Abstract
Inconsistent detection of plant-based dietary small RNAs in circulation has thwarted the use of dietary RNA therapeutics. Here we demonstrate mice consuming diets rich in vegetables displayed enhanced serum levels of the plant specific small RNA MIR2911. Differential centrifugation, size-exclusion chromatography and proteinase K treatment of plant extracts suggest this RNA resides within a proteinase K-sensitive complex. Plant derived MIR2911 was more bioavailable than the synthetic RNA. Furthermore, MIR2911 exhibited unusual digestive stability compared with other synthetic plant microRNAs. The characteristics of circulating MIR2911 were also unusual as it was not associated with exosomes and fractionated as a soluble complex that was insensitive to proteinase K treatment, consistent with MIR2911 being stabilized by modifications conferred by the host. These results indicate that intrinsic stability and plant-based modifications orchestrate consumer uptake of this anomalous plant based small RNA and invite revisiting plant-based microRNA therapeutic approaches.
Similar content being viewed by others
Introduction
Evidence has been presented that small RNAs ingested from plant-based foods act as bioactives1,2,3,4. Concurrently, numerous groups have failed to detect any dietary RNAs in plasma5,6,7. The most compelling evidence regarding activity of exogenous plant based small RNAs called microRNAs (miRNAs) from plant-based foods is work done with rice-based miRNA MIR168a and the herb-based 26S ribosomal RNA-derived small RNA, termed miRNA MIR29111,2. One group has reported that both MIR168a and MIR2911 are absorbed by cells of the gastrointestinal (GI) tract of consuming animals, packaged into exosomes and trafficked via the bloodstream to a variety of tissues, where they are capable of regulating mammalian genes1,2.
While, numerous groups have failed to detect any dietary RNAs in plasma5,6,7, independent studies have specifically replicated the detection of circulating plant-derived MIR2911 which is found in high levels in various herbs8,9,10. The intensity of detection of MIR2911 in murine sera and urine are approximately correlated with dietary intake9. However, rote abundance is not the sole determinate of apparent RNA uptake, as gavage-feeding large doses of synthetic MIR2911 permitted only small transient increases in serum levels9. Moreover, the fact that MIR2911 is one of the few dietary plant RNAs whose uptake by animal consumers has been replicated poses the question of whether the efficacy of MIR2911 delivery to animals might be attributed to unique characteristics of this small RNA. Specifically, unique plant derived packaging and/or the intrinsic stability conferred by the nucleotide sequence and composition of MIR2911 could contribute to its enhanced uptake compared to other miRNAs.
RNAs in circulation are considered to be unstable molecules that are subject to prompt degradation by endogenous RNases and rapid renal clearance11. However, intact RNAs of various types have been detected in circulation12. Characterization of the extracellular miRNAs in peripheral blood plasma suggests nuclease resistant miRNAs exist in different forms, such as in exosomes or in association with components of the RNA-induced silencing complex (RISC), e.g. Argonaute 2 (AGO2)13,14. However, in disagreement with others, our previous work shows that circulating dietary MIR2911 does not co-immunoprecipitate with AGO21,9. Further characterization of circulatory traits of MIR2911 is critical to our understanding of the mechanisms of dietary plant-based small RNA uptake.
We report here an investigation of the physical state of MIR2911 in both plants and in circulation. We measured digestive stability and uptake of the plant-derived MIR2911 in comparison to synthetic MIR2911 and other plant miRNA species. Together the results of the present study demonstrate dietary MIR2911 is packaged and bioavailable from numerous widely consumed vegetables. Furthermore, the MIR2911 complex maybe modified by the host to facilitate circulatory stability. Our results highlight unique plant-based modifications, RNA stability and host specific alterations as possible prerequisites for detection of a circulating plant-based small RNA and offer a plausible explanation for the inability of numerous groups to detect dietary miRNAs in animal sera.
Results
Assaying 2911 levels in vegetables
Various herbs that are not widely consumed in the western diet have been shown to contain high levels of MIR29119. Here, different vegetables, purchased from a local grocery store, were assayed for levels of MIR2911 via quantitative reverse transcription-polymerase chain reaction (qRT-PCR) (Table 1). Cauliflower displayed the highest levels of MIR2911 at 549 fmoles/g while broccoli had 302 fmoles/g, cabbage had 380 (fmoles/g), spinach had 228 fmoles/g and lettuce had 84 fmoles/g of MIR2911.
Uptake of plant-based mir2911 in animals fed vegetable diets
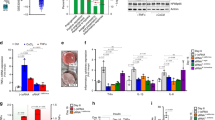
Using a modified chow-based diet supplemented with vegetables, we tested whether MIR2911 could be detected from sera of animals consuming these diets. MIR2911 was measured seven days after initiating feeding. Fold differences in circulating MIR2911 levels in vegetable diet-fed animals compared to chow-fed animals were as follows: spinach, 13-fold higher (69 femtomolar (fM)); cauliflower, 11-fold higher (60 fM); cabbage, 10-fold higher (52 fM); broccoli, 3-fold higher (17 fM). A small change was detected in lettuce fed mice (9 fM) (Fig. 1).
