieve density multiplication by a factor of 10, greatly enhancing the pattern resolution. The rapid assembly kinetics, minimal topography and broad processing window demonstrate the advantages of inorganic chemical patterns composed of hard surfaces.
Similar content being viewed by others
Introduction
The self-assembly of block copolymers through microphase separation yields patterns with various morphologies and feature sizes ranging from 3 to 100 nm1,2,3,4,5. Defect-free and well-registered block copolymer domains can be created by directed self-assembly on patterned topographic features, known as graphoepitaxy6,7,8, or on patterns with controlled chemical contrast, known as chemoepitaxy9,10,11,12,13. Directed self-assembly can improve the line-edge roughness of the lithographically-defined template14, correct defects in the underlying template via a self-healing mechanism15 and enhance the pattern resolution compared to the periodicity of the template via density multiplication13,16.
The Liu-Nealey (LiNe) flow is a paradigm of chemoepitaxy, as it seamlessly integrates with state-of-the-art 193 nm immersion lithography17,18. Chemical patterns based on cross-linkable polystyrene (X-PS) are commonly used in the LiNe flow because X-PS preferentially wets the polystyrene (PS) domains in the widely studied polystyrene-block-poly(methyl methacrylate) (PS-b-PMMA) system19. After the X-PS mat (6–8 nm in thickness) is patterned to form line-space arrays, the guiding stripes are typically trimmed with a plasma etch. Then a random copolymer brush with hydroxyl groups is grafted into the void spaces between the guiding lines, followed with solvent rinsing to remove excess brush material. This process, known as backfilling, controls the chemistry of the background regions to minimize the interfacial energy between the block copolymer film and the patterned surface and reduces the step height of the chemical pattern19. However, these processing steps can complicate the chemical pattern. For example, plasma trimming oxidizes the sidewalls of the X-PS stripes and forms a trapezoidal shape, consequently changing the guiding scheme of the chemical pattern into one that has three tones20,21. Furthermore, the grafted brush layer can cause interdigitation of the polymer chains with the random copolymer brush, decreasing their diffusivity and resulting in slowed chain dynamics during assembly22.
Alternatively, if a material that wets PS or PMMA is patterned on a neutral surface, only lithography and plasma etching are needed to prepare the chemical contrast pattern. Here, monolayer graphene grown on a germanium wafer is proposed as an appealing template to perform directed self-assembly. Graphene has similar wetting behavior as an X-PS mat23,24, but is a single atom in thickness, forming nearly no topography and providing a smooth, flat and rigid surface for the assembly of block copolymers. Graphene also has a well-defined step height and its inert surface interacts relatively weakly with block copolymers, which may enhance the mobility of polymer chains during segregation and result in more rapid assembly kinetics. Due to its sp2 bonded lattice with carbon-carbon bond distance of 1.42 Å, graphene also has high mechanical strength25, thermal stability26, chemical inertness and impermeability27, making it a robust template that is compatible with a broad range of manufacturing conditions. Continuous graphene films can be deposited over the wafer scale via chemical vapor deposition (CVD)28,29,30, which ensures uniform surface chemistry and monolayer thickness over large areas. In previous work, chemically-modified graphene has been used as a suitable surface for the self-assembly of block copolymers into random fingerprint features31,32. However, the use of a two-dimensional material as a template for directed assembly of block copolymers into ordered patterns would extend the capabilities of block copolymer lithography.
In this work, we demonstrate that atomically-thin graphene stripe arrays can be used as chemical patterns for directed assembly of block copolymers, resulting in well-registered lamellae patterns with complex architectures. The graphene/germanium chemical patterns enable block copolymer assembly on templates with incommensurate periods and in 90° bending structures. Faster assembly dynamics are observed on graphene/germanium templates than on conventional patterns based on polymer mats and brushes. Density multiplication by a factor of 10 is also demonstrated, which means that the period of the assembled block copolymer is one-tenth the template periodicity. These results indicate that graphene/germanium chemical patterns enable robust, reproducible directed self-assembly of block copolymers.
