Abstract
Naturally occurring compounds are considered as attractive candidates for cancer treatment and prevention. Quercetin and ellagic acid are naturally occurring flavonoids abundantly seen in several fruits and vegetables. In the present study, we evaluate and compare antitumor efficacies of quercetin and ellagic acid in animal models and cancer cell lines in a comprehensive manner. We found that quercetin induced cytotoxicity in leukemic cells in a dose-dependent manner, while ellagic acid showed only limited toxicity. Besides leukemic cells, quercetin also induced cytotoxicity in breast cancer cells, however, its effect on normal cells was limited or none. Further, quercetin caused S phase arrest during cell cycle progression in tested cancer cells. Quercetin induced tumor regression in mice at a concentration 3-fold lower than ellagic acid. Importantly, administration of quercetin lead to ~5 fold increase in the life span in tumor bearing mice compared to that of untreated controls. Further, we found that quercetin interacts with DNA directly and could be one of the mechanisms for inducing apoptosis in both, cancer cell lines and tumor tissues by activating the intrinsic pathway. Thus, our data suggests that quercetin can be further explored for its potential to be used in cancer therapeutics and combination therapy.
Similar content being viewed by others
Introduction
Among different diseases, cancer is still considered as the most detrimental for survival of humans. Chemotherapy is considered as the most promising modality for treating cancer. Although chemotherapy against cancer was introduced more than five decades ago, its application is still very limited in most of the cancers. Ideally, a chemotherapeutic drug should eradicate cancer cells by targeting a particular receptor, protein or DNA specific to the neoplastic cells and should reduce tumor burden by inducing cytotoxic and/or cytostatic effects, with least collateral damage to adjacent normal cells.
Naturally occurring compounds are considered as the most interesting agents to test for cancer prevention and therapy, due to their anticipated multimodal actions and limited toxicity. Phytochemicals may also affect the signaling pathways within the cells, including those regulating cell proliferation, activation of apoptosis etc. In addition, combined regimens of naturally occurring compounds with standard chemotherapeutic drugs are very promising in providing additive or synergistic efficacy. Among various naturally occurring compounds, polyphenols are known to be present in various edible fruits including grapes, berries, walnut, pomegranate, apples, etc. Among these, flavonoids consist of a large group of natural, small molecular weight compounds, ubiquitously present in almost all fruits and vegetables.
Polyphenols are reported to have anticarcinogenic properties against different cancers. There are several studies that address the efficiency of polyphenol containing foods in cancer prevention and therapy. However, there are only limited studies to identify individual components responsible for the anticancer properties of polyphenols. Similarly, little is known about mechanism by which such small molecule inhibitors purified from plants act on tumor cells.
Quercetin (3,3′,4′,5,7- pentahydroxy-flavone), is one of the most abundant flavonoids found in fruits and vegetables (Suppl. Fig. 1). It has been shown to exert anticancer and antiinflammatory effects1,2. Due to its antiproliferative nature and relevance in antihypertensive and neurotropic activity, it has been chemically synthesized and commercially sold3,4,5. There are contradicting reports on its antioxidant as well as oxidative properties6,7,8, which need further evaluation. The ability of quercetin to induce cell cycle arrest also warrants additional investigation as conflicting findings have been reported. For example, it appears that quercetin treatment could lead to cell cycle arrest at G0/G1 in leukemia9,10, or S phase in colorectal carcinoma11 or G2/M phases of the cell cycle in breast carcinoma, leukemia and oesophageal adenocarcinoma cell lines12,13,14. Ellagic acid is also one of the phenolic compounds abundantly present in fruits and nuts and is reported to inhibit cell mobility and cell invasion (Suppl. Fig. 1)15. It is known to cause cell cycle arrest at G1 phase and induce apoptosis at a concentration of 100 μM16.
Quercetin has been used in combination with many naturally occurring compounds like resveratrol1, 2-methoxyestradiol17, luteolin derivatives18, ellagic acid2, as well as synthetic drugs used in chemotherapy e.g., doxorubicin19, cisplatin20 etc. In several cases, combination therapy with quercetin resulted in the synergistic effects as well1,17,21.
In spite of various studies suggesting the cytotoxicity of quercetin and ellagic acid in various cancer cell lines, the mechanism of action of these flavonoids during tumor regression is largely unclear. Therefore, in the present study we investigated anticancer properties of quercetin in a systematic manner, using ex vivo, in vitro and in vivo model systems. We found that it induces several fold higher levels of cytotoxicity in cancer cells than ellagic acid. Quercetin also induced cytotoxicity in breast cancer cells apart from leukemic cells, however, its effect on normal cells was limited or none. Further, we showed that quercetin induces S phase arrest; though, we could not observe any detectable ROS production. Quercetin induced tumor regression in mice more effectively than ellagic acid and led to ~5 fold increase in survival. Quercetin induced apoptosis both in cancer cell lines and tumor tissues by activating intrinsic pathway. Thus our data indicates that quercetin could be developed further as a potential anti-cancer agent, both in conventional and combination therapy.
Results
Quercetin induces cytotoxicity in cancer cells
Cytotoxic effect of quercetin and ellagic acid was examined in three leukemic cell lines (CEM, K562 and Nalm6), two breast cancer cell lines (T47D and EAC) and two normal cell lines (293T and MEF1). Cells were treated with increasing concentration of quercetin and ellagic acid (10, 50, 100 and 250 μM for 48 h) and cytotoxic effect was assessed by either MTT or trypan blue and in some cases both. Results showed that quercetin induced cytotoxicity in all the leukemic cell lines in a dose-dependent manner while ellagic acid exhibited only limited sensitivity (Fig. 1). Among leukemic cells, Nalm6 exhibited maximum sensitivity showing reduction in cell viability even at lowest concentration of quercetin (10 μM). IC50 value of quercetin was estimated to be 20, 40 and 55 μM at 48 h, in Nalm6, K562 and CEM, respectively. For ellagic acid, IC50 was 170, >250 and 183 μM for Nalm6, K562 and CEM, respectively. Thus, our results showed that quercetin induced significantly higher toxicity in all cancer cell lines tested.
Determination of the cytotoxic effect of quercetin and ellagic acid on leukemic cell lines.
Cell viability and cell proliferation was determined by MTT and trypan blue assays respectively, on CEM, K562 and Nalm6 cell lines. Cell lines were treated with quercetin (QR) (A) or ellagic acid (EA) (B) for 48 h. DMSO treated cells served as vehicle control (denoted as ‘C’) in both the cases. Concentrations of compound used were 10, 50, 100 and 250 μM. ‘ns’ is ‘not significant’ while ‘*’ represents significance (*P < 0.05, **P < 0.01, ***P < 0.001). Data represents SEM of three independent repeats.
