Abstract
We conducted a 2-year hospital-based study on norovirus gastroenteritis among children and adults between August 2012 and September 2014. A total of 1,146 norovirus cases were identified. Young children (aged ≤ 5 years) accounted for a majority (53.3%) of cases. Hospitalization incidence exhibited a U-shaped pattern with the highest rate in young children (1,475 per 100,000 person-years), followed by the elderly aged > 84 years (581 per 100,000 person-years). A subset (n = 395, 34.5%) of cases were selected for norovirus genotyping and noroviral load measurement. Non-GII.4 infections were more commonly observed in young children than in older adults (aged > 65 years) (20.5% versus 9.2%; p < 0.05). In young children, the median noroviral load of GII.4 and non-GII.4 cases was indistinguishably high (cycle threshold value, median [interquartile range]: 16.6 [15.2–19.3] versus 16.6 [14.9–21.6]; p = 0.45). Two age-specific non-GII.4 genotypes (GII.3 and GII.6) were identified among young children. These findings may have implications in norovirus vaccination strategy.
Similar content being viewed by others
Introduction
Noroviruses are a large group of viruses associated with non-bacterial acute gastroenteritis in community and healthcare settings worldwide affecting all age groups1. Noroviruses are classified into at least 5 genogroups (GI–GV) and nearly 40 genotypes, in which GI, GII and GIV infect humans. Despite the genetic diversity, cross-reactive immune responses following norovirus infections have been demonstrated2,3. Yet, indirect evidence from a recent case report of sequential natural infections of norovirus GII.4 and GII.6 genotypes suggests that at least some genotypes may represent distinct serotype and induce only limited cross-reactive immune response4. One particular genotype, known as genogroup II genotype 4 (GII.4), has long been recognized as a leading circulating genotype that causes acute gastroenteritis in developed countries since mid-1990s5,6. While generally regarded as mild infections in otherwise healthy individuals, norovirus gastroenteritis has been estimated to result in over 14,000 hospitalizations and nearly 1 million health care visits in the United States7 and almost 200,000 deaths in developing countries1 among young children per year. These figures suggest the need for a norovirus vaccine which is currently under development8,9. Yet, the broad genetic and probably serotypic diversity of norovirus complicates selection of candidate vaccine strain(s). Studies on the diversity of circulating virus genotypes and the associated virus burden in hospitalized cases of norovirus gastroenteritis that represent the more severe end of the spectrum of disease have been limited10,11,12. The need for these information to assess the disease burden attributed to different norovirus genotypes is warranted. Our group has been conducting hospital-based study on norovirus gastroenteritis since 201212. Here, we reported our latest findings on the genetic diversity and virus burden in hospitalized norovirus gastroenteritis.
Results
Description of Norovirus Cases
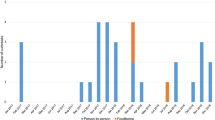
A total of 1,146 norovirus cases were identified during the 2-year study period. Nine (0.8%) cases had re-infections and all but 3 were young children aged ≤ 5 years. The male-to-female ratio was 1.11. Age distribution of cases is shown in Fig. 1A. The median age was 4 years (interquartile range [IQR], 1–66 years). Young children aged ≤ 5 years accounted for a majority (53.3%; 611/1,146) of cases. The age-stratified hospitalization incidence exhibited a U-shaped pattern with the highest rate in young children aged < 5 years (1,475 per 100,000 person-years), followed by the elderly aged > 84 years (581 per 100,000 person-years); adults aged 20–59 years were minimally affected (≤20 cases per 100,000 person-years) (Fig. 1A).
Age distribution of norovirus gastroenteritis.
(A) Age-stratified hospitalization incidence and number of cases of norovirus gastroenteritis. (B) Proportion of norovirus genogroup II genotype 4 (GII.4) and non-GII.4 cases in each age group. (C) Age distribution of GII.4 and non-GII.4 cases shown as cumulative case percentage. N, number; yr, year.