Results and Discussion
Directed assembly on graphene chemical patterns
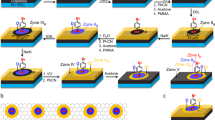
First, we show that chemical patterns consisting of graphene guiding stripes on germanium can direct the assembly of block copolymers into straight, parallel lamellae structures. In this process, which is shown schematically in Fig. 1a–e, continuous monolayer graphene films are grown directly on germanium wafers via CVD (Fig. 1a,b). Scanning electron microscopy (SEM) and atomic force microscopy (AFM) show that the graphene surface is uniform with low roughness of 1–2 nm over 10 × 10 μm2 (Supplementary Fig. S1). The underlying germanium surface consists of terraces and steps, which are predominately one atomic layer in height and are induced during synthesis and the graphene film contains sparse wrinkles that are formed during post-growth cooling. Raman spectroscopy (Fig. 1i, top blue spectrum) confirms that the graphene is predominately a single layer in thickness, as indicated by the integrated 2D:G ratio of ~4 and the Raman 2D full-width-at-half-maximum of ~26 cm−1 33. The negligible Raman D band intensity indicates that the graphene is of high quality, with a relatively low defect density34.
Characterization of the graphene/germanium chemical pattern.
(a–e) Process flow of the block copolymer directed assembly on graphene/germanium chemical patterns. Graphene is grown directly on germanium via CVD (a,b). Resist is spin-coated onto the graphene and patterned into stripe arrays (c). The exposed graphene is etched using a reactive ion plasma and the resist is removed in solvents and via thermal annealing, resulting in a graphene/germanium stripe array (d). The block copolymer is spin-coated on the chemical pattern and thermally annealed to direct assembly (e). (f) SEM image of a graphene stripe array (bright lines) on germanium (dark lines). Inset shows a magnified image highlighting the edges of the graphene stripes. (g,h) AFM topographic map (g) and height, h, profile plotted against surface distance, d, (h) along the dashed line in (g). Scale bars in (f,g) are 250 nm. Height scale bar in (g) is 8 nm. (i), Raman spectrum of an as-synthesized monolayer graphene film on germanium (top blue spectrum) and a graphene stripe array on germanium (bottom red spectrum). The sharp peaks at ~1555 and ~2330 cm−1 correspond to ambient oxygen and nitrogen, respectively.
Line arrays are patterned into a poly(methyl methacrylate) (PMMA) mask over the continuous graphene monolayer films using electron-beam lithography (Fig. 1c). The exposed graphene regions are subsequently etched with reactive oxygen ion plasma, resulting in the formation of isolated graphene guiding stripes (Fig. 1d,f–h). Mild etching conditions (10 sccm O2, 50 W, 10 mtorr, 1–2 s) are used to ensure that the germanium surface is not etched and that the single-atom step height of the graphene stripes is preserved (Fig. 1g,h). The samples are sonicated and rinsed in chlorobenzene and toluene and then annealed at 350–400 °C at ~10−5 torr to remove residue and contamination introduced during patterning. This cleaning ensures a pristine, atomically-thin chemical pattern, which increases the fidelity and reproducibility of the directed block copolymer assembly. The height profile along the graphene stripe array (Fig. 1g,h) indicates that the average step height of the chemical pattern is ~5 Å, consistent with a single layer of graphene. After etching, the Raman D:G ratio increases to ~1 (Fig. 1i, bottom red spectrum), consistent with increased graphene edge density due to ribbon formation.
Finally, PS-b-PMMA is directed to assemble on the graphene/germanium chemical patterns via thermal annealing (Fig. 1e). PS-b-PMMA is chosen for the directed assembly studies because it is among the most commonly studied and well understood systems used for block copolymer lithography4,9,19,35. The molecular weight of the PS-b-PMMA is 85k-b-91k g mol−1, corresponding to a bulk lamellar period (L0) of 78 nm. The graphene ribbon arrays are spin-coated with ~60 nm of PS-b-PMMA and the sample is annealed at 250 °C for 5–10 min in an inert N2 environment (O2 < 1 ppm and H2O < 1 ppm). Figure 2d shows that the PS-b-PMMA assembles into lamellae that closely follow the underlying graphene chemical pattern and are not disrupted by the germanium terracing. In Fig. 2, the period of the patterned graphene stripes, Ls, is chosen to be equivalent to L0 = 78 nm to ensure that the PS-b-PMMA assembles under optimum conditions.