Since quercetin exhibited higher toxicity in leukemic cell lines, its effect was evaluated further in the human breast cancer cell line, T47D using MTT assay. Results showed a limited sensitivity to quercetin with an IC50 of 160 μM in T47D cells (Suppl. Fig. 2A). Interestingly, when quercetin was tested against mouse breast cancer cell line, EAC, it showed much higher sensitivity (IC50 of 50 μM), while no such effect was observed when ellagic acid was used (Suppl. Fig. 2B). However, when normal cell lines (293T and MEF-1) were analyzed using MTT assay, quercetin was found to be insensitive (Suppl. Fig. 2A). Evaluation of Nalm6 cells treated with quercetin (20 and 50 μM) by live dead cell assay following staining with Calcein-A/PI confirmed elevated cytotoxicity induced by quercetin in Nalm6 cells (Suppl. Fig. 3).
Hence, our results show that quercetin induced significant toxicity in both leukemic and breast cancer cell lines, however, its effect on normal cells was minimal.
Quercetin induces S phase arrest during cell cycle progression followed by apoptosis
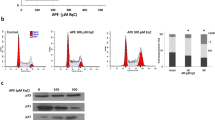
Nalm6 cells were subjected to flow cytometric analyses following treatment with quercetin (20 μM) for 8, 16 and 24 h. Results showed a distinct S phase arrest following 16 h of quercetin treatment in Nalm6 cells (Fig. 2A,B). Besides, a dose-dependent increase in apoptotic cell population was observed in Sub G1 phase when Nalm6 cells were treated with increasing concentrations of quercetin (10, 50, 100, 250 μM for 48 h) as compared to vehicle control (Fig. 2C,D). Interestingly, cell cycle arrest at S phase was observed only at lower time point (16 h) while a dose dependent increase in Sub G1 population was observed at 48 h (Fig. 2C,D). Thus, results show that quercetin induces S phase arrest followed by apoptosis in cancer cells.
Effect of quercetin on cell cycle progression in Nalm6 cells.
(A) Nalm6 cells (0.75 × 105 cells/ml) were incubated at 37 °C with quercetin (20 μM) for 8, 16 and 24 h. Following incubation, cells were harvested, fixed and stained with propidium iodide and analysed by flow cytometry. (B) Bar diagram showing the percentage of cells in S phase of cell cycle. (C) Cell cycle distribution of Nalm6 cells after treating with increasing concentrations (10, 50, 100, 250 μM) of quercetin for 48 h. Histogram resulted after flow cytometry analysis is shown. (D) Bar diagrams showing percentage of cells in each phase of cell cycle. C represents cells alone, while VC stands for DMSO treated vehicle control. In each case M1 represents G1 phase, M2 represents G2 phase, M3 represents S phase and M4 represents Sub G1 phase. Error bars represents SEM of two independent repeats and ‘*’indicates the significance, *P < 0.05.
Quercetin binds to DNA by intercalation
DNA binding ability of quercetin was tested by incubating with calf thymus (CT) DNA, followed by CD spectroscopy. A native B-DNA conformation was observed when CT DNA was studied alone, while a negative shift in peak was observed when DNA was incubated with increasing concentrations of quercetin (50, 100, 150 μM for 1 h, RT) (Fig. 3A) suggesting an intercalation activity. Ethidium bromide stained DNA served as a positive control in which a positive shift was observed at 275 nm and a negative shift at 248 nm (Fig. 3A). Gel mobility shift assay was performed using supercoiled and linearized (digested with EcoRI) form of pUC18 DNA. A shift in the mobility of linearized pUC18 on agarose gel was observed when incubated with increasing concentrations of quercetin (10, 50, 100, 150, 200, 250 μM) (Fig. 3B). Besides, when covalently closed plasmid DNA was incubated with quercetin, a shift in mobility was observed in the case of nicked circle, while an increase in intercalation was observed for supercoiled form in a concentration (10, 50, 100, 150, 200, 250 μM) dependent manner. Ethidium bromide served as positive control for the experiment (Fig. 3C). Therefore, our data suggest that quercetin can directly interact with DNA.
Determination of mode of action of quercetin.
(A) Conformational changes induced by quercetin in DNA was assessed by incubating calf thymus (CT) DNA with different concentrations of quercetin (50, 100, 150 μM) for 1 h at room temperature. Following incubation, CD spectra was recorded at wavelength of 210–320 nm. Ethidium bromide (10, 20 μM) stained DNA served as a positive control. ‘0 μM’ is the untreated CT DNA. Boxed region represents the shift in DNA peak due to intercalation of the compounds. (B,C) Gel mobility shift assay was performed using 100 ng pUC18 DNA. The linearized (digested with EcoRI) (B) and supercoiled (C) pUC18 DNA were incubated at room temperature for 1 h with increasing concentrations of quercetin (10, 50, 100, 150, 200, 250 μM) and products were resolved. Ethidium bromide (2.5, 10, 25 μM) stained DNA served as positive control for the assay. Stained DNA was resolved on 1.2% agarose gel at 30 V. Boxes indicate the shift in the DNA band position due to intercalation.
Quercetin treatment leads to significant reduction in tumor volume
Breast adenocarcinoma developed in Swiss albino mice was used for evaluating antitumor effects of quercetin and ellagic acid. On 12th day of injection of Ehrlich Ascites Carcinoma (EAC) cells, when visible tumor was detectable, mice were orally administered with 6 doses of either quercetin (1 mg/kg) or ellagic acid (3 mg/kg) on every third day. The respective doses of compounds were selected based on a pilot study (data not shown). Result showed a remarkable reduction in tumor volume when treated with each of the compounds as compared to untreated control animals bearing tumor after 10th day of treatment (Fig. 4). However, the reduction in tumor size was more significant in case of quercetin treated animals, since the effect is pronounced at a dose, which is three fold lower than that of ellagic acid (Fig. 4).
Evaluation of effect of quercetin and ellagic acid on tumor growth in mice.
Solid tumor was induced in female Swiss albino mice by injecting EAC cells intramuscularly. (A) Six doses of quercetin (1 mg/kg body wt) and (B) ellagic acid (3 mg/kg body wt) were administered every third day from 12th day of EAC cell injection. Data shows the tumor volume at different time intervals with and without treatment of the compound. Data was collected from three independent experiments with a set of five animals each. Error bars indicate the standard deviation (SD) of three independent experiments. P value was calculated by comparing the mean of untreated control group (EAC alone) and with mean of quercetin treated group, *P < 0.05, ***P < 0.001.
Administration of quercetin improves the lifespan of tumor bearing mice, with no side effects
Increase in life span was evaluated in case of quercetin treated (1 mg/kg) tumor bearing mice as described in Materials and Methods. While untreated control mice survived for a maximum of only ~50 days, treatment with quercetin led to a ~5 fold increase in the life span for atleast 40% of mice (Fig. 5A). This confirms that administration of quercetin did not result in any side effects. Thus, our data shows that quercetin treatment in mice leads to lower tumor load, increased survival and minimal side effects.
Evaluation of effect of quercetin on survival of tumor bearing mice and its side effects.
