Abstract
The dependence of the glass transition in mixtures on mixing thermodynamics is examined by focusing on enthalpy of mixing, ΔHmix with the change in sign (positive vs. negative) and magnitude (small vs. large). The effects of positive and negative ΔHmix are demonstrated based on two isomeric systems of o- vs. m- methoxymethylbenzene (MMB) and o- vs. m- dibromobenzene (DBB) with comparably small absolute ΔHmix. Two opposite composition dependences of the glass transition temperature, Tg, are observed with the MMB mixtures showing a distinct negative deviation from the ideal mixing rule and the DBB mixtures having a marginally positive deviation. The system of 1, 2- propanediamine (12PDA) vs. propylene glycol (PG) with large and negative ΔHmix is compared with the systems of small ΔHmix and a considerably positive Tg shift is seen. Models involving the properties of pure components such as Tg, glass transition heat capacity increment, ΔCp and density, ρ, do not interpret the observed Tg shifts in the systems. In contrast, a linear correlation is revealed between ΔHmix and maximum Tg shifts.
Similar content being viewed by others
Introduction
Glass transition is a typical liquid-solid non-equilibrium transition occurred in all kinds of materials, however, the physics of the phenomenon remains unsolved and debated1,2,3. The glass transition behaviors are found to be quite sensitive to even subtle modification in molecular structure and intermolecular interactions4. Binary glass forming mixtures are simple and appealing systems to investigate the glass transition due to the ease in modifying the intermolecular interactions. Miscible mixtures usually show a single glass transition temperature, Tg5, which might not necessarily be the mean value of the pure components' Tgs produced by the ideal mixing rule, but is related to the mixture composition (e.g. mole fraction, x)6,7.
The composition dependence of Tg in mixtures has been extensively studied in a number of systems and diverse observations in the Tg-x relationships have been reported, showing positive, negative and zero deviations from the ideal mixing rule5,6,8,9,10,11,12,13. Several models are proposed in search of the explanation for such deviations14, such as the most widely used Couchman - Karasz (CK) and Gordon - Taylor (GT) models15,16. The CK model takes the glass transition as an Ehrenfest second order transition and assumes the changes upon mixing in enthalpy (ΔHmix), entropy (ΔSmix) and volume (ΔVmix) for both liquid and glass to be negligible at Tg. Moreover, the glass transition heat capacity increment, ΔCp, is assumed to be temperature-independent. The GT model is proposed based on ideal volume of mixing and the linear change in volume with temperature. The two models have the similar expression as, Tg = (x1Tg1 + kix2Tg2)/(x1 + kix2), where kCK = ΔCp1/ΔCp2 is determined for the CK model while for the GT model kGT = ρ2Tg2/ρ1Tg1 and xi and ρi (i = 1, 2) denote the mole fraction and density of pure components17. Obviously, the two models emphasize the significance of two components' properties including Tg, ΔCp and ρ. Although the two models have successfully explained some experimental measurements18,19, there are a number of systems that show pronounced discrepancy between experimental measurements and the theoretical calculation made by the models6,13,20. Alternatively, mixing thermodynamics is argued to determine the glass transition behaviors in mixtures and studies found that positive enthalpy of mixing generally corresponds to negative Tg deviation5 and even a value of ΔHmix as low as 200 J/mol can result in a remarkably negative Tg shift21. However, the evaluation of the weighted contributions to the Tg shifts from both mixing thermodynamics and the properties of pure components have not been available. Moreover, systematic studies of the dependence of Tg on mixing thermodynamics have not been done. Therefore, the studies of the impact of ΔHmix on the Tg shifts in mixtures by changing both magnitude (small vs. large) and sign (positive vs. negative) would help gain insight into the glass transition behaviors in mixtures. Unfortunately, such studies are not accessible, in particular, the effect of small ΔHmix.