Serum MIR2911 in mice fed various vegetable-containing diets.
Detection of MIR2911 in sera from mice fed various vegetable-containing diets. Mice were fed diets for 7 days before blood collection and qRT-PCR analysis. N = 5; error bars = S.E.M. *p < 0.05; **p < 0.01 between chow and vegetable diet fed mice. Experiment replicated at least three times with each diet.
Content of MIR2911 in edible plant derived exosome-like nanoparticles (EPDENs) and the form of MIR2911 in cabbage extracts
Previous work has shown that edible plant derived exosome-like nanoparticles (EPDENs) contain miRNAs and inferred that they may serve as a vehicle to deliver plant dietary miRNAs to animals15,16. We sought to determine if MIR2911 within plants was encapsulated in these exosome-like particles. Using standard ultracentrifugation-based exosome purification techniques16, we isolated EPDENs from cabbage extract. The particles were characterized as exosome-like based on electron microscopic examination of sucrose gradient purified bands (Supplementary Fig. 1a). However, the majority of MIR2911 was not detected in the EPDENs but found mainly in the non-vesicular fractions (Fig. 2a). As an alternative approach, size exclusion chromatography (SEC) was applied to the cabbage extract (Fig. 2b) to discern the complexation of MIR2911 in the plant. Comparing the MIR2911 elution profile with those of the size standards and free synthetic RNA (Supplementary Fig. 2, Fig. 2b), the data suggested that the size of MIR2911 found in cabbage extracts is consistent with that of a protein-containing complex. Indeed, MIR2911 from the cabbage extract appeared to be sensitive to proteinase K treatment that reduced MIR2911 content by more than 99%, while only 18% of the MIR2911 was degraded in the control (Fig. 2c). These studies demonstrated that MIR2911 is not contained within EPDENs in plants, but is complexed, possibly with a protein.
Forms of MIR2911 in cabbage extract.
(a) Edible Plant Derived Exosome-like Nanoparticles (EPDENs) were purified from clarified cabbage lysate via ultracentrifugation. Levels of MIR2911 were quantified by qRT-PCR in various fractions: cabbage leaves liquefied in a blender (Liquefied Cabbage); Supernatant from 10,000 × g centrifugation (Slow Clarification); Supernatant from 150,000 × g ultracentrifugation (Ultra Clarification); EPDEN bands at 30% 45% interface (EPDEN band1) and 45% 60% interface (EPDEN band2) of sucrose gradient fractionation of total vesicles. (b) Size exclusion chromatography analysis of MIR2911 in ultracentrifugation-clarified cabbage lysate (c) qRT-PCR analysis of MIR2911 levels in proteinase K-treated or control (PBS-treated) ultracentrifugation-clarified cabbage lysate.
Assaying stability of MIR2911 using in vitro digestion
The plant-based modifications of MIR2911 may render it more resistant to digestion and contribute to the enhanced serum detection reported previously8,9. It also remains uncertain whether post-translational miRNA modifications (such as 2′ O-methylation at the 3′ nucleotide) protect the plant-based small RNA from degradation and whether the sequence or nucleotide composition of MIR2911 confers stability since compared with other miRNAs, MIR2911 appeared to be more robustly absorbed than other plant miRNAs8,9. To address these questions, an artificial in vitro digestion system that simulates mammalian gastric and intestinal conditions was employed17. Three samples were compared: sample one contained 1 mL of cabbage extract that contained 10 pmoles of plant derived MIR2911 as measured by qRT-PCR and 10 pmoles of spiked-in synthetic 2′ O-methylated MIR168a and artificial miRNA termed C7; sample two contained 1 mL of phosphate buffered saline (PBS) with 10 pmoles each of synthetic 2′ O-methylated MIR2911, MIR168a and C7; sample three contained 1 mL of PBS with 10 pmoles each of synthetic MIR2911, MIR168a and C7 without the 2′ O-methylation. Time course analysis of the miRNA content was done during both gastric and intestinal phases of digestion. Most miRNAs displayed modest resistance in the acidic gastric environment, while in the intestinal phase the levels of all miRNAs were drastically reduced after five minutes. However, regardless of synthesis origin and the 2′ O-methylation, the digestive stability of MIR2911 was 10–100 fold higher than that of the other miRNAs tested. For MIR2911, the most stable form was the plant derived form in the cabbage extract, as 0.044% survived, compared to 0.0059% for the 2′ O-methylated form and 0.0037% for the non-modified form (Fig. 3).
