Abstract
Time calibration derived from the fossil record is essential for molecular phylogenetic and evolutionary studies. Fossil mice and rats, discovered in the Siwalik Group of Pakistan, have served as one of the best-known fossil calibration points in molecular phylogenic studies. Although these fossils have been widely used as the 12 Ma date for the Mus/Rattus split or a more basal split, conclusive paleontological evidence for the nodal assignments has been absent. This study analyzes newly recognized characters that demonstrate lineage separation in the fossil record of Siwalik murines and examines the most reasonable nodal placement of the diverging lineages in a molecular phylogenetic tree by ancestral state reconstruction. Our specimen-based approach strongly indicates that Siwalik murines of the Karnimata clade are fossil members of the Arvicanthini-Otomyini-Millardini clade, which excludes Rattus and its relatives. Combining the new interpretation with the widely accepted hypothesis that the Progonomys clade includes Mus, the lineage separation event in the Siwalik fossil record represents the Mus/Arvicanthis split. Our test analysis on Bayesian age estimates shows that this new calibration point provides more accurate estimates of murine divergence than previous applications. Thus, we define this fossil calibration point and refine two other fossil-based points for molecular dating.
Similar content being viewed by others
Introduction
Divergence time estimates in phylogenetic studies have become increasingly valuable for addressing questions regarding lineage diversification rates, evolutionary patterns and historical biogeography, among others1,2. Fossil data are the most common type of calibration used to estimate absolute node ages for trees. However, inherent problems exist as a source of error when incorporating fossil priors for calibrating trees, including completeness of the fossil record, age accuracy of fossils or strata and correct phylogenetic placement of fossil calibration3,4,5,6. In this study, we use a specimen-based approach to show that one of the most widely-used fossil dates for mammal phylogenies, the Mus/Rattus split (mice/rats divergence)7,8,9, had been placed at the incorrect node.
The paleontological event on which this datum (~12 Ma) is based is the first appearance of the fossil genus Progonomys in geologic time10,11,12. This event evolutionarily represents the first split from the earliest definitive murine, Antemus and the acquisition of derived dental characters of crown Murinae (connections of anterostyle and enterostyle with corresponding medial cusps)10,11,12. Owing to frequent citations of Jacobs and Downs10, fossil murine rodents from the Siwalik Group of Pakistan are among the most prominent paleontological records utilized as fossil calibration (Fig. S1). As paleontologists suggested10,11,12, the Siwalik fossil-based date was widely accepted as the Mus/Rattus split in molecular phylogenetic studies in the 90’s and early 2000’s13,14,15,16,17,18,19,20,21,22. More recently, molecular phylogenetic studies have clarified evolutionary relationships of the Murinae and confirmed that the earliest split within the subfamily is the divergence between the Phloeomyini and core Murinae, followed by the Mus/Rattus split within core Murinae9,22,23,24,25,26,27,28,29,30. As a result, this 12 Ma fossil-based date is now more commonly assigned to the Phloeomys/core Murinae split9,24,26,27,29,30,31. However, accepting the transition from Antemus to Progonomys as a calibration point for the Phloeomys/core Murinae split is entirely dependent on molecular tree topology and direct morphological evidence has not been demonstrated in the fossil record. None of the Siwalik specimens possess derived dental characters (a cordate-shaped anteroconid on lower first molars and a well-developed posterostyle on upper first molars) shared by members of the Phloeomyini32.
The rationale for considering the first appearance of Progonomys as the Mus/Rattus split is based on the simplified phylogenetic hypothesis of Siwalik murine rodents by Jacobs33 and Jacobs and Downs10, which proposed dichotomous lineages (the Progonomys clade and the Karnimata clade), deriving from Antemus (Fig. 1A, see SI Discussion for details). Mus was confidently placed in the Progonomys clade based on observation of gradual morphological change from older Progonomys to younger Mus auctor in Siwalik murines10,33,34. On the other hand, Rattus was placed in the Karnimata clade with some uncertainty10, along with extant genera of the Arvicanthini (Arvicanthis, Pelomys, Mylomys and Golunda), heavily relying on the anterior displacement of the anterostyle10,33,34 (dental terminology in Fig. 1B). No further morphological evidence is found for the evolutionary relationship between Rattus and Siwalik species of the Karnimata clade35.
Phylogenetic hypothesis, dental terminology and lineage separation in Siwalik Murinae.
