Abstract
RNA interference (RNAi) is a powerful and convenient tool for sequence-specific gene silencing and it is triggered by double-stranded RNA (dsRNA). RNAi can be easily achieved in many eukaryotes by either injecting or feeding dsRNAs. This mechanism has demonstrated its potential in fundamental research on genetics, medicine and agriculture. However, the possibility that insects might develop refractoriness to RNAi remains unexplored. In this study, we report that the oriental fruit fly, Bactrocera dorsalis, became refractory to RNAi using orally administered dsRNA targeting endogenous genes. Furthermore, refractoriness to RNAi is not gene-specific and its duration depends on the dsRNA concentration. RNAi blockage requires the endocytic pathway. Fluorescence microscopy indicated that in RNAi refractory flies, dsRNA uptake is blocked. Genes involved in the entry of dsRNAs into cells, including chc, cog3, light and others, are down-regulated in RNAi refractory flies. Increasing the endocytic capacity by improving F-actin polymerization disrupts RNAi refractoriness after both primary and secondary dsRNA exposures. Our results demonstrate that an insect can become refractory to RNAi by preventing the entry of dsRNA into its cells.
Similar content being viewed by others
Introduction
RNAi is a conserved mechanism by which endogenous genes are silenced by dsRNAs in a sequence-specific manner1. dsRNAs can be delivered into animals by various methods, including injection2, feeding3 and transgenic expression4. These dsRNAs are then processed by a member of the RNase III family, Dicer, into siRNAs of approximately 21 nucleotides in length5. The siRNA works as a guide and is loaded into the RNA-induced silencing complex (RISC), leading to sequence-specific mRNA cleavage6,7. RNAi can be cell-autonomous or non-cell-autonomous8. Cell-autonomous RNAi refers to RNAi that occurs within a single cell. Non-cell-autonomous RNAi refers to the ability of dsRNA to trigger RNAi in cells that are distant from the initial site of RNAi or the location where the dsRNA was introduced8. For the efficient use of RNAi in pest control, the focus must be on non-cell-autonomous RNAi caused by the feeding of dsRNAs9. The use of RNAi in pest management requires the dsRNA to be ingested in the lumen without being degraded, then taken up in the intestinal cells. Then, the dsRNA molecules can pass through the intestinal cells and into the body cavity, where they can act on other tissues, such as muscles8.
Because RNAi is easy to induce and highly efficient, it has been widely used throughout the scientific field. It has become a basic method in functional genetic studies. Genome-wide screens for genes involved in many biological pathways have been successfully carried out using RNAi-based methods10,11. Although RNAi-based therapy has not been fully realised, several achievements in related areas still provide hope for success12. RNAi is also a promising tool in agricultural science and especially in pest management, as an environmentally friendly pesticide13.
RNAi experiments have been carried out in various insect orders, including the Diptera14, Coleoptera15, Lepidoptera16, Hemiptera17 and Isoptera18. Two landmark articles demonstrated the feasibility of the oral administration of dsRNA in insects19,20, supporting the use of RNAi in insect pest control. However, RNAi in insects has yielded varying results. Among the insects in which RNAi has been investigated, some appear to be RNAi-insensitive21. For example, feeding dsRNAs to adults of the Dipteran species Drosophila melanogaster failed to elicit RNAi22. In the Lepidopteran species Spodoptera litura, gene silencing using dsRNA was observed only with injection; the feeding method failed23. An attempt to silence the nitroporin 2 gene in 4th instar larvae of Rhodnius prolixus failed despite the large quantity of dsRNA that was used (80 μg)17.
It has been demonstrated that both dsRNAs and siRNAs activate the type I interferon (IFN) system in mammalian cells24. Unlike in vertebrates, it is generally believed that IFN responses do not exist in invertebrates because the critical genes or major effectors of the IFN pathway are absent in these species25. Recent studies have shown that the RNAi pathway plays an important role in invertebrate viral immunity26. Robalino et al. (2004) injected dsRNAs derived from vertebrate immunoglobulin genes, fish non-coding genomic DNA, bacterial vector sequences and the Taura syndrome virus into marine shrimp. Each of these sequences induced protection against infection with the white spot syndrome virus (WSSV)25. Administration of dsRNA that targets either virus-specific or non-specific sequences can trigger an antiviral response that controls viral infections in honey bees27. However, importantly, this viral immunity was not sequence-specific; it could be activated by dsRNAs derived from any sequence25.
To date, there is little, if any, evidence indicating that an organism can become refractory to dsRNA-induced RNAi. Here, we report that the insect B. dorsalis can become refractory to RNAi triggered by feeding. Refractoriness is caused by a decrease in the endocytic entry of dsRNA into the intestinal cells, thus preventing RNAi. Digital gene expression (DGE) and qPCR analysis show that several genes involved in endocytosis are down-regulated. Increasing the endocytic capacity by promoting actin assembly can reverse the refractoriness. The mechanism of RNAi refractoriness uncovered here might explain why RNAi is difficult to achieve in certain insects.