Superior Stability of MIR2911 compared to other plant microRNAs in in vitro digestion.
The stability of MIR2911, MIR168 and C7 were assayed in an in vitro digestion system that includes a gastric digestion phase and a intestinal digestion phase. Three different samples were digested: (a) cabbage lysate containing a measured amount of 10 pmoles of plant-derived MIR2911 and spiked-in 10 pmoles each of synthetic 2′ O-methylated MIR168 and C7; (b) 10 pmoles each of synthetic 2′ O-methylated MIR2911, MIR168 and C7; (c) 10 pmoles each of synthetic MIR2911, MIR168 and C7 without 2′ O-methylations. Aliquots of digestion reaction were removed at various time points for analysis: 30 min, 60 min in gastric phase, 5 min, 30 min and 75 min in intestinal phase. Levels of surviving miRNAs were measured by qRT-PCR. The surviving percentages were calculated by comparing the levels of miRNAs at each time point to their starting levels before digestion.
MIR2911 stability ex vivo and in vivo
MIR2911 displayed greater stability during in vitro digestion (Fig. 3), we thus sought to test whether MIR2911 was more stable than other miRNAs in the murine gut. 10 pmoles of synthetic 2′ O-methylated MIR2911, MIR168a and C7 were digested in the ex vivo intestinal fluids and were tested for surviving miRNA levels. The results from the ex vivo digestion agreed with the in vitro digestion in that MIR2911 appeared more stable after 2 hours of digestion than the other miRNAs tested (Fig. 4).
Stability of miRNAs assayed in ex vivo intestinal fluids.
10 pmoles of synthetic 2′ O-methylated MIR2911, MIR168a and C7 were incubated in mouse ex vivo intestinal fluids. Surviving miRNA levels were assayed at 5 min, 60 min and 120 min during digestion; percentage of survival were calculated by dividing surviving levels to the measured pre-digestion levels.
A greater efficiency for MIR2911 uptake was previously observed through feeding plant based diets than through gavage feeding of synthetic RNAs9. This observation prompted a further comparison between the stability of MIR2911 from a plant-based diet and synthetic MIR2911 gavage fed to the mice, by directly measuring MIR2911 levels in the small intestines of mice under these feeding conditions. It was found MIR2911 levels in the small intestines of mice fed the plant-based diet (daily dietary MIR2911 intake 10 pmoles) were more than a 100-fold higher than those of mice gavage fed a single 400 pmole dose of the synthetic MIR2911 (Fig. 5). MIR168a, a miRNA present in both plant-based diets and chow, was detected in the small intestines at a level 100-fold lower than that of MIR2911 (Fig. 5). Together, these results substantiate that MIR2911, especially MIR2911 derived from plants, is more stable than other plant-based miRNAs.
Mice fed plant diets contain substantially higher MIR2911 in the small intestines than MIR168a and than mice gavage fed a single dose of microRNAs.
Levels of MIR2911 and MIR168 in the small intestines were measured by qRT-PCR from mice fed honeysuckle diet (Plant), mice gavage fed synthetic miRNAs (Gavage), or mice fed the control chow diet (Chow). N = 3; error bars = S.E.M. *p < 0.05 between Plant and Gavage, or Plant and Chow groups.
Uptake of plant-derived MIR2911 vs synthetic MIR2911
Stability of plant-derived MIR2911 in cabbage extracts was greater than that of the synthetic MIR2911 (Fig. 3); however does this translate into improved bioavailability? To test this, we sought to compare and contrast uptake efficiency of MIR2911 in these two forms by gavage feeding. To achieve a higher feeding dose, cabbage extracts were concentrated to contain 12.5 pmoles of MIR2911 per 500 μL gavage volumes. As a comparison, similar quantities of synthetic MIR2911 should be used; however, this dose has proven to be insufficient to render detectable MIR2911 levels in the sera of animals9. Hence we chose higher dosages of synthetic MIR2911 (400 pmoles per mouse, 32-fold more than the cabbage MIR2911 dosage). An approximate 2-fold increase in the serum MIR2911 levels from the mice gavage fed cabbage extract was detected compared to the animals receiving synthetic 2′ O-methylated MIR2911 (Fig. 6). This suggested that MIR2911 derived from plants is more bioavailable than the synthetic RNA.