Distribution of Norovirus Genotypes
All Selected Cases
A total of 395 (34.5%) cases were selected for noroviral load measurement and genotyping. The age distribution of the selected cases was highly comparable with that of all cases (median [IQR]: 3 [1–60] versus 4 [1–66], years; p = 0.13, Mann-Whitney U test). Noroviral load was determined. Norovirus genotype was successfully determined in 321 (81.3%) cases. Failed genotyping was associated with lower noroviral load (Ct value, median [IQR]: 27.8 [24.2–30.8] versus 17.8 [15.8–20.8]; p < 0.0001, Mann-Whitney U test). By age, young children aged ≤ 5 years exhibited the highest successful rate of genotyping (86.7%; 195/225), followed by older adults aged > 65 years (77.4%; 65/84) and lowest in older children and adults aged 6–65 years (70.9%; 61/86). Norovirus genotyping successful rate tended to be higher in young children aged ≤5 years than in older adults aged > 65 years (p = 0.055; Fisher’s exact test). Two hundred and fifty seven (80.0%; 257/321) cases were genotyped as GII.4 in which all but four (98.4%) cases belonged to the Sydney 2012 strain. There were 2 cases of GII.4 Den Haag 2006 and 2 cases of GII.4 New Orleans 2009. Among the 64 non-GII.4 cases, 7 (11%) belonged to GI and 58 (91%) to GII with two cases of inter-genogroup and intra-genogroup coinfection. The 3 most common non-GII.4 genotypes were GII.6 (5.9%; 19/321), GII.3 (4.7%; 15/321) and GII.8 (1.6%; 5/321). A list of norovirus non-GII.4 genotype detected in this study is shown in Table 1.
Young Children and Older Adults
Among cases of known genotype, GII.4 strains accounted for 79.5% (155/195) of cases in young children aged ≤ 5 years. In contrast, 90.8% (59/65) of cases in older adults aged >65 years were caused by a GII.4 strain (Fig. 1B). Non-GII.4 infections were more commonly observed in young children aged ≤ 5 years than in older adults aged > 65 years (20.5% versus 9.2%; p < 0.05, Fisher’s exact test). In both GII.4 and non-GII.4 infections, a majority of cases were from young children aged ≤5 years (GII.4: 60.3% [155/257]; non-GII.4: 63% [40/64]) (Fig. 1C). Greater norovirus genotype diversity was observed in young children aged ≤ 5 years than in older adults aged >65 years. In young children aged ≤ 5 years, 10 genotypes were detected among the 40 non-GII.4 cases and nearly 70% of cases were caused by GII.6 and GII.3 strains (Table 1). In older adults aged > 65 years, 4 genotypes were detected among 6 non-GII.4 cases (Table 1).
Comparison of Noroviral Load
All Genotyped Cases
The distribution of noroviral load on presentation with age in GII.4 and non-GII.4 cases was shown in Fig. 2A. In general, noroviral load decreased with increasing age (GII.4: r = 0.17, p < 0.01; non-GII.4: r = 0.34, p < 0.01; Spearman correlation). The slope of the best-fit line of non-GII.4 cases (0.057) was greater than that of GII.4 cases (0.025), indicating the decline in noroviral load with age was more pronounced in non-GII.4 cases. The median noroviral load of GI cases was over 1,000-fold lower than that of other non-GII.4 cases (Ct value, median [IQR]: 28.0 [25.0–30.7] versus 17.4 [15.0–21.8]; p < 0.001, Mann-Whitney U test).
Fecal viral load of norovirus genogroup II genotype 4 (GII.4) and non-GII.4 gastroenteritis on case presentation by
(A) age and (B) age group. Cycle threshold (Ct) value of reverse transcription–polymerase chain reaction (RT-qPCR) assay was determined for each case to represent noroviral load. Higher Ct values indicate lower noroviral loads. Blue dotted lines indicate best-fit lines. In panel B, for clarity only p-values (Mann-Whitney U test) of comparisons between GII.4 and non-GII.4 cases were shown. Other comparisons were described in the main text where appropriate. Red horizontal lines denote medians. NS, not significant; r, Spearman’s rank correlation coefficient; *, p < 0.05.
