Abstract
Overexpresssion of HER-2 in the MDA-MB-435/LCC6 (LCC6HER-2) tumour model is associated with significantly increased hypoxia and reduced necrosis compared to isogenic control tumours (LCC6Vector); this difference was not related to tumour size or changes in vascular architecture. To further evaluate factors responsible for HER-2-associated changes in the tumour microenvironment, small animal magnetic resonance imaging (MRI) and positron emission tomography (PET) were used to measure tumour tissue perfusion and metabolism, respectively. The imaging data was further corroborated by analysis of molecular markers pertaining to energy homeostasis and measurements of hypoxia and glucose consumption. The results showed a strong trend towards higher perfusion rates (~58% greater, p = 0.14) and significantly higher glucose uptake in LCC6HER-2 (~2-fold greater; p = 0.025), relative to control tumours. The expression of proteins related to energy stress (P-AMPK, P-ACC) and glucose transporters (GLUT1) were lower in LCC6HER-2 tumours (~2- and ~4-fold, respectively). The in vitro analysis showed that LCC6HER-2 cells become more hypoxic in 1% oxygen and utilise significantly more glucose in normoxia compared to LCC6Vectorcells (p < 0.005). Amalgamation of all the data points suggests a novel metabolic adaptation driven by HER-2 overexpression where higher oxygen and glucose metabolic rates produce rich energy supply but also a more hypoxic tumour mass.
Similar content being viewed by others
Introduction
Amplification of the HER-2 gene (c-erbB2 or neu gene) is associated with poor prognosis1,2,3,4,5,6,7 and is a therapeutic target of interest in various cancers3,7,8. HER-2 signalling is known to trigger pathways that increase cell proliferation and enhance cell survival, culminating in resistance to anti-cancer therapies2,3. Our group showed that overexpressing HER-2 in the aggressive MDA-MB-435/LCC6 tumour model9 (LCC6HER-2) increased the tumour’s viability and levels of hypoxia compared to isogenic control tumours transfected with vector alone (LCC6Vector)10. High levels of hypoxia would reduce the effectiveness of radiation treatment and chemotherapy, but more importantly is associated with an increased chance of metastasis and lower survival rates11,12. We now report that LCC6HER-2 cells consume more oxygen than their non-HER-2 counterparts which may explain the observed increase in hypoxic cell fraction found in LCC6HER-2 tumours (relative to control LCC6Vector tumours). Hypoxia in LCC6HER-2 tumours is present concomitantly with increased glucose consumption, suggesting higher rates of glycolysis. The data also indicate that HER-2 expression is associated with improved cell bioenergetics which may provide a survival advantage for HER-2 overexpressing tumours.
Methods
LCC6 cells, Tumours and In Vitro Oxygen and Glucose Consumption
The identity of the LCC6 parental cell line, MDA-MB-435, was determined to be of melanoma origin rather than breast following a period of controversy and confusion13,14. Although MDA-MB-435 cells may have been misidentified as breast cancer cells in studies examining the effects of HER-2 overexpression in tumour biology, they are still useful models of altered gene expression15,16. In our laboratory, the accessibility of MDA-MB-435/LCC6 (LCC6) transfected variants and its consistent tumour take rates in animals provided an opportunity to study the in vivo effects of HER-2 overexpression (LCC6HER-2) against an isogenic vector transfected tumours (LCC6Vector)10,17.
Animal study and imaging protocols were reviewed and approved by the animal care committee at the University of British Columbia working under the auspices of the Canadian Animal Care Council. All animal procedures and monitoring were carried out in accordance with the approved protocols. Tumours were grown subcutaneously to 200 mm3 in immuno-compromised female Rag2M mice10 and size matched LCC6HER-2 and LCC6Vector tumours were imaged with magnetic resonance imaging (MRI) and positron emission tomography (PET) to evaluate in situ tumour tissue perfusion and glucose uptake rates, respectively. Tumours were harvested after imaging and processed for Western Blotting. In vitro glucose consumption rates were measured using Amplex® Red Galactose/Galactose Oxidase Assay (Invitrogen); cells were seeded in 6-well plates (5 × 105 cells/3 mL media; DMEM supplemented with 10% FBS (DMEM 10%FBS), Stemcell Technologies, Vancouver, BC) and allowed to adhere overnight, washed twice with glucose free DMEM media, then incubated in DMEM 10%FBS containing 6000, 1000, 500 or 200 μM glucose. The remaining glucose concentrations at 0, 6 and 24 hours were determined and expressed as a percentage of the initial glucose concentrations. The hypoxia marker EF5, which is reduced in the absence of oxygen and forms covalent cellular adducts18, was used to evaluate oxygen consumption in a sealed system. Cells (2, 4 and 6 × 106/mL) were suspended in EF5-containing DMEM 10% FBS media (200 μM) and incubated in air-tight tubes for two hours and after staining with anti-EF5 antibody analysed with flow cytometry, as previously described10. The fluorescence intensity of the samples is proportional to the average number of EF5 adducts present in the cell population and is a surrogate measure of oxygen depletion under these conditions.