Characterization of circulating MIR2911
We initially hypothesized that dietary MIR2911 is present within circulating Argonaute complexes. However, in earlier work we failed to co-immunoprecipitate MIR2911 with antibodies against a mouse AGO2 protein9. Here we tested whether circulating MIR2911 is primarily encapsulated in exosomes or stabilized by other proteins. We used both differential ultracentrifugation and PEG precipitation-based methods to purify circulating vesicles from mice fed cabbage and various herbs. The recovery of vesicles consistent in size and morphology with exosomes and microvesicles was confirmed by transmission electron microscopy (TEM) (Supplementary Fig. 1b) and an exosome quantification kit (Supplementary Fig. 1c). Vesicle pellets and the vesicle-poor supernatants were assayed via qRT-PCR for miR-16, a well characterized circulating endogenous miRNA14. In the context of either of the two exosome isolation protocols, the majority (over 90%) of the circulating miR-16 and MIR2911 were not found in the exosome vesicles (Fig. 7a). To further understand the circulatory forms of MIR2911, size exclusion chromatography (SEC) was employed. Given the large size of exosome associated miRNAs, they should elute from SEC columns early relative to protein associated miRNAs such as miR-16. Using pooled MIR2911-rich mouse sera, miRNA-containing particles were fractionated. The copies of MIR2911, miR-16 and let-7a (an exosome-associated miRNA), were quantified in each fraction by qRT-PCR. The results showed that let-7a eluted earlier than both MIR2911 and miR-16. The fractionation of let-7 and miR-16 were consistent with previous work in human plasma14 while MIR2911 fractionated in the vicinity of the protein associated miRNA miR-16 (Fig. 7b).
Circulatory forms of MIR2911 in mice fed plant-containing diets.
(a) Sera were obtained from mice fed either chamomile, honeysuckle, or chow diet. Exosomes were isolated from these mouse sera via ultracentrifugation protocol (UC) or PEG precipitation protocol (PEG). Levels of miR-16 and MIR2911 were quantified by qRT-PCR in the exosome pellets, or the supernatant fractions. (b) Size exclusion chromatography analysis of Let-7a, miR-16 and MIR2911 levels in control (untreated) serum samples from mice fed vegetable diets. (c) Time course analysis of miR-16 and MIR2911 levels in proteinase K-treated or control (untreated) serum samples from mice fed vegetable diets. (d) Size exclusion chromatography analysis of Let-7a, miR-16 and MIR2911 levels in proteinase K-treated serum samples from mice fed vegetable diets. Note: in Panel (b) and (d), the scale for Y axis for the 3 miRNA targets are set independently. E.g. for miR-16, the peak1 in panel (b) (arrow) represents similar concentration as the peak1 in panel (d) (arrow).
A protease-insensitive complex stabilizes circulating MIR2911
We sought to further address the putative protein association of MIR2911 in sera by proteolytic digestion. If a protein complex protects MIR2911 then proteinase K treatment would render MIR2911, like miR-16, sensitive to degradation by plasma RNases14. In our assay conditions miR-16, previously shown to bind to AGO2, was rapidly degraded in the presence of proteinase K; however, MIR2911 was insensitive to this treatment (Fig. 7c). When the proteinase K-treated sera were run on the SEC column, the non-exosome elution peaks of miR-16 and Let-7a disappeared compared to the control sera; however, the eluting pattern of MIR2911 remained largely unchanged (Fig. 7b,d). This alteration in the physical state of MIR2911, transitioning from a proteinase K-sensitive complex in cabbage extracts (Fig. 2c) to a proteinase K-resistant complex in circulation implies a host modification of the dietary small RNA during uptake.
Discussion
Exposure to dietary MIR2911 can come from a variety of popular vegetables (Table 1). This plant-derived small RNA delivered by typical dietary ingestion had a digestive robustness that appears to help make it more bioavailable compared to other miRNAs (Figs 3 and 4). In milk, specific miRNAs appear to be encapsulated in exosomes conferring protection against degradation and facilitating uptake18,19. We hypothesize that a plant specific protein can facilitate uptake as well as enhancing stability during digestion. Our results implicate modification of the MIR2911 complex during consumer uptake. During digestion or absorption, the proteinase K-sensitive cabbage complex (Fig. 2) is transformed into a serum-stable proteinase K-resistant complex (Fig. 6). We speculate this protein “shield” of MIR2911may be modified in transit, possibly within intestinal epithelial cells. It remains an open question whether the plant binding protein is completely exchanged with an animal protein, or it is only chemically modified.
Different plant-based miRNAs are present within the cellular matrix of the plant material and may be associated with other plant molecules such as proteins and polysaccharides20. These components along with post-translational modifications may provide a conduit for dietary plant-derived miRNAs uptake20. In previous in vitro experiments, miR168a in soybean and mir166 in rice display a greater resistance to degradation than miR168a from rice and mir166 from soybean20. Given that there are no sequence differences between these soybean and rice miRNAs it has been proposed that plant-specific mechanisms are in play that offer varying protection of miRNAs from degradation. Here we also demonstrate the importance of the primary sequence, as MIR2911 is approximately 100-fold more tolerant to simulated mammalian intestinal digestion conditions than MIR168a (Fig. 3). This tolerance is independent of the 2′ O-methylation as the synthetic non-methylated form is equally stable (Fig. 3).