(A) Kaplan-Meier survival curve of female Swiss albino mice treated with quercetin (1 mg/kg, six doses). The experiment was performed in two independent batches with a set of five animals each. ‘**’ represents significance, **P < 0.01. (B) Histopathologic analysis of tumor following quercetin treatment. Histological sections of thigh of tumor bearing mouse with (treated tumor) and without treatment (control tumor) of quercetin after 30th day (d–f) and (a–c) and 45th day (j–l) and (g–i) of tumor development. Final magnifications shown are 100× (a, d, g and j panel), 200× (b, e, h and k panel) and 400× (c, f, i and l panel). ‘M’ is muscle, ‘TC’ is tumor cells and ‘ITC’ stands for infiltering tumor cells.
Histopathological studies were done on tumor section of control and quercetin treated mice at 30th and 45th days of treatment, by hematoxylin and eosin staining (Fig. 5B). High nuclear staining by hematoxylin was observed in control tumor animals (Fig. 5B; a–c) thus showing the presence of large number of proliferating cells as compared to treated tumor (Fig. 5B; d–f). While control tumor muscle at 30th day of tumor development showed distorted histology and infiltration of tumor cells (Fig. 5B; a–c), following quercetin treatment, cell proliferation and damage in muscle architecture was limited (Fig. 5B; d–f). This was further reduced on 45th day of quercetin treatment (Fig. 5B; j–l) in comparison to control (Fig. 5B; g–i).
TUNEL staining was observed in quercetin treated tumor tissue section (30th day) in comparison to untreated control tumor tissues (Fig. 6A,B) suggesting the presence of DNA fragmentation, which is a characteristic feature of apoptosis. Thus, our data suggest that quercetin treatment might activate apoptosis in tumor tissues.
Evaluation of quercetin mediated cytotoxicity and its mechanism in tumor tissues.
(A) TUNEL assay showing DNA fragmentation on 30th day of quercetin treated tumor tissues in comparison to untreated control tumor. Brown color nuclei indicate DAB staining showing DNA breaks, while nuclei with intact DNA were stained with methyl green. Final magnifications shown are 200× and 400×. (B) Bar diagram with SEM (n = 5) represents % TUNEL positive nuclei in tumor control and quercetin treated mice tissues. ‘*’ represents P < 0.05. (C) Immuno staining was performed on paraffin embedded tissue sections for Ki-67, p-p53 and p53 using respective antibodies in control tumor tissues and quercetin treated tumor tissues after 30th day of treatment. Final magnification shown is 400×. (D) The images of IHC were quantified and plotted as SEM (n = 5). ‘*’represents significance (*P < 0.05, **P < 0.01).
Immunohistochemical analysis showed strong nuclear staining of Ki-67 (Fig. 6C; b), a cellular proliferation marker, in untreated tumor tissue sections (30th day). In contrast, substantially less number of Ki67 positive cells were observed in quercetin treated tissue sections (Fig. 6Cc,D). Further, we observed significant increase in p53 and p-p53 (phospho-p53) positive cells in quercetin treated tumor sections (Fig. 6Cf,i,D), as compared to untreated controls (30th day) (Fig. 6Ce,h) suggesting activation of apoptotic pathway. Tissue sections with no primary antibody served as negative control in each case (Fig. 6Ca,d,g).
Quercetin treatment results in depolarization of mitochondrial membrane potential
Depolarization in mitochondrial membrane potential was studied by staining treated and untreated cells with JC1 dye. The depolarization leads to a shift from red to green fluorescence leading to the release of CYTOCHROME C. Interestingly, a significant shift from red to green fluorescence was observed when Nalm6 cells were treated with increasing concentrations of quercetin (10, 20, 50, 100 μM) for 12 h as compared to vehicle control. 2,4 dinitro phenol (2,4-DNP) was used as a positive control (Fig. 7A). Hence our data suggest the involvement of mitochondrial pathway of apoptosis during quercetin mediated cytotoxicity.
Assessment of effect of quercetin on mitochondrial transmembrane potential (ΔψM) and apoptosis.
(A) Nalm6 cells were treated with quercetin (10, 20, 50 and 100 μM) for 12 h. Cells were harvested, stained with JC1 dye for 20 min at 37o C and analyzed using flow cytometer. Depolarization in mitochondrial membrane potential was assessed by a shift from red to green fluorescence, where red indicates JC1 aggregates in intact mitochondria and green represents green fluorescing monomers in cytosol depicting loss in mitochondrial potential. VC represents DMSO treated vehicle control. Bar diagram shows percentage of depolarized population after quercetin treatment. ‘*’represents significance where *P < 0.05, **P < 0.01. 2,4 DNP treated cells served as positive control. (B) Annexin V/PI dual staining of Nalm6 cells for the detection of apoptotic stages following treatment with quercetin. Nalm6 cells were stained with annexin V-FITC/PI after treatment with quercetin (0, 20 and 50 μM) for 6, 12, 18, 24 and 48 h. Cells were analysed quantitatively as well as qualitatively. Histogram showing distribution of annexin V-FITC/PI stained cells. In each panel lower left quadrant shows the cells negative to both annexin V-FITC and PI staining, lower right panel shows the cells stained with annexin V (early apoptotic cells), upper left quadrant shows cells stained with PI alone (necrotic cells) and upper right shows the cells positive for both annexin V and PI (late apoptotic cells). Histogram showing distribution of early and late apoptotic cells as well as necrotic cells following treatment with quercetin for 6, 12, 18, 24 and 48 h, respectively. Error bar represents standard error mean (SEM) of atleast two independent repeats. P value has been calculated as compared to DMSO control in each case where *P < 0.05, **P < 0.01, ***P < 0.001.
Accumulation of ROS after treatment with antitumor compounds is one of the factors responsible for cells undergoing DNA damage and apoptosis. We were interested in checking whether Nalm6 cells were undergoing cell death due to ROS production. In order to test this, the cells were treated with quercetin (25 μM) for different time points and subjected to flow cytometry. H2O2 induced ROS production served as a positive control. Results showed that quercetin did not induce any significant change in the levels of ROS (Suppl. Fig. 4A,B) suggesting that cell death induced by quercetin was not due to ROS production.
Quercetin treatment leads to apoptosis
In order to confirm whether the cytotoxicity induced by quercetin is indeed due to apoptosis, rather than necrosis, annexin V-FITC/PI staining was performed following quercetin treatment. Nalm6 cells treated with quercetin (0, 20, 50 μM for 6, 12, 18, 24 and 48 h) were subjected to annexin V-FITC/PI double-staining followed by flow cytometry (Fig. 7B). Apoptotic cells can be detected by virtue of binding of annexin V-FITC to phosphotidyl serine (PS) present on the outer side of the cell membrane. In normal cells PS is predominantly present on cytosolic side of the membrane, while it gets translocated towards outer side of the membrane when cells undergo apoptosis. Results showed a concentration dependent increase in early apoptotic cells upon treatment with quercetin for 6 and 12 h, while an increase in late apoptotic cells were observed at 18, 24 and 48 h of treatment suggesting activation of apoptotic pathways (Fig. 7B). Further, confocal microscopic analysis showed cells stained with annexin V-FITC (green fluorescence), following treatment with quercetin (20 μM) suggesting cells in early apoptotic phase (Suppl. Fig. 5). In contrast, most of the cells were stained with annexin V-FITC/PI (green and red) at higher concentrations of quercetin (50 μM) indicating that these cells were undergoing late apoptosis (Suppl. Fig. 5).