Results and Discussion
Feeding of dsRNAs targeting endogenous genes induces protection against secondary RNAi
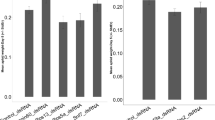
Unlike vertebrates, invertebrates lack acquired immunity28. However, growing empirical evidence suggests that previous exposure to a parasite can lead to increased protection in response to a subsequent challenge29. In invertebrates, this phenomenon is termed “immune priming”29. In this study, we aimed to determine if primary RNAi could further influence secondary RNAi in B. dorsalis. First, we tested the effects of prior RNAi using a dsRNA sequence targeting a non-endogenous gene on a subsequent RNAi by exposure of dsRNA targeting an endogenous gene. These dsRNAs were derived from enhanced green fluorescent protein (egfp) (186 bp), Discosoma sp. red fluorescent protein (dsred) (192 bp), the hly gene from Listeria monocytogenes (hly) (207 bp). The flies were divided into two groups. The challenged group (Ch) was fed an artificial diet containing one of the above dsRNAs for 6 hr. The other group, referred to as the naive group (Nv), was fed a normal artificial diet. Five days post-first exposure (dpe), both the challenged group and the naive group were orally administered a dsRNA targeting either the sex peptide receptor (spr) gene (280 bp) or the ribosomal protein L19 (rpl19) gene (205 bp). rpl19 is housekeeping genes encoding ribosomal protein L19 subunit. Our results showed that rpl19 is uniformly expressed in all tissues in B. dorsalis (Figure S1). spr mediates post-mating behaviour changes in female flies. spr is highly expressed in the midgut and is also expressed in the head and the reproductive organs in B. dorsalis (data unpublished). The results showed that, in the first round of RNAi, none of the three exogenous dsRNAs affected the expression of spr or rpl19 (Figure S2A, B). In addition, spr dsRNA and rpl19 dsRNA did not have mutual effect on each other expression (Figure S2A, B). In the secondary RNAi assay, both the naive group and the challenged group exhibited strong RNAi; the expression of the target genes was reduced by more than 60%, demonstrating the efficiency of the RNAi (Figure 1A, B). These results suggest that exposure to a dsRNA targeting a non-endogenous gene did not reduce the effect of subsequent exposure to a dsRNA against an endogenous gene. We also tested if dsRNA length could affect the secondary RNAi outcome. The results showed that exposure to 92 bp and 494 bp egfp dsRNAs did not change the effect of a secondary RNAi targeting either rpl19 or spr. Both the naïve and the challenged group exhibited strong RNAi (Figure 1C, D). It indicates that the length of dsRNAs targeting exogenous genes do not affect the outcome.
Feeding dsRNA that targets exogenous genes do not affect subsequent RNAi.
(A) The effect of dsRNA that targets exogenous genes on a second exposure to rpl19 dsRNA. (B) The effect of dsRNA that targets exogenous genes on a second exposure to spr dsRNA. (C) The effect of 92 bp and 494 bp egfp dsRNA on a second exposure to rpl19 dsRNA. (D) The effect of 92 bp and 494 bp egfp dsRNA on a second exposure to spr dsRNA. Normalised target gene expression is reported relative to the expression after egfp dsRNA treatment, which was set to 1. All error bars represent the SE of the mean of three independent biological replicates. * indicates a statistically significant difference in spr or rpl19 expression between rpl19 dsRNA or spr dsRNA and the control egfp dsRNA treatments (P < 0.01).
Next, we used rpl19 dsRNA as the first exposure for the challenged group, whereas egfp dsRNA was used for the naive group. rpl19 expression decreased by 65.2% after the first exposure (Figure S3A). The RNAi effect disappeared four days after the first RNAi (Figure S3A). As expected, the naive group showed efficient RNAi after secondary exposure to a dsRNA targeting rpl19; in this group, rpl19 expression decreased by 66% at 5 dpe (Figure 2A). However, in the challenged group, after secondary RNAi, depletion of rpl19 could not be observed at 5 dpe. This phenomenon suggests that an initial exposure to rpl19 dsRNA prevents RNAi-induced gene silencing after a second exposure to the same dsRNA.
Feeding dsRNA that targets endogenous genes induces protection against subsequent RNAi.
(A) RNAi refractoriness to secondary rpl19 dsRNA exposure, primed by rpl19 dsRNA. (B) RNAi refractoriness to secondary spr dsRNA exposure, primed by rpl19 dsRNA. (C) RNAi refractoriness to secondary spr dsRNA exposure, primed by spr dsRNA. (D) RNAi refractoriness to secondary rpl19 dsRNA exposure, primed by spr dsRNA. Normalised target gene expression is reported relative to the expression after egfp dsRNA treatment, which was set to 1. All error bars represent the SE of the mean of three independent biological replicates. * indicates a statistically significant difference in spr or rpl19 expression between rpl19 dsRNA or spr dsRNA and the control egfp dsRNA treatments (P < 0.01). Different letters indicate a significant difference in rpl19 or spr expression among the rpl19 dsRNA or spr dsRNA treatments (P < 0.01).
We next examined the duration of RNAi refractoriness. We set the time lag between the two RNAi exposures to 10, 20 or 30 days (Figure S4). First we examined rpl19 and spr gene expression in the untreated flies. Our results suggest that there is no expression level fluctuation for spr and rpl19 during 30 days in the untreated flies (Figure S5A, B). qPCR results of secondary RNAi suggested that, although RNAi refractoriness decreased with time, it was still observable after secondary RNAi at 10 and 20 dpe. After a secondary oral administration of rpl19 dsRNA, rpl19 mRNA levels were only down-regulated by 25% and 39% at 10 and 20 dpe, respectively, in the challenged group (Figure 2A). By contrast, insects that were first treated with either egfp dsRNA or rpl19 dsRNA exhibited strong rpl19 depletion after 30 dpe, indicating that the RNAi refractory period lasted no more than 30 days after initial exposure (Figure 2A). These results demonstrated that exposure to orally administered rpl19 dsRNA induced a change in the insect host that made it refractory to the effects of subsequent RNAi targeting rpl19. Our results show that feeding B. dorsalis a dsRNA against a specific gene reduced the RNAi efficiency of a second exposure to dsRNA against the same gene.