Endogenous extracellular miRNAs are often associated with ribonucleoprotein complexes or exosomes in the circulation as this packaging protects the small RNA from RNases14. We discovered here that the majority of plant-derived MIR2911 appeared to circulate independently from either of these mechanisms. Using SEC fractionation of sera from mice on vegetable diets, MIR2911 did not migrate with the exosome-associated miRNA let-7a (Fig. 6b,d). Meanwhile, although MIR2911 did fractionate with miR-16, an AGO2 associated miRNA, MIR2911 was not proteinase K sensitive (Fig. 6c,d). Together these results suggested the majority of MIR2911 in circulation was in a unique form distinct from these endogenous circulating miRNAs. Additional studies will be needed to unravel mechanisms in the GI tract that facilitate this dietary small RNA uptake and identify how this small RNA is stabilized by the host to facilitate its circulatory stability. Previously, we have sequenced MIR2911 from the sera to show that consumer uptake does not alter the sequence of the small RNA9. However, our methodologies are only measuring levels of full length MIR2911 and truncations and mutant versions of MIR2911 may also be bioavailable.
The unique properties of MIR2911, especially the superior stability against digestion, are not universal to all plant-based miRNAs (Figs 3 and 4) and thus might explain the discordance in detection frequencies among dietary plant-based miRNAs in circulation21. This could explain why serum detection of plant miRNAs has proven problematic5,6,7. Edible plant derived exosome-like nanoparticles (EPDENs) appear to mediate interspecies communication16 but it remains an open question if these effects are mediated by small RNAs. In our case, the majority of MIR2911 did not appear to be physically associated with EPDENs (Fig. 2).
Considering the superior uptake efficiency of the anomalous MIR2911, it can serve as a model for reverse engineering of therapeutic small RNAs that are stable in peripheral blood. Future work can focus on investigating mechanisms of digestive stability and uptake and testing the functionality of dietary plant small RNAs in consumers.
Methods
Animal studies
All studies involving mice were carried out in accordance to the approved guidelines of the Institutional Animal Care and Use Committee (IACUC) of Baylor College of Medicine. The experimental protocol involving mice was approved by IACUC of Baylor College of Medicine. Specifically, the institutional animal protocols AN-2624, AN-6438 and AN-6454 cover the experiments performed in this study. Male 7- to 8-week-old ICR mice were used and were obtained from the Center for Comparative Medicine at Baylor College of Medicine. All feeding studies were replicated at least three times and the results shown are representative of the biological replicates. Vegetables used in the feeding studies were obtained from a local grocery store and were freeze-dried to 30% of the fresh weight before they were finely ground and used to prepare diets. The plant-chow diets were prepared by mixing finely ground chow, plant material and water at 2:1:2 weight ratios9. Each day 5 g of the plant-chow diet that contained 1 g of dried plant material was fed to each mice. The daily intake of plant material per each mouse is equivalent to 3.3 g of fresh weight.
Serum collection and RNA extraction
Blood was collected via retro-orbital bleeding of mice and was allowed to coagulate at room temperature for 1 h9. Sera were separated by centrifugation at 800 × g for 10 min at room temperature followed by centrifugation at 10,000 × g at 4 °C for 10 min to remove all blood cells and debris. Total RNA was extracted from 100 μL of sera using the miRNeasy Mini Kit from Qiagen (Valencia, CA) following manufacturer’s recommendations. Exogenous control RNAs were spiked in at 1 pmole per sample after addition of Qiazol to the samples.
Analysis of miRNA levels by qRT-PCR
Taqman microRNA Assays for let-7d,g,i22, let-7a, miR-16, MIR161, MIR2911, MIR168a and C7 were obtained from Life Technologies (Grand Island, NY). For serum samples, total RNA equivalent to 10 μL of sera was used in each RT reaction. Of the 10 μL RT product, 1 μL was used for each triplicated qPCR reaction. To quantify miRNA levels in the plants, 10–30 mg of fresh plant material were ground to a fine powder in liquid nitrogen and then subjected to RNA isolation using the miRNEASY kit (Qiagen, Valencia, CA); 1 pmole of synthetic artificial miRNA C7 was spiked into the plant Qiazol lysate as an exogenous RNA control. The quantification result was normalized to the weight of starting plant material. qRT-PCR was performed using a Biorad CFX96 Real-Time PCR Detection System and data were analyzed using Biorad CFX software9. The Delta-Delta-Ct method was used to calculate relative levels of miRNAs. Absolute concentrations of miRNAs were calculated based on standard curves obtained from serial dilutions of synthetic miRNAs. The quantification Ct values all fall in the linear range of taqman assays as determined by the synthetic microRNA standards.