(A) Simplified phylogenetic hypothesis for Siwalik murine rodents proposed by Jacobs33 and Jacobs and Downs10 (after Kimura et al.34). (B) Dental terminology used in this study. 1. protocone, 2. anterostyle, 3. enterostyle, 4. hypocone. (C) Quantitative assessment of lineage separation in Siwalik murine rodents using geometric morphometric analysis on tooth outline of the upper first molars (details are found in Kimura et al.34).
In more recent studies, we have shown that diverging tooth morphology in the Siwalik lineages was initiated by 11.2 Ma, with decreasing morphological overlap through time34 between the Progonomys and Karnimata clades (Fig. 1C) and that the 11.2 Ma stratigraphic occurrence can be considered as the evidence of lineage separation under the initial split criterion36. In this study, we examine the most reasonable placement of this separation event in a molecular phylogenetic tree by presenting new qualitative characters that define the lineage separation and tracing ancestral states of the characters in a molecular phylogenetic tree of extant species.
Here we show that progressive acquisition of new dental characters in the Karnimata clade is strong evidence for Siwalik murine rodents representing a younger node, the Mus/Arvicanthis split, than previous applications. The finding of this study constrains the timing of the Mus/Arvicanthis split to ~11.2 Ma as a stratigraphic occurrence and to 11.36 Ma by a molecular divergence estimate and further allows revision of calibration dates for two additional clades. These fossil-based dates are exceptionally highly resolved, compared to any other estimate thus far, because the divergence event is morphologically tracked in geologic time and because of well-constrained age estimates of Siwalik localities within both biostratigraphic and paleomagnetic frameworks.
Results
Frequency distributions of the size and inclination of the metacone (Fig. 2A–E) and the presence of the posterostyle were evaluated for each Siwalik species (Fig. 3, Tables S1–S3, Dataset S1). A large metacone that is posteriorly inclined parallel to the paracone (Fig. 2A) is plesiomorphic for the Murinae. In the Karnimata clade, ?Karnimata conserves this plesiomorphic condition in 60% of the specimens at 11.2 Ma (Table S2). By 8.2 Ma, this plesiomorphic condition disappears from the Karnimata clade. Instead, a new combination of character states, a small metacone in vertical orientation (Fig. 2E), appears for the first time in 24% of Karnimata darwini at 9.2 Ma (Fig. 3, Table S2). The frequency of the derived condition (small, vertical) exceeds half of the specimens at 8.8 Ma and reaches 85% by 7.4 Ma. In contrast, the plesiomorphic condition of the metacone persisted in the Progonomys clade (Fig. 3). The frequency of the plesiomorphic condition continuously increases as the frequency of the ‘large, slightly inclined’ state decreases through time. By 8.8 Ma, more than 90% of the specimens have this plesiomorphic condition. None of the specimens in the Progonomys clade have the derived condition of the metacone (Table S3). As expected from the observations in each clade, the correlation between the two characters (size, inclination) is stronger in the Karnimata clade (Goodman-Kruskal’s gamma = −0.76) than in the Progonomys clade (Goodman-Kruskal’s gamma = −0.39). The null hypothesis of independence between the variables was rejected for the Karnimata clade (M2 = 23.9, df = 2, p < 0.0001) but was accepted for the Progonomys clade (M2 = 1.92, df = 2, p = 0.38).
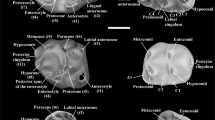
Size and inclination of the metacone on the labial side of the M1 tooth.
(A) As large as the paracone. (B) Smaller than the paracone. (C) Inclined posteriorly parallel to the axis of the paracone. (D) Slightly inclined posteriorly but not parallel to the axis of the paracone. (E) Not inclined posteriorly (=vertical). Colors correspond to those in Fig. 4.
Percent frequency distribution of the size and inclination of the metacone in Siwalik murine rodents.
Species that are likely in ancestor-descendant relationships10,33 are connected by a solid line. Open symbols correspond to those in Fig. 1A and colors of the ‘size and inclination’ legend correspond to those in Fig. 4. Different from the previous phylogenetic hypothesis (Fig. 1A), Progonomys hussaini is interpreted to appear after the initiation of lineage separation34. See Tables S1–S3 for numeric percentages and the number of individuals observed in this study and SI Discussion for age determination. All localities but DP 13 have Y as a prefix.