Quercetin activates intrinsic pathway of apoptosis
In order to find out the mechanism by which quercetin induces apoptosis, western blotting studies were performed to evaluate the alterations in the protein expression. Nalm6 cells were treated with increasing concentrations of quercetin (0, 10, 20 μM for 24 h), lysate was prepared and analyzed for various apoptotic pathway proteins. Results showed an increase in level of p53 and p-p53 along with cleavage of MCL1, an apoptotic marker (Fig. 8A; Suppl. Fig. 6). A reduction in level of anti-apoptotic proteins BCL2 and BCL-xL was observed while a concomitant increase in level of BAX, a proapoptotic protein was also noted (Fig. 8A). Further, the release of CYTOCHROME C in conjunction with SMAC/DIABLO indicated the activation of mitochondrial intrinsic pathway resulting in apoptosis (Fig. 8A). The observed increase in the level of activated CASPASE 3, cleaved CASPASE 9 and PARP1 further confirmed such a conclusion (Fig. 8A). The data was quantitated, normalized with respect to tubulin and represented as bar diagram with standard error bar (Suppl. Fig. 6).
Effect of quercetin on expression of apoptotic proteins and mechanism of action.
(A) Expression level of apoptotic proteins following treatment with quercetin. Cell lysate was prepared after treating Nalm6 cells with quercetin (0, 10, 20 μM, for 24 h). Protein lysate was resolved on SDS-PAGE and immunoblotting was performed using appropriate primary and secondary antibodies. Expression pattern was studied for p53, p-p53, BCL2, BCL-xL, BAX, MCL 1, CYTOCHROME C, SMAC/DIABLO, activated CASPASE 3, CASPASE 9, PARP1 and Tubulin. Ponceau stained PVDF membrane after protein transfer acted as a control for equal protein loading. (B) Detection of DNA fragmentation in Nalm6 cells following treatment with quercetin. Chromosomal DNA was extracted after treatment with quercetin (10, 50, 100, 250 μM). The purified DNA was resolved on 2% agarose gel at 50 V for 5 h. M is hyper ladder I (Bioline). 5-fluorouracil (5-FU; 100 μM) was used as positive control. (C) Proposed model for quercetin induced apoptosis leading to cytotoxicity. Treatment of cancer cells with quercetin leads to apoptosis through activation of mitochondrial intrinsic pathway. Since quercetin is a DNA intercalator, it might induce DNA damage, which leads to the upregulation of p53. This results in the down regulation of antiapoptotic protein, BCL2 and cleavage of MCL1. CYTOCHROME C and SMAC/DIABLO are released due to loss in mitochondrial membrane potential. SMAC/DIABLO inhibits IAPs (inhibitors of apoptosis proteins), while CYTOCHROME C release leads to cleavage of CASPASE 9, which in turn cleaves CASPASE 3. CASPASE 3 leads to fragmentation and degradation of cellular DNA and activation of PARP1 resulting in apoptosis.
DNA fragmentation is one of the hallmarks of apoptosis differentiating between the necrotic and apoptotic modes of cell death. To test for the presence of DNA fragmentation, Nalm6 cells were treated with different concentrations of quercetin (0, 10, 50, 100, 250 μM) for 48 h. An increase in endonuclease mediated cleaved DNA ladder intensity was observed with increasing concentration of quercetin after 48 h further confirming the apoptosis (Fig. 8B). 5-fluorouracil (5-FU), a compound known for inducing DNA fragmentation was used as the positive control for the assay.
Discussion
Development of safe, novel and effective drugs with less toxicity and lesser side effects is needed for cancer therapeutics. DNA double strand breaks, if not repaired can also lead to genomic instability and cancer. Hence targeting DSB repair genes is one of the methods for cancer therapeutics22. Previous studies from our lab have shown cytotoxic properties of extracts prepared from fruits such as strawberry23 and sapota24, Methyl Angolensate purified from callus of Red wood tree25,26,27 and many synthetic compounds like MPTQ28,29, SCR722,30,31, Levamisole derivative (4a)32, ASHD33, hydantoin derivatives DFH and DCH34 etc. on various cancer cells. These compounds have been shown to be antiproliferative and interfered with different cellular signaling pathways to induce apoptosis.
Phytochemicals are extensively used for evaluation of their cancer therapeutic properties35. Quercetin and ellagic acid are two such compounds studied for their anticancerous and antiproliferative properties as single molecule or in combination1,2,21,36. These natural flavonoids are thought to act as antioxidants and provide defense against several pathologies.
Previous reports showed that both quercetin and ellagic acid induced cytotoxicity in various human cancer cell lines, with varying sensitivity1,12,16,37. In our ex vivo assays, we find that quercetin induces cytotoxicity in leukemic cells effectively; pre-B cell line, Nalm6 being most sensitive with an IC50 value of 20 μM (Fig. 1). A varying range of IC50 for quercetin was reported earlier38,39,40,41,42,43, which ranges from 10 μM to >100 μM. In our studies, quercetin was more cytotoxic compared to ellagic acid. Between the breast cancer cell lines tested, the one derived from mouse showed more sensitivity. However, normal cells derived from human embryonic kidney as well as mouse embryonic fibroblasts were insensitive to quercetin. The ability of quercetin to spare normal cells has also been reported previously44. Chronic myelogenous leukemia cell line, K562 was reported to display resistance to most of the anticancerous drugs. However, we observe that it was sensitive towards quercetin, which was consistent with a previous study8.
A prominent cell cycle arrest at S phase was noted upon treatment with quercetin in Nalm6 cells at early time points (16 h). Besides, an increase in cells in Sub G1 phase was observed in a dose dependent manner, when Nalm6 cells were treated for 48 h. This suggests that quercetin might induce DNA damage, which need to be corrected before cell division takes place. Considering the ability of quercetin to interact with DNA, the observation can be easily explained (see below). A similar S phase arrest has been observed previously as well5,11. However, as reported by others, we did not observe either G0/G1 or G2/M arrest9,10,12,13,14. Difference in the cancer type used may account for such a disparity in the observation.
Consistent with the cytotoxic effect induced by quercetin, we find that even in the tumor tissues, it induced 3-fold more cytotoxicity than ellagic acid. While an impressive, 1 mg/kg quercetin was sufficient for significant inhibition of tumor growth in breast adenocarcinoma bearing mice, for an equivalent effect we had to administer 3 mg/kg of ellagic acid. The dose at which tumor regression was observed in the current work was 10 times lower than that used in past studies19,45. This discrepancy could be due to differences in the cancer cell types or mouse strains used for developing tumor. EAC cells used in the present study are undifferentiated malignant cell lines derived from mice breast adenocarcinoma, commonly used for inducing tumors in mice and used to assess the anticancer effect of various compounds24,28,31,46,47,48. Thus we conclude that quercetin is more effective than ellagic acid in inducing cytotoxicity in different cancer cells.