Young Children and Older Adults
Noroviral load by age group is shown in Fig. 2B. Highest median noroviral load was observed in young children aged ≤ 5 years. In GII.4 infections, the median noroviral load in young children aged ≤ 5 years was ~5-fold higher than that in case aged > 5 years (Ct value, median [IQR]: 16.6 [15.2–19.3] versus 19.0 [16.7–21.6]; p < 0.0001, Mann-Whitney U test). In non-GII.4 infections, the median noroviral load in young children aged ≤ 5 years was ~23-fold higher than that in cases aged > 5 years (Ct value, median [IQR]: 16.6 [14.9–21.6] versus 21.1 [17.6–24.5]; p < 0.05, Mann-Whitney U test). In young children aged ≤5 years, the median noroviral load of GII.4 and non-GII.4 cases was indistinguishably high (Ct value: 16.6 versus 16.6; p = 0.45, Mann-Whitney U test). In cases aged > 5 years, the median noroviral load of non-GII.4 cases was ~4-fold lower than that of GII.4 cases (Ct value: 21.1 versus 19.0; p < 0.05, Mann-Whitney U test). When analyzing older children and adults aged 6–65 years and older adults aged >65 years separately, the median noroviral load of non-GII.4 cases was consistently lower than that of GII.4 cases, although the difference did not reach statistical significance.
Characteristics of Norovirus GII.6, GII.3 and GII.8 Infections
Norovirus GII.6, GII.3 and GII.8 strains were the 3 most common non-GII.4 genotypes detected in our study. Their age distribution and fecal noroviral load are shown in Table 1. The median age of GII.6 and GII.3 cases were 2 years (IQR: 1–4 years) and 1 year (IQR: 1–4 year), respectively. Young children aged ≤5 years accounted for 79% (15/19) and 80% (12/15) of GII.6 and GII.3 cases, respectively. In contrast, the median age of GII.8 cases was 88 years (IQR: 27–96 years). No GII.8 cases were observed in young children aged ≤5 years. The median age of the 5 GII.8 cases was higher than that of GII.3 and GII.6 cases (both p < 0.01; Kruskal-Wallis test). The median noroviral load of GII.8 (Ct value: 20.1) cases was lower than that of GII.6 (Ct value: 16.8) and GII.3 (Ct value: 16.6) but the difference did not reach statistical significance.
Norovirus Re-infections
There were 9 cases of re-infection and 2 cases were genotyped. The first genotyped case was a one-year-old girl. Her first episode of gastroenteritis was attributed to a GII.6 strain and the second episode was caused by a GII.4 Sydney 2012 strain separated by a 4-month period. Both stool specimens showed high noroviral loads (Ct values: 18.1 [GII.6] and 20.1 [GII.4]). The second genotyped case was a male infant aged <1 year. He was first infected with a GII.4 Sydney 2012 strain with high virus shedding (Ct value: 16.0) and was admitted again for norovirus gastroenteritis after 1 month. The noroviral load of the second episode was too low for virus genotyping (Ct value: 33.3).
Discussion
Studies on hospitalization of norovirus gastroenteritis have focused on either young children or older adults7,10,13,14,15. Our study, to the best of our knowledge, is comprehensive in investigating genetic diversity and virus burden of norovirus gastroenteritis in both children and adults in a hospital setting. There are three key observations: (1) young children aged ≤5 years accounted for a majority (>60%) of norovirus GII.4 and non-GII.4 infections requiring hospitalization, extending the findings of our previous study12; (2) non-GII.4 genotypes accounted for 20.5% and 9.2% of hospitalized cases of young children aged ≤5 years and older adults aged >65 years, respectively; and (3) both GII.4 and non-GII.4 infections exhibited indistinguishably high noroviral load (both with median Ct values of 16.6) in young children aged ≤5 years. It has long been recognized that there is a high degree of norovirus genotype diversity in young children16,17. With the recent advancement in the development of monovalent and bivalent norovirus vaccines8,18,19, one important unanswered question is whether to include a second genotype together with the contemporarily predominant GII.4 genotype as a vaccine component. Our findings that non-GII.4 infections attained virtually identical high noroviral load as in GII.4 infections in young children clearly indicates that non-GII.4 genotypes are replication competent in young children. In our study, norovirus GII.6 and GII.3 genotypes are common in young children which is in agreement with other epidemiological studies11,14. Notably, studies on historical stool specimens dated back to the 1970 s and 1980 s suggested that GII.3, rather than GII.4, was the main circulating norovirus genotype at that time, at least in hospitalized pediatric diarrheal cases20. This suggests that non-GII.4 genotypes such as GII.3 may be a candidate vaccine genotype. While a monovalent GII.4 vaccine could cover a large majority of hospitalized norovirus infections, a multivalent vaccine supplemented with circulating non-GII.4 genotype(s) may offer additional protection, especially for young children and should be considered. Notably, immune escape norovirus GII.4 strains emerged every 2 to 4 years. Our study was performed soon after the emergence of the then novel GII.4 Sydney 2012 strain which remained as the major circulating strain throughout the study period. This allows us to explore how emergent GII.4 strains influence the prevalence and seasonality of non-GII.4 genotypes. Continuous epidemiological surveillance of non-GII.4 infections is necessary as the herd immunity of non-GII.4 genotypes may influence norovirus circulating patterns21.