RNAi refractoriness is not sequence-specific and is influenced by the concentration of the priming dsRNA
We next examined if RNAi refractoriness primed by rpl19 dsRNA was also effective against secondary RNAi targeting spr at 5 dpe. The results showed that prior ingestion of rpl19 dsRNA provided protection against subsequent RNAi targeting spr. Feeding the naive group with spr dsRNA lead to an approximately 70% down-regulation of spr expression after secondary RNAi (Figure 2B). By contrast, the group initially challenged with rpl19 dsRNA showed no reduction in spr gene expression after secondary spr RNAi at 5 dpe (Figure 2B). There was no significant difference between the naive group and the challenged group when secondary spr gene silencing was assessed at 20 dpe (Figure 2B). We then tested if a dsRNA targeting a gene other than rpl19 could elicit RNAi refractoriness. To address this question, we chose spr as a target gene for initial dsRNA exposure. The results showed that, in the challenged group, the initial exposure to spr dsRNA induced refractoriness towards to secondary exposure to both spr dsRNA and rpl19 dsRNA (Figure 2C, D). The refractory state primed by spr dsRNA protected B. dorsalis for 5 dpe after secondary exposure to both spr and rpl19 dsRNA. However, this refractory period was much shorter than that provoked by rpl19 dsRNA. There was no difference in gene expression between the challenged and the naive groups fed either rpl19 dsRNA or spr dsRNA after 10 dpe (Figure 2C, D). These results indicate that a refractory state can be provoked by dsRNAs with different sequences. Because we did not observe this refractory state using egfp or other dsRNAs targeting exogenous genes, we assume that only dsRNAs targeting endogenous genes can trigger RNAi refractoriness.
We next explored if the concentration of dsRNA used in the first exposure influenced RNAi refractoriness. The results showed that 100 ng/μl rpl19 dsRNA decreased rpl19 expression by 38%, but that 10 ng/μl rpl19 dsRNA did not decrease rpl19 expression (Figure 3A). Both dsRNA concentrations could block RNAi-mediated gene silencing after secondary exposure to two different dsRNAs 5 dpe. Nevertheless, the refractory periods were shorter than those induced by the 1000 ng/μl treatment. In the group treated with 100 ng/μl, secondary exposure 10 dpe decreased rpl19 expression by 40% in the challenged group and by 70% in the naive group (Figure 3B). No difference could be observed between the naive and challenged groups after secondary exposure 20 dpe (Figure 3B). This shortened period of refractoriness was more obvious when analysing its effect on a second exposure to spr dsRNA; in this case, the RNAi refractory period lasted no longer than 10 days (Figure 3C). The refractoriness induced by the 10 ng/μl rpl19 dsRNA were even shorter than those induced by 100 ng/μl rpl19 dsRNA (Figure 3D, E). These results clearly illustrated that the duration of RNAi refractoriness to dsRNAs is correlated with the dsRNA concentration used for priming. In addition, we show that effective target gene silencing after the first exposure was not a prerequisite for RNAi refractoriness. Since that only the dsRNAs targeting endogenous genes could induce the RNAi refractoriness, this process might involves the dsRNA and target mRNA interaction. In addition, the fact that target gene silencing is not necessary for the RNAi refractoriness demonstrated that low level of dsRNA and target mRNA interaction, although not enough to silence target genes, is enough to elicit RNAi refractoriness. Thus, the high level of this interaction caused by high concentration of dsRNA guarantees a prolonged refractoriness.
RNAi refractoriness primed by different concentrations of rpl19 dsRNA.
(A) The RNAi effect induced by oral administration of different concentrations of rpl19 dsRNA. (B) RNAi refractoriness to secondary rpl19 dsRNA exposure, primed by 100 ng/μl rpl19 dsRNA. (C) RNAi refractoriness to secondary spr dsRNA exposure, primed by 100 ng/μl rpl19 dsRNA. (D) RNAi refractoriness to secondary rpl19 dsRNA exposure, primed by 10 ng/μl rpl19 dsRNA. (E) RNAi refractoriness to secondary spr dsRNA exposure, primed by 10 ng/μl rpl19 dsRNA. Normalised target gene expression is reported relative to expression in the egfp dsRNA control, which was set to 1. All error bars represent the SE of the mean of three independent biological replicates. * indicates a statistically significant difference in spr or rpl19 expression between rpl19 dsRNA or spr dsRNA and the control egfp dsRNA treatments (P < 0.01). Different letters indicate a significant difference in rpl19 or spr expression among the rpl19 dsRNA or spr dsRNA treatments (P < 0.01).
Disruption of the endocytic pathway inhibits dsRNA entry
We next investigated the molecular mechanisms underlying RNAi insensitivity. In our conditions, the midgut is expected to be involved in dsRNA uptake13. Cy3-labelled dsRNAs were used to track dsRNA in the midgut cells of naive and challenged insects. Fluorescence microscopy revealed differences between the rpl19 dsRNA-challenged group and the egfp dsRNA naive group after secondary exposure to rpl19 dsRNA (Figure 4A). In the naive group, dsRNA begins to enter the cell after 30 minutes of incubation and after another 30 minutes, the dsRNAs accumulate in a spot near the nucleus. This indicates that egfp dsRNA treatment did not impair the ability of dsRNAs from the second exposure to enter the midgut cells. However, in the rpl19 dsRNA-challenged group, the dsRNAs failed to enter the midgut cells, as indicated by labelled dsRNAs accumulating outside the cells after both 30 and 60 min. These fluorescence microscopy observations demonstrate that the challenged group failed to respond to dsRNA feeding due to impaired dsRNA cellular uptake.