Comparison of assembly kinetics
We compare the assembly kinetics of PS-b-PMMA on graphene/germanium templates and conventional chemical patterns based on X-PS (Fig. 2). The latter control samples consist of a patterned X-PS mat on a silicon oxide (SiO2) substrate. The graphene and X-PS films are patterned into similar line-space arrays with Ls = L0. The samples are spin-coated with PS-b-PMMA and annealed at 250 °C for 2–30 min to reach equilibrium. Directed assembly occurs more rapidly on the graphene-based chemical pattern than on the X-PS-based chemical pattern. Within 2 min, the block copolymer is largely ordered, following the underlying graphene template on >65% of the surface area (Fig. 2a); while the directed assembly remains local and is limited to short segments on the X-PS-based pattern (Fig. 2e). The block copolymer is well-assembled over >95% of the graphene/germanium chemical pattern after 5 min (Fig. 2b) and becomes completely ordered over the entire surface at 10 min (Fig. 2c). On the other hand, it takes 30 min to achieve the same degree of order on the X-PS-based chemical pattern (Fig. 2h). These results may indicate that the smooth, crystalline, rigid surfaces of graphene and germanium enable the polymers to diffuse more efficiently, resulting in faster assembly kinetics with fewer defects22.
Directed assembly on chemical patterns with mismatched periodicity
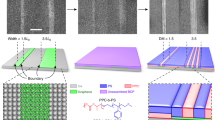
The ability of the block copolymer to conform, relative to L0, to the periodicity of the underlying chemical pattern during assembly is critical for applications such as bit patterned media36,37. PS-b-PMMA is assembled on graphene line arrays with Ls varying between 65 and 88 nm (Fig. 3a–d). The PS-b-PMMA is able to assemble on templates with 0.846 < Ls/L0 < 1.13, allowing a broad commensurability window for designing the chemical pattern.
Directed assembly on incommensurate graphene/germanium chemical patterns.
(a–d) Directed assembly of PS-b-PMMA on graphene/germanium chemical patterns with Ls of 66 nm (a), 70 nm (b), 80 nm (c) and 88 nm (d) corresponding to Ls/L0 of 0.846, 0.897, 1.03 and 1.13, respectively. Scale bars are 200 nm. (e,f) Directed assembly of PS-b-PMMA on graphene/germanium chemical templates that are patterned into 90° bending structures with Ls of 85 nm (e) and 75 nm (f). Scale bars are 200 nm.
In addition to using an array of straight lines as the chemical pattern, directed assembly of PS-b-PMMA is also demonstrated on graphene/germanium chemical patterns with complex architectures (Fig. 3e,f). Bends with 90° angles, in which the line-to-line distance at the corner is 41% longer than L0, are patterned. Despite this large degree of incommensurability, the assembled PS-b-PMMA follows the graphene-based chemical pattern with 90° turns, as shown in Fig. 3e,f. In order to achieve such abrupt features using other types of chemical patterns, homopolymer/block copolymer blends are typically required to fill the gaps near the areas where the block copolymer domains bend10,38.