A 5-fold increase in life span was observed in tumor bearing animals following treatment with quercetin compared to that of untreated tumor bearing mice. This indicates that quercetin induced cytotoxicity in tumor cells without significantly affecting the normal cells. Remarkably, at least 40% of the tumor bearing animals survived upto 250 days. Histological studies and Ki-67 staining confirmed a reduction in proliferative cells and a low damage in muscle architecture in quercetin treated tumor animals in comparison to the untreated control.
Intercalation of small molecules causes conformational changes in DNA, which can affect physiological processes such as replication, transcription, translation and repair. CD spectroscopic and gel shift studies showed that quercetin could intercalate to DNA (Fig. 3), which was also consistent with a recent report38. DNA intercalation is one of the mechanisms by which anticancerous drugs cause DNA damage, accumulation of which could culminate in apoptosis. The observed cell cycle arrest at S phase was also consistent with the plausible induction of DNA damage following DNA intercalation within the cells. However, we could not observe any ROS generation suggesting that it might act as an antioxidant, which was also consistent with another study49.
Depolarization in mitochondrial membrane potential in Nalm6 cells, TUNEL staining positive cells in tumor tissues, Annexin V/PI staining showing early and late apoptotic cells and DNA fragmentation as detected by gel assays suggest activation of apoptosis following exposure to quercetin (Fig. 8C). Apoptosis involve downregulation of antiapoptotic proteins and upregulation of proapoptotic proteins50. Treatment with quercetin resulted in the upregulation of p53, which was consistent with an earlier report51. Downregulation of antiapoptotic protein BCL2 and BCL-xL, upregulation of BAX, p-p53, release of CYTOCHROME C, SMAC/DIABLO as well as cleavage of CASPASE 3, CASPASE 9, PARP1 and MCL1 suggested the involvement of intrinsic mitochondrial pathway (Fig. 8C).
Quercetin is suggested to affect multiple cell signaling processes in cancers and various other diseases (Fig. 8C). Heterogeneous nuclear ribonucleoprotein A1 (hnRNPA1) was identified as a target of quercetin in prostate cancer cell line, PC352. Besides, quercetin shown to inhibit hedgehog signaling pathway in prostate cancer53. Previous studies also showed that quercetin can enhance TRAIL mediated apoptosis in colon adenocarcinoma cells54. Another study suggested set of kinases including ABL1, Aurora-A, B, C as its target55. Therefore, it is possible that quercetin treatment might affect multiple targets within the cells, including DNA by direct intercalation as shown in the present study.
In conclusion, quercetin can induce cytotoxicity through multiple routes of action. Our results show that quercetin, a DNA intercalator induces cytotoxicity in cancer cell lines and tumor tissues in mice by activating intrinsic pathway of apoptosis. Therefore, our results suggest that quercetin is a promising candidate in cancer therapeutics.
Materials and Methods
Cell lines and cell culture
Human chronic mylegenous leukemia cell line (K562), human acute lymphoblastic leukemia cell line (CEM); human embryonic kidney epithelial cell line (HEK 293T), human breast cancer cell lines (T47D), mouse breast cancer cell lines (EAC) and mouse embryonic fibroblast cell line (MEF-1) were purchased from National Centre for Cell Science, Pune, India. Human B cell leukemia cell line (Nalm6) was a gift from M. Lieber (USA). K562, CEM, T47D and Nalm6 were cultured in RPMI1640 (Sera Lab, USA) with 10% FBS (GIBCO, BRL), 100 U of Penicillin G/ml and 100 μg of streptomycin/ml at 37 °C in humidified atmosphere containing 5% CO2. 293T, EAC and MEF were cultured in DMEM containing 10% FBS (GIBCO, BRL), 100 U of Penicillin G/ml and 100 μg of streptomycin/ml.
Trypan blue exclusion assay
Effect of quercetin and ellagic acid on cell viability was determined on CEM, K562 and Nalm6 cell lines by trypan blue exclusion assay as described29,34. Cells were seeded at a density of 0.75 × 105 cells/ml for 24 h and treated with different concentrations of quercetin and ellagic acid (0, 10, 50, 100 and 250 μM) for 48 h. IC50 value was determined by counting the cells after trypan blue staining using DMSO treated cells as vehicle control. The experiment was repeated a minimum of three independent times and data was presented as bar diagram with standard error mean (SEM).
MTT assay
Effect of quercetin and ellagic acid on cell proliferation was assessed by MTT assay as described earlier31,56. Leukemia cell lines (CEM, K562, Nalm6), breast cancer cell lines (EAC and T47D) or normal cell lines (293 T and MEF-1) were seeded (0.75 × 105 cells/ml) for 24 h at 37 °C and then treated with 0, 10, 50, 100 and 250 μM of quercetin or ellagic acid for 48 h and subjected to MTT assay. Experiment was repeated a minimum of three times independently, each with duplicate sets of reaction and presented as bar diagram along with standard error mean (SEM).
Cell cycle analysis by flow cytometry
Cell cycle analyses was performed in Nalm6 cell line as described29,33. A time course experiment was conducted on Nalm6 cells following treatment with quercetin (20 μM) for 8, 16 and 24 h. Besides Nalm6 cells were also treated with increasing concentrations of quercetin (0, 10, 50, 100, 250 μM) for 48 h. In both the experiments cells were seeded at 0.75 × 105 cells/ml, cells were harvested after quercetin treatment, washed, fixed and incubated with RNase A. Cells were then stained with propidium iodide (50 μg/ml) and readings were acquired in flow cytometer (BD Biosciences FACS Calibur, USA). A minimum of 10,000 cells were acquired per sample and data was analyzed by using WinMDI 2.9 software. Experiments were repeated a minimum of two independent times and data is presented along with error bars.
Circular Dichroism (CD)
DNA binding ability of quercetin was studied using CD as described earlier31. Quercetin (0, 50, 100, 150 μM) was incubated with sheared calf thymus (CT) DNA in buffer (10 mM Tris HCl, 15 mM NaCl) for 1 h and spectra were recorded at a wavelength of 210–320 nm on JASCO J-810 spectropolarimeter. Spectra for buffer alone and buffer with corresponding concentrations of quercetin without DNA were subtracted from the experimental data. CD spectra of ethidium bromide (0, 10, 20 μM) incubated with CT DNA served as a positive control.
DNA mobility shift assay
The assay was performed as described earlier31. 100 ng of supercoiled and linearized pUC18 plasmid was incubated with quercetin (0, 10, 50, 100, 150, 200, 250 μM) at room temperature for 1 h and resolved on agarose gel. For further details refer supplementary methods.