The cellular entry of dsRNA in rpl19 dsRNA-challenged flies is disrupted.
(A) Subcellular localization of Cy3-labelled dsRNA in the egfp dsRNA-treated naive group and the rpl19 dsRNA-treated challenged group over time. (B) Subcellular localization of Cy3-labelled dsRNA after incubation in the presence of Baf. (C) Expression levels of genes required for the endocytic entry of dsRNAs. (D) Microinjection of rpl19 dsRNA in the second RNAi. *, P < 0.05 and **, P < 0.01. Normalised expression of the target genes is given relative to their expression in the egfp dsRNA-treated control, which was set to 1. All error bars represent the SE of the mean of three independent biological replicates.
Two mechanisms of dsRNA uptake have been identified: one is mediated by the SID-1 transmembrane protein and the other is mediated by endocytosis30. However, there is no sid-1 gene in several insect genomes, including that of D. melanogaster9. Two independent works have shown that D. melanogaster relies on receptor-mediated endocytosis to take up dsRNA31,32. Bafilomycin A1 (Baf), a specific inhibitor of vacuolar proton ATPases, is often employed to demonstrate the requirement for low endosomal pH. Our results showed that pretreating the B. dorsalis midgut with baf blocked dsRNA entry into the midgut cells (Figure 4B). This strongly supports the notion that, in B. dorsalis, the entry of dsRNA into the cells requires endocytosis. We next examined the expression of genes reported to be responsible for the cellular entry of dsRNA in D. melanogaster31. These genes, such as chc, rab7, light and saposin, influence several crucial steps of endocytosis, including vesicle formation and transport, intracellular transport and lipid metabolism. Saleh et al. have shown that, in cell culture assays, RNAi against each of these genes inhibits dsRNA entry31. qPCR results showed that, in the rpl19 dsRNA-challenged group, most of these genes were repressed 24 hr after secondary exposure (Figure 4C). For example, chc, a key gene required for clathrin-mediated endocytosis that encodes the clathrin heavy chain protein, was down-regulated by more than 60%. A similar level of down-regulation was observed for the rab7, arf72a, light (a vacuolar protein sorting Vsp41 orthologue) and vacuolar H+-ATPase (V-H-ATPase) genes, which encode components of the endocytic vesicle trafficking and protein sorting pathways. Other genes, including members of the Golgi complex (COG) family (e.g., ldlCp, cog3 and bet3) were also down-regulated. nina c, which is required for actin polymerization and cytoskeletal organization, was also down-regulated after the second exposure. These results raised the hypothesis that RNAi refractoriness is caused by a decrease in endocytosis. In order to find if the RNAi refractoriness is systemic, we directly injected rpl19 dsRNA into the flies to see if it could bypass RNAi refractoriness. The result showed that, in the challenged group, direct injection of dsRNA could not reduce the expression of the target gene at 5 dpe. This result demonstrated that this refractoriness is systemic (Figure 4D).
Actin assembly has been shown to be an essential element of endocytosis33. Hydrogen peroxide (H2O2) induces the formation of cellular F-actin in a dose-dependent manner34. To test the role of endocytosis in RNAi refractoriness, we investigated the ability of H2O2 to disrupt RNAi refractoriness. Applying H2O2 during the secondary exposure disrupted RNAi refractoriness; a second exposure with 100 ng/μl rpl19 dsRNA and 5% H2O2 resulted in a 41.1% decrease in target gene expression compared with the egfp dsRNA treatment (Figure 5A). This is also supported by fluorescence microscopy; we found that dsRNA successfully entered the cells after co-incubation with H2O2 (Figure 5B). Because increasing the endocytic capacity with H2O2 led to a higher level of RNAi-mediated gene silencing, our results strongly suggest that refractoriness to RNAi is mediated by a decrease in endocytosis.
H2O2 can disrupt RNAi refractoriness.
(A) Co-feeding of 100 ng/μl rpl19 dsRNA with H2O2 in the second exposure. (B) Subcellular localization of Cy3-labelled dsRNA in the flies co-incubated with 5% H2O2 and rpl19 dsRNA in challenged group. (C) Co-feeding of 10 ng/μl rpl19 dsRNA with H2O2 in the first exposure. ** (P < 0.01) indicate significant differences in gene expression between the rpl19 dsRNA treatment and the control. The normalised expression of the target gene is given relative to its expression in the egfp dsRNA control, which was set to 1. All error bars represent the SE of the mean of three independent biological replicates.
DGE analysis reveals that RNAi refractoriness is intrinsic
Our results showed that 10 ng/μl rpl19 dsRNA could not efficiently reduce rpl19 expression (Figure 3A). Therefore, we tried to determine if RNAi insensitivity could also occur during the first dsRNA exposure. RNAseq analysis was used to identify genes that were differentially expressed after the first dsRNA exposure. RNA samples extracted at 8, 12 and 24 hr after exposure to 10 ng/μl rpl19 dsRNA or egfp dsRNA were mixed together and sequenced.