Magnetic Resonance Imaging (MRI)
In situ tumour perfusion rates were measured using an animal MRI scanner (Bruker, Germany) equipped with a 7T/30 cm horizontal bore magnet. Tumour perfusion measurements were carried out as described previously19. Animals were anaesthetized with isofluorane and positioned in a specially designed holder constructed in-house. The animal’s body temperature was maintained at physiological levels and respiratory rates at ~100 breaths/min during the scan. A catheter was inserted in a tail vein for remote injection of the contrast reagent, Gd-DTPA. A 3 turn solenoid coil surrounding the tumour was used for spin excitation and signal reception. The volume transfer constant (Ktrans) between blood plasma the extra-vascular space in the tumour was calculated from the rapid acquisition of T1-weighted sequences, which monitor the passage of Gd-DTPA in viable tissue, using a two-compartment pharmacokinetic model20. When vessels are highly permeable, as in tumours, Ktrans values approximate blood flow per unit volume of tissue.
Positron Emission Tomography (PET)
The Concorde-CTI microPET®Focus™ 120 scanner21 was used with 18F-fluorodeoxyglucose (FDG), a fluorinated analogue of glucose to evaluate glucose uptake in LCC6Vector and LCC6HER-2 tumours. FDG is taken up by cellular glucose transporters and phosphorylated to FDG-6-phosphate and trapped in the cytoplasm22,23. Regions of interest (ROI) were only drawn on areas of FDG uptake in the PET images to exclude necrotic tissue and the mean activity from three adjacent planes normalized to the ROI area (mm2). Paired sets of mice (5 sets) bearing LCC6Vector and LCC6HER-2 tumours were injected intravenously with FDG and scanned simultaneously after 60 minutes.
Western Blotting
Tumour lysates were analysed by Western blotting as described previously10 using antibodies for HER-2, P-AMPKThr172 and P-ACCSer79 (Cell Signaling Technology) and GLUT1 (Chemicon Int.). Protein expression was reported relative to β-actin in the same lane. Western blot analysis was repeated at least twice for each protein.
Statistics
Statistical analyses were performed with STATISTICA software. One way ANOVA analysis of variance was used to calculate p values. Differences were considered significant at p ≤ 0.05. Images of the original Western blots are shown in the supplementary material.
Results and Discussion
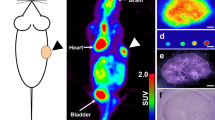
Previously, we showed that HER-2 overexpression in the LCC6 (LCC6HER-2) tumour model increases tissue hypoxia and overall tumour viability without changing vascular parameters; i.e., vessel density, numbers of functional Hoechst 33342 perfused vessels and the median distance of tissue to blood vessels (MDV)10. The classical model of tumour hypoxia is based on an oxygen gradient that emanates outwards from the blood vessel to tissue24; thus an increase in hypoxia without changes in the vascular parameters would imply that factors other than oxygen delivery are at play. A possible reason for this could be impaired tissue perfusion; Ktrans, a surrogate marker of tissue perfusion was thus measured with MRI. The MR images show that LCC6HER-2 tumours have more areas of active blood flow (bright pixels) than LCC6Vector controls (Fig. 1A). The Ktrans values reported here represent the relative blood flow rates in tumour tissue. The median Ktrans value in LCC6HER-2 tumours was on average 58% greater than LCC6Vector tumours (0.059 ± 0.011 and 0.034 ± 0.0041 (n = 4) ml/g tissue/min, respectively) although the difference was not statistically significant (p = 0.14, Fig. 1B). However, these results strongly support the notion that the emergence of hypoxia in LCC6HER-2 tumours is not due to poor blood flow and suggest that oxygen and nutrients may be supplied equally and diffuse the same distance in both tumours. In fact, given the strong trend to higher perfusion in LCC6HER-2 tumours, a decrease in hypoxia might be expected.