Preparation of synthetic miRNAs
Synthetic miRNAs were obtained from Integrated DNA Technologies (Coralville, IA). The sequence of the miRNAs are as follows: MIR-2911 5′-GGC CGG GGG ACG GGC UGG GA-3′; MIR-168a 5′-UCG CUU GGU GCA GAU CGG GAC-3′; C7 5′-GGA UCA UCU CAA GUC UUA CGU3′; MIR161 5′- UCA AUG CAU UGA AAG UGA CUA-3′. For gavage feeding, the RNAs were diluted in RNase-free PBS and each animal was fed 400 pmoles of each RNA in 500 μL volume.
Preparation and concentration of cabbage extract and isolation of Edible Plant Derived Exosome-like Nanoparticles (EPDENs)
Fresh cabbage was purchased from a local market, mixed with ice-cold water at 1 g/mL and then blended in a mixer. This slush was sequentially centrifuged at 1000 × g for 10 min, 3000 × g for 20 min and 10,000 × g for 20 min to remove large particles. The supernatant was then centrifuged at 150,000 × g for 90 min. Supernatant from the ultracentrifuged sample was denoted cabbage extract. MIR2911 was concentrated from 50 mL of cabbage extract to 5 mL using an EMD Millipore (Billerica, MA) Amicon protein concentrator with a molecular weight cutoff of 30 kD following the manufacturer’s specifications. EPDENs were isolated and purified from the pellet from the ultracentrifugation according to the methods as described previously16. The pellet was resuspended in PBS, transferred to a sucrose step gradient (15%/30%/45%/60%) and centrifuged at 150,000 × g for 120 min using a previously described protocol16. The bands between the 8%/30% layer and 30%/45% layer were harvested separately and noted as EPDENs. The protein concentration of these fractions was determined using the Bio-Rad protein quantification assay kit (Bio-Rad Laboratories, Hercules, CA, USA). The purified specimens were prepared for electron microscopy using conventional procedures14 and observed using an Hitachi H7500 Transmission Electron Microscope. Photomicrographs were taken using a AMT XR-16 digital camera.
In vitro digestion of miRNAs
In vitro digestion conditions were based on a previously described protocol14,14. Briefly, the gastric phase was composed of a gastric electrolyte solution (7.8 mM K+, 72.2 mM Na+, 70.2 mM Cl-, 0.9 mM H2PO4-, 25.5 mM HCO3-, 0.1 mM Mg2+, 1.0 mM NH4+, 0.15 mM Ca2+) with pH adjusted by 1N HCl to 2.0 and porcine pepsin (80 mg/mL) (Sigma, St. Louis, MO); the intestinal phase was formed by adding to the gastric phase an intestinal electrolyte solution (7.8 mM K+, 72.2 mM Na+, 124.4 mM Cl-, 55.5 H2PO4-, 85 mM HCO3-, 0.33 mM Mg2+, 0.6 mM Ca2+), 24 mg/mL of bile extract (Sigma, St. Louis, MO) and 4 mg/mL of porcine pancreatin (Sigma, St. Louis, MO) and 1N NaOH to adjust the pH to 7.0. 1 mL of cabbage extract spiked in with synthetic miRNAs or synthetic miRNAs in a 1 mL PBS solution were first mixed with 1 mL gastric phase and digested with slow rotation at 37 °C for 60 min. The digestion mixture was then mixed with the intestinal phase and digested with slow rotation at 37 °C for an additional 75 min. 100 μL of samples were removed at 30 min, 60 min of the gastric phase and 5 min, 30 min and 75 min of the intestinal phase for analysis of surviving miRNA levels. 100 μL of pre-digestion samples were used as controls for calculating the percentage of surviving miRNAs.
Proteinase K treatment
Mouse sera or cabbage extract were mixed with proteinase K (P1807S, New England Biolabs, Ipswich, MA) at a final concentration of 1 mg/mL, or with the control PBS buffer and were incubated at 55 °C for 1 hour.
Digestion of miRNAs in ex vivo intestinal fluids; assaying miRNA levels in the small intestines in vivo
Intestines from chow-fed mice were excised and flushed with 1 mL PBS to collect the contents of the intestines. The supernatant derived from centrifugation at 10,000 × g for 5 min was denoted ex vivo intestinal fluid. 10 pmoles of synthetic 2 O’-methylated MIR2911, MIR168a and C7 were incubated in mouse ex vivo intestinal fluids with slow rotation at 37 °C. Residual intact miRNA levels were assayed at 5 min, 60 min and 120 min of digestion; percentage of residual intact miRNA was calculated by dividing surviving levels to the measured pre-digestion levels. For assaying miRNAs from small intestines, mice were fed a honeysuckle diet or chow diet for 3 days8. The mice were then sacrificed and intestinal contents collected and miRNA levels measured. Simultaneously, chow-fed mice were gavaged 400 pmoles of MIR2911 and MIR168a. miRNA levels from the intestine were assessed 2 hours post gavage.