Animal and ethics statement
Mice were maintained in strict accordance with the principles and guidelines of the ethical committee for animal care of Indian Institute of Science (IISc) in accordance with Indian National Law on animal care and use. The experiments designed for the present study were approved by Institutional Animal Ethical Committee (Ref. CAF/Ethics/289/2012) IISc, Bangalore, India. For other details refer supplementary methods.
Preparation of EAC cells and tumor induction
Fixed number of ehrlich ascites carcinoma cells (1 × 106 cells/animal) were injected in the peritoneal cavity of donor mouse and allowed to multiply. The cells were withdrawn, diluted in 1X PBS, counted and re-injected to the right thigh of the experimental animals (1 × 106 cells/animal) for developing solid tumor.
Evaluation of anticancer effects of quercetin and ellagic acid on tumor development
Total of 15 mice were taken in a batch, of which 10 were injected with EAC cells in their right thigh to develop solid tumor. 5 mice served as no tumor control, which did not receive quercetin or ellagic acid treatment (Group I). 10 mice with tumor were divided into two groups based on size of the tumor so that mean tumor size of each group remain equal, one of which served as tumor control and received no treatment (Group II), while quercetin (1 mg/kg) or ellagic acid (3 mg/kg) was administered to the other by oral gavage method on 12th day of EAC injection when small tumor was visible (Group III). This group received 6 doses of compound every 3 days. The study was repeated for three independent batches. The dose selected for final study was based on a pilot study conducted using different doses of quercetin and ellagic acid (1, 3 and 10 mg/kg).
Tumor volume was determined by measuring tumor diameter after every 4 days for each animal using vernier calipers as described24,28,31. Tumor volume was calculated by using the formula, V = 0.5ab2 where ‘a’ and ‘b’ represents major and minor diameter, respectively31. After 30th and 45th days one animal was sacrificed from each group by cervical dislocation and tissues were fixed separately for normal, control and treated animals from multiple batches.
Determination of effect of quercetin on survival of tumor animals
The % survival of quercetin treated mice was calculated and compared with control animals. Five animals bearing tumor were taken in each control and quercetin treated groups. The study was done in two independent batches. The death pattern of control and treated animals was reported and percentage increase in life span was calculated using the formula [(T-C)/C] × 100, where T indicates the time of survival of treated animals while C represents survival time of control animals24,28,46. The treated animals were monitored for at least 250 days.
Histological evaluation
Tumor tissues from control and quercetin treated animals were collected. Tissues were embedded in paraffin wax and sections of 5 μm were cut in rotary microtome (Leica Biosystems, Germany). Sections were de-paraffinized and hematoxylin and eosin staining was done as described23,57. Sections were evaluated for structural changes using light microscope (Carl Zeiss, Germany) and images were captured.
TUNEL assay
Terminal deoxynucleotidyl transferase (TdT) dUTP Nick-End Labeling (TUNEL) assay was performed using DNA fragmentation Detection Kit (Calbiochem, USA). For further details refer supplementary methods.
Immunohistochemistry (IHC)
Immunohistochemical staining was performed on quercetin treated tissues sections as described before31,57. For further details refer supplementary methods.
JC1 staining to detect change in mitochondrial membrane potential
5,5’,6,6’-tetrachloro-1,1’,3,3’ tetraethylbenzimidazolylcarbocyanine iodide/chloride (JC-1), a fluorescent carbocyanin dye, was used to check the transmembrane potential (ΔψM) of mitochondria25. For further details refer supplementary methods.
Annexin V-FITC/propidium iodide (PI) staining for apoptotic stages
Annexin V-FITC/PI staining was carried out to detect early and late apoptotic cellular stages, as described34. For further details refer supplementary methods.
Immunoblotting
Nalm6 cell lines were seeded at a density of 0.75 × 105 cells/ml for 24 h. Following treatment with quercetin (0, 10, 20 μM, for 24 h), cells were harvested and lysate was prepared in RIPA buffer (25 mM Tris (pH 7.6), 150 mM NaCl, 1% NP-40, 1% sodium deoxycholate and 0.1% SDS) as described34,57. Protein concentration was determined by Bradford assay. Approximately, 30 μg lysate was resolved on 10–12% SDS-PAGE and transferred onto the PVDF membrane (Millipore, USA). Membrane was blocked and incubated with respective primary antibodies (BCL2, BAX, MCL1, CASPASE 9, activated CASPASE 3, CYTOCHROME C, p53, p-p53, SMAC/DIABLO, BCL-xL, PARP1, Tubulin) at 4 °C overnight. Blots were washed, incubated with HRP-conjugated secondary antibodies, developed using chemiluminescent solution (ImmobilonTM western, Millipore, USA) and scanned by using gel documentation system (ImageQuant LAS4000, GE, USA). The experiment was repeated a minimum of three independent times.
DNA fragmentation assay
Nalm6 cells were treated with quercetin (0, 10, 50, 100, 250 μM) as described above and DNA fragmentation analysis was performed58. For further details refer supplementary methods.
Statistical analysis
Data was expressed as mean ± SEM for normal and experimental animals. Statistical analysis was performed by two way ANOVA and student’s t-test using software Graph pad prism 5.1. Values were considered significant when P ≤ 0.05. Kaplien-Meier survival curve was also plotted using Graph pad prism 5.1.
Additional Information
How to cite this article: Srivastava, S. et al. Quercetin, a Natural Flavonoid Interacts with DNA, Arrests Cell Cycle and Causes Tumor Regression by Activating Mitochondrial Pathway of Apoptosis. Sci. Rep. 6, 24049; doi: 10.1038/srep24049 (2016).
References
Mertens-Talcott, S. U. & Percival, S. S. Ellagic acid and quercetin interact synergistically with resveratrol in the induction of apoptosis and cause transient cell cycle arrest in human leukemia cells. Cancer Lett 218, 141–151 (2005).
Mertens-Talcott, S. U., Talcott, S. T. & Percival, S. S. Low concentrations of quercetin and ellagic acid synergistically influence proliferation, cytotoxicity and apoptosis in MOLT-4 human leukemia cells. J Nutr 133, 2669–2674 (2003).
Haghiac, M. & Walle, T. Quercetin induces necrosis and apoptosis in SCC-9 oral cancer cells. Nutr Cancer 53, 220–231 (2005).
Senthilkumar, K. et al. Quercetin regulates insulin like growth factor signaling and induces intrinsic and extrinsic pathway mediated apoptosis in androgen independent prostate cancer cells (PC-3). Mol Cell Biochem 344, 173–184 (2010).
Zhang, H. et al. Antitumor activities of quercetin and quercetin-5′,8-disulfonate in human colon and breast cancer cell lines. Food Chem Toxicol 50, 1589–1599 (2012).
Kim, G. N. & Jang, H. D. Protective mechanism of quercetin and rutin using glutathione metabolism on HO-induced oxidative stress in HepG2 cells. Ann N Y Acad Sci 1171, 530–537 (2009).