A total of 5,973,846 and 6,512,317 raw paired-end reads with a length of 100 bp, corresponding to 10 ng/μl rpl19 dsRNA treatment and 10 ng/μl egfp dsRNA treatment libraries were generated respectively. A total of 5,793,444 and 6,363,092 clean reads were left after removing reads with adaptors, reads containing poly N and low quality reads from raw data (Table S1). We mapped the sequences of two DGE libraries to the reference transcriptome dataset of B. dorsalis35, which contains 48,876 unigenes (Figure S6). This analysis identified 190 genes whose expression varied in response to feeding rpl19 dsRNA compared with egfp dsRNA treatment. The gene set comprised 26 up-regulated and 164 down-regulated genes. Gene Ontology (GO) enrichment analysis indicated that the genes were enriched for 5 biological processes (p < 0.001), including translation regulator activity, cell motility and transport (Table S2). KEGG analysis of the differentially expressed genes showed that they function in several processes (Figure S7). The up-regulated genes mainly function in metabolism and translation, whereas the down-regulated genes primarily function in metabolism, translation and transcription, folding, sorting and degradation and transport and catabolism.
Importantly, several genes that play a crucial role in endocytosis were found to be down-regulated in the 10 ng/μl rpl19 dsRNA-treated flies (Table 1). chc and hsc70, which encode factors that work with auxin to uncoat CCV from cargo36, as well as saposin, were found to be down-regulated. In addition, the expression of three genes encoding different F-actin isoforms, actin 3, actin 4 and actin 5, was reduced. The dynamic polymerization of actin has a central role in clathrin-mediated endocytosis, which reshapes the plasma membrane37. The expression of three kinases, hexo kinase 2, map2k1 and pgk1, which regulate endocytosis38, was also repressed. However, we failed to identify genes that had been previously characterised as involved in insect immunity; these genes encode components of the Toll, Imd and Jak/STAT pathways. This is consistent with the findings of Flenniken and Andino27, who used microarrays to identify genes involved in the dsRNA-mediated antiviral response in honey bees; they did not identify any classical immunity genes. These results, as well as ours, suggest that dsRNA-mediated antiviral defence may involve unique genes and signal transduction cascades27. In addition, we did not find core genes in the RNAi machinery, like dicer 2 and argonaute 2, in the DGE analysis, indicating the RNAi refractoriness is not due to the different activity of the RNAi machinery. The RNAseq results imply that endocytosis-mediated RNAi refratoriness also occurs at the first dsRNA exposure. To validate this hypothesis, we co-fed flies with 5% H2O2 and 10 ng/μl rpl19 dsRNA, a dsRNA concentration that does not reduce rpl19 expression. However, rpl19 expression still decreased 33.5% relative to the egfp dsRNA treatment (Figure 5C). This result demonstrates that H2O2, which influences endocytosis, can also influence RNAi-induced gene silencing. This observation reinforces our conclusion that variation in endocytic capacity can influence RNAi and that RNAi insensitivity is linked to a decrease in endocytic activity.
Conclusions
Taken together, our findings indicate that B. dorsalis possesses a mechanism to down-regulate dsRNA-mediated RNAi. We also demonstrated that primed RNAi refractoriness involves clathrin-mediated endocytosis. We hypothesize that our findings will extend to other insect species. In line with this assumption, other works have already shown that receptor-mediated endocytosis influenced dsRNA entry in D. melanogaster31. Our conclusion is also supported by the work of Whyard22, who showed that feeding dsRNA encapsulated by transfection reagents could induce RNAi in four different Drosophila species, whereas direct dsRNA feeding did not work. This suggests that a mechanism involved in dsRNA entry is impaired in Drosophila species. We speculate that this defect is linked to a reduction in endocytosis. A recent report focused on the early response of Drosophila S2 cells to viruses39. Interestingly, this work showed that receptors such as Sr-CI, Eater and TepI were significantly down-regulated after pathogenic virus treatments and significant changes in phagocytic activity were observed. It is known that together, Sr-CI and Eater contribute to more than 90% of dsRNA uptake into S2 cells. Considering the evolutionary conservation and functional relevance of the dsRNA entry pathway in intact organisms31, this change in membrane transport capacity might be common in host-pathogen interactions.
RNAi is an important viral defence mechanism in insects. The mechanism that we uncovered might affect the viral defences of insects and could explain the variability observed between the various physiological states that influence endocytosis. It also suggests that infection by a virus could influence RNAi silencing against a second virus or against a second exposure to the first virus. Furthermore, our work also has important consequences for the use of RNAi in other insects; it could explain why RNAi is difficult to achieve in some insect species. It also provides a solution to this problem by showing that promoting endocytosis enhances RNAi. In summary, our findings provide a perspective into mechanisms that allow invertebrates to protect their genetic information from non-self dsRNAs.
Methods
Fly rearing
B. dorsalis flies were reared at the Institute of Urban and Horticultural Pests at Huazhong Agricultural University. Adult flies were maintained in cages at 28°C under a 12 hr light:12 hr dark photoperiod and fed an artificial diet consisting of 2.5% yeast extract, 7.5% sugar, 2.5% honey, 0.4% agar and 87% H2O.