In this work, we studied the glass transitions in binary glass forming molecular systems and systematic comparison is shown with typical scenarios of small vs. large and positive vs. negative ΔHmix. A global picture of the ΔHmix effect on the Tg shifts in mixtures is presented. The results are expected to benefit the understanding of the glass transition as well as the precise evaluation of Tg in poor glass formers.
Results
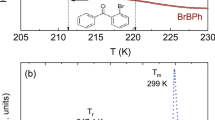
Fig. 1 presents the Cp curves around Tg for the glass forming o- vs. m- MMB and o- vs. m- DBB systems. The composition dependence of ΔCp obtained from all the Cp curves are plotted in Fig. 2(a) and (b) for the two systems. The ΔCp values of the pure components are estimated by extrapolating the mixtures' ΔCp and 0.619, 0.673, 0.35 and 0.38 J/(g–K) are obtained for o-MMB, m-MMB, o-DBB and m-DBB. The composition dependences of Tg are plotted in Fig. 3(a) and (b). A markedly negative Tg deviation is immediately visible for the mixtures of o- vs. m- MMB, although the deviation is small. In contrast, the evaluation of the Tg shift for the system of o- vs. m-DBB in Fig. 3(b) is of a bit difficulty. The fitting with marginally positive deviation or a linear fitting seems to be able to explain the experimental points, as shown by the black solid line and the light cyan solid line. The bottom line for o- vs. m-DBB is that the deviation is, by no means, negative. The pure components' Tgs can be evaluated by the numeric extrapolation in Fig. 3, yielding 156.1, 147.5, 160 and 157.2 K for o-MMB, m-MMB, o-DBB and m-DBB. The ΔHmix values are also presented in Fig. 3(c) and (d), reported for the mixtures of o- vs. m- MMB and o- vs. m- DBB, with ΔHmix maxima to be 3.42 and −4.98 J/mol, respectively22. Fig. 4(a) and (b) show the composition dependences of ΔCp and Tg for the system of 12PDA vs. PG over the whole composition, where ΔHmix maximum of 12PDA vs. PG is recorded to be −3876 J/mol23. A remarkably positive Tg deviation is seen in the system. The ΔCp values for 12PDA and PG are determined to be 1.36 and 0.86 J/(g–K) while the Tgs to be 146.8 and 169.7 K.
Composition dependence of Tg and ΔHmix in the isomeric mixtures of o- vs. m- methoxymethylbenzene (MMB) and o- vs. m- dibromobenzene (DBB).
In the upper frames (a) and (b), the red solid squares and circles are experimental values. The fitting line for the o- vs. m- methoxymethylbenzene (MMB) is given by the black solid line, while the fitting line for the o- vs. m- dibromobenzene (DBB) is expressed by the black or the light cyan solid line. The predicted values by the CK and GT models are shown by the blue dotted (CK) and magenta dash-dot-dot (GT) lines. The lower frames (c) and (d) show ΔHmix values for the two isomeric systems recorded in Ref. 22. The horizontal axes show the mole fraction of m- MMB (x) and DBB (x).
The explanations of the Tg values in the mixtures using the CK and GT equations with the extrapolated Tg and ΔCp values of the pure components are presented by the blue dotted (CK) and magenta dash-dot-dot (GT) lines in Fig. 3(a), (b) and Fig. 4(b). It appears that the calculated Tg curves in terms of the CK and GT models do not satisfactorily explain the experimental measurements. Similar observations have been reported in early studies of a number of glass forming mixtures13,20. The large discrepancy suggests that, notwithstanding the importance of the properties (Tg, ΔCp and ρ) of the pure components, the exact explanation of the Tg - x relationship for most cases asks for more considerations.