Robaszkiewicz, A., Balcerczyk, A. & Bartosz, G. Antioxidative and prooxidative effects of quercetin on A549 cells. Cell Biol Int 31, 1245–1250 (2007).
Brisdelli, F., Coccia, C., Cinque, B., Cifone, M. G. & Bozzi, A. Induction of apoptosis by quercetin: different response of human chronic myeloid (K562) and acute lymphoblastic (HSB-2) leukemia cells. Mol Cell Biochem 296, 137–149 (2007).
Yuan, Z. et al. Quercetin-induced apoptosis of HL-60 cells by reducing PI3K/Akt. Mol Biol Rep 39, 7785–7793 (2012).
Yoshida, M., Yamamoto, M. & Nikaido, T. Quercetin arrests human leukemic T-cells in late G1 phase of the cell cycle. Cancer Res 52, 6676–6681 (1992).
Richter, M., Ebermann, R. & Marian, B. Quercetin-induced apoptosis in colorectal tumor cells: possible role of EGF receptor signaling. Nutr Cancer 34, 88–99 (1999).
Choi, J. A. et al. Induction of cell cycle arrest and apoptosis in human breast cancer cells by quercetin. Int J Oncol 19, 837–844 (2001).
Lee, T. J. et al. Quercetin arrests G2/M phase and induces caspase-dependent cell death in U937 cells. Cancer Lett 240, 234–242 (2006).
Zhang, Q., Zhao, X. H. & Wang, Z. J. Flavones and flavonols exert cytotoxic effects on a human oesophageal adenocarcinoma cell line (OE33) by causing G2/M arrest and inducing apoptosis. Food Chem Toxicol 46, 2042–2053 (2008).
Pitchakarn, P. et al. Ellagic acid inhibits migration and invasion by prostate cancer cell lines. Asian Pac J Cancer Prev 14, 2859–2863 (2013).
Narayanan, B. A., Geoffroy, O., Willingham, M. C., Re, G. G. & Nixon, D. W. p53/p21(WAF1/CIP1) expression and its possible role in G1 arrest and apoptosis in ellagic acid treated cancer cells. Cancer Lett 136, 215–221 (1999).
Yang, F. et al. Combination of Quercetin and 2-Methoxyestradiol Enhances Inhibition of Human Prostate Cancer LNCaP and PC-3 Cells Xenograft Tumor Growth. Plos One 10, e0128277 (2015).
Ravishankar, D. et al. Exploring quercetin and luteolin derivatives as antiangiogenic agents. Eur J Med Chem 97, 259–274 (2015).
Wang, G., Zhang, J., Liu, L., Sharma, S. & Dong, Q. Quercetin potentiates doxorubicin mediated antitumor effects against liver cancer through p53/Bcl-xl. Plos One 7, e51764 (2012).
Nessa, M. U., Beale, P., Chan, C., Yu, J. Q. & Huq, F. Synergism from combinations of cisplatin and oxaliplatin with quercetin and thymoquinone in human ovarian tumour models. Anticancer Res 31, 3789–3797 (2011).
Akagi, K. et al. Modulating effects of ellagic acid, vanillin and quercetin in a rat medium term multi-organ carcinogenesis model. Cancer Lett 94, 113–121 (1995).
Srivastava, M. R. S. DNA double-strand break repair inhibitors as cancer therapeutics. Chem Biol 22, 17–29 (2015).
Somasagara, R. R. et al. Extracts of strawberry fruits induce intrinsic pathway of apoptosis in breast cancer cells and inhibits tumor progression in mice. Plos One 7, e47021 (2012).
Srivastava, M. et al. Sapodilla plum (Achras sapota) induces apoptosis in cancer cell lines and inhibits tumor progression in mice. Sci Rep 4, 6147 (2014).
Chiruvella, K. K. et al. Methyl angolensate, a natural tetranortriterpenoid induces intrinsic apoptotic pathway in leukemic cells. FEBS Lett 582, 4066–4076 (2008).
Chiruvella, K. K., Panjamurthy, K., Choudhary, B., Joy, O. & Raghavan, S. C. Methyl angolensate from callus of Indian redwood induces cytotoxicity in human breast cancer cells. Int J Biomed Sci 6, 182–194 (2010).
Chiruvella, K. K. & Raghavan, S. C. A natural compound, methyl angolensate, induces mitochondrial pathway of apoptosis in Daudi cells. Invest New Drugs 29, 583–592 (2011).
Sharma, S. et al. A novel DNA intercalator, 8-methoxy pyrimido[4′,5′:4,5]thieno (2,3-b)quinoline-4(3H)-one induces apoptosis in cancer cells, inhibits the tumor progression and enhances lifespan in mice with tumor. Mol Carcinog 52, 413–425 (2013).
Shahabuddin, M. S., Nambiar, M., Choudhary, B., Advirao, G. M. & Raghavan, S. C. A novel DNA intercalator, butylamino-pyrimido[4′,5′:4,5]selenolo(2,3-b)quinoline, induces cell cycle arrest and apoptosis in leukemic cells. Invest New Drugs 28, 35–48 (2010).
John, F. et al. Pluronic copolymer encapsulated SCR7 as a potential anticancer agent. Faraday Discuss 177, 155–161 (2015).
Srivastava, M. et al. An inhibitor of nonhomologous end-joining abrogates double-strand break repair and impedes cancer progression. Cell 151, 1474–1487 (2012).
Hegde, M. et al. Novel levamisole derivative induces extrinsic pathway of apoptosis in cancer cells and inhibits tumor progression in mice. Plos One 7, e43632 (2012).
Kavitha, C. V. et al. Propyl-2-(8-(3,4-difluorobenzyl)-2′,5′-dioxo-8-azaspiro[bicyclo[3.2.1] octane-3,4′-imidazolidine]-1′-yl) acetate induces apoptosis in human leukemia cells through mitochondrial pathway following cell cycle arrest. Plos One 8, e69103 (2013).
Kavitha, C. V. et al. Novel derivatives of spirohydantoin induce growth inhibition followed by apoptosis in leukemia cells. Biochem Pharmacol 77, 348–363 (2009).
Overby, A., Zhao, C. M. & Chen, D. Plant phytochemicals: potential anticancer agents against gastric cancer. Curr Opin Pharmacol 19C, 6–10 (2014).
Khanduja, K. L., Gandhi, R. K., Pathania, V. & Syal, N. Prevention of N-nitrosodiethylamine-induced lung tumorigenesis by ellagic acid and quercetin in mice. Food Chem Toxicol 37, 313–318 (1999).
Umesalma, S., Nagendraprabhu, P. & Sudhandiran, G. Ellagic acid inhibits proliferation and induced apoptosis via the Akt signaling pathway in HCT-15 colon adenocarcinoma cells. Mol Cell Biochem 399, 303–313 (2014).
Bishayee, K. et al. Quercetin induces cytochrome-c release and ROS accumulation to promote apoptosis and arrest the cell cycle in G2/M, in cervical carcinoma: signal cascade and drug-DNA interaction. Cell Prolif 46, 153–163 (2013).