Tissue perfusion rates are higher in LCC6HER-2 tumours relative to LCC6Vector tumours.
(A) Tumour perfusion maps of Gd-DTPA concentrations in edge to edge axial slices of LCC6Vector and LCC6HER-2 tumours (top and bottom row, respectively). The bright pixels within the images show areas of tumours which are actively perfused; the intensities correspond to Gd-DTPA concentrations. The areas of perfused tissue are more evenly distributed in LCC6HER-2 tumours compared to LCC6Vector tumours. (B) Ktrans values show a strong trend towards higher perfusion rates in LCC6HER-2(■) tumours compared to LCC6Vector (□) tumours (median ± SD, p = 0.14, n = 4). (C) LCC6Vector (□) and LCC6HER-2 (■) cells were incubated in air-tight containers containing 1% oxygen in the presence of the hypoxia marker EF5 for two hours followed by staining with anti-EF5 antibody and flow cytometric analysis; fluorescence intensity (arbitrary units) indicates the relative amount of EF5 adducts present in the cells (mean ± SD). The graph shows that LCC6HER-2 cells have significantly more adducts than LCC6Vector cells indicating that under these conditions, they consume more oxygen and become hypoxic faster. Significant differences (p < 0.001) are indicated by the asterisk (*).
LCC6HER-2 overexpressing tumours could also develop more hypoxia than controls because of differences in oxygen metabolism. This alternative interpretation is corroborated by the in vitro data showing that LCC6HER-2, compared to the LCC6Vector cells, become more hypoxic in a 1% oxygen environment (Fig. 1C; p < 0.001). The hypoxia marker EF5 is used here as a surrogate measure of oxygen depletion in the closed system. The data show that HER-2 overexpressing cells have higher levels of EF5 staining suggesting they consume more oxygen than the vector controls. In turn, higher oxygen consumption reflects increased rates of oxidative processes (e.g. oxidative metabolism of energy substrates such as glucose or fatty acids) for producing ATP. In a tumour, the demand for oxygen by tumour cells could outstrip the supply and create areas of hypoxia even in vascularized tissue.
Increased hypoxia associated with HER-2 overexpression is not unique to the LCC6 model; overexpressing HER-2 in the MCF7 breast cancer tumour model resulted in higher hypoxia levels as shown with pimonidazole staining of tumour sections and lower tissue oxygenation measured directly using an intratumoural pO2 probe25. Moreover, as in our study, vascular parameters including MDV were similar in HER-2 overexpressing and control MCF7 tumours and inhibiting HER-2 with trastuzumab resulted in better oxygenation25. However, tumour tissue perfusion rates were not measured in this study so the status of oxygen delivery in these tumours is unknown. Interestingly, recent studies reported that the transcription factor NF-κB is an important physiological regulator of mitochondrial respiration26. The authors suggested that increased mitochondrial respiration is a metabolic adaptation to maintain energy homeostasis in growing cells and tumours under conditions where nutrients are in high demand to meet cellular needs of proliferating cells. As previously shown10, LCC6HER-2 cells and tumours express NF-κB at much higher levels than LCC6Vector controls which may account for higher oxidative metabolism in HER-2 overexpressing LCC6 tumours.
In contrast to our results showing the association of HER-2 expression with increased oxygen consumption (Fig. 1C), studies from other groups showed that expression of HER-2 in various cell lines promotes a metabolic shift from mitochondrial respiration (oxidative metabolism) to glycolysis via intermediary molecules15,16,27,28 and the data suggested that HER-2 overexpression was associated with lower oxygen consumption compared to controls15,16,27,28. The different methods of measuring oxygen consumption, culture conditions, cell type specific pathways regulating metabolism in vitro and potential interference from molecular silencing of metabolic pathways used in these studies, however, make a direct comparison between these results15,16,27,28 and ours difficult. Moreover, the in situ oxygenation or hypoxia levels in tumours were not measured in these reports so it is difficult to conclude whether the observed in vitro differences due to HER-2 activity15,16,27 would be present in vivo as well.