Increased pattern resolution via density multiplication
In order to enhance the resolution of the lithographically-defined template, assembly of block copolymers on graphene/germanium chemical patterns with density multiplication is explored (Fig. 4). The graphene is patterned into stripe arrays with Ls that are integer multiples, n, of L0 (where n = 1, 4, 5, 6, 8, 9 and 10). The line width of the graphene guiding stripes is chosen to be 1.5 L0 for n > 1 to clearly identify the underlying graphene stripes with SEM after directed assembly. The PS-b-PMMA is spin-coated onto the graphene ribbon array and the sample is annealed at 250 °C for 5–10 min to induce assembly. Despite the lack of guiding stripes for n periods of L0, the block copolymer segregates into regular domains between the graphene ribbons. On the wide graphene stripes, the PS and PMMA domains assemble into lamellae in which the bottom domains are nearly parallel to the surface, creating a U-shaped cross-section; this likely occurs because the PS preferentially wets the graphene surface when the width of a stripe is greater than L0, as previously reported on X-PS based chemical patterns39. The surface affinity of the graphene/germanium chemical pattern to PS-b-PMMA is further characterized, below. Density multiplication by a factor of 10 is achieved using the graphene chemical pattern (Fig. 4h), which is greater than the largest factor of 4 previously obtained using PS-b-PMMA with smaller molecular weight18,39. In addition to achieving density multiplication with 85k-b-91k PS-b-PMMA, the feature size can be further reduced to 12.5 nm by performing density multiplication using PS-b-PMMA with smaller molecular weight of 22k-b-22 k g mol−1, corresponding to L0 of 25 nm (Supplementary Fig. S2).
Density multiplication on graphene/germanium chemical patterns.
(a-f) Directed assembly of PS-b-PMMA on graphene/germanium chemical patterns with density multiplication by a factor of 1 (a), 4 (b), 5 (c), 6 (d), 8 (e) and 9 and 10 (f). In f, the regions to the left and right of the dashed line are patterned with 9X and 10X density multiplication, respectively. Scale bars are 200 nm.
Surface affinity measurements
Investigations of water contact angles and the wetting behavior of block copolymer films40 are conducted to gain insight into the mechanism governing the directed assembly (Supplementary Fig. S3). The water contact angle on graphene, germanium, PS, PMMA and a random PS-PMMA (PS-r-PMMA) film is determined to provide an indirect measurement of their surface affinity towards each of the domains in PS-b-PMMA. The water contact angle on a continuous monolayer graphene film (81°) is similar to that of the PS film (91°). Furthermore, the contact angle on a bare germanium surface after plasma treatment (72°) is similar to that of the PMMA film (68°). These results suggest that PS and PMMA preferentially wet the graphene and germanium regions, respectively, during assembly.
The wetting behavior of the block copolymer domains on graphene and germanium is further verified with a hole/island experiment40. PS-b-PMMA (molecular weight of 22k-b-22 k g mol−1) thin films with thicknesses of 1.5 L0, 1.75 L0 and 2.0 L0 are spin-coated onto continuous graphene films after growth on germanium and onto bare germanium surfaces after being treated with an oxygen plasma to etch the graphene. The samples are annealed at 190 °C for 1 h to induce self-assembly and AFM is used to resolve the equilibrium morphology of the block copolymer films. At 190 °C, PS has lower surface energy than PMMA, so it is energetically favored to wet the free surface. Thus, from the morphology of the self-assembled films, the surface affinity of graphene and germanium toward each block copolymer domain can be determined.
The morphologies of the assembled PS-b-PMMA films are shown in Fig. 5. After assembly on the graphene surface (left column in Fig. 5), films with thickness of 1.5 L0 and 1.75 L0 have featureless terraces with step heights of 1.0 L0, whereas films with thickness of 2.0 L0 are flat and featureless. These results are consistent with symmetric wetting of the PS-b-PMMA, as shown in the assembly schematics in Fig. 5 and indicate that the graphene surface is preferential to PS domains. In contrast, after assembly on the germanium surface (right column in Fig. 5), fingerprint lamellae form at each thickness. Uniform fingerprint lamellae are obtained when the film thickness is 1.75 L0, which is consistent with assembly on a nearly neutral surface40. However, both featureless regions and fingerprint lamellae form when the film thickness is 1.5 L0 and 2.0 L0. The film is flat for the 1.5 L0 case, whereas the featureless regions are higher than the fingerprint regions for the 2.0 L0 case. As shown in the schematic of the assembled block copolymer domains in Fig. 5, this step height indicates that the germanium surface is slightly preferential to PMMA domains.