Borska, S. et al. In vitro effect of quercetin on human gastric carcinoma: targeting cancer cells death and MDR. Food Chem Toxicol 50, 3375–3383 (2012).
Huang, S. L., Hsu, C. L. & Yen, G. C. Growth inhibitory effect of quercetin on SW 872 human liposarcoma cells. Life Sci 79, 203–209 (2006).
Wenzel, U., Herzog, A., Kuntz, S. & Daniel, H. Protein expression profiling identifies molecular targets of quercetin as a major dietary flavonoid in human colon cancer cells. Proteomics 4, 2160–2174 (2004).
Huang, Y. T. et al. Effects of luteolin and quercetin, inhibitors of tyrosine kinase, on cell growth and metastasis-associated properties in A431 cells overexpressing epidermal growth factor receptor. Br J Pharmacol 128, 999–1010 (1999).
Yoshida, M. et al. The effect of quercetin on cell cycle progression and growth of human gastric cancer cells. FEBS Lett 260, 10–13 (1990).
Son, Y. O. et al. Selective effects of quercetin on the cell growth and antioxidant defense system in normal versus transformed mouse hepatic cell lines. Eur J Pharmacol 502, 195–204 (2004).
Pratheeshkumar, P. et al. Quercetin inhibits angiogenesis mediated human prostate tumor growth by targeting VEGFR- 2 regulated AKT/mTOR/P70S6K signaling pathways. Plos One 7, e47516 (2012).
Noaman, E., Badr El-Din, N. K., Bibars, M. A., Abou Mossallam, A. A. & Ghoneum, M. Antioxidant potential by arabinoxylan rice bran, MGN-3/biobran, represents a mechanism for its oncostatic effect against murine solid Ehrlich carcinoma. Cancer Lett 268, 348–359 (2008).
Gekeler, V., Epple, J., Kleymann, G. & Probst, H. Selective and synchronous activation of early-S-phase replicons of Ehrlich ascites cells. Mol Cell Biol 13, 5020–5033 (1993).
Kumar, A. et al. Antiangiogenic and antiproliferative effects of substituted-1,3,4-oxadiazole derivatives is mediated by down regulation of VEGF and inhibition of translocation of HIF-1alpha in Ehrlich ascites tumor cells. Cancer Chemother Pharmacol 64, 1221–1233 (2009).
Vidya Priyadarsini, R. et al. The flavonoid quercetin induces cell cycle arrest and mitochondria-mediated apoptosis in human cervical cancer (HeLa) cells through p53 induction and NF-kappaB inhibition. Eur J Pharmacol 649, 84–91 (2010).
Hengartner, M. O. The biochemistry of apoptosis. Nature 407, 770–776 (2000).
Chan, S. T., Yang, N. C., Huang, C. S., Liao, J. W. & Yeh, S. L. Quercetin enhances the antitumor activity of trichostatin A through upregulation of p53 protein expression in vitro and in vivo. Plos One 8, e54255 (2013).
Ko, C. C. et al. Chemical proteomics identifies heterogeneous nuclear ribonucleoprotein (hnRNP) A1 as the molecular target of quercetin in its anti-cancer effects in PC-3 cells. J Biol Chem 289, 22078–22089 (2014).
Slusarz, A. et al. Common botanical compounds inhibit the hedgehog signaling pathway in prostate cancer. Cancer Res 70, 3382–3390 (2010).
Psahoulia, F. H., Drosopoulos, K. G., Doubravska, L., Andera, L. & Pintzas, A. Quercetin enhances TRAIL-mediated apoptosis in colon cancer cells by inducing the accumulation of death receptors in lipid rafts. Mol Cancer Therapeut 6, 2591–2599 (2007).
Boly, R. et al. Quercetin inhibits a large panel of kinases implicated in cancer cell biology. Int J Oncol 38, 833–842 (2011).
Freimoser, F. M., Jakob, C. A., Aebi, M. & Tuor, U. The MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] assay is a fast and reliable method for colorimetric determination of fungal cell densities. Appl Environ Microbiol 65, 3727–3729 (1999).
Sharma, S., Choudhary, B. & Raghavan, S. C. Efficiency of nonhomologous DNA end joining varies among somatic tissues, despite similarity in mechanism. Cell Mol Life Sci 68, 661–676 (2011).
Sahu, U. et al. A Novel Anticancer Agent, 8-Methoxypyrimido[4′,5′:4,5]thieno(2,3-) Quinoline-4(3H)-One Induces Neuro 2a Neuroblastoma Cell Death through p53-Dependent, Caspase-Dependent and -Independent Apoptotic Pathways. Plos One 8, e66430 (2013).
Acknowledgements
We thank Dr. M. Nambiar, Mr. R. Sebastian, Ms. M. Pandey and other members of SCR laboratory for discussions and comments on the manuscript. We thank Central Animal Facility, Dr. Uttara Chakraborty and other members of FACS Facility and Confocal Facility, IISc for their help. This work was supported by grants from IISc-DBT partnership programme [DBT/BF/PR/INS/2011–12/IISc]. SS and RRS are supported by Dr. D. S. Kothari Postdoctoral Fellowship from UGC, India and NM is supported by SRF from CSIR.
Author information
Authors and Affiliations
Contributions
S.C.R., S.S. and R.R.S. conceived and designed the experiments; S.S., R.R.S., M.H., N.M., T.S.K., B.C. and M.S. performed the experiments and analysed the data; S.C.R. and S.S. wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Srivastava, S., Somasagara, R., Hegde, M. et al. Quercetin, a Natural Flavonoid Interacts with DNA, Arrests Cell Cycle and Causes Tumor Regression by Activating Mitochondrial Pathway of Apoptosis. Sci Rep 6, 24049 (2016). https://doi.org/10.1038/srep24049
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep24049
This article is cited by
-
Targeting Aminoglycoside Acetyltransferase Activity of Mycobacterium tuberculosis (H37Rv) Derived Eis (Enhanced Intracellular Survival) Protein with Quercetin
The Protein Journal (2024)
-
Anticancer Properties of Different Varieties of Date Palm (Phoenix dactylifera L.) Leaf Extracts in Human Tumor Cells: a Comparative Study
Plant Foods for Human Nutrition (2024)
-
Recombined Egg Yolk Granules with Controllable Particle Sizes for Quercetin Delivery – Effects of Particle Size on Physicochemical Stability, Bioaccessibility, and Bioactivity of Quercetin
Food Biophysics (2024)
-
Hydroxyapatite microbeads containing BMP-2 and quercetin fabricated via electrostatic spraying to encourage bone regeneration
BioMedical Engineering OnLine (2023)
-
A Comprehensive Study on the Anti-cancer Effects of Quercetin and Its Epigenetic Modifications in Arresting Progression of Colon Cancer Cell Proliferation
Archivum Immunologiae et Therapiae Experimentalis (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.