Isolation of serum exosomes
Exosomes were isolated by two methods: ultracentrifugation14 and PEG precipitation (Serum Total Exosome Isolation Kit (Invitrogen, Carlsbad, CA)23. Briefly, for the ultracentrifugation method, 1 mL of pooled sera was diluted by DEPC-treated sterile PBS to 3 mL and was then clarified at 12,000 × g for 15 min. The supernatant was then centrifuged at 190,000 × g for 2 h. The vesicle pellet was resuspended in 400 μL PBS. For the PEG precipitation, 200 μL of pooled serum was clarified at 2000 × g for 30 min after which the exosomes were isolated according to the manufacturer’s instructions. As quality controls, exosome vesicles were fixed, contrasted with uranyl oxalate and visualized by transmission electron microscopy. Additionally, the exosome preparation were quality-checked and quantified by a commercially available EXOCET kit (SystemBio, Mountain View, CA) following manufacturer’s instructions.
Size-exclusion chromatography
A Sephacryl S-500 column (GE Healthcare, Pittsburgh, PA) was loaded with 1 mL of pooled mouse serum, or cabbage extract and eluted with PBS solution (pH 7.4) at room temperature14. To verify the efficacy of the SEC column, the elution pattern of size standards including thyroglobulin (660 kD), BSA (66.5 kD) and tyrosine (0.181 kD) and a mouse serum sample were analyzed. The results agreed with a previous report, demonstrating that the SEC column is adequate to separate vesicle and protein fractions14. The copies of MIR2911, miR-16 and let-7a were quantified in each fraction by qRT-PCR and normalized to spiked-in 161 miRNAs. Protein abundance of each fraction was measured by absorbance at 280 nm. Fractions were stored at −20 °C before use.
Statistical analysis
Statistical analyses were performed with the student T-Test formula in Microsoft Excel. Significance was set at P < 0.05. Data are presented as means ± SEMs.
Additional Information
How to cite this article: Yang, J. et al. Anomalous uptake and circulatory characteristics of the plant-based small RNA MIR2911. Sci. Rep. 6, 26834; doi: 10.1038/srep26834 (2016).
References
Zhou, Z. et al. Honeysuckle-encoded atypical microRNA2911 directly targets influenza A viruses. Cell Research 25, 39–49, doi: 10.1038/cr.2014.130 (2015).
Zhang, L. et al. Exogenous plant MIR168a specifically targets mammalian LDLRAP1: Evidence of cross-kingdom regulation by microRNA. Cell Research 22, 107–126, doi: 10.1038/cr.2011.158 (2012).
Mlotshwa, S. et al. A novel chemopreventive strategy based on therapeutic microRNAs produced in plants. Cell Research 25, 521–524, doi: 10.1038/cr.2015.25 (2015).
Hirschi, K. D., Pruss, G. J. & Vance, V. Dietary delivery: A new avenue for microRNA therapeutics? Trends Biotechnology 33, 431–432, doi: 10.1016/j.tibtech.2015.06.003 (2015).
Snow, J. W., Hale, A. E., Isaacs, S. K., Baggish, A. L. & Chan, S. Y. Ineffective delivery of diet-derived microRNAs to recipient animal organisms. RNA biology 10, 1107–1116, doi: 10.4161/rna.24909 (2013).
Witwer, K. W., McAlexander, M. A., Queen, S. E. & Adams, R. J. Real-time quantitative PCR and droplet digital PCR for plant miRNAs in mammalian blood provide little evidence for general uptake of dietary miRNAs: limited evidence for general uptake of dietary plant xenomiRs. RNA biology 10, 1080–1086, doi: 10.4161/rna.25246 (2013).
Dickinson, B. et al. Lack of detectable oral bioavailability of plant microRNAs after feeding in mice. Nature Biotechnology 31, 965–967, doi: 10.1038/nbt.2737 (2013).
Yang, J., Farmer, L. M., Agyekum, A. A. A. & Hirschi, K. D. Detection of dietary plant-based small RNAs in animals. Cell Research 25, 517–520, doi: 10.1038/cr.2015.26 (2015).
Yang, J., Farmer, L. M., Agyekum, A. A. A., Elbaz-Younes, I. & Hirschi, K. D. Detection of an abundant plant-based small RNA in healthy consumers. PLoS One 10, e0137516, p. 0137511-0137514, doi: 10.1371/journal.pone.0137516 (2015).
Yang, J., Hirschi, K. D. & Farmer, L. M. Dietary RNAs: New stories regarding oral delivery. Nutrients 7, 3184–3199, doi: 10.3390/nu7053184 (2015).
Etheridge, A., Gomes, C. P., Pereira, R. W., Galas, D. & Wang, K. The complexity, function and applications of RNA in circulation. Frontiers in genetics 4, 115, doi: 10.3389/fgene.2013.00115 (2013).