Determination of the surface affinity of graphene and germanium to the block copolymer domains with a hole/island experiment.
Self-assembly behavior of 22k-b-22k PS-b-PMMA (L0 = 25 nm) on unpatterned graphene films on germanium (left half) and bare germanium (right half) surfaces. On each surface, an AFM height image, AFM phase image and schematic of the assembled block copolymer domains are displayed side-by-side. These data are shown for PS-b-PMMA films with thickness of 1.5 L0 (top row), 1.75 L0 (middle row) and 2.0 L0 (bottom row). Line scans over a surface distance, d, for samples with PS-b-PMMA thickness of 1.5 L0 and 1.75 L0 on graphene show that the height, h, across the terrace boundary is 25 nm, ~1.0 L0. The corresponding schematics show the proposed arrangement of the block copolymer domains after assembly on graphene (preferential to PS) and germanium (slightly preferential to PMMA) surfaces. PS (PMMA) domains are red (blue). The height scale bars are 40 nm, the phase scale bars are 5° and the size of each scan is 1.2 × 1.2 μm2.
The difference in surface energy between the graphene and the germanium likely provides the chemical contrast needed for directed assembly; the PS and PMMA preferentially wet the graphene and germanium regions, respectively. This chemical contrast also provides a favorable template for directed self-assembly with density multiplication, as both the PS and PMMA domains can wet the nearly neutral germanium substrate to form well-ordered patterns, despite the lack of guiding stripes every L019. Furthermore, the large degree of commensurability that is provided by the graphene guiding stripes may also be attributed to this chemical contrast, which can allow compensation of the energetic penalty when the block copolymers assemble on guiding stripes with LS that deviates from L011. Unlike the chemical affinity of X-PS, which can be altered by processing steps such as plasma trimming and brush backfilling20, the surface energy of graphene should be relatively unchanged during processing due to its high chemically inertness.
Large-area directed assembly on graphene/SiO2 chemical patterns
The above results were obtained using graphene guiding stripes that are grown directly on germanium. The direct synthesis of the graphene chemical pattern on a target substrate is desirable for wafer-scale assembly of block copolymers because (1) CVD is an inherently scalable process, yielding uniform continuous graphene films over large areas that are only limited in extent by the size of the substrate or the size of the reaction chamber41 and (2) direct growth yields relatively pristine graphene films42,43, providing a clean, highly reproducible template on which to conduct directed assembly.
In order to demonstrate the generality of the directed assembly using graphene-based chemical patterns, we also transfer large-area monolayer graphene films grown via CVD onto alternative substrates using a sacrificial polymer support28 and subsequently conduct directed assembly experiments. SiO2 is chosen as the target substrate because it is preferential to the PMMA domains over the PS domains and thus provides suitable chemical contrast and surface energetics with respect to graphene to direct the assembly of PS-b-PMMA. After transfer onto SiO2, the continuous graphene films are patterned into guiding stripes using electron-beam lithography and reactive ion etching, as described above. The 85k-b-91k PS-b-PMMA is directed to assemble into aligned lamellae on the graphene guiding stripes on SiO2 wafers with Ls of 78 nm (Supplementary Fig. S4). Notably, we also demonstrate directed assembly on these graphene/SiO2 chemical patterns over large areas (100 × 75 μm2) by patterning the guiding stripes using extreme ultraviolet (EUV) lithography. After directed assembly, the PS-b-PMMA is well-ordered over the entire patterned region, demonstrating that our approach is compatible with state-of-the-art planar lithographic techniques used for high-throughput patterning (Supplementary Fig. S4).