The duration and cross-protection of norovirus immunity have been topics of hot debate. Experimental volunteer challenge study by a GI strain known as Norwalk virus suggests that homotypic immunity to norovirus infection may last for 8 weeks to 2 years22. A much longer duration of immunity from 4 to 8 years has been estimated from mathematical modeling that used population-based incidence data23. In our 2-year study, norovirus re-infections that required hospitalization were rarely observed. This is consistent with the notion that norovirus immunity may endure 2 years or longer. It is possible that we might have missed some cases of norovirus re-infections if the second episode of gastroenteritis was not severe enough to trigger medical attention and hospitalization as a result of partial immunity. In a recent longitudinal community-based follow-up study of a birth cohort in Peru, multiple infections by different norovirus genotypes were commonly observed in nearly half of cases24. Re-infections may be more likely to be detected in setting of milder norovirus gastroenteritis. We genotyped two paediatric cases of norovirus re-infection in which one case was confirmed to be infected with two different genotypes (GII.6 and GII.4) within a short period of 4 months. This suggests that protective heterotypic immunity may not occur following natural infection4. Interestingly, noroviral loads of both GII.4 and non-GII.4 cases decreased with age. One interpretation is that previous norovirus exposure may confer partial immunity which is limited in strength. Alternatively, longer delay in seeking medical care in older patients may confound noroviral load data.
Our study has limitations. Firstly, we did not determine patients’ Vesikari score to evaluate whether norovirus GII.4 and non-GII.4 infections in young children lead to similar clinical severity of gastroenteritis despite their similarly high viral loads that has been shown to associate with longer duration of diarrhea25,26. Huhti and colleagues have shown that the GII.4 genotype was associated with more protracted diarrhea and vomiting than non-GII.4 genotypes27. It should be, however, noted that in the study of Huhti the predominant non-GII.4 genotypes were GII.b (now known as GII.P21/GII.3), GII.2 and GII.7 instead of GII.6 and GII.3 as in our study. There is a possibility that different norovirus genotypes display distinct clinical characteristics. Likewise, it is also elusive whether differential viral loads among different age groups are associated with clinical severity. Secondly, non-GII.4 cases aged >5 years had the lowest noroviral load. This may lead to underestimation of the prevalence of non-GII.4 cases in older cases as genotyping is more likely to fail due to insufficient noroviral load. Thirdly, it is unclear whether our findings can be generalized to other settings such as community settings and foodborne outbreaks5,28,29. Nevertheless, hospitalized cases are likely to represent more severe infections among all-cause norovirus gastroenteritis and they are one of main indexes in evaluating efficacy of a vaccine and success of a vaccination program.
In summary, among hospitalized cases of norovirus gastroenteritis, non-GII.4 infections are common in young children, usually with high virus burden and are not uncommon in older adults. Both norovirus GII.4 and non-GII.4 infections exhibit indistinguishably high virus burden in young children. There may be a need for a multivalent norovirus vaccine targeted to young children and probably older adults who will most likely experience severe infections in the future. Continuous epidemiological surveillance of non-GII.4 infections is necessary to guide the selection of norovirus vaccine strain.