Valadi, H. et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nature cell biology 9, 654–659, doi: 10.1038/ncb1596 (2007).
Zampetaki, A., Willeit, P., Drozdov, I., Kiechl, S. & Mayr, M. Profiling of circulating microRNAs: from single biomarkers to re-wired networks. Cardiovasc Res 93, 555–562, doi: 10.1093/cvr/cvr266 (2012).
Arroyo, J. D. et al. Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proceedings of the National Academy of Sciences USA 108, 5003–5008, doi: 10.1073/pnas.1019055108 (2011).
Ju, S. et al. Grape exosome-like nanoparticles induce intestinal stem cells and protect mice from DSS-induced colitis. Molecular Therapy : J American Society of Gene Therapy 21, 1345–1357, doi: 10.1038/mt.2013.64 (2013).
Mu, J. et al. Interspecies communication between plant and mouse gut host cells through edible plant derived exosome-like nanoparticles. Molecular Nutrition & Food Ressearch 58, 1561–1573, doi: 10.1002/mnfr.201300729 (2014).
Minekus, M. et al. A standardised static in vitro digestion method suitable for food-an international consensus. Food & function 5, 1113–1124, doi: 10.1039/c3fo60702j (2014).
Baier, S. R., Nguyen, C., Xie, F., Wood, J. R. & Zempleni, J. MicroRNAs are absorbed in biologically meaningful amounts from nutritionally relevant doses of cow milk and affect gene expression in peripheral blood mononuclear cells, HEK-293 kidney cell cultures and mouse livers. Journal of Nutrition 144, 1495–1500, doi: 10.3945/jn.114.196436 (2014).
Zempleni, J., Baier, S. R. & Hirschi, K. D. Diet-responsive MicroRNAs are likely exogenous. Journal of Biological Chemistry 290, 25197, doi: 10.1074/jbc.L115.687830 (2015).
Philip, A., Ferro, V. A. & Tate, R. J. Determination of the potential bioavailability of plant microRNAs using a simulated human digestion process. Molecular Nutrition & Food Ressearch 59, 1962–72, doi: 10.1002/mnfr.201500137 (2015).
Witwer, K. W. & Hirschi, K. D. Transfer and functional consequences of dietary microRNAs in vertebrates: Concepts in search of corroboration: Negative results challenge the hypothesis that dietary xenomiRs cross the gut and regulate genes in ingesting vertebrates, but important questions persist. Bioessays 36, 394–406, doi: 10.1002/bies.201300150 (2014).
Chen, X. et al. A combination of Let-7d, Let-7g and Let-7i serves as a stable reference for normalization of serum microRNAs. PLoS One 8, e79652, doi: 10.1371/journal.pone.0079652 (2013).
Wang, Y. et al. Diagnostic and prognostic value of circulating miR-21 for cancer: A systematic review and meta-analysis. Gene 533, 389–397, doi: 10.1016/j.gene.2013.09.038 (2014).
Acknowledgements
We thank Debra Townley and the image core facility at Baylor College of Medicine for the TEM preparation and imaging of the samples. We thank Dr. Lisa Farmer for her valuable input and pilot experiments with plant EPDENs. We thank Dr. Kurt Bohren for his suggestions on the design of the size exclusion chromatography experiments. We appreciate valuable comments and suggestions on the design of these studies and writing of the paper from Natee Kongchan and Dr. Joel Neilson.
Author information
Authors and Affiliations
Contributions
J.Y. and K.D.H. designed all the studies. M.Y. co-designed size exclusion chromatography experiments. J.Y. performed most of the experiments. T.H. and M.Y. carried out size exclusion chromatography. L.B. and I.-Y.E. conducted animal husbandry and feeding studies. J.Y. and K.D.H wrote the paper.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Yang, J., Hotz, T., Broadnax, L. et al. Anomalous uptake and circulatory characteristics of the plant-based small RNA MIR2911. Sci Rep 6, 26834 (2016). https://doi.org/10.1038/srep26834
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep26834
This article is cited by
-
Plant-derived extracellular vesicles (PDEVs) in nanomedicine for human disease and therapeutic modalities
Journal of Nanobiotechnology (2023)
-
Dietary bovine milk miRNAs transported in extracellular vesicles are partially stable during GI digestion, are bioavailable and reach target tissues but need a minimum dose to impact on gene expression
European Journal of Nutrition (2022)
-
Plant-derived phosphocholine facilitates cellular uptake of anti-pulmonary fibrotic HJT-sRNA-m7
Science China Life Sciences (2019)
-
Prediction of plant-derived xenomiRs from plant miRNA sequences using random forest and one-dimensional convolutional neural network models
BMC Genomics (2018)
-
MicroRNAs from plants to animals, do they define a new messenger for communication?
Nutrition & Metabolism (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.