Plasmid construction
Total RNA was extracted from adult flies using RNAiso Plus reagent (TaKaRa, Japan). First strand cDNA was synthesised using a PrimeScript 1st Strand cDNA Synthesis Kit (TaKaRa, Japan). PCR fragments from each gene were cloned into the SacI and HindIII sites of the L4440 plasmid. The egfp and dsred fragment were cloned from the PUbnlsEGFP plasmid and the PUbnlsRED palsmid. All the primers used in the experiments were listed in Table S3. All the HT115 (DE3) competent cells lacking RNase III were prepared using standard CaCl2 methodology and were transformed with recombinant plasmid DNA.
dsRNA preparation and quantification
A single HT115 (DE3) colony was cultured overnight in LB at 37°C with shaking at 220 rpm. The culture was diluted 100-fold in 800 mL 2 × YT supplemented with 75 μg/mL ampicillin and 12.5 μg/mL tetracycline and cultured at 37°C to an OD600 of 0.5. Production of T7 polymerase was induced with 0.4 mM IPTG and the bacteria were incubated with shaking for an additional 4 hr at 37°C. Total nucleic acids were extracted40. The bacterial pellets were resuspended in 1 M ammonium acetate/10 mM EDTA and an additional volume of phenol:chloroform:isoamyl alcohol (25:24:1) was added. The samples were incubated at 65°C for 30 min and centrifuged at 12,000 g for 15 min. The upper phase was mixed with isopropanol, incubated at −20°C overnight and centrifuged at 12,000 g for 30 min. The nucleic acid pellet was resuspended in TE. For dsRNA quantification, the nucleic acids were treated with RQ1 RNase-free DNase (Promega, USA) and RNase A solution (Promega, USA). The concentration was determined using a NanoDrop 1000 (Thermo, USA). The dsRNAs were loaded onto a 2% agarose gel, stained with ethidium bromide and photographed to determine integrity.
Microinjection
Needles were prepared with a puller at 60°C (PC-10, Narishige, Tokyo Japan). Microinjection was performed using an Eppendorf micromanipulation system. The injection condition was set to a Pi of 300 hpa and a Ti of 0.3 s. A total of 100 ng egfp dsRNA or rpl19 dsRNA was injected into the flies in the challenged group.
Bioassays
Flies were collected five to ten days after emergence. For each treatment, flies were dehydrated/starved for 24 hr. The artificial diet was cut into circular pieces with 6 cm diameters. Each piece was covered with 400 μl of dsRNA solution prepared as described above. Unless mentioned, the concentration of dsRNA is 1000 ng/μl in the feeding bioassay. The flies were fed the artificial diet supplemented with dsRNA starting at 8:00 am and were returned to a normal artificial diet at 14:00 pm the same day. The experiments were performed in triplicate. For immune priming experiments, the flies were divided into two groups. For the first RNAi exposure, one group was fed rpl19 dsRNA or spr dsRNA as described above. These flies were referred to as the challenged group. The other group, referred to as the naïve group, was fed egfp dsRNA. We set different lag times between the first and second dsRNA exposures (5, 10, 20 and 30 days). At each time point, we collected flies from both the challenged and naïve groups and examined their responses to a second dsRNA exposure (either rpl19 dsRNA or spr dsRNA). In this experiment, egfp dsRNA was used as the control to determine the RNAi effect. In all cases, samples were collected 24 hr after the second exposure. All experiments were repeated in triplicate. For the RNAi refractoriness disruption experiments, 5% H2O2 was co-fed with the dsRNA.
Real-time PCR
For each treatment, 10 flies were collected for RNA extraction. RNA was extracted using RNAiso Plus reagent (TaKaRa, Japan). cDNA was synthesised from 100 ng total RNA using Transcript RT Master Mix (TaKaRa, Japan) according to the manufacturer's instructions. Real-time RT-PCR was performed using BioRad SYBR Green qPCR mix (BioRad, USA) according to the manufacturer's instructions, on a BioRad MyIQ2 machine. All RNA samples were analysed in triplicate. The reactions included 2 μl cDNA, 10 μl SYBR Green mix (BioRad, USA), 0.8 μl each of forward and reverse primers and 6.4 μl ddH2O. The reactions were set up in 96-well real-time PCR plates (BioRad, USA). The thermocycler conditions were 95°C for 30 s, followed by 40 cycles at 95°C for 15 s and 60°C for 30 s. Melting curve analysis was performed at the end of each amplification run to confirm the presence of a single peak. The thermocycler conditions for the melting curve analysis were 55°C for 60 s, followed by 81 cycles starting at 55°C for 10 s with a 0.5°C increase each cycle. To avoid off-target effects, qPCR primers were designed to detect the parts of the transcript outside the dsRNA target. sdha was used as a reference gene and it was selected from a pool of candidate genes using geNorm. The data were analysed using the 2−ΔΔCT method41 using IQ5 standard edition ver. 2.1 (BioRad, USA). The expression of rpl19 and spr was quantified relative to the levels of rpl19 and spr in the flies fed egfp dsRNA. Biological experiments were performed independently and in triplicate. All results from experimental replicates were analysed using Student's t-test or a one-way analysis of variance (ANOVA) and a Duncan's test using SPSS 20 (IBM Corporation, USA).