To investigate if changes in oxygen consumption associated with HER-2 expression affected glucose metabolism we measured glucose consumption in LCC6HER-2 and LCC6Vector cells. Our results showed that LCC6HER-2 cells have higher glucose consumption in an aerobic environment (Fig. 2). This finding was consistent with the findings published in the same reports referenced above15,16,27,28. Interestingly, in the LCC6 model, the rate of consumption appears to be dependent on the glucose concentration; at 6000 and 1000 μM glucose, significant differences in glucose consumption between LCC6Vector and LCC6HER-2 cells were noted at 24 hours (88% vs. 82%; p = 0.03 and 43% vs. 29%; p = 4.8 × 10−7, for 6000 and 1000 μM, respectively), whereas at 500 and 200 μM, significant differences were apparent by 6 hours (71% vs. 64%; p = 0.001 and 83% vs. 58%; p = 0.001). These data imply that HER-2 overexpressing LCC6 cells may respond faster to changes in its environment, i.e., glucose deprivation.
LCC6HER-2 cells consume more glucose than LCC6Vector cells.
Graphs (A–D) show the consumption of glucose by LCC6Vector (□) and LCC6HER-2 (■) cells over a 24 hour period (results expressed as the percentage of glucose remaining in the media). The rates of glucose consumption are dependent on the glucose present in the media:at 6000 and 1000 μM glucose (A and B, respectively) differences in consumption rates become significant after 24 hours; however at 500 and 200 μM glucose (C and D, respectively), the differences are apparent by 6 hours. Significant differences (p < 0.005) are indicated by the asterisk (*).
Most importantly, we also confirm higher rates of glucose metabolism in vivo by measuring in situ metabolic activity in LCC6HER-2 and LCC6Vector tumours using PET. The data show that FDG signal in LCC6HER-2 is more evenly distributed and more intense than in LCC6Vector tumours (Fig. 3A). The in situ accumulation of FDG per unit area of viable tissue show that LCC6HER-2 tumours utilize on average ~2-fold more glucose than LCC6Vector tumours (2.09 × 10-4 ± 4.26 × 10−5 Bq/mm2 and 9.89 × 10−5 ± 1.4 × 10−5 Bq/mm2 respectively; p = 0.025; n = 5 for each tumour group; Fig. 3B). Malignant tissue is known to use more glucose than normal tissue29 due to the Warburg effect30. Cells transformed with oncogenes, including HER-2, have also been shown to elevate levels of glucose transporters and glucose consumption in vitro, although the molecular pathways involved were not elucidated31,32,33. HER-2 overexpression was also shown to enhance the activation of proteins involved in glycolytic, metabolic, stress-responses and detoxification processes in the breast tumour microenvironment34. Others demonstrated a glycolytic shift based on higher levels of lactate dehydrogenase A in tumours derived from cell lines obtained from mammary gland tumours of neu-transgenic mice28. To the best of our knowledge, we provided the first direct, functional measurements showing increased glucose uptake in vivo in the HER-2 background in a set of otherwise isogenic tumours. It is uncertain to what extent this increase is a reflection of a glycolytic shift in the oxygenated tumour tissue or an increased metabolic activity of a hypoxic fraction present in LCC6HER-2 tumours. However, considering the results showing increased glucose consumption in aerobic conditions in LCC6HER-2 compared to LCC6Vector cells (Fig. 3) it is reasonable to speculate that HER-2 mediates a glycolytic shift in oxygenated tissue in LCC6HER-2 tumours.
FDG uptake is higher in LCC6HER-2 tumours.
(A) PET images of in situ FDG accumulation in LCC6Vector and LCC6HER-2 tumours; the tumour is circled in yellow and the intensity of green areas in the tumour reflects the activity of phosphorylated FDG trapped in cells. In the color scale used, purple is low activity, red is high activity. (B) Total accumulation of FDG present in regions of interest (viable tissue only) drawn on PET images of LCC6Vector (□) and LCC6HER-2 (■) tumours (mean ± SD; p = 0.025, n = 5). Significant differences (p < 0.05) are indicated by the asterisk (*).