Materials and Methods
Catchment Population and Case Selection
This is a 2-year hospital-based study on norovirus gastroenteritis conducted between August 2012 and September 2014. Our study site, the Prince of Wales Hospital, is the norovirus diagnosis laboratory for Hospital Authority’s New Territories East Cluster that comprises of 3 major acute general hospitals, serving a population of about 1.2 million and covering nearly 20% of entire population in Hong Kong. Stool specimens were collected from patients admitted with acute gastroenteritis (diarrhea [loose/watery stool] or vomiting with a frequency of 3 or more in 24 hours) and tested for norovirus upon clinical suspicion (without other alternative explanation such as underlying bowel disorder) of norovirus infections. Diagnostic testing was performed twice weekly in batches. Patients tested positive for norovirus were identified. For this study, a subset of norovirus cases were selected for noroviral load measurement and genotyping as follows: during August 2012–February 2014, all consecutive cases tested positive for norovirus in the first test batch of each week were selected; during March 2014–September 2014, all consecutive cases tested positive in both weekly batches were selected as part of enhanced surveillance for emergent norovirus GII.4 strain. Cases with norovirus detected in separate occasions with more than 30 days apart were regarded as having re-infections. Patients’ demographic information of age and sex were retrieved from the computerized clinical management system. For calculating age-stratified norovirus hospitalization incidence, the catchment population was used as the at-risk population. Catchment population statistics was compiled from the most recent Hong Kong 2011 Population Census (http://www.census2011.gov.hk/en/district-profiles.html). Ethics approval for this study and waiver of consent of using de-identified leftover diagnostic specimens were obtained from the joint Chinese University of Hong Kong–New Territories East Cluster clinical research ethics committee (reference number CRE-2013.330). All experiments were performed in accordance with relevant guidelines and regulations.
Norovirus Detection and Noroviral Load Quantification
Viral RNA was extracted and purified from ~10% (0.1 gram) stool suspension using MagMAX Viral RNA Isolation Kit (Life Technologies). Norovirus detection and noroviral load quantification was performed by using a quantitative, genogroup-specific, probe-based real-time reverse transcription–polymerase chain reaction (RT-qPCR) assay that targeted the junction between RNA-dependent RNA polymerase (RdRp) and viral protein 1 (VP1) genes of norovirus as previously described30. Briefly, 10 μL of purified RNA was used as template in a 25-μL total reaction volume using SuperScript III Platinum One-Step qRT-PCR kit (Life Technologies). No template (negative) control was included in every run. To control for batch-to-batch variation, a positive control of known Ct value was included in each run. Assay cycle threshold (Ct) value was determined for each case to represent noroviral load. Higher Ct values indicated lower noroviral loads.
Norovirus Genotyping
Viral RNA was converted into complementary DNA (cDNA) using SuperScript III reverse transcriptase in the presence of random hexamers (Life Technologies). Norovirus genogroup-specific PCR amplification of a short fragment (~500 bp) comprising partial RdRp and VP1 genes of the virus was generated using primers GIFF/G1SKR (for norovirus GI) and G2FB/G2SKR (for norovirus GII)30. Viral RNA that failed to generate a PCR product was subjected to a separate round of reverse transcription using SuperScript III reverse transcriptase in the presence of a tagged oligo-dT primer, followed by a genogroup-specific long PCR of the 3’ end of the virus genome that covered VP1 and VP2 genes and 3’-untranslated region using primers COG1F/Tag (for norovirus GI) and COG2F/Tag (for norovirus GII). PCR products were purified by treatment with ExoSAP-IT (Affymetrix) and then directly sequenced by the forward PCR primer using Sanger sequencing. Chromatograms were manually checked using ChromasPro version 1.7.6 (Technelysium). Virus genotype assignment based on the partial VP1 gene was performed using RIVM’s online norovirus typing tool (http://www.rivm.nl/mpf/norovirus/typingtool)31. Non-GII.4 genotypes in this study were defined to include all genotypes of GI and GII except GII.4.
Statistical Analysis
Categorical variables were compared using Fisher’s exact test. Continuous variables were compared using Mann-Whitney U test (between 2 groups) or Kruskal-Wallis test with Dunn’s post hoc method to correct for multiple comparison (among 3 groups or more). Correlation between two continuous variables was measured by Spearman correlation. All statistical tests were performed using Prism version 5.04 (GraphPad). A two-tailed p-value < 0.05 was considered to be statistically significant.
Additional Information
How to cite this article: Chan, M. C.W. et al. Virus Genotype Distribution and Virus Burden in Children and Adults Hospitalized for Norovirus Gastroenteritis, 2012-2014, Hong Kong. Sci. Rep. 5, 11507; doi: 10.1038/srep11507 (2015).
References
Patel, M. M. et al. Systematic literature review of role of noroviruses in sporadic gastroenteritis. Emerg. Infect. Dis. 14, 1224–1231 (2008).
Lindesmith, L. C. et al. Heterotypic humoral and cellular immune responses following Norwalk virus infection. J. Virol. 84, 1800–1815 (2010).
Swanstrom, J., Lindesmith, L. C., Donaldson, E. F., Yount, B. & Baric, R. S. Characterization of blockade antibody responses in GII.2.1976 Snow Mountain virus-infected subjects. J. Virol. 88, 829–837 (2014).
Parra, G. I. & Green, K. Y. Sequential gastroenteritis episodes caused by 2 norovirus genotypes. Emerg. Infect. Dis. 20, 1016–1018 (2014).
Vega, E. et al. Genotypic and epidemiologic trends of norovirus outbreaks in the United States, 2009 to 2013. J. Clin. Microbiol. 52, 147-155 (2014).
Hasing, M. E. et al. Emergence of a new norovirus GII.4 variant and changes in the historical biennial pattern of norovirus outbreak activity in Alberta, Canada, from 2008 to 2013. J. Clin. Microbiol. 51, 2204–2211 (2013).
Payne, D. C. et al. Norovirus and medically attended gastroenteritis in U.S. children. N. Engl. J. Med. 368, 1121–1130 (2013).
Atmar, R. L. et al. Norovirus vaccine against experimental human Norwalk Virus illness. N. Engl. J. Med. 365, 2178–2187 (2011).
Debbink, K., Lindesmith, L. C. & Baric, R. S. The State of Norovirus Vaccines. Clin. Infect. Dis. (2014).
Ahmed, S. M. et al. Global prevalence of norovirus in cases of gastroenteritis: a systematic review and meta-analysis. Lancet Infect. Dis. 14, 725–730 (2014).
Franck, K. T., Fonager, J., Ersboll, A. K. & Bottiger, B. Norovirus epidemiology in community and health care settings and association with patient age, Denmark. Emerg. Infect. Dis. 20, 1123–1131 (2014).
Chan, M. C., Leung, T. F., Kwok, A. K., Lee, N. & Chan, P. K. Characteristics of patients infected with norovirus GII.4 Sydney 2012, Hong Kong, China. Emerg. Infect. Dis. 20, 658–661 (2014).
Haustein, T., Harris, J. P., Pebody, R. & Lopman, B. A. Hospital admissions due to norovirus in adult and elderly patients in England. Clin. Infect. Dis. 49, 1890–1892 (2009).
Puustinen, L. et al. Noroviruses as a major cause of acute gastroenteritis in children in Finland, 2009-2010. Scand. J. Infect. Dis. 43, 804–808 (2011).
Ouyang, Y. et al. Etiology and epidemiology of viral diarrhea in children under the age of five hospitalized in Tianjin, China. Arch. Virol. 157, 881–887 (2012).
Hoa Tran, T. N., Trainor, E., Nakagomi, T., Cunliffe, N. A. & Nakagomi, O. Molecular epidemiology of noroviruses associated with acute sporadic gastroenteritis in children: global distribution of genogroups, genotypes and GII.4 variants. J. Clin. Virol. 56, 185–193 (2013).
Park, S. et al. Characterization of norovirus infections in Seoul, Korea. Microbiol. Immunol. 56, 700–707 (2012).
Bernstein, D. I. et al. Norovirus Vaccine Against Experimental Human GII.4 Virus Illness: A Challenge Study in Healthy Adults. J. Infect. Dis. 211, 870–878 (2014).
Treanor, J. J. et al. A Novel Intramuscular Bivalent Norovirus Virus-Like Particle Vaccine Candidate-Reactogenicity, Safety and Immunogenicity in a Phase 1 Trial in Healthy Adults. J. Infect. Dis. 210, 1763–1771 (2014).
Boon, D. et al. Comparative evolution of GII.3 and GII.4 norovirus over a 31-year period. J. Virol. 85, 8656–8666 (2011).
Sakon, N. et al. Impact of Genotype-Specific Herd Immunity on the Circulatory Dynamism of Norovirus: A 10-Year Longitudinal Study of Viral Acute Gastroenteritis. J. Infect. Dis. 211, 879–888 (2014).
Parrino, T. A., Schreiber, D. S., Trier, J. S., Kapikian, A. Z. & Blacklow, N. R. Clinical immunity in acute gastroenteritis caused by Norwalk agent. N. Engl. J. Med. 297, 86–89 (1977).
Simmons, K., Gambhir, M., Leon, J. & Lopman, B. Duration of immunity to norovirus gastroenteritis. Emerg. Infect. Dis. 19, 1260–1267 (2013).
Saito, M. et al. Multiple norovirus infections in a birth cohort in a Peruvian Periurban community. Clin. Infect. Dis. 58, 483–491 (2014).
Furuya, D. et al. Age, viral copy number and immunosuppressive therapy affect the duration of norovirus RNA excretion in inpatients diagnosed with norovirus infection. Jpn. J. Infect. Dis. 64, 104–108 (2011).
Lee, N. et al. Fecal viral concentration and diarrhea in norovirus gastroenteritis. Emerg. Infect. Dis. 13, 1399–1401 (2007).
Huhti, L. et al. Norovirus GII-4 causes a more severe gastroenteritis than other noroviruses in young children. J. Infect. Dis. 203, 1442–1444 (2011).
Vega, E. et al. Novel surveillance network for norovirus gastroenteritis outbreaks, United States. Emerg. Infect. Dis. 17, 1389–1395 (2011).
Leshem, E. et al. Effects and Clinical Significance of GII.4 Sydney Norovirus, United States, 2012-2013. Emerg. Infect. Dis. 19, 1231–1238 (2013).
Kageyama, T. et al. Broadly reactive and highly sensitive assay for Norwalk-like viruses based on real-time quantitative reverse transcription-PCR. J. Clin. Microbiol. 41, 1548–1557 (2003).
Kroneman, A. et al. An automated genotyping tool for enteroviruses and noroviruses. J. Clin. Virol. 51, 121–125 (2011).
Acknowledgements
This study was jointly supported by institutional direct grant for research (to MCWC; reference number 2013.1.045) and departmental funding.
Author information
Authors and Affiliations
Contributions
M.C.W.C. and P.K.S.C. conceived and designed the study. M.C.W.C. supervised the study. T.F.L, E.A.S.N. and N.L. coordinated specimen collection and provision of patient care. M.C.W.C., T.W.S.C. and A.K.K. did laboratory testing. M.C.W.C. analyzed data and drafted the manuscript. All authors critically reviewed the manuscript and approved the final version. M.C.W.C. has access to all data and is responsible for the integrity of the study.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Chan, M., Leung, T., Chung, T. et al. Virus Genotype Distribution and Virus Burden in Children and Adults Hospitalized for Norovirus Gastroenteritis, 2012–2014, Hong Kong. Sci Rep 5, 11507 (2015). https://doi.org/10.1038/srep11507
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep11507
This article is cited by
-
Profiling of humoral immune responses to norovirus in children across Europe
Scientific Reports (2022)
-
Molecular epidemiology and genetic diversity of norovirus among hospitalized children with acute gastroenteritis in Tianjin, China, 2018–2020
BMC Infectious Diseases (2021)
-
Genomic and biological characterization of a pandemic norovirus variant GII.4 Sydney 2012
Virus Genes (2020)
-
Chimeric GII.3/GII.6 norovirus capsid (VP1) proteins: characterization by electron microscopy, trypsin sensitivity and binding to histo-blood group antigens
Archives of Virology (2018)
-
Genomic characterization of GII.3 noroviruses isolated from children in Zhengzhou city, China, 2015/16
Archives of Virology (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.