DGE analysis
A mixed sample of flies was collected 8, 12 and 24 hr after exposure to 10 ng/μl dsRNA. Total RNA was extracted and digested with DNase I (Ambion, USA). mRNA was purified with a Micropoly(A)Purist™ mRNA purification kit(Ambion, USA)following the manual's instructions. cDNA synthesis was performed with a SuperScript Double-stranded cDNA Synthesis Kit (Invitrogen, USA). cDNA was purified using Ampure beads (Agencourt, USA). The purified cDNA was used to prepare a library using a TruSeq™ DNA sample Prep Kit-Set A (Illumina, USA) and PCR amplification was performed using a TruSeq PE Cluster Kit (Illumina, USA). Finally, the products were sequenced on an Illumina HiSeqTM 2000 System (Illumina, USA) and 100 bp pair-end reads were generated. Clean reads were mapped to a B. dorsalis transcriptome dataset35. Expression values were calculated in units of RPKM. Statistical analysis to identify differentially expressed genes was performed using an MA plot-based method with a random sampling model in DEGseq42.
Immunofluoresence microscopy
dsRNA was fluorescently labelled using a Silencer siRNA Labelling Kit with Cy3 (Ambion, USA). Labelling of dsRNA was verified by decreased electrophoretic mobility compared with unlabelled dsRNA on an agarose gel. B. dorsalis midgut tissue from both the challenged and naïve groups were incubated with labelled dsRNA. For bafilomycin A1 treatment, midgut tissue was first incubated with 0.2 μM Baf for 30 minutes. Cy3-labelled dsRNA was then added to the reaction. The tissue was fixed for 20 min in 4% formaldehyde. Actin was visualized with Acti-stain™ 488 fluorescent phalloidin (Cytoskeleton, Inc., USA) following the instruction manual. Nuclei were counterstained with DAPI. Images were captured on an Olympus IX71 microscope driven by cellSens Dimension software (Olympus, Japan). All images were imported into and processed in Adobe Photoshop (Adobe, USA).
References
Fire, A. et al. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 391, 806–811 (1998).
Kennerdell, J. R. & Carthew, R. W. Use of dsRNA-mediated genetic interference to demonstrate that frizzled and frizzled 2 act in the wingless pathway. Cell 95, 1017–1026 (1998).
Turner, C. T. et al. RNA interference in the light brown apple moth, Epiphyas postvittana (Walker) induced by double-stranded RNA feeding. Insect Mol Biol 15, 383–391 (2006).
Dietzl, G. et al. A genome-wide transgenic RNAi library for conditional gene inactivation in Drosophila. Nature 448, 151–156 (2007).
Bernstein, E., Caudy, A. A., Hammond, S. M. & Hannon, G. J. Role for a bidentate ribonuclease in the initiation step of RNA interference. Nature 409, 363–366 (2001).
Hannon, G. J. RNA interference. Nature 418, 244–251 (2002).
Hammond, S. M. S., Bernstein, E. E., Beach, D. D. & Hannon, G. J. G. An RNA-directed nuclease mediates post-transcriptional gene silencing in Drosophila cells. Nature 404, 293–296 (2000).
Whangbo, J. S. & Hunter, C. P. Environmental RNA interference. Trends in Genetics 24, 297–305 (2008).
Huvenne, H. & Smagghe, G. Mechanisms of dsRNA uptake in insects and potential of RNAi for pest control: A review. Journal of Insect Physiology 56, 227–235 (2010).
Cheng, L. W. Use of RNA interference in Drosophila S2 cells to identify host pathways controlling compartmentalization of an intracellular pathogen. Proc. Natl. Acad. Sci. U.S.A. 102, 13646–13651 (2005).
Dorner, S. et al. A genomewide screen for components of the RNAi pathway in Drosophila cultured cells. Proc. Natl. Acad. Sci. U.S.A. 103, 11880–11885 (2006).
Davidson, B. L. & McCray, P. B. Current prospects for RNA interference-based therapies. Nat Rev Genet 12, 329–340 (2011).
Price, D. R. G. & Gatehouse, J. A. RNAi-mediated crop protection against insects. Trends in Biotechnology 26, 393–400 (2008).
Li, X., Zhang, M. & Zhang, H. RNA interference of four genes in adult Bactrocera dorsalis by feeding their dsRNAs. PLoS ONE 6, e17788 (2011).
Bolognesi, R. et al. Characterizing the mechanism of action of double-stranded RNA activity against western corn rootworm (Diabrotica virgifera virgifera LeConte). PLoS ONE 7, e47534 (2012).
Terenius, O. et al. RNA interference in Lepidoptera: An overview of successful and unsuccessful studies and implications for experimental design. Journal of Insect Physiology 57, 231–245 (2011).
Araujo, R. N. et al. RNA interference of the salivary gland nitrophorin 2 in the triatomine bug Rhodnius prolixus (Hemiptera: Reduviidae) by dsRNA ingestion or injection. Insect Biochemistry and Molecular Biology 36, 683–693 (2006).
Zhou, X., Wheeler, M. M., Oi, F. M. & Scharf, M. E. RNA interference in the termite Reticulitermes flavipes through ingestion of double-stranded RNA. Insect Biochemistry and Molecular Biology 38, 805–815 (2008).
Baum, J. A. et al. Control of coleopteran insect pests through RNA interference. Nat Biotechnol 25, 1322–1326 (2007).
Mao, Y.-B. et al. Silencing a cotton bollworm P450 monooxygenase gene by plant-mediated RNAi impairs larval tolerance of gossypol. Nat Biotechnol 25, 1307–1313 (2007).
Bellés, X. Beyond Drosophila: RNAi in vivo and functional genomics in insects. Annu. Rev. Entomol. 55, 111–128 (2010).
Whyard, S., Singh, A. D. & Wong, S. Ingested double-stranded RNAs can act as species-specific insecticides. Insect Biochemistry and Molecular Biology 39, 824–832 (2009).