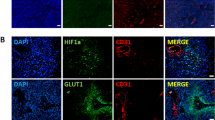
Higher oxygen consumption is also a reflection of a greater ATP demand, thus, a parallel increase in oxidative and glycolytic metabolism would appear to be a versatile and advantageous adaptation to balance bioenergetic stress in HER-2 overexpressing tumours to sustain proliferation and survival of cancer cells. In line with this rationale molecular factors germane to cell bioenergetics were examined. Expression levels of phosphorylated-5′-AMP-activated protein kinase (P-AMPK) the activated form of AMPK, an important regulator of energy processes in cells35 provide further insight into the bioenergetics of the LCC6 tumours. AMPK is phosphorylated when the ratio of AMP to ATP in the cell is high – i.e. when the concentration of ATP is too low to sustain cell functions and the cell enters a state of energy stress36. P-AMPK subsequently activates various pathways that produce energy and minimizes cellular energy expenditure in an overall bid to raise ATP levels35,36. The analysis of activated AMPK (P-AMPK) showed that expression of this protein was lower (~2-fold) in LCC6HER-2 compared to LCC6Vector tumours; levels of phosphorylated Acetyl CoA Carboxylase (P-ACC), a downstream effector of AMPK, also trended lower (Fig. 4A). These data provided further insight into the bioenergetics of the LCC6 tumours and suggested that LCC6HER-2 tumours appear to have sufficient ATP to meet the energy demand in contrast to control LCC6Vector tumours.
(A) Expression levels of P-AMPK, an indicator of energy stress, are lower in LCC6HER-2 relative to LCC6Vector tumours; as expected, the expression levels of a downstream effector of P-AMPK, P-ACC, follow the same trend (P-AMPK: phosphorylated AMPK; P-ACC: phosphorylated ACC). (B) Western blot analysis shows that LCC6HER-2 tumours have lower expression levels of GLUT1, the major transmembrane transporter for glucose, relative to LCC6Vector tumours. Representative results from two tumours are shown in the figure. For clarity, the bands in the figure were cropped from the original gel. Gels were run under the same experimental conditions. The images of the full gels are available in the supplementary material.
The lower energy stress in LCC6HER-2 tumours was also reflected in greatly reduced (~4-fold) expression of glucose transporter-1 (GLUT1) in vivo (Fig. 4B). Since GLUT1 is responsible for transporting glucose (and FDG), across the cell membrane22,23, this result appears counterintuitive considering that FDG uptake was greater in LCC6HER-2 compared to LCC6Vector tumours. However, we argue that low expression levels of P-AMPK in LCC6HER-2 tumours indicate that tumours are replete with ATP and thus well-energized; consequently there is no need to upregulate GLUT1 levels in LCC6HER-2 tumours. In contrast, existing energy stress in LCC6Vector tumours possibly results in sufficient expression of GLUT1 in order to balance energy needs, as described in other systems37. Based on our data it is reasonable to hypothesize that the high uptake of FDG in LCC6HER-2 tumours is not due solely to GLUT1 levels, but also to activation of glycolytic enzymes by HER-234 that shuttle glucose into glycolytic pathways at a higher rate23,38,39. The net result is an increase in overall glucose flux through the cell consistent with the higher levels of FDG uptake in LCC6HER-2 tumours.
In conclusion, we hypothesize, on the basis of the present data, that overexpression of HER-2 in the LCC6 tumour model is associated with higher rates of oxidative metabolism in addition to increased levels of glycolytic activity. Joining these two functions ultimately produces a more hypoxic tumour, but one that efficiently meets its energy demand. Most importantly, the present data establish a link between HER-2 signalling and cellular metabolic processes which warrants further investigation in other tumour models where HER-2 overexpression is clinically relevant. On-going work in our laboratory is focussed on the putative roles that HER-2 activation play in metabolic processes as in the long-term such studies may identify novel metabolic targets for the treatment of HER-2 positive cancers.
Additional Information
How to cite this article: Dragowska, W. H. et al. Overexpression of HER-2 in MDA-MB-435/LCC6 Tumours is Associated with Higher Metabolic Activity and Lower Energy Stress. Sci. Rep. 6, 18537; doi: 10.1038/srep18537 (2016).
References
Slamon, D. J. et al. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 235, 177–182 (1987).
Hynes, N. E. & Lane, H. A. ERBB receptors and cancer: the complexity of targeted inhibitors. Nat Rev Cancer 5, 341–354 (2005).
Arteaga, C. L. et al. Treatment of HER2-positive breast cancer: current status and future perspectives. Nature reviews. Clinical oncology 9, 16–32, 10.1038/nrclinonc.2011.177 (2012).