Conclusion
In conclusion, we demonstrate that an atomically-thin monolayer graphene/germanium chemical pattern can be easily prepared and successfully used to direct the assembly of PS-b-PMMA. Graphene-based chemical patterns offer several unique advantages compared to conventional templates based on polymer mats and brushes. Specifically, graphene-based chemical patterns (1) provide faster assembly kinetics, (2) enable assembly on patterns with high degree of incommensurability, (3) allow assembly on irregular and complex templates and (4) provide density multiplication by a factor of 10, even for relatively high molecular weight block copolymers. In future work, chemical contrast may be alternatively patterned by exposing the sample to chemical or plasma treatment, by forming a self-assembled monolayer, or by depositing a second two-dimensional atomic layer (e.g. by lateral growth, van der Waals epitaxy, or stacking). These graphene-based chemical patterns may enable the directed self-assembly of ultranarrow block copolymer domains for manufacturing of high resolution features, both in the semiconductor electronics industry and in high density magnetic media. This work also opens the door for directed assembly studies on chemical patterns based on the large library of two-dimensional materials.
Methods
Graphene synthesis
Ge(111) (Semiconductor Wafer, Inc) and Ge(110) (University Wafer) substrates are used to catalyze graphene growth, as described previously42,43. The germanium samples are sonicated in acetone and isopropyl alcohol for 15 min to clean their surfaces and then etched in deionized water at 90 °C for 15 min. The germanium substrates are placed into a horizontal tube furnace with a quartz tube inner diameter of 34 mm and the system is evacuated to ~10−5 torr. The chamber is filled to atmospheric pressure with a flow of 200 sccm of Ar (99.999%) and 100 sccm of H2 (99.999%). The samples are annealed for 30 min at 910 °C before introducing 4.6 sccm of CH4 (99.99%), which serves as the carbon precursor, to start the graphene synthesis. Growth occurs for 12 h to ensure complete graphene coverage on the germanium surface. The samples are rapidly cooled by sliding the furnace away from the growth region, which terminates growth.
Characterization
AFM (Veeco MultiMode SPM) in tapping mode and SEM (Zeiss LEO 1530) are used to characterize the samples after graphene growth, after patterning of graphene into guiding stripes and after directed assembly of PS-b-PMMA. The quality and thickness of graphene after growth and after patterning is assessed via Raman spectroscopy (Thermo Scientific DXRxi) using an excitation wavelength of 532 nm, power of 10 mW and spot size of 0.6 μm. Contact angle measurements (Dataphysics OCA 15) are conducted using deionized water.
Additional Information
How to cite this article: Chang, T.-H. et al. Directed self-assembly of block copolymer films on atomically-thin graphene chemical patterns. Sci. Rep. 6, 31407; doi: 10.1038/srep31407 (2016).
References
Seidel, R. et al. Directed self-oriented self-assembly of block copolymers using chemically modified surfaces In Directed self-assembly of block co-polymers for nano-manufacturing (ed. Gronheid, R. & Nealey, P. ) 129–170 (Woodhead, 2015).
Bang, J., Jeong, U., Ryu, D. Y., Russell, T. P. & Hawker, C. J. Block copolymer nanolithography: translation of molecular level control to nanoscale patterns. Adv. Mater. 21, 4769–4792 (2009).
Hawker, C. J. & Russell, T. P. Block copolymer lithography: Merging “bottom-up” with “top-down” processes. MRS Bull. 30, 952–966 (2005).
Darling, S. B. Directing the self-assembly of block copolymers. Prog. Polym. Sci. 32, 1152–1204 (2007).
Park, S. et al. Macroscopic 10-terabit-per-square-inch arrays from block copolymers with lateral order. Science 323, 1030–1033 (2009).
Segalman, R. A., Yokoyama, H. & Kramer, E. J. Graphoepitaxy of spherical domain block copolymer films. Adv. Mater. 13, 1152–1155 (2001).
Han, E., Kang, H., Liu, C.-C., Nealey, P. F. & Gopalan, P. Graphoepitaxial assembly of symmetric block copolymers on weakly preferential substrates. Adv. Mater. 22, 4325–4329 (2010).
Park, S. M. et al. Directed assembly of lamellae-forming block copolymers by using chemically and topographically patterned substrates. Adv. Mater. 19, 607–611 (2007).