The analysis of ancestral state reconstruction shows that a combination of the ‘small’ and ‘vertical’ states of the metacone is concentrated in the Arvicanthini (Fig. 4A, Fig. S2 for black-and-white). Maximum likelihood unambiguously placed it as the most probable ancestral state (93.0%) of the metacone for the Arvicanthini. In the node of the Arvicanthini and Otomyini, a small metacone in vertical orientation is predicted as the most probable ancestral state (80.3%) but is marginally insignificant over the second most probable ancestral state, a large metacone in parallel orientation (12.2%, difference of log likelihood = 1.89). For the node of the Arvicanthini, Otomyini and Millardini (hereafter the Arvicanthini-Otomyini-Millardini clade), prediction for vertical orientation of the metacone (71.9%) is greater than that for parallel orientation (26.3%).
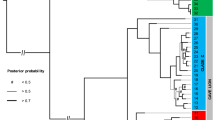
Siwalik murine rodents documenting the Mus/Arvicanthis split and divergence time estimates.
(A) Size and inclination of the metacone, as well as the presence of the posterostyle, in modern murine rodents on a cladogram based on the maximum likelihood tree of Fabre et al.28. Pie charts indicate the probability of ancestral states of the combined characters of the metacone at a given node. Systematic nomenclature of the tribes follows Lecompte et al.22. The “?” state is assigned for the species in which the inclination of the metacone is not recognizable because the metacone is fused with the paracone or hypocone. (B) Maximum clade credibility tree from the BEAST analysis of the Fabre et al.29 data using the 11.2 Ma calibration point for the Mus/Arvicanthis split. Node bars indicate the 95% credible interval of the posterior density of divergence times. Number on the nodes represents the posterior mean of divergence times.
Discussion
Siwalik murines of the Karnimata clade demonstrate progressive acquisition of a derived condition of the metacone (a combination of small size and vertical orientation Fig. 2E), which appeared as a minor variation at 9.2 Ma and became a dominant character state by 8.8 Ma. In contrast, this condition does not occur in any individuals of the Siwalik Progonomys clade. Ancestral state reconstruction suggests that a small meta-cone in vertical orientation is a synapomorphic character for the Arvicanthini-Otomyini-Millardini clade (Fig. 4A). Inside that clade, three arvicanthine genera (Golunda, Mylomys, Stochomys) and the Otomyini secondarily lost the character in acquiring their specialized tooth morphologies (Fig. 4A, see SI Discussion for details and Fig. S3 for photographs). Outside the Arvicanthini-Otomyini-Millardini clade, a vertical metacone was independently acquired by five genera, associated with the development of the posterostyle (Fig. 4A, see SI Discussion for details and Fig. S3 for photographs). In contrast, all genera of the Arvicanthini-Otomyini-Millardini clade (except Thamnomys and Grammomys) and Siwalik species of the Karnimata clade lack the posterostyle. Thus, based on the increased frequency of the derived condition of the metacone in the Karnimata clade (Fig. 3) and a continuous absence of the posterostyle, Siwalik species of the Karnimata clade are placed definitively as members of the Arvicanthini-Otomyini-Millardini clade. Importantly, the possibility of close relationships between the Karnimata clade and the Rattini is rejected. Combining the new observations and the widely-accepted hypothesis that the Progonomys clade includes Mus10,11,33, it is most logical to conclude that the progressive morphological divergence between the Progonomys and Karnimata clades represents the Mus/Arvicanthis split, which is more internal than the Mus/Rattus split at the tribal level. The independent acquisition of a vertically-oriented small metacone in the Apodemini within the Progonomys clade (Fig. 4A) is consistent with the paleontological hypothesis that European species of Progonomys is ancestral to Apodemus37.
Absolute nodal ages were estimated with the 11.2 Ma fossil date of the Mus/Arvicanthis split using the published data of Fabre et al.29 (Figs 4B and S4) and were then compared with the results of Fabre et al.29 (Tables S4). In three separate analyses of Fabre et al.29, the 12 Ma fossil date (i.e., the first appearance of Progonomys) was applied to two older nodes (the Mus/Rattus split and the Phloeomys/core Murinae split, respectively) and was excluded from analysis for cross-validation of the fossil constraint. Newly calibrated ages in this study are consistently older than the three applications of Fabre et al.29 and our molecular estimates are more congruent with paleontological evidence compared to any previous studies9,22,26,29,30 (Table S4). For example, the posterior mean age for the Phloeomys/core Murinae split (i.e., the node of the crown Murinae) estimated to be 13.6 Ma matches with the fossil evidence (13.8 Ma) of the first definitive murine rodents, which is outside the 95% CI of Fabre et al.29 (Fig. 4). This new calibration point for the most diverse group of modern mammals is unique in that a lineage separation event is identified based on morphological divergence, rather than the stratigraphic occurrence of the most basal taxa and that the rich fossil record within a magnetostratigraphic framework constrains the bounding of the molecular divergence dating.
Based on our results, we explicitly define the lineage separation event of the Siwalik murines as a fossil calibration point of the Mus/Arvicanthis split and newly refine two fossil-dates for the Arvicanthini-Otomyini-Millardini clade and for the Murini, using Siwalik fossils. We follow Parham et al.38, who proposed a standard system for introducing new fossil calibrations, including a series of steps with an emphasis on the use of museum specimens to clarify the phylogenetic position of the fossil calibration.
The Mus/Arvicanthis split
Referred Specimens — M1 specimens from localities Y791 and Y797, which are assigned to ?Karnimata.
Paleontological Event — The first appearance of ?Karnimata, interpreted to be close to the initiation of lineage separation between the Progonomys and Karnimata clades36 (Fig. 1C).
Stratigraphic Occurrence (Age) — Localities Y791 and Y797 (median: 11.2 Ma); Nagri Formation, Siwalik Group, Potwar Plateau, northern Pakistan.
Age Determination — Magnetic polarity stratigraphy correlated to the Geomagnetic Polarity Time Scale of Ogg and Smith39.
Minimum Age Constraints — 11.1 Ma; the upper ages of Y791 and Y797, associated with the first appearance of ?Karnimata.
Maximum Age Constraints — 12.3 Ma; the lower age of locality Y634 (median: 12.3 Ma), associated with the first appearance of Progonomys.
Reference — This study and Kimura et al.36.
Most recent common ancestor of the Arvicanthini, Otomyini and Millardini
Referred Specimen — M1 specimens of Karnimata darwini from Y182 that have a small metacone in vertical orientation.
Paleontological Event — The first occurrence of the derived condition (small, vertical) of the metacone in a chronoclinal assemblage of the Karnimata clade (Fig. 3).
Stratigraphic Occurrence (Age) — Locality Y182 (median: 9.2 Ma); Dhok Pathan Formation, Siwalik Group, Potwar Plateau, northern Pakistan.
Age Determination — Magnetic polarity stratigraphy correlated to the Geomagnetic Polarity Time Scale of Ogg and Smith39.
Minimum Age Constraints — 8.7 Ma; the upper age of Y388 (median: 8.8 Ma), associated with >50% frequency occurrence of the derived condition of the metacone in Karnimata sp. (Fig. 3).
Maximum Age Constraints — 10.1 Ma; the lower age of locality Y311 (median: 10.1 Ma), which is stratigraphically the youngest locality with no occurrence of the derived condition of the metacone (Fig. 3).
Reference — This study.
Most recent common ancestor of the Murini
Referred Specimen — Lower m1 specimen of Mus sp. from Y547.
Paleontological Event — The first appearance of the genus Mus in geologic time.
Stratigraphic Occurrence (Age) — Locality Y547 (median: 8.0 Ma); Dhok Pathan Formation, Siwalik Group, Potwar Plateau, northern Pakistan.
Age Determination — Magnetic polarity stratigraphy correlated to the Geomagnetic Polarity Time Scale of Ogg and Smith39.
Minimum Age Constraints — 7.3 Ma; the upper age of Y931 (median: 7.4 Ma), which yields more Mus specimens than Y547.
Maximum Age Constraints — 8.3 Ma; the lower age of locality Y24 (median: 8.2 Ma), which is stratigraphically the youngest locality with abundant murine specimens, yet lacking Mus specimens.
Other note — Stable isotope values are associated with the specimen40.
Reference — This study and Kimura et al.40.
Paleomagnetic and biostratigraphic studies in the Siwalik group designate each small mammal fossil locality as belonging to a chronologically controlled stratigraphic bin of 100,000 years maximum duration (see SI Discussion for details). The bins were constructed by subdividing stratigraphic intervals between magnetochron boundaries41,42. This dating precision to ≤100,000 years, in combination with the fine-scale fossil record, is the great strength of Siwalik Muridae for molecular clock calibration studies, providing narrow intervals for the new fossil calibrations.
While the usage of molecular divergence dating is vast, a priori evaluation of fossil calibration quality is crucial for accurate Bayesian estimates of divergence times2,3,4,5,6. A comparative simulation study shows that an a posteriori cross-validation approach may select highly precise but inaccurate calibration points that are not consistent with fossil evidence5. Our results better corroborate paleontological estimates and greatly reduce the range of the prior age for the calibration point from the 95% credible interval of ~10 million years29, an interval nearly equaling to most of Murine evolutionary history (~14 million years), to that of ~2 million years. We further emphasize the fundamental importance of a well-dated and documented fossil record for evolutionary questions concerning modern organisms that rely on molecular time estimates. Such questions include but are not limited to diversification rates, evolutionary patterns and biogeographic events, which are influenced by geological processes and climate changes through time.
Methods
Fossil specimens (n = 272) of upper first molars (M1), ranging in age from 14.3 to 6.5 Ma, were examined in this study (Dataset S1). They were collected from the Potwar Plateau, northern Pakistan, in the 1970’s to 2000 and are currently on long-term loan from the Geological Survey of Pakistan at the Peabody Museum of Archaeology and Ethnology, Harvard University. We follow Kimura et al.36 for systematic classification of Siwalik murines. Ages of the Siwalik localities derived from paleomagnetic stratigraphy are based on Geomagnetic Polarity Time Scale 200443. Modern specimens (n = 500) examined in this study (Dataset S2) include individuals of 70 genera and 79 species, comprising 54% (70/130) of the total genera of Murinae and 79% (31/39) of the genera within the Mus/Arvicanthis split22,29,44. These specimens are curated in the Museum of Comparative Zoology (MCZ), Harvard University and in the Smithsonian Institution National Museum Natural History (USNM). Digital images of upper molars of these species are provided in Fig. S3.
First, we examined change in the frequency distribution of the size and inclination of the metacone relative to the paracone on M1 of the Siwalik fossil species. Schematic diagrams of the character states, to which each specimen was referred for scoring characters, are shown in Fig. 2. The size of the metacone was observed as the width of the metacone on the labial side of the tooth relative to that of the paracone, scored as (a) as large as the paracone or (b) smaller than the paracone (Fig. 2A,B). The inclination of the metacone was scored as the axis of the metacone is (c) inclined posteriorly parallel to that of the paracone, (d) slightly inclined posteriorly but not parallel to that of the paracone and (e) not inclined posteriorly (=vertical) (Fig. 2C–E). The size of the posterostyle was scored relative to that of the enterostyle in occlusal view. These character states were also scored for modern species. We preferred the qualitative assessments, rather than quantitative measurements, in this study because large-scale systematic differences were expected to appear as fixed characters. For the Karnimata and Progonomys clades, independence and correlation of the two metacone characters were tested and measured by a generalized Cochran-Mantel-Haenszel (CMH) test and Goodman-Kruskal’s gamma.
Then, we traced ancestral states of these metacone characters on the maximum likelihood (ML) tree of Fabre et al.28 by the ML method of ancestral state reconstruction in Mesquite 2.7545. Topologies of molecular trees are largely congruent for Murinae22,24,27,28,30, but the chronogram of Fabre et al.28 most comprehensively covers murine species to date. The Markov k-state 1 parameter model (Mk1) was chosen, which takes the rate of character change as a single parameter and assumes equal probability for any particular character change. The likelihood decision threshold of 2.0 was adopted as a cutoff for the significance of the likelihood ratio between two character states. Eight genera (Thamnomys, Dephomys, Hapalomys, Papagomys, Echiothrix, Lenothrix, Margaretamys, Pithecheir) were excluded from the ancestral character state reconstruction due to low bootstrap values (<70%) on their nodes.
Finally, we applied the new fossil calibration point (the Mus/Arvicanthis split) to a published dataset to test whether the new calibration point gives more accurate estimates of absolute nodal ages than previous applications. The data of Fabre et al.29 were chosen for the test analysis because they provide detailed comparisons of estimated ages obtained by different applications of the 12 Ma fossil date. In our analysis, we replaced these calibration points with the Mus/Arvicanthis split. The fossil date of the Apodemurini was excluded because its credible interval (CI) greatly overlaps with the newly proposed date for the Mus/Arvicanthis split. All other conditions were identical to those in Fabre et al.29. The analysis was conducted in BEAST (v 1.8.0)46 via the CIPRES Science Gateway47. We bounded the lower and upper limits of this prior (median age: 11.63 Ma, 95% CI: 11.16–14.03 Ma) based on the stratigraphic occurrence of ?Karnimata and Progonomys hussaini at the lower bound and the stratigraphic occurrence of “near Progonomys” at the upper bound, respectively, as described in the Discussion. We set the minimum age of the fossil locality in the 5% quantile of the lognormal curve. SI Materials and Methods provide further details of the methods used in this study.
Additional Information
How to cite this article: Kimura, Y. et al. Corrected placement of Mus-Rattus fossil calibration forces precision in the molecular tree of rodents. Sci. Rep. 5, 14444; doi: 10.1038/srep14444 (2015).
References
Ho, S. Y. W. & Phillips, M. J. Accounting for calibration uncertainty in phylogenetic estimation of evolutionary divergence times. Syst. Biol. 58, 367–80 (2009).
Ho, S. Y. W. & Duchêne, S. Molecular-clock methods for estimating evolutionary rates and timescales. Mol. Ecol. 23, 5747–5965 (2014).
Graur, D. & Martin, W. Reading the entrails of chickens: molecular timescales of evolution and the illusion of precision. Trends Genet. 20, 80–86 (2004).
Donoghue, P. C. J. & Benton, M. J. Rocks and clocks: calibrating the Tree of Life using fossils and molecules. Trends Ecol. Evol. 22, 424–431 (2007).
Warnock, R. Calibration uncertainty in molecular dating analyses: there is no substitute for the prior evaluation of time priors. Proc. R. Soc. London B 282, 20141013 (2015).
Dos Reis, M. & Yang, Z. The unbearable uncertainty of Bayesian divergence time estimation. J. Syst. Evol. 51, 30–43 (2013).
Benton, M. J. & Donoghue, P. C. J. Paleontological evidence to date the tree of life. Mol. Biol. Evol. 24, 26–53 (2007).
Benton, M. J., Donoghue, P. C. J. & Asher, R. J. in The Timetree of Life (eds. Hedges, S. B. & Kumar, S. ) 35–86 (Oxford University Press, 2009).
Steppan, S., Adkins, R. M. & Anderson, J. Phylogeny and divergence-date estimates of rapid radiations in muroid rodents based on multiple nuclear genes. Syst. Biol. 53, 533–553 (2004).
Jacobs, L. L. & Downs, W. R. in Rodent and Lagomorph Families of Asian Origins and Diversification (eds. Tomida, Y., Li, C. & Setoguchi, T. ) 149–156 (National Science Museum Monographs 8, 1994).
Jacobs, L. L. & Flynn, L. J. in Interpreting the Past: Essays on Human, Primate and Mammal Evolution in Honor of David Pilbeam (eds. Lieberman, D. E., Smith, R. J. & Keller, J. ) 63–80 (Brill Academic Publishers, 2005).
Jacobs, L. L. & Pilbeam, D. Of mice and men: Fossil-based divergence dates and molecular ‘clocks’. J. Hum. Evol. 9, 551–555 (1980).
She, J. X., Bonhomme, F., Boursot, P., Thaler, L. & Catzeflis, F. Molecular phylogenies in the genus Mus: Comparative analysis of electrophoretic, scnDNA hybridization and mtDNA RFLP data. Biol. J. Linn. Soc. 41, 83–103 (1990).
Chevret, P., Denys, C., Jaeger, J.-J., Michaux, J. & Catzeflis, F. Molecular and palaeontological aspects of the tempo and mode of evolution in Otomys (Otomyinae: Muridae: Mammalia). Biochem. Syst. Ecol. 21, 123–131 (1993).
Chevret, P., Veyrunes, F. & Britton-Davidian, J. Molecular phylogeny of the genus Mus (Rodentia: Murinae) based on mitochondrial and nuclear data. Biol. J. Linn. Soc. 84, 417–427 (2005).
Smith, M. F. & Patton, J. L. Phylogenetic relationships and the radiation of sigmodontine rodents in South America: evidence from cytochrome b. J. Mamm. Evol. 6, 89–128 (1999).
Ducroz, J. F., Volobouev, V. & Granjon, L. An assessment of the systematics of arvicanthine rodents using mitochondrial DNA sequences: evolutionary and biogeographical implications. J. Mamm. Evol. 8, 173–206 (2001).
Michaux, J., Reyes, A. & Catzeflis, F. Evolutionary history of the most speciose mammals: molecular phylogeny of muroid rodents. Mol. Biol. Evol. 18, 2017–2031 (2001).
Salazar-Bravo, J., Dragoo, J. W., Tinnin, D. S. & Yates, T. L. Phylogeny and evolution of the neotropical rodent genus Calomys: inferences from mitochondrial DNA sequence data. Mol. Phylogenet. Evol. 20, 173–184 (2001).
Huchon, D. et al. Rodent phylogeny and a timescale for the evolution of Glires: evidence from an extensive taxon sampling using three nuclear genes. Mol. Biol. Evol. 19, 1053–1065 (2002).
Suzuki, H., Shimada, T., Terashima, M., Tsuchiya, K. & Aplin, K. Temporal, spatial and ecological modes of evolution of Eurasian Mus based on mitochondrial and nuclear gene sequences. Mol. Phylogenet. Evol. 33, 626–646 (2004).
Lecompte, E. et al. Phylogeny and biogeography of African Murinae based on mitochondorial and nuclear gene sequences, with a new tribal classification of the subfamily. BMC Evol. Biol. 8, 199 (2008).
Watts, C. & Baverstock, P. Evolution in the Murinae (Rodentia) assessed by microcomplement fxation of albumin. Aust. J. Zool. 43, 105–118 (1995).
Steppan, S. J., Adkins, R. M., Spinks, P. Q. & Hale, C. Multigene phylogeny of the Old World mice, Murinae, reveals distinct geographic lineages and the declining utility of mitochondrial genes compared to nuclear genes. Mol. Phylogenet. Evol. 37, 370–388 (2005).
Jansa, S. A. & Weksler, M. Phylogeny of muroid rodents: relationships within and among major lineages as determined by IRBP gene sequences. Mol. Phylogenet. Evol. 31, 256–276 (2004).
Jansa, S. A., Barker, F. K. & Heaney, L. R. The pattern and timing of diversification of Philippine endemic rodents: evidence from mitochondrial and nuclear gene sequences. Syst. Biol. 55, 73–88 (2006).
Rowe, K. C., Reno, M. L., Richmond, D. M., Adkins, R. M. & Steppan, S. J. Pliocene colonization and adaptive radiations in Australia and New Guinea (Sahul): multilocus systematics of the old endemic rodents (Muroidea: Murinae). Mol. Phylogenet. Evol. 47, 84–101 (2008).
Fabre, P.-H., Hautier, L., Dimitrov, D. & Douzery, E. J. P. A glimpse on the pattern of rodent diversification: a phylogenetic approach. BMC Evol. Biol. 12, 88 (2012).
Fabre, P. et al. A new genus of rodent from Wallacea (Rodentia: Muridae: Murinae: Rattini) and its implication for biogeography and Indo-Pacific Rattini systematics. Zool. J. Linn. Soc. 169, 408–447 (2013).
Schenk, J. J., Rowe, K. C. & Steppan, S. J. Ecological opportunity and incumbency in the diversification of repeated continental colonizations by muroid rodents. Syst. Biol. 62, 837–64 (2013).
Rowe, K. C., Aplin, K. P., Baverstock, P. R. & Moritz, C. Recent and rapid speciation with limited morphological disparity in the genus Rattus. Syst. Biol. 60, 188–203 (2011).
Musser, G. G. & Heaney, L. Philippine rodents: definitions of Tarsomys and Limnomys plus a preliminary assessment of phylogenetic patterns among native Philippine murines (Murinae, Muridae). Bull. Am. Museum Nat. Hist. 211, 1–138 (1992).
Jacobs, L. L. Fossil rodents (Rhizomyidae and Muridae) from Neogene Siwalik deposits, Pakistan. Museum North. Arizona Press Bull. Ser. 52, 1–103 (1978).
Kimura, Y., Jacobs, L. L. & Flynn, L. J. Lineage-specific responses of tooth shape in murine rodents (Murinae, Rodentia) to late Miocene dietary change in the Siwaliks of Pakistan. PLoS One 8, e76070 (2013).
Patnaik, R. Phylogeny of Siwalik murine rodents: Implications for Mus-Rattus divergence time. J. Paleontol. Soc. India 59, 15–28 (2014).
Kimura, Y., Flynn, L. J. & Jacobs, L. L. A paleontological case study for species delimitation in diverging fossil lineages. Hist. Biol. (2015).
Martín-Suárez, E. & Mein, P. Revision of the genera Parapodemus, Apodemus, Rhagamys and Rhagapodemus (Rodentia, Mammalia). Geobios 31, 87–97 (1998).
Parham, J. F. et al. Best practices for justifying fossil calibrations. Syst. Biol. 61, 346–359 (2012).
Ogg, J. G. & Smith, A. G. in A Geologic Time Scale 2004 (eds. Gradstein, F. M., Ogg, J. G. & Smith, A. G. ) 63–86 (Cambridge University Press, 2004).
Kimura, Y. et al. Fossil mice and rats show isotopic evidence of niche partitioning and change in dental ecomorphology related to dietary shift in Late Miocene of Pakistan. PLoS One 8, e69308 (2013).
Barry, J. C., et al. Faunal and environmental change in the late Miocene Siwaliks of northern Pakistan. Paleobiol. Mem. 3, 1–71 (2002).
Flynn, L.J., et al. The Siwaliks of Pakistan: Time and faunas in a Miocene terrestrial setting. J. Geol. 98, 589–604 (1990).
Gradstein, F. M., Ogg, J. G. & Grastein, F. M. Geologic Time Scale 2004–why, how and where next! Lethaia 37, 175–181 (2004).
Musser, G. G. & Carleton, M. D. in Mammal species of the World: A taxonomic and Geographic Reference (eds. Wilson, D. E. & Reeder, D. M. ) 894–1531 (Johns Hopkins University Press, 2005).
Maddison, W. P. & Maddison, D. R. Mesquite: a modular system for evolutionary analysis. version 2.75. (2011).
Drummond, A. J., Suchard, M. A., Xie, D. & Rambaut, A. Bayesian phylogenetics with BEAUti and the BEAST 1.7. Mol. Biol. Evol. 29, 1969–1973 (2012).
Miller, M. A., Pfeiffer, W. & Schwartz, T. Creating the CIPRES science gateway for inference of large phylogenetic trees. Proc. Gatew. Comput. Environ. Work. (GCE), New Orleans, LA 14, 1–8 (2010).
Acknowledgements
This research was supported by the Institute for the Study of Earth and Man (travel grants to YK) at Southern Methodist University, the Society of Vertebrate Paleontology (Mary R. Dawson Grant to YK) and the Geological Society of America (Graduate Student Research Grant to YK). Specimens were collected mainly under grants to David Pilbeam. We thank James E. Brooks for constructive suggestions and logistical advice. We would like to acknowledge Rajeev Patnaik for observations on a specimen of Cremnomys and Pierre-Henri Fabre for providing his tree data. YK is grateful to Judith M. Chupasko, Mark D. Omura and Darrin Lunde for access to specimens at MCZ and USNM. The anonymous reviewers provided useful comments to improve the quality of the manuscript.
Author information
Authors and Affiliations
Contributions
Y.K. – designed the study, performed the analyses, interpreted the results, prepared the manuscript. MTRH – performed the analyses, prepared the manuscript. M.M.M. – interpreted the results, prepared the manuscript. L.L.J. and L.J.F. – interpreted the results, prepared the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Kimura, Y., Hawkins, M., McDonough, M. et al. Corrected placement of Mus-Rattus fossil calibration forces precision in the molecular tree of rodents. Sci Rep 5, 14444 (2015). https://doi.org/10.1038/srep14444
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep14444
This article is cited by
-
Molecular phylogeny and historical biogeography of Iranian murids (Rodentia: Muridae)
Mammalian Biology (2024)
-
Conservation and divergence of canonical and non-canonical imprinting in murids
Genome Biology (2023)
-
Species and genetic diversity of Bandicota (Murinae, Rodentia) from Myanmar based on mitochondrial and nuclear gene sequences
Mammal Research (2020)
-
First levantine fossil murines shed new light on the earliest intercontinental dispersal of mice
Scientific Reports (2019)
-
Similarities and differences in patterns of germline mutation between mice and humans
Nature Communications (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.