Kern, J. A. et al. p185neu expression in human lung adenocarcinomas predicts shortened survival. Cancer research 50, 5184–5187 (1990).
Santin, A. D. et al. Overexpression of HER-2/neu in uterine serous papillary cancer. Clinical cancer research: an official journal of the American Association for Cancer Research 8, 1271–1279 (2002).
Wen, Y. et al. HER-2/neu promotes androgen-independent survival and growth of prostate cancer cells through the Akt pathway. Cancer research 60, 6841–6845 (2000).
Yan, M., Parker, B. A., Schwab, R. & Kurzrock, R. HER2 aberrations in cancer: implications for therapy. Cancer treatment reviews 40, 770–780, 10.1016/j.ctrv.2014.02.008 (2014).
Bang, Y. J. et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet 376, 687–697, 10.1016/S0140-6736(10)61121-X (2010).
Neve, R. M. et al. A collection of breast cancer cell lines for the study of functionally distinct cancer subtypes. Cancer Cell 10, 515–527 (2006).
Dragowska, W. H. et al. HER-2/neu overexpression increases the viable hypoxic cell population within solid tumors without causing changes in tumor vascularization. Mol Cancer Res 2, 606–619 (2004).
Coleman, C. N., Mitchell, J. B. & Camphausen, K. Tumor hypoxia: chicken, egg, or a piece of the farm? J Clin Oncol 20, 610–615 (2001).
Gilkes, D. M. & Semenza, G. L. Role of hypoxia-inducible factors in breast cancer metastasis. Future Oncol 9, 1623–1636, 10.2217/fon.13.92 (2013).
Rae, J. M., Creighton, C. J., Meck, J. M., Haddad, B. R. & Johnson, M. D. MDA-MB-435 cells are derived from M14 melanoma cells—a loss for breast cancer, but a boon for melanoma research. Breast Cancer Res Treat 104, 13–19 (2007).
Christgen, M. & Lehmann, U. MDA-MB-435: the questionable use of a melanoma cell line as a model for human breast cancer is ongoing. Cancer biology & therapy 6, 1355–1357 (2007).
Ding, Y. et al. Receptor tyrosine kinase ErbB2 translocates into mitochondria and regulates cellular metabolism. Nature communications 3, 1271, 10.1038/ncomms2236 (2012).
Zhao, Y. H. et al. Upregulation of lactate dehydrogenase A by ErbB2 through heat shock factor 1 promotes breast cancer cell glycolysis and growth. Oncogene 28, 3689–3701, 10.1038/onc.2009.229 (2009).
Warburton, C. et al. Treatment of HER-2/neu overexpressing breast cancer xenograft models with trastuzumab (Herceptin) and gefitinib (ZD1839): drug combination effects on tumor growth, HER-2/neu and epidermal growth factor receptor expression and viable hypoxic cell fraction. Clinical cancer research: an official journal of the American Association for Cancer Research 10, 2512–2524 (2004).
Koch, C. J. Measurement of absolute oxygen levels in cells and tissues using oxygen sensors and 2-nitroimidazole EF5. Methods Enzymol 352, 3–31 (2002).
Kozlowski, P., Wong, J. & Goldenberg, S. L. Serial tumour blood-flow measurements in androgen-dependent and -independent Shionogi tumour models. BJU Int 95, 644–649 (2005).
Tofts, P. S. et al. Estimating kinetic parameters from dynamic contrast-enhanced T(1)-weighted MRI of a diffusable tracer: standardized quantities and symbols. J Magn Reson Imaging 10, 223–232 (1999).
Tai, Y. C. et al. Performance evaluation of the microPET focus: a third-generation microPET scanner dedicated to animal imaging. J Nucl Med 46, 455–463 (2005).
Yamada, K., Brink, I., Bisse, E., Epting, T. & Engelhardt, R. Factors influencing [F-18] 2-fluoro-2-deoxy-D-glucose (F-18 FDG) uptake in melanoma cells: the role of proliferation rate, viability, glucose transporter expression and hexokinase activity. J Dermatol 32, 316–334 (2005).
Moreno-Sanchez, R., Rodriguez-Enriquez, S., Marin-Hernandez, A. & Saavedra, E. Energy metabolism in tumor cells. The FEBS journal 274, 1393–1418, 10.1111/j.1742-4658.2007.05686.x (2007).