Rajagopal, R., Sivakumar, S., Agrawal, N., Malhotra, P. & Bhatnagar, R. K. Silencing of midgut aminopeptidase N of Spodoptera litura by double-stranded RNA establishes its role as Bacillus thuringiensis toxin receptor. J. Biol. Chem. 277, 46849–46851 (2002).
Sioud, M. RNA interference and innate immunity. Advanced Drug Delivery Reviews 59, 153–163 (2007).
Robalino, J. et al. Induction of antiviral immunity by double-stranded RNA in a marine invertebrate. J. Virol. 78, 10442–10448 (2004).
Galiana-Arnoux, D., Dostert, C., Schneemann, A., Hoffmann, J. A. & Imler, J.-L. Essential function in vivo for Dicer-2 in host defense against RNA viruses in drosophila. Nat Immunol 7, 590–597 (2006).
Flenniken, M. L. & Andino, R. Non-specific dsRNA-mediated antiviral response in the honey bee. PLoS ONE 8, e77263 (2013).
Rowley, A. F. & Powell, A. Invertebrate immune systems specific, quasi-specific, or nonspecific? J. Immunol. 179, 7209–7214 (2007).
Rodrigues, J., Brayner, F. A., Alves, L. C., Dixit, R. & Barillas-Mury, C. Hemocyte differentiation mediates innate immune memory in Anopheles gambiae mosquitoes. Science 329, 1353–1355 (2010).
Gu, L. & Knipple, D. C. Recent advances in RNA interference research in insects: Implications for future insect pest management strategies. Crop Protection 45, 36–40 (2013).
Saleh, M.-C. et al. The endocytic pathway mediates cell entry of dsRNA to induce RNAi silencing. Nat. Cell Biol. 8, 793–802 (2006).
Ulvila, J. et al. Double-stranded RNA is internalized by scavenger receptor-mediated endocytosis in Drosophila S2 cells. J. Biol. Chem. 281, 14370–14375 (2006).
Galletta, B. J. & Cooper, J. A. Actin and endocytosis: mechanisms and phylogeny. Current Opinion in Cell Biology 21, 20–27 (2009).
Omann, G. M., Harter, J. M., Burger, J. M. & Hinshaw, D. B. H2O2-induced increases in cellular F-actin occur without increases in actin nucleation activity. Arch. Biochem. Biophys. 308, 407–412 (1994).
Zheng, W., Peng, T., He, W. & Zhang, H. High-throughput sequencing to reveal genes involved in reproduction and development in Bactrocera dorsalis (Diptera: Tephritidae). PLoS ONE 7, e36463 (2012).
Chang, H. C. Hsc70 is required for endocytosis and clathrin function in Drosophila. The Journal of Cell Biology 159, 477–487 (2002).
Kaksonen, M., Toret, C. P. & Drubin, D. G. Harnessing actin dynamics for clathrin-mediated endocytosis. Nat Rev Mol Cell Biol 7, 404–414 (2006).
Pelkmans, L. et al. Genome-wide analysis of human kinases in clathrin- and caveolae/raft-mediated endocytosis. Nature 436, 78–86 (2005).
Zhu, F., Ding, H. & Zhu, B. Transcriptional profiling of Drosophila S2 cells in early response to Drosophila C virus. Virology Journal 10, 1–1 (2013).
Timmons, L. & Fire, A. Specific interference by ingested dsRNA. Nature 395, 854–854 (1998).
Livak, K. J. & Schmittgen, T. D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2 − ΔΔCT Method. Methods 25, 402–408 (2001).
Wang, L., Feng, Z., Wang, X., Wang, X. & Zhang, X. DEGseq: an R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics 26, 136–138 (2010).
Acknowledgements
This work was supported by the earmarked fund for the China Agricultural Research System (No. CARS-27) and Fundamental Research Funds for the Central Universities (No. 2014PY005). We thank Professor Bruno Lemaitre from the Global Health Institute, EPFL, Switzerland and Professor Alfred M. Handler from the USDA-ARS, USA for insightful advice and comments that improved the manuscript.
Author information
Authors and Affiliations
Contributions
H.Z. and X.L. conceived the study and participated in its design. H.Z. provided the materials for the study. X.L. and X.D. performed the bioassays and DGE analysis. X.L. and C.Z. performed the immunofluorescence microscopy and H2O2 bioassays. H.Z. and X.L. performed the data analysis. H.Z. and X.L. wrote the manuscript. All authors read and approved the final manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Li, X., Dong, X., Zou, C. et al. Endocytic pathway mediates refractoriness of insect Bactrocera dorsalis to RNA interference. Sci Rep 5, 8700 (2015). https://doi.org/10.1038/srep08700
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep08700
This article is cited by
-
RNAi-directed knockdown in the cnidarian fish blood parasite Sphaerospora molnari
Scientific Reports (2024)
-
Ingestion of bacteria expressing dsRNA to maggots produces severe mortality and deformities in fruit fly, Bactrocera dorsalis (Hendel) (Diptera: Tephritidae)
Egyptian Journal of Biological Pest Control (2021)
-
Innate and adaptive resistance to RNAi: a major challenge and hurdle to the development of double stranded RNA-based pesticides
3 Biotech (2021)
-
Advances in exogenous RNA delivery techniques for RNAi-mediated pest control
Molecular Biology Reports (2020)
-
Identification of highly effective target genes for RNAi-mediated control of emerald ash borer, Agrilus planipennis
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.