Ouk Kim, S. et al. Epitaxial self-assembly of block copolymers on lithographically defined nanopatterned substrates. Nature 424, 411–414 (2003).
Stoykovich, M. P. et al. Directed assembly of block copolymer blends into nonregular device-oriented structures. Science 308, 1442–1446 (2005).
Edwards, E. W., Montague, M. F., Solak, H. H., Hawker, C. J. & Nealey, P. F. Precise control over molecular dimensions of block-copolymer domains using the interfacial energy of chemically nanopatterned substrates. Adv. Mater. 16, 1315–1319 (2004).
Stoykovich, M. P. et al. Directed self-assembly of block copolymers for nanolithography: Fabrication of isolated features and essential integrated circuit geometries. ACS Nano 1, 168–175 (2007).
Ruiz, R. et al. Density multiplication and improved lithography by directed block copolymer assembly. Science 321, 936–939 (2008).
Stoykovich, M. P. et al. Remediation of line edge roughness in chemical nanopatterns by the directed assembly of overlying block copolymer films. Macromolecules 43, 2334–2342 (2010).
Cheng, J. Y., Rettner, C. T., Sanders, D. P., Kim, H.-C. & Hinsberg, W. D. Dense self-assembly on sparse chemical patterns: Rectifying and multiplying lithographic patterns using block copolymers. Adv. Mater. 20, 3155–3158 (2008).
Bita, I. et al. Graphoepitaxy of self-assembled block copolymers on two-dimensional periodic patterned templates. Science 321, 939–943 (2008).
Rincon-Delgadillo, P., Craig, G., Gronheid, R. & Nealey, P. F. Scale-up of a chemo-epitaxy flow for feature multiplication using directed self-assembly of block-copolymers. J. Photopolym. Sci. Technol. 26, 831–839 (2013).
Delgadillo, P. A. R. et al. Implementation of a chemo-epitaxy flow for directed self-assembly on 300-mm wafer processing equipment. J. Micro. Nanolithogr. MEMS MOEMS 11, 031302 (2012).
Liu, C.-C. et al. Fabrication of lithographically defined chemically patterned polymer brushes and mats. Macromolecules 44, 1876–1885 (2011).
Williamson, L., Lin, G., Cao, Y., Gronheid, R. & Nealey, P. Tuning the strength of chemical patterns for directed self-assembly of block copolymers. Proc. SPIE Alternative Lithographic Technologies VI 90491B (2014).
Williamson, L. D. et al. Three-tone chemical patterns for block copolymer directed self assembly. ACS Appl. Mater. Interfaces 8, 2704–2712 (2016).
Zhao, J. & Granick, S. How polymer surface diffusion depends on surface coverage. Macromolecules 40, 1243–1247 (2007).
Li, H. & Zeng, X. C. Wetting and interfacial properties of water nanodroplets in contact with graphene and monolayer boron–nitride sheets. ACS Nano 6, 2401–2409 (2012).
Li, Z. et al. Effect of airborne contaminants on the wettability of supported graphene and graphite. Nat. Mater. 12, 925–931 (2013).
Lee, C., Wei, X., Kysar, J. W. & Hone, J. Measurement of the elastic properties and intrinsic strength of monolayer graphene. Science 321, 385–388 (2008).
Campos-Delgado, J. et al. Thermal stability studies of CVD-grown graphene nanoribbons: Defect annealing and loop formation. Chem. Phys. Lett. 469, 177–182 (2009).
Bunch, J. S. et al. Impermeable atomic membranes from graphene sheets. Nano Lett. 8, 2458–2462 (2008).
Li, X. et al. Large-area synthesis of high-quality and uniform graphene films on copper foils. Science 324, 1312–1314 (2009).
Lee, J.-H. et al. Wafer-scale growth of single-crystal monolayer graphene on reusable hydrogen-terminated germanium. Science 344, 286–289 (2014).
Wang, G. et al. Direct growth of graphene film on germanium substrate. Sci. Rep. 3, 2465, 10.1038/srep02465 (2013).
Kim, B. H. et al. Surface energy modification by spin-cast, large-area graphene film for block copolymer lithography. ACS Nano 4, 5464–5470 (2010).
Kim, J. Y. et al. Flexible and transferrable self-assembled nanopatterning on chemically modified graphene. Adv. Mater. 25, 1331–1335 (2013).
Graf, D. et al. Spatially resolved raman spectroscopy of single- and few-layer graphene. Nano Lett. 7, 238–242 (2007).
Ferrari, A. C. Raman spectroscopy of graphene and graphite: Disorder, electron-phonon coupling, doping and nonadiabatic effects. Solid State Commun. 143, 47–57 (2007).
Stoykovich, M. P. & Nealey, P. F. Block copolymers and conventional lithography. Mater. Today 9, 20–29 (2006).
Albrecht, T. R. et al. Bit patterned media at 1 Tdot/in2 and beyond. IEEE Trans. Magn. 49, 773–778 (2013).
Ruiz, R., Dobisz, E. & Albrecht, T. R. Rectangular patterns using block copolymer directed assembly for high bit aspect ratio patterned media. ACS Nano 5, 79–84 (2011).
Stenbock-Fermor, A., Knoll, A. W., Böker, A. & Tsarkova, L. Enhancing ordering dynamics in solvent-annealed block copolymer films by lithographic hard mask supports. Macromolecules 47, 3059–3067 (2014).
Liu, C.-C. et al. Chemical patterns for directed self-assembly of lamellae-forming block copolymers with density multiplication of features. Macromolecules 46, 1415–1424 (2013).
Suh, H. S., Kang, H. M., Nealey, P. F. & Char, K. Thickness dependence of neutral parameter windows for perpendicularly oriented block copolymer thin films. Macromolecules 43, 4744–4751 (2010).
Bae, S. et al. Roll-to-roll production of 30-inch graphene films for transparent electrodes. Nat. Nanotechnol. 5, 574–578 (2010).
Jacobberger, R. M. et al. Direct oriented growth of armchair graphene nanoribbons on germanium. Nat. Commun. 6, 8006, 10.1038/ncomms9006 (2015).
Kiraly, B. et al. Electronic and mechanical properties of graphene-germanium interfaces grown by chemical vapor deposition. Nano Lett. 15, 7414–7420 (2015).
Acknowledgements
T.-H.C., S.M. and Z.M. acknowledge support from the Office of Naval Research (No. N00014-09-1-0803). The program manager at the Office of Naval Research is Dr. Chagaan Baatar. S.M. also acknowledges support from a Winslow Sargeant Fellowship. S.X., H.S.S., C.-C.L. and P.F.N. acknowledge support from the ASTC and the Institute for Molecular Engineering at the University of Chicago. R.M.J. and M.S.A. acknowledge support from the Department of Energy Office of Science Early Career Research Program through the Office of Basic Energy Sciences (No. DE-SC0006414) and R.M.J. also acknowledges support from the Department of Defense Air Force Office of Scientific Research through the National Defense Science and Engineering Graduate Fellowship (No. 32 CFR 168a).
Author information
Authors and Affiliations
Contributions
T.-H.C., S.X. and R.M.J. conceived the experimental design, performed the experiments, characterized the samples, analyzed the results and prepared the manuscript. H.S.S. assisted in designing and analyzing the hole/island experiment. C.-C.L. contributed to the EUV scaling experiment in Supplementary Figure S4. S.M. provided graphene samples on copper in Supplementary Figure S4. D.G. obtained the AFM data in Supplementary Figure S4. X.W., M.S.A., Z.M. and P.F.N. supervised the project. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Chang, TH., Xiong, S., Jacobberger, R. et al. Directed self-assembly of block copolymer films on atomically-thin graphene chemical patterns. Sci Rep 6, 31407 (2016). https://doi.org/10.1038/srep31407
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep31407
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.