The relative Tg shift, ΔTg/Tg, for the systems of o- vs. m- MMB, o- vs. m- DBB and 12PDA vs. PG are shown in Fig. 5, where ΔTg is the Tg difference between the measured values and the ones calculated in terms of the linear average of the pure components' Tgs. Two more typical systems recorded in our recent work, benzil (BZL) vs. m-nitroaniline (MNA)21 and methyl m-toluate (MMT) vs. methyl o-toluate (MOT)24, are also included in Fig. 5 to represent the mixing scenarios of larger positive ΔHmix and ideal mixing, respectively. It is seen in Fig. 5 that the five systems differ greatly in the Tg deviations from the ideal mixing rule. The system of 12PDA vs. PG has a remarkably large and positive Tg deviation with the maximum ΔTg/Tg being as high as 14.5%. In contrast, the system of BZL vs. MNA has a large and negative Tg deviation with the maximum of ΔTg/Tg of −2.3%21. For systems with smaller positive or negative ΔHmix, ΔTg/Tg becomes smaller.
Relative Tg differences, ΔTg/Tg, for five typical glass forming systems, representing various mixing modes.
(1) ΔHmix ≫ 0 (benzil (BZL) vs. m- nitroaniline (MNA)), (2) ΔHmix ≥ 0 (o- vs. m- methoxymethylbenzene (MMB)), (3) ΔHmix = 0 (methyl m-toluate (MMT) vs. methyl o-toluate (MOT)), (4) ΔHmix ≤ 0 (o- vs. m- dibromobenzene (DBB)) and (5) ΔHmix ≪ 0 (1, 2- propanediamine (12PDA) vs. propylene glycol (PG)).
Discussion
The mixing thermodynamics for the BZL vs. MNA system (Fig. 5) is recently measured showing the ΔHmix maximum of ~200 J/mol21, while the MMT vs. MOT system is regarded as an ideal mixing case with ΔHmix = 024,25. Accordingly, the five cases in Fig. 5 represent the typical model systems of ΔHmix ≫ 0 (BZL vs. MNA), ΔHmix ≥ 0 (o- vs. m- MMB) ΔHmix = 0 (MMT vs. MOT), ΔHmix ≤ 0 (o- vs. m- DBB) and ΔHmix ≪ 0 (12PDA vs. PG). It is therefore evident that systems with large absolute values of ΔHmix, either positive or negative, show remarkable negative or positive Tg shifts. Similar observations have been recorded in earlier experimental measurements. For example, the binary mixtures constituted by monoalcohols and amines with large and negative ΔHmix26,27,28 generally show large and positive Tg shifts29. Likewise, large and negative ΔHmix is also detected in the mixtures composed of water and alkoxy alcohols, such as the aqueous systems of 2-methoxyethanol30 and 2-ethoxyethanol31 and large and positive Tg deviations are observed in such mixtures10.
The results in this work allow us to check the presumably quantitative correlation between Tg and ΔHmix in binary mixtures. In addition to the experimental measurements reported here, more Tg8,21,25,29,32 and ΔHmix values21,22,23,33,34,35,36,37 are collected. Fig. 6 shows a plot between ΔTg/Tg and ΔHmix measured at 298 K for 13 systems with ΔHmix ranging from 1044 J/mol to −3900 J/mol. Strikingly, a nearly linear correlation is revealed and positive ΔTg/Tg values of mixtures are found to be enlarged with increasingly negative ΔHmix. Note that although systems with positive ΔHmix are relatively fewer, the available data in the three systems basically follow the correlation showing that increasingly positive ΔHmix corresponds to enhanced negative Tg deviation (ΔTg/Tg). Note that in Fig. 6 there are a few systems that somehow deviate from the master line such as methanol vs. triethylamine29,33. This may arise partly from the error in ΔHmix values, since several different ΔHmix values can be found for a mixing system38. However, the minor errors do not affect the demonstration of the explicit relationship between the thermodynamics and the glass transition behaviors in binary mixtures. Compared with the results in Figs. 3 and 4 expressed by the CK and GT models, Fig. 6 consequently implies that ΔHmix could be a more decisive factor in affecting the Tg behaviors in mixtures.
Correlation between ΔHmix and ΔTg/Tg based on 13 binary molecular glass forming systems.
(1) 1- butanol vs. 1- