Abstract
Virus-vector relationships can be complex and diverse as a result of long-term coevolution. Understanding these interactions is crucial for disease and vector management. Rice stripe virus (RSV) is known to be transovarially transmitted within its vector, Laodelphax striatellus, and causes serious rice stripe disease. In RSV-infected L. striatellus, we found contrasting changes in vector fecundity, physiology, primary endosymbionts (i.e. yeast-like symbionts, YLS) and feeding behavior that can interact to affect the spread of RSV. RSV-infected L. striatellus exhibited a significant decrease in fecundity that could lead a reduction of viruliferous individuals in populations. As a potential response to this loss, RSV infection also significantly shortened nymphal stage duration, which can strengthen RSV vertical circulation in L. striatellus populations and promote RSV spreading by adult migration and dispersal. Down-regulated JHAMT and up-regulated CYP307A1 in the juvenile hormone and ecdysteroid pathways, respectively, were linked to accelerated development. RSV-infected adults were also found to have higher body weight in conjunction with increased YLS abundance. Furthermore, prolonged host plant phloem exposure to salivation by RSV-infected adults should further enhance RSV horizontal transmission. Our study highlights potential strategies of RSV in enhancing its transmission and provides new insights into the complexity of virus-vector interactions.
Similar content being viewed by others
Introduction
Approximately 80% of plant viruses are known to spread through insect vectors1. The physiology, behavior and even endosymbionts of insect vectors can have profound ecological and evolutionary implications for the transmission of a plant virus to a new host. The migratory small brown planthopper, Laodelphax striatellus, is an insect with a high dispersal ability that has been an increasing threat to rice yield since the 1960s and acts as the main insect vector of rice stripe virus (RSV)2,3,4. As a member of the genus Tenuivirus, RSV is persistent across generations and transovarially transmitted in a circulative-propagative manner in L. striatellus5. Its transmission to rice has caused severe reductions in yield over the last few decades in China. It was reported that RSV ribonucleoproteins are distributed throughout the ovum, lumen and epithelial cells of midgut and even muscles6 and consistent with its transovarial transmission, RSV particles are also found in follicular cells of ovarioles of L. striatellus6. Repeated transovarial passage of RSV was demonstrated to last for 40 generations in planthoppers and L. striatellus females were more efficient in RSV transmission than males7. Nevertheless, many fundamental questions about RSV-L. striatellus interactions remain to be addressed including the nature and consequences (if any) of other phenotypic effects of RSV infection on L. striatellus.
Recently vitellogenin (Vg) was demonstrated to play a critical role in assisting the virus to enter nurse cells in the germarium in L. striatellus via Vg-mediated endocytosis8. After incorporating into oocytes, Vg is stored in a crystalline form of Vitellin (Vn) and reserved as a food source for the future embryo9. Thus, Vg is very important for the propagation and transmission of RSV due to the direct relationship between Vg production and the vector’s fecundity and abundance at the population level. Importantly, most hemipteran insects use only juvenile hormone (JH) to regulate Vg gene transcription10. JH and the main ecdysteroid, 20-hydroxy-ecdysone (20E), are highly versatile insect hormones that coordinate development, growth and reproduction of insects11. It has also been reported that 20E and JH play critical roles in innate immunity12,13. Therefore, in this study we use expression patterns of three genes, JHAMT in the juvenile hormone pathway, CYP307A1 in the ecdysteroid pathway and Vg to assess the potential immune response to RSV infection as well as the effects of RSV infection on the development and fecundity of L. striatellus at the transcriptional level.
The potential for interactions between RSV and yeast-like symbionts (YLS) in the L. striatellus system remains an open question. As primary endosymbionts that are critical to L. striatellus biology, YLS are also transovarially transmitted in L. striatellus14,15,16. YLS provide their host with nutritional benefits and their absence can have adverse impacts on planthopper performance17,18,19. For instance, L. striatellus can obtain sterols from those synthesized by YLS18,19,20, whereas insects that do not harbor YLS have to ingest exogenous sterols from food21 that are necessary for cellular membrane structures and as precursors for ecdysteroids20,22. As YLS and RSV are both located in the same tissues6 and transovarially transmitted in L. striatellus8,15, we tested for potential interactions between them during the life cycle of L. striatellus.
Evidence currently indicates that the relationships between a virus and vector can be either mutualistic23,24 or antagonistic25,26 and the virus may affect the vector’s physiology and behavior to influence its likelihood of transmission27,28,29,30,31. Nevertheless, the lack of comprehensive and integrated consideration of vector physiology and behavior after virus infection has limited our understanding of the evolutionary significance and mechanisms underlying their interactions. In this study, we systemically examined the changes in physiology (including development, body weight and fecundity), primary endosymbionts (i.e. YLS) and feeding behavior of RSV-infected L. striatellus to more comprehensively understand the interactions in the RSV-L. striatellus-YLS system.
Results
Effects of RSV infection on L. striatellus egg hatching and nymphal stage
There was no significant difference in hatching period between the viruliferous population and the non-viruliferous strains (means: 8.20 vs. 8.35; P = 0.34; see Supplementary Table S2 and Supplementary Fig. S2 online). However, RSV infection significantly shortened the duration of the 1st (P = 0.001), 3rd (P = 0.03), 4th (P = 0.01), 5th (P = 0.005) and total nymphal stages (P < 0.001). Significant effects of sex on the duration of the 2nd (P < 0.001), 3rd (P = 0.047), 5th (P = 0.03) and total nymphal stages (P < 0.001) were also found. RSV infection significantly reduced the 5th instar and total nymphal stage development time by 10.78% and 6.69%, respectively, for females and 7.17% and 6.69% for males (all differences P < 0.05; Table 1, Fig. 1). Moreover, for males, the 1st and 3rd instar durations were significantly shortened by 7.87% and 9.23%, respectively (all differences P < 0.05; Fig. 1). Among RSV-infected 3rd instar nymphs, the male duration was significantly shorter than the female duration (mean difference: -7.87%; P < 0.05; Fig. 1), while no significant difference was found between the male and female durations in the absence of RSV infection (P > 0.05; Fig. 1). Among the 5th instar nymphs, the only significant difference was a shorter developmental period for males versus females in the absence of RSV infection (mean difference: -10.40%; P < 0.05; Fig. 1).
Durations of the 1st-5th instar nymphs of females and males of L. striatellus with (RSV) and without RSV (No RSV) infection.
n = 184 and 181 for the infected females and males, respectively; n = 156 and 166 for the uninfected females and males, respectively. Only significant differences are marked with letters. Different lowercase and uppercase letters show significant differences between the infected and uninfected L. striatellus for females or males and between females and males for the infected or uninfected L. striatellus by LSD test at P < 0.05.
Dynamic expression levels of genes in ecdysteroid and juvenile hormone (JH) pathways
Given that there were significant differences in the duration of the 5th instar between L. striatellus with and without RSV infection for both females and males, we examined the expression of two key genes in the KEGG insect hormone biosynthesis pathway, CYP307A1 in the ecdysteroid pathway and JHAMT in the JH pathway, across this developmental stage. We measured expression levels at 0 h, 24 h, 48 h, 60 h and 72 h after molting into the 5th instar. For both CYP307A1 and JHAMT, we found significant main effects of RSV infection (CYP307A1: P < 0.001; JHAMT: P = 0.02), sampling time (P < 0.001; P < 0.001) and their interactions (P = 0.004; P < 0.001) on gene expression levels (see Supplementary Table S3 online). Figure 2 shows that CYP307A1 expression levels were significantly higher in nymphs with RSV infection at 0 h (mean difference: +157.15%), 48 h (mean difference: +31.26%), 60 h (mean difference: +77.78%) and 72 h (mean difference: +101.98%) after molting (all differences P < 0.05; Fig. 2). Differences in CYP307A1 expression levels between 5th instar nymphs with and without RSV infection were not significant at 24 h (mean relative expression: 0.77 vs. 0.87) (P > 0.05; Fig. 2). JHAMT expression levels were significantly higher in nymphs with RSV infection at 0 h immediately after molting (mean difference: +65.04%), but significantly lower at 60 h (mean difference: –36.10%) and 72 h (mean difference: –43.35%) after molting (all differences P < 0.05; Fig. 2). Differences in JHAMT expression levels between 5th instar nymphs with and without RSV infection were not significant at 24 h (mean relative expression: 0.86 vs. 1.08) and 48 h (mean relative expression: 1.66 vs. 1.70) (both comparisons P > 0.05; Fig. 2).
The temporal dynamics of gene expression levels of CYP307A1 and JHAMT at 0 h, 24 h, 48 h, 60 h and 72 h after molting in the 5th instar nymphs of L. striatellus, with (RSV) and without RSV (No RSV) infection.
Thirty individual 5th instar nymphs were randomly mixed as one sample for each sampling time with three repeats. Only significant differences are marked with letters. Different lowercase showed significant differences between the infected and uninfected L. striatellus with RSV by LSD test at P < 0.05.
Effects of RSV infection on the adult weight of L. striatellus
Significant effects of RSV infection (P = 0.001) and sex (P < 0.001) on the body weight of newly emerged adult were found, but no significant interaction between RSV infection and sex was observed (P = 0.63) (see Supplementary Table S2 online). Compared with non-viruliferous individuals, RSV infection significantly increased body weight of the newly emerged female adults (mean difference: +6.82%; P < 0.05; Table 1). Newly emerged RSV-infected male adults were also heavier than non-RSV males (means: 0.689 mg vs. 0.677 mg), but the difference was not significant (P > 0.05; Table 1). Overall, the body weight of newly emerged female adults was significantly greater than that of newly emerged male adults both with (mean difference: +18.29%) and without (mean difference: +12.70%) RSV infection (both comparisons P < 0.05; Table 1).
Effects of RSV infection on L. striatellus female fecundity and Vg gene expression
RSV infection significantly affected the number of eggs laid per female (P = 0.007; see Supplementary Table S2 online) as well as the gene expression level of Vg in newly emerged adult females (P = 0.04; see Supplementary Table S3 online). The mean number of eggs laid per female was significantly reduced by 15.05% in RSV-infected individuals compared to those without RSV infection (P < 0.05; Fig. 3A). The level of Vg gene expression in newly emerged female adults with RSV infection was significantly lower than that of L. striatellus females without RSV infection (mean difference: -66.32%; P < 0.05; Fig. 3B).
The number of eggs laid per female adults (A) and the relative transcript levels of Vg in newly emerged female adults (B) of L. striatellus with (RSV) and without RSV (No RSV) infection.
n = 15 for both the infected and uninfected female adults in panel A; Ten newly emerged female adults were randomly mixed as one sample with nine repeats for each treatment in panel B. Only significant differences are marked with letters. Different lowercase letters showed significant differences between the L. striatellus with RSV infection and those without RSV infection by LSD test at P < 0.05.
Effects of RSV infection on the abundance of YLS in L. striatellus adults
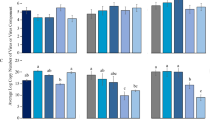
The relative and absolute abundances of YLS harbored in newly emerged adults of L. striatellus were significantly affected by RSV infection (P < 0.001 for both relative and absolute abundance), sex (P < 0.001 for both relative and absolute abundance) and their interactions (relative abundance: P = 0.01; absolute abundance: P < 0.001) (see Supplementary Table S2 online). RSV infection significantly increased the abundance of YLS in female adults (mean difference relative abundance: +23.15%; absolute abundance: +33.02%) (both comparisons P < 0.05; Fig. 4), but it only marginally increased the abundance of YLS in male adults (mean difference relative abundance: +4.71%; absolute abundance: +10.58%) (both comparisons P > 0.05; Fig. 4). Overall, YLS abundances in female adults were significantly higher than in male adults both with RSV (mean difference relative abundance: +71.91%; absolute abundance: +53.06%) and without RSV infection (mean difference relative abundance: +45.88%; absolute abundance: +76.92%) (all comparisons P < 0.05; Fig. 4).
The relative and absolute abundance of yeast-like symbionts (YLS) harbored in female and male adults of L. striatellus with (RSV) and without RSV (No RSV) infection.
n = 184 and 181 for the infected female and male adults, respectively; n = 156 and 166 for the uninfected female and male adults, respectively. Only significant differences are marked with letters. Different lowercase and uppercase letters show significant differences between the infected and uninfected L. striatellus for females or males and between the females and males for infected or uninfected L. striatellus by LSD test at P < 0.05.
Relationships between RSV and YLS abundance, nymphal stage duration and adult weight of L. striatellus with RSV infection
The indirect enzyme linked immunosorbent assay (ELISA) method was used to determine the relative abundance of RSV harbored in female and male adults of L. striatellus. The results showed that there were no significant differences in the relative abundance of RSV between females and males (mean OD405: 1.619 vs. 1.622; P = 0.88; see Supplementary Table S2 and Supplementary Fig. S3 online). Pearson correlation analysis (Table 2) indicated a significant positive correlation between the relative abundance of RSV and YLS in female adults (n = 170, r = 0.30, two-tailed, P < 0.001), but no significant correlations were found between the relative abundance of RSV and nymphal stage duration (n = 170, |r| < 0.10, two-tailed, P > 0.24), or between the relative abundance of RSV and adult weight (n = 170, |r| < 0.07, two-tailed, P > 0.44) for the females and males of L. striatellus with RSV infection.
Effects of RSV infection on the feeding behavior of L. striatellus
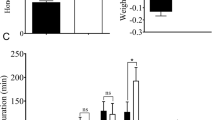
There was a significant interaction effect between RSV infection and sex on the total number of P waveforms (P = 0.03) and on the duration of NP (P < 0.001), P (P = 0.004) and N4b (P < 0.001) waveforms. A significant effect of RSV infection (P = 0.02) on the duration of N4a waveform was also observed (see Supplementary Table S2 online). The total number of P waveforms was significantly lower for the female adults with RSV infection compared to those without RSV infection (mean difference: -47.08%; P < 0.05; Fig. 5). Moreover, RSV infection significantly decreased the duration of NP waveform (mean difference: -71.85%) and significantly increased the duration of N4b waveform (mean difference: +67.20%) for the female adults (both comparisons P < 0.05; Fig. 5). The effects of RSV infection on the duration of NP (mean difference: 1176.83%) and N4b (mean difference: -56.44%) waveforms for the males were opposite to the females (both comparisons P < 0.05; Fig. 5), showing the existence of sexual dimorphism in the duration of NP and N4b waveforms of L. striatellus feeding in response to RSV infection. Significantly increased durations of N4a waveforms were also found for female adults with RSV infection compared to those without RSV infection (mean difference: 32.25%; P < 0.05; Fig. 5).
The EPG experiment of feeding behavior for the female and male adults of L. striatellus with (RSV) and without RSV (No RSV) infection.
NP: Non Penetration waveform; P: Pathway phase, sum of irregular mixed and transition phase prior to N4a; N4a: Sieve element salivation waveform; N4ab: Transition phase between N4a and N4b; N4b: Sieve element ingestion waveform; N5: Xylem feeding waveform; N7: Potential cell penetration; Nx: Unclear waveform. n = 15–20 for each treatment; Only significant differences are marked with letters. Different lowercase and uppercase letters show significant differences between the infected and uninfected L. striatellus for females or males and between the females and males for the infected or uninfected L. striatellus by LSD test at P < 0.05.
Discussion
RSV infection of its vector, L. striatellus, reduced female fecundity, which would be predicted to negatively impact the spread of the virus across generations and to new hosts. As a way to potentially compensate for this negative effect, RSV infection accelerated nymphal development, increased the weight, enhanced abundance of YLS and modified the feeding behavior of L. striatellus. These virus-mediated changes in vector physiology, endosymbionts and behavior could compensate for reduced fecundity by favoring the transmission of RSV. Based on the known and inferred effects of RSV infection on L. striatellus, a conceptual model describing interactions and their potential consequences in the RSV-L. striatellus-YLS system is provided in Fig. 6.
Potential comprehensive interactions between in the RSV-L. striatellus-YLS system.
Interactions as based the effects of RSV infection on the development, physiology and feeding behavior of its vector L. striatellus and on the virus transmission. Generation: A—B: egg stage; B—C: nymph stage; C—A: adult stage; Abbreviation: Vg: vitellogenin gene; VP: Viruliferous population; VC: vertical circulation; HP: Hatching period; ND: Nymphal stage duration; JH: Juvenile hormone; MH: Molting hormone (Ecdysteroids); YLS: Yeast-like symbiotes; HT: horizontal transmission.
Decreases observed in the expression levels of the Vg gene in newly emerged female adults along with a decrease in the number of eggs laid per female confirmed previous speculation that RSV infection has deleterious effects on female fecundity32,33,34,35. It has been reported that JH regulates Vg gene transcription in most hemipterans10 and topical application of JH III up-regulated Vg gene expression in the brown planthopper, Nilaparvata lugens36. Given that positive correlations have been found between JHAMT expression levels and JH titer37, our results are consistent with the same positive relationship linking gene expression levels of JHAMT and Vg.
JH and the main ecdysteroid, 20-hydroxy-ecdysone (20E), are commonly known for coordinating development and growth of insects11. Positive correlation between CYP307A1 expression and 20E in Anopheles gambiae was revealed by Pondeville et al. (2013)38. It was also reported that 20E blocked innate immunity at the onset of metamorphosis in Drosophila12 and that JH and ecdysteroids dynamically regulate innate immunity in the fat body during Bombyx postembryonic development, with JH acting as an immune-activator and 20E inhibiting innate immunity13. The up-regulation of JHAMT at 0 h and marginal down-regulation of CYP307A1 at 24 h, suggested an early immunity activation in RSV-infected L. striatellus. However, the dynamic gene expression levels of JHAMT and CYP307A1 that we observed later in L. striatellus are consistent with a dominant immunosuppression triggered by RSV infection, with JHAMT down-regulated at 60 h and 72 h and CYP307A1 up-regulated at 0 h, 48 h, 60 h and 72 h. Given that interactions between JH and ecdysteroids play major roles in growth, the immune response characterized by the down-regulation of JH and up-regulation of 20E level should be related to the significantly accelerated nymphal development we observed. Accelerated nymphal development leads to a shorter generation time among RSV-infected individuals. This could favor RSV infection as the shortened generation time might promote RSV spreading by earlier adult migration and dispersal. The shortened generation time could be particularly beneficial in accelerating the vertical cycle of RSV transovarial transmission in the viruliferous population. (i.e., virus vertical circulation). L. striatellus is a migratory pest that is able to overwinter in diapause2 and the transovarial transmission of RSV may last for as many as 40 generations with a transovarial transmission rate ranging from about 60% to 100% when fed on rice Oryza sativa L.5. Considering this, the cumulative effects over generations would further magnify this beneficial effect triggered by RSV. Importantly, a shorter juvenile development period is also widely assumed to reduce the risk of predation (or exposure to other mortality factors) prior to reproduction39. This could be an additional non-mutually exclusive consequence of RSV infection that further contributes to RSV transmission. Moreover, a recent study showed that knockdown of Vg expression due to RNA interference could result in inhibition of the invasion of ovarioles by RSV8. Thus from an evolutionary perspective, significantly decreased Vg expression in this study may be an adaptive response to the immunosuppression. In light of the above interactions, RSV-triggered changes in physiology and development to compensate for reduced fecundity in L. striatellus could represent the outcome of co-evolutionary interactions between the virus and its vector, since fitness of RSV relies heavily on its vectors’ fitness and ability to move itself to new hosts.
A delay in egg development attributable to RSV infection has been reported in L. striatellus by Li et al. (2015)34, which seems contradictory to our results showing no effect of RSV infection on hatching time, but an effect of accelerated nymphal development. Contrasting effects of RSV infection on L. striatellus fecundity have also been reported elsewhere35,40 and may be due to differences in experimental conditions including temperature, photoperiod, rice cultivar, insect rearing, treatment method, sampling times and ratios of RSV-positive individuals in the progeny from viruliferous mothers41. These variable results imply that the effects on development and reproduction triggered by RSV infection on L. striatellus might be influenced by environmental or genetic factors. Further studies must be performed to address the differences.
In addition to the beneficial changes in nymphal development associated with RSV infection, we also observed potentially beneficial virus-associated changes in interactions between L. striatellus and its primary endosymbionts, YLS. To our knowledge, this is the first such report. It has previously been shown that significantly higher weights were found in the symbiont-harbouring Nilaparvata lugens compared with aposymbiotic individuals42. Thus the higher weight found in L. striatellus may be attributed to the significantly higher abundance of YLS triggered by RSV infection. We found a significant positive correlation between the relative abundance of RSV and the abundance of YLS and significant increases in both the abundance of YLS and the body weight of adult L. striatellus infected with RSV, but only in females. This relationship suggests that RSV and YLS are likely to interact in the process of transovarial transmission in female adults. RSV infection may stimulate YLS propagation, which in turn may supply additional nutritional resources to support insect growth17,18,19. In this way, RSV may indirectly benefit both its vector and itself by increasing the abundance and associated nutritional benefits of YLS17,18,19,20,21,22. This finding represents an initial step in elucidating the interaction between persistent propagative viruses and the primary endosymbionts in their insect vector hosts. These interactions warrant further study due to their implications for the coordinated control of plant diseases and their vectors.
The behavior of insect vectors has profound ecological and evolutionary implications for plant virus transmission43. In most cases, the acquisition and inoculation of a plant virus often occurs through the insect vector feeding process. As such, the ability of a plant virus to alter its vectors’ feeding behavior in a manner that facilitates its own transmission would be selectively advantageous31. We found that the feeding behavior of L. striatellus with and without RSV infection exhibited a sexual dimorphism in the number and duration of NP and N4b waveforms. N4a is the preparation period just prior to N4b and virtually all studies of feeding behavior of herbivorous insects support the idea that saliva secretion happens prior to ingestion of cells or phloem sap (i.e. N4b in L. striatellus)31,44,45,46. Thus, our findings of extended duration of N4a (phloem salivation waveforms) in RSV-infected females during the period that is essential for inoculation of persistently transmitted viruses47, is directly related to a higher probability of RSV transmission to the host plant. Furthermore, the significant decrease in the number of P waveforms in RSV-infected adult females and the significant decrease in the duration of NP waveform could both save time on non-ingestion probes before the stylet gets to the phloem. Once the stylet reaches the phloem, RSV-infected female L. striatellus could sustain ingestion and thus lead to significantly longer durations of N4a and N4b waveforms compared with the females without RSV infection. As a result, the likelihood of horizontal transmission of RSV infection is changed by alterations in the feeding behavior of infected L. striatellus and enhanced RSV transmission would most likely happen in female adults. This scenario is further supported by the agreement between our findings and those of Gingery (1988) in which a difference in RSV transmission efficiency between the females and males was found7. A similar case of sexual differences in virus transmission has recently been found in western flower thrips, in which the males spread Tomato spotted wilt virus more efficiently than females as a result of feeding behavior modification31,48.
Taken together, the interactions in the RSV-L. striatellus-YLS system we have shown suggest that the RSV-triggered changes in insect development, behavior and endosymbiont abundance could be considered as viral adaptations that counter the potentially negative effects of RSV infection on its insect vector, as have been observed in this and other studies32,33,34,35. To our knowledge, this is the first reported evidence of a link between RSV infection of its insect vector, L. striatellus and changes that positively affect the likelihood of viral transmission across generations and between hosts. Our study also suggests that the nature of relationships between a plant virus and its insect vector cannot be simply classified as mutualistic or antagonistic; a more comprehensive and integrated assessment is necessary. The recent whole genome sequencing of the other closely-related rice virus vector, the brown planthopper, Nilaparvata lugens (Stål), along with its fungal and bacterial endosymbionts4,49 provides an excellent rice planthopper model for further investigating plant virus-insect-endosymbionts interactions and the underlying mechanisms.
Methods
Small brown planthopper, Laodelphax striatellus
Both the high-viruliferous (RSV-carrying rate: ≥90%) and non-viruliferous L. striatellus strains originated from an initially half-viruliferous strain (nearly 50% RSV-carrying rate) and were reared for multiple generations on rice seedlings (cv. TN1) separately in glass beakers (diameter: height = 10 cm: 15 cm) as stock populations in a growth incubator (HPG280H; Orient Electronic, Harbin, China) at 26°C with a photoperiod of 14 h/10 h (light/dark). The rice seedlings were fertilized with Kimura-B culture solution and the glass beakers were enclosed with a piece of nylon mesh after introducing insects onto 2–3 cm high rice seedlings. All rice planthoppers in the colony were provided with fresh seedlings every 10–14 days for sufficient nutrition.
Hatching period, nymphal stage duration and adult weight and fecundity
Seventy and 60 male-female pairs of high-viruliferous and non-viruliferous L. striatellus, respectively, were reared separately and mated on rice seedlings in glass tube cages (diameter: length = 3 cm: 15 cm) for 2 days. Each mated female was transferred into another glass tube with new rice seedlings to oviposit for one day and then all adults were removed. Glass tubes were checked daily for newly hatched 1st instar nymphs to quantify hatching period (days) of L. striatellus. RSV infection was detected by dot-immunoblot assay (DIBA)50,51 when the 1st instar nymphs grew to 5th instar nymphs. Other newly hatched 1st instar nymphs were transferred into numbered glass tubes (diameter: length = 3 cm: 15 cm) with one nymph per tube and the nymphs were daily checked for molting. The exuvia was removed and the ecdysis date was recorded to quantify L. striatellus nymphal duration. Once the adults emerged, they were identified as females and males and weighed using precision scales with an accuracy of ± 0.1 μg (Mettler Toledo XP2U). The adults were then transferred into 1.5 ml clear microtubes (Axygen MCT-150-C) for quantification of YLS and RSV as described below. RSV detection was conducted on all adults. The insects chosen for measuring nymphal stage duration and adult weight were used to quantify the YLS and RSV. To test RSV effects on fecundity, 15 male-female pairs of adults with and without RSV infection were reared separately and mated following the same protocol as described above for hatching period. Since female adult L. striatellus mates multiple times with male adult, a new male was added into the glass tube when the original male died before the female. One rice seedling per glass tube was planted in Kimura-B culture solution. Glass tubes were enclosed with a piece of nylon mesh and culture solution was added along the tube wall every 3 days for sufficient nutrition. Cages were checked daily for hatched nymphs and unviable eggs were counted by dissecting rice stems under a stereomicroscope (MOTIC SMZ-168) until the female adults died. RSV detection was conducted by DIBA when the female adult died. The fecundity of L. striatellus with and without RSV infection was calculated as the sum of unviable eggs and hatched nymphs.
RSV detection—Detection of RSV by dot-immunoblot assay (DIBA)
A dot-immunoblot assay (DIBA)50,51 was conducted to detect RSV infection in L. striatellus chosen for measuring the hatching period, nymphal stage duration, adult weight and adult fecundity. Three replicate samples per individual were made and RSV was confirmed by two positives out of three replications.
RSV detection—Detection of RSV by RT-PCR
Total RNA was isolated following the standard protocol of TRIzol reagent (Invitrogen) from individual newly-molted to 3 day-old (i.e., 0, 24, 48, 60 and 72 h post-ecdysis) 5th instar nymphs and individual newly emerged female adult L. striatellus which were used for the quantitative real-time PCR (qRT-PCR) experiment. The concentration and quality of total RNA were determined by a NanoDrop spectrophotometer (Thermo Scientific). The cDNA was synthesized by using the PrimeScrip RT reagent kit (TaKaRa) after treatment with DNAse I (TaKaRa) to remove contaminated genomic DNA. PCR was conducted with 2 × EasyTaq PCR SuperMix (TransGen), using a RSV specific primer pair and RNA specific control primers in duplex PCR as described by Cai et al. (2003)52. PCR products were analyzed by 1% agarose gels. Amplification fragments were verified by DNA sequencing. Detection of RSV by RT-PCR was conducted before the quantitative real-time PCR (qRT-PCR) analysis of Vitellogenin, CYP307A1 and JHAMT as described below.
Quantification of YLS and RSV in female and male adults of L. striatellus
The insects were transferred into 1.5 ml clear microtubes (Axygen MCT-150-C) once the adult L. striatellus emerged. They were numbered & identified as females or males, cooled to –22 °C for 80 s and weighed using precision scales with an accuracy of ± 0.1 μg (Mettler Toledo XP2U) and kept in microtubes separately at −80°C in an ultra-cold storage freezer (Thermo Scientific Forma 702, USA) for experiments. To quantify YLS and RSV in the same L. striatellus, a modification of indirect ELISA assays was conducted: A 200 μL aliquot of carbonate buffer (50 mM at pH 9.0) was added to each 1.5 ml clear microtube containing L. striatellus individuals and all samples were homogenized in a Tissue Lyser II (Qiagen) by shaking for 1.5 min at 20 Hz with two steel balls. This parameter set can ensure that all tissues and cells can be lysed as fully as possible to release YLS into the buffer. After homogenization, the steel balls were removed with a magnet and washed with 60 μL carbonate buffer to make a final volume of 260 μL in each 1.5 ml clear microtube. YLS in the sample tubes were sedimented by centrifugation at 12000 g for 2 min (5810R centrifuge, Eppendorf). RSV remained in supernatant and was transferred to an ELISA plate with 70 μL supernatant in each well. The supernatant was extracted carefully to prevent the loss of YLS in the sediment), covered with parafilm and incubated overnight at 4°C, three replications were made for each individual. The blocking, washing and incubation steps were same as the indirect ELISA, except that the color reaction was developed by a conventional alkaline phosphatase-conjugated rabbit anti-mouse IgG (A4312, Sigma Immunochemicals, Labkemi, Stockholm, Sweden) 70 μl/well, diluted 1:20000 in block buffer followed by 70 μl/well pNPP-substrate (Sigma Immunochemicals) diluted in diethanolamine buffer to a final concentration of 1 mg/ml. The reaction was stopped after a 40-min incubation in the dark by the addition of 70 μl/well of 0.1 M EDTA (EDTA: sc-29092), pH 7.5. Negative controls were made using non-viruliferous L. striatellus and positive controls made using RSV purified from infected rice plants. The plates were read with a microtiter plate reader (Model 550; Bio-Rad, Hercules, California, USA) at OD405 to quantify the relative abundance or RSV. The remained 50 μL sediment that contained the YLS was fully homogenized with a vortex to ensure the even distribution and three replications (10 μL/replication) were made for each individual. Then YLS was quantified by the commonly used hemacytometry according to the method described by Chen et al. (2006b)53. Excluding the loss, 10 μL homogenate per microtube was used to redetect RSV with DIBA assay for accuracy.
Quantitative real-time PCR (qRT-PCR) analysis of Vitellogenin, CYP307A1 and JHAMT
For qRT-PCR analysis of vitellogenin (Vg) gene, total RNA was extracted with Trizol (Invitrogen) from newly emerged female adults of the high-viruliferous and non-viruliferous L. striatellus strains. For the qRT-PCR analysis of the CYP307A1 (Genebank ID: KC701468.1) and JHAMT (primers provided by Prof. Li, G.Q. from the Department of Entomology, Nanjing Agricultural University) genes, RNA was similarly extracted from newly-molted to 3 day-old (i.e., 0, 24, 48, 60 and 72 h post-ecdysis) 5th instar nymphs of high-viruliferous and non-viruliferous strains of L. striatellus (see Supplementary Table S1 for primer sequences). First-strand complementary DNA was synthesized using the PrimeScript™ RT reagent kit (TaKaRa). RSV was detected with RT-PCR as described above and the qRT-PCR was performed using SYBR® Premix Ex Taq™ (Tli RNaseH Plus) (TaKaRa) in combination with a 7500 Real-Time PCR Detection System. Reactions were performed in a 20 μl final volume reaction, using primers in a final concentration of 200 nM and 1 μl of the undiluted L. striatellus cDNA template to make the Ct value fall within the suitable range of 15 to 35 based on preliminary experiments. No template was added to negative control reactions. A total of 30 infected and uninfected samples were separately mixed as 1 repeat and 3 repeats were made for each treatment in the qRT-PCR analysis of the CYP307A1 and JHAMT. A total of 10 infected and uninfected samples were separately mixed as 1 repeat and 9 repeats were made for each treatment in the qRT-PCR analysis of the Vg. ACTIN1 and ARF2 were used as housekeeping genes. The specific primers for all genes used in the qRT-PCR assays are shown in Supplementary Table S1.
The electrical penetration graph (EPG) recordings
The EPG technique was conducted to monitor feeding behavior of male and female L. striatellus adults with and without RSV infection using a GIGA-8 DC EPG amplifier system (EPG system, Wageningen University) as introduced by Tjallingii54. Only brachypterous female and male adults were selected from the high-viruliferous and non-viruliferous strains of L. striatellus. L. striatellus individuals were carefully connected to a gold wire (diameter: length = 18.5 μm: 3 cm) with conductive silver glue on their dorsum. After 1 h starvation, L. striatellus individuals were linked to the amplifier. To complete the electronic circuit, they were connected to the same position of stem area of each rice plant above the soil at 2nd or 3rd internode. The experiment was conducted in a greenhouse at 26.5 ± 1°C with 70 ± 10% humidity at a photoperiod of 14 h/10 h (light/dark). In order to reduce technical error, recordings were made on 4 channels simultaneously. Probing behavior was recorded for 6 h continuously and 4 h of records starting from the beginning of feeding were analyzed using EPG Stylet + a software (Wageningen Agricultural University, 2012). At least 15 replicates (15–20) per treatment were conducted. All recorded signals were analyzed.
Supplementary Fig. S1 shows typical DC-EPG waveform patterns produced by L. striatellus feeding on rice seedlings, based on the analyses in relation to other planthopper studies46,55,56,57. In this analysis, non-penetration (NP) waveform correlated with the absence of feeding. Pathway phase (P) indicated that the stylet of L. striatellus was inserted into the plant; it was irregular with increased amplitude as reported by Seo et al. (2009) in BPH46. The N5 waveform occurred occasionally during the pathway period and it exhibited a consistent shape close to that found by Seo et al. (2009), which is identified as xylem feeding waveform46. N4a has been suggested as sieve element salivation waveform and N4b has been confidently attributed to sieve element feeding phase46,56,57,58. N4ab was found as a transition phase between the waveforms of N4a and N4b and a similar phase was also found by Seo et al. (2009)46. The N7 waveform was classified as potential drops since it suddenly dropped from active pathway activities described by Ghaffar et al. (2011)56 and it was correlated with cell penetration of aphids described by Tjallingii (1988)58. The Nx waveform occasionally occurring during the period of N4b waveform (Fig. 1) was first found in our study.
Data analysis
Statistical analysis of all data was performed using SPSS 20.0. Levene’s test was used to test the homoscedasticity of variances (P > 0.10) and Shapiro–Wilk test was analyzed for normality (P > 0.05). Data were transformed if necessary to meet normality assumptions. The developmental durations of the 1st–5th instar nymphs, adult body weights, YLS abundance and EPG waveforms (square-root transformation for total number and natural log transformation for total duration of specific waveforms to improve model fit) of the female and male adults were analyzed separately using two-way analysis of variance (ANOVA), with RSV as main factor (infected vs. uninfected) and sex (female vs. male) as sub-factor. Since the 1st–5th instar nymphal stage durations of L. striatellus were measured on the same individuals over time, nymphal durations were also analyzed by two-way repeated measures ANOVA with RSV as main factor (infected vs. uninfected) and sex (female vs. male) as sub-factor and nymphal instars as repeat effects. Two-way ANOVAs were also used to analyze the effects of RSV infection on the gene expression levels of CYP307A1 and JHAMT for the 5th instar nymphs across sampling times from newly molted to the 3 days post-molt. One-way ANOVAs were also used to analyze the effects of RSV (infected vs. uninfected) on the developmental duration of eggs (hatching period), the number of eggs laid per female and the gene expression level of Vg in female adults of L. striatellus. If significant effects of RSV infection, sex (sampling time) or their interactions on the above variables were found, the least significant difference (LSD) test was further used to compare the means between the infected and uninfected L. striatellus or between females and males of L. striatellus at P < 0.05. For the L. striatellus with RSV infection, Pearson correlation analysis was conducted to investigate the relationships between the relative abundance of RSV with YLS abundance, adult weight and nymphal stage duration for both females and males of L. striatellus. Absolute abundance (YLS/adult) rather than relative abundance (YLS/mg adult) of YLS was chosen for representative of the YLS abundance to avoid the influence from the parameter of L. striatellus adult weight in the Pearson correlation analysis. Necessary test was conducted before the Pearson correlation analysis to rule out the self-correlation (Durbin-Watson) and collinearity (Collinearity diagnosis).
Additional Information
How to cite this article: Wan, G. et al. Rice stripe virus counters reduced fecundity in its insect vector by modifying insect physiology, primary endosymbionts and feeding behavior. Sci. Rep. 5, 12527; doi: 10.1038/srep12527 (2015).
References
Hohn, T. Plant virus transmission from the insect point of view. Proc. Natl. Acad. Sci. U.S.A. 104, 17905–17906, 10.1073/pnas.0709178104 (2007).
Sun, J. T. et al. Evidence for high dispersal ability and mito-nuclear discordance in the small brown planthopper, Laodelphax striatellus. Sci. Rep. 5, 8045, 10.1038/srep08045 (2015).
Zhang, F. J. et al. Massively parallel pyrosequencing-based transcriptome analyses of small brown planthopper (Laodelphax striatellus), a vector insect transmitting rice stripe virus (RSV). BMC genomics 11, 303, 10.1186/1471-2164-11-303 (2010).
Hibino, H. Biology and epidemiology of rice viruses. Annu Rev Phytopathol 34, 249–274, 10.1146/annurev.phyto.34.1.249 (1996).
Falk, B. W. & Tsai, J. H. Biology and molecular biology of viruses in the genus Tenuivirus. Annu Rev Phytopathol 36, 139–163, 10.1146/annurev.phyto.36.1.139 (1998).
Deng, J. H., Li, S., Hong, J., Ji, Y. H. & Zhou, Y. J. Investigation on subcellular localization of rice stripe virus in its vector small brown planthopper by electron microscopy. Virol. J. 10, 310, 10.1186/1743-422X-10-310 (2013).
Gingery, R. E. in The plant viruses, Vol. 4 (ed. Milne, R. G. ) Ch. 9, 297–329 (Springer US, 1988).
Huo, Y. et al. Transovarial transmission of a plant virus is mediated by vitellogenin of its insect vector. PLoS Path 10, e1003949, 10.1371/journal.ppat.1003949 (2014).
Tufail, M. & Takeda, M. Molecular characteristics of insect vitellogenins. J. Insect Physiol. 54, 1447–1458, 10.1016/j.jinsphys.2008.08.007 (2008).
Tufail, M., Nagaba, Y., Elgendy, A. M. & Takeda, M. Regulation of vitellogenin genes in insects. Entomol. Sci. 17, 269–282, 10.1111/ens.12086 (2014).
Flatt, T. et al. Hormonal regulation of the humoral innate immune response in Drosophila melanogaster. J. Exp. Biol. 211, 2712–2724, 10.1242/jeb.014878 (2008).
Beckstead, R. B., Lam, G. & Thummel, C. S. The genomic response to 20-hydroxyecdysone at the onset of Drosophila metamorphosis. Genome Biol. 6, R99, 10.1186/gb-2005-6-12-r99 (2005).
Tian, L. et al. Genome-wide regulation of innate immunity by juvenile hormone and 20-hydroxyecdysone in the Bombyx fat body. BMC genomics 11, 10.1186/1471-2164-11-549 (2010).
Chen, F. J. et al. Morphological difference of the yeast-like endosymbiotes in adult planthoopers of Nilaparvata lugens (Stål), Laodelphax striatellus (Fallén) and Sogatella furcifera (Horvath). Acta Zootaxonomica Sinica 31, 728–735 (2006).
Cheng, D. J. & Hou, R. F. Histological observations on transovarial transmission of a yeast-like symbiote in Nilaparvata lugens Stål (Homoptera, Delphacidae). Tissue Cell 33, 273–279, 10.1054/tice.2001.0173 (2001).
Noda, H. Histological and histochemical observation of intracellular yeastlike symbiotes in the fat body of the smaller brown planthopper, Laodelphax striatellus (Homoptera: Delphacidae). Appl. Entomol. Zool. 12, 134, 10.1303/aez.12.134 (1977).
Noda, H. & Koizumi, Y. Sterol biosynthesis by symbiotes: cytochrome P450 sterol C-22 desaturase genes from yeastlike symbiotes of rice planthoppers and anobiid beetles. Insect Biochem. Mol. Biol. 33, 649–658, 10.1016/s0965-1748(03)00056-0 (2003).
Chen, Y. H., Bernal, C. C., Tan, J., Horgan, F. G. & Fitzgerald, M. A. Planthopper “adaptation” to resistant rice varieties: changes in amino acid composition over time. J. Insect Physiol. 57, 1375–1384, 10.1016/j.jinsphys.2011.07.002 (2011).
Wan, P. J. et al. Constructing the major biosynthesis pathways for amino acids in the brown planthopper, Nilaparvata lugens Stål (Hemiptera: Delphacidae), based on the transcriptome data. Insect Mol. Biol. 23, 152–164, 10.1111/imb.12069 (2014).
Eya, B. K. et al. Chemical association in symbiosis - sterol donors in planthoppers. J. Chem. Ecol. 15, 373–380, 10.1007/Bf02027797 (1989).
Clark, A. J. & Block, K. The absence of sterol synthesis in insects. J. Biol. Chem. 234, 2578–2582 (1959).
Behmer, S. T. & Nes, W. D. Insect sterol nutrition and physiology: A global overview. Adv In Insect Phys 31, 1–72, 10.1016/S0065-2806(03)31001-X (2003).
Jiu, M. et al. Vector-virus mutualism accelerates population increase of an invasive whitefly. PLoS ONE 2, e182, 10.1371/journal.pone.0000182 (2007).
Maris, P. C., Joosten, N. N., Goldbach, R. W. & Peters, D. Tomato spotted wilt virus infection improves host suitability for its vector Frankliniella occidentalis. Phytopathology 94, 706–711, 10.1094/PHYTO.2004.94.7.706 (2004).
Mann, R. S., Sidhu, J. S. & Butter, N. S. Deleterious effects of cotton leaf curl virus on longevity and fecundity of whitefly, Bemisia tabaci (Gennadius). J. Entoml. 6, 62–66, 10.3923/je.2009.62.66 (2009).
Deangelis, J. D., Sether, D. M. & Rossignol, P. A. Survival, development and reproduction in western flower thrips (Thysanoptera, Thripidae) exposed to impatiens necrotic spot virus. Environ. Entomol. 22, 1308–1312, 10.1093/ee/22.6.1308 (1993).
Ingwell, L. L., Eigenbrode, S. D. & Bosque-Perez, N. A. Plant viruses alter insect behavior to enhance their spread. Sci. Rep. 2, 578, 10.1038/srep00578 (2012).
Moreno-Delafuente, A., Garzo, E., Moreno, A. & Fereres, A. A plant virus manipulates the behavior of its whitefly vector to enhance its transmission efficiency and spread. PLoS ONE 8, e61543, 10.1371/journal.pone.0061543 (2013).
Bragard, C. et al. Status and prospects of plant virus control through interference with vector transmission. Annu Rev Phytopathol 51, 177–201, 10.1146/annurev-phyto-082712-102346 (2013).
Gutiérrez, S., Michalakis, Y., Van Munster, M., Blanc, S. & Biere, A. Plant feeding by insect vectors can affect life cycle, population genetics and evolution of plant viruses. Funct Ecol 27, 610–622, 10.1111/1365-2435.12070 (2013).
Stafford, C. A., Walker, G. P. & Ullman, D. E. Infection with a plant virus modifies vector feeding behavior. Proc. Natl. Acad. Sci. U.S.A. 108, 9350–9355, 10.1073/pnas.1100773108 (2011).
Maramorosch, K. & Jensen, D. D. Harmful and Beneficial Effects of Plant Viruses in Insects. Annu. Rev. Microbiol. 17, 495–530, 10.1146/annurev.mi.17.100163.002431 (1963).
Zhou, Y. J. et al. Rice stripe virus disease. (Jiangsu Science and Technology Press, 2010).
Li, S. et al. Rice stripe virus affects the viability of its vector offspring by changing developmental gene expression in embryos. Sci. Rep. 5, 7883, 10.1038/srep07883 (2015).
Nasu, S. Studies on some leafhoppers and planthoppers which transmit virus diseases of rice plant in Japan. Bull. Kyushu Agric. Exp. Stn 8, 153–349 (1963).
Tufail, M. et al. Molecular cloning, transcriptional regulation and differential expression profiling of vitellogenin in two wing-morphs of the brown planthopper, Nilaparvata lugens Stål (Hemiptera: Delphacidae). Insect Mol. Biol. 19, 787–798, 10.1111/j.1365-2583.2010.01035.x (2010).
Niwa, R. et al. Juvenile hormone acid O-methyltransferase in Drosophila melanogaster. Insect Biochem. Mol. Biol. 38, 714–720, 10.1016/j.ibmb.2008.04.003 (2008).
Pondeville, E. et al. Microarray and RNAi analysis of P450s in Anopheles gambiae male and female steroidogenic tissues: CYP307A1 is required for ecdysteroid synthesis. PLoS ONE 8, e79861, 10.1371/journal.pone.0079861 (2013).
Nylin, S. & Gotthard, K. Plasticity in life-history traits. Annu Rev Entomol 43, 63–83, 10.1146/annurev.ento.43.1.63 (1998).
Kisimoto, R. On the transovarial passage of the rice stripe virus through the small brown planthopper, Laodelphax striatelllus Fallén. In Conference on relationships between arthropods and plant-pathogenic viruses, Tokyo. 73-90 (1965).
Gui, Q. Q., Shi, X. M. & Liu, F. Differences in rice stripe virus transmission abilities of Laodelphax striatellus (Homoptera: Delphacidae) from four geographical populations. J. Asia Pac. Entomol. 17, 595–600, 10.1016/j.aspen.2014.06.001 (2014).
Wilkinson, T. L. & Ishikawa, H. On the functional significance of symbiotic microorganisms in the Homoptera: a comparative study of Acyrthosiphon pisum and Nilaparvata lugens. Physiol. Entomol. 26, 86–93, 10.1046/j.1365-3032.2001.00214.x (2001).
Stafford, C. A., Walker, G. P. & Ullman, D. E. Hitching a ride: Vector feeding and virus transmission. Commun Integr Biol 5, 43–49, 10.4161/cib.18640 (2012).
Jin, S., Chen, Z. M., Backus, E. A., Sun, X. L. & Xiao, B. Characterization of EPG waveforms for the tea green leafhopper, Empoasca vitis Göthe (Hemiptera: Cicadellidae), on tea plants and their correlation with stylet activities. J. Insect Physiol. 58, 1235–1244, 10.1016/j.jinsphys.2012.06.008 (2012).
Alvarez, A. E. et al. Infection of potato plants with potato leafroll virus changes attraction and feeding behaviour of Myzus persicae. Entomol. Exp. Appl. 125, 135–144, 10.1111/j.1570-7458.2007.00607.x (2007).
Seo, B. Y., Kwon, Y. H., Jung, J. K. & Kim, G. H. Electrical penetration graphic waveforms in relation to the actual positions of the stylet tips of Nilaparvata lugens in rice tissue. J. Asia Pac. Entomol. 12, 89–95, 10.1016/j.aspen.2009.02.002 (2009).
Prado, E. & Tjallingii, W. F. Aphid activities during Sieve element punctures. Entomol. Exp. Appl. 72, 157–165 (1994).
Rotenberg, D. et al. Variation in tomato spotted wilt virus titer in Frankliniella occidentalis and its association with frequency of transmission. Phytopathology 99, 404–410, 10.1094/PHYTO-99-4-0404 (2009).
Xue, J. et al. Genomes of the rice pest brown planthopper and its endosymbionts reveal complex complementary contributions for host adaptation. Genome Biol. 15, 521, 10.1186/s13059-014-0521-0 (2014).
Hawkes, R., Niday, E. & Gordon, J. A dot-immunobinding assay for monoclonal and other antibodies. Anal. Biochem. 119, 142–147, 10.1016/0003-2697(82)90677-7 (1982).
Qin, W. S., Gao, D. M. & Chen, S. X. Studies on techniques of rapid detecting rice stripe virus in Laodelphax striatellus. Acta Agric. Zhejiangensis 6, 226–229 (1994).
Lijun, C. et al. Detecting rice stripe virus (RSV) in the small brown planthopper (Laodelphax striatellus) with high specificity by RT-PCR. J. Virol. Methods 112, 115–120, 10.1016/S0166-0934(03)00200-3 (2003).
Chen, F. J. et al. Individual size and abundance of the yeast-like endosymbiote in different geographic populations of brown planthopper, Nilaparvata lugens. Kun Chong Zhi Shi 4, 460–465 (2006).
Tjallingii, W. F. Electronic recording of penetration behaviour by aphids. Entomol. Exp. Appl. 24, 721–730, 10.1111/j.1570-7458.1978.tb02836.x (1978).
He, Y. et al. Influence of pymetrozine on feeding behaviors of three rice planthoppers and a rice leafhopper using electrical penetration graphs. J. Econ. Entomol. 104, 1877–1884, 10.1603/ec11180 (2011).
Ghaffar, M. B., Pritchard, J. & Ford-Lloyd, B. Brown planthopper (N. lugens Stå1) feeding behaviour on rice germplasm as an indicator of resistance. PLoS ONE 6, e22137, 10.1371/journal.pone.0022137 (2011).
Kimmins, F. M. Electrical penetration graphs from Nilaparvata lugens on resistant and susceptible rice varieties. Entomol. Exp. Appl. 50, 69–79, 10.1111/j.1570-7458.1989.tb02317.x (1989).
Tjallingii, W. F. in Aphids, their biology, natural enemies and control, Vol. 2B (eds Minks, A. K. & Harrewijn, P. ) “Electrical recording of stylet penetration activities”, 95–108 (Elsevier, 1988).
Acknowledgements
This research was supported by the National Basic Research Program of China “973” (2010CB126200), the National Nature Science Foundations of China (31272051, 31170362 and 31070755), the Special Fund for Agro-scientific Research in the Public Interest (201303021) and the Fok Ying Tung Education Foundation (122033).
Author information
Authors and Affiliations
Contributions
G.J.W. and F.J.C. designed the study; G.J.W., S.L.J. and W.J.W. performed the experiments; X.R.T. supplied insect and monoclonal antibody; G.Q.L. supplied gene information of JHAMT; G.J.W., F.J.C., X.R.T., G.A.S., W.D.P. and S.L.J. analysed the data; G.J.W. wrote the manuscript; G.J.W., F.J.C., X.R.T., G.A.S., W.D.P. and G.Q.L. reviewed and polished the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Wan, G., Jiang, S., Wang, W. et al. Rice stripe virus counters reduced fecundity in its insect vector by modifying insect physiology, primary endosymbionts and feeding behavior. Sci Rep 5, 12527 (2015). https://doi.org/10.1038/srep12527
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep12527
This article is cited by
-
Impacts of elevated CO2 on exogenous Bacillus thuringiensis toxins and transgene expression in transgenic rice under different levels of nitrogen
Scientific Reports (2017)
-
Modeling population dynamics of yeast-like symbionts (Ascomycota: Pyrenomycetes: Clavicipitaceae) of the planthopper Delphacodes kuscheli (Hemiptera: Delphacidae)
Symbiosis (2017)
-
Small interfering RNA pathway modulates persistent infection of a plant virus in its insect vector
Scientific Reports (2016)
-
Viruliferous rate of small brown planthopper is a good indicator of rice stripe disease epidemics
Scientific Reports (2016)
-
Differential proteomics profiling of the ova between healthy and Rice stripe virus-infected female insects of Laodelphax striatellus
Scientific Reports (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.