Brown, J. M. & Wilson, W. R. Exploiting tumour hypoxia in cancer treatment. Nat Rev Cancer 4, 437–447, 10.1038/nrc1367 (2004).
Hardee, M. E. et al. Her2/neu signaling blockade improves tumor oxygenation in a multifactorial fashion in Her2/neu + tumors. Cancer chemotherapy and pharmacology 63, 219–228, 10.1007/s00280-008-0729-3 (2009).
Mauro, C. et al. NF-kappaB controls energy homeostasis and metabolic adaptation by upregulating mitochondrial respiration. Nature cell biology 13, 1272–1279, 10.1038/ncb2324 (2011).
Eichner, L. J. et al. miR-378(*) mediates metabolic shift in breast cancer cells via the PGC-1beta/ERRgamma transcriptional pathway. Cell metabolism 12, 352–361, 10.1016/j.cmet.2010.09.002 (2010).
Fantin, V. R., St-Pierre, J. & Leder, P. Attenuation of LDH-A expression uncovers a link between glycolysis, mitochondrial physiology and tumor maintenance. Cancer Cell 9, 425–434, 10.1016/j.ccr.2006.04.023 (2006).
Shaw, R. J. Glucose metabolism and cancer. Curr Opin Cell Biol 18, 598–608 (2006).
Warburg, O. On the origin of cancer cells. Science 123, 309–314 (1956).
Flier, J. S., Mueckler, M. M., Usher, P. & Lodish, H. F. Elevated levels of glucose transport and transporter messenger RNA are induced by ras or src oncogenes. Science 235, 1492–1495 (1987).
Sistonen, L., Holtta, E., Lehvaslaiho, H., Lehtola, L. & Alitalo, K. Activation of the neu tyrosine kinase induces the fos/jun transcription factor complex, the glucose transporter and ornithine decarboxylase. J Cell Biol 109, 1911–1919 (1989).
Cairns, R. A., Harris, I. S. & Mak, T. W. Regulation of cancer cell metabolism. Nat Rev Cancer 11, 85–95, 10.1038/nrc2981 (2011).
Zhang, D. H. et al. Proteomic study reveals that proteins involved in metabolic and detoxification pathways are highly expressed in HER-2/neu-positive breast cancer. Mol Cell Proteomics 4, 1686–1696, 10.1074/mcp.M400221-MCP200 (2005).
Hardie, D. G. Minireview: the AMP-activated protein kinase cascade: the key sensor of cellular energy status. Endocrinology 144, 5179–5183 (2003).
Hardie, D. G. The AMP-activated protein kinase pathway—new players upstream and downstream. J Cell Sci 117, 5479–5487 (2004).
Pauwels, E. K. et al. FDG accumulation and tumor biology. Nucl Med Biol 25, 317–322 (1998).
DeBerardinis, R. J., Lum, J. J., Hatzivassiliou, G. & Thompson, C. B. The biology of cancer: metabolic reprogramming fuels cell growth and proliferation. Cell metabolism 7, 11–20, 10.1016/j.cmet.2007.10.002 (2008).
Szablewski, L. Expression of glucose transporters in cancers. Biochimica et biophysica acta 1835, 164–169, 10.1016/j.bbcan.2012.12.004 (2013).
Acknowledgements
The work was supported by Rethink Breast Cancer (DTTY) and the Canadian Breast Cancer Alliance (MB); imaging platforms and infrastructure were funded through the Canadian Institutes of Health Research and the Canadian Foundation for Innovation. The PET and MRI studies were supported in part by the Cancer Research Society Inc. Salary support was provided by The Swiss National Foundation (MG). The authors thank Dana Masin and the members of the Investigational Drug Program for expert technical assistance in animal and imaging studies.
Author information
Authors and Affiliations
Contributions
W.D., M.B. and D.Y. conceived and designed experiments. M.G. helped design experiments and collected data. P.K. and A.Y. carried out M.R. imaging studies. T.R., M.A. and V.S. facilitated PET experiments. W.D., M.G. and D.Y. prepared and wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Dragowska, W., Ginj, M., Kozlowski, P. et al. Overexpression of HER-2 in MDA-MB-435/LCC6 Tumours is Associated with Higher Metabolic Activity and Lower Energy Stress. Sci Rep 6, 18537 (2016). https://doi.org/10.1038/srep18537
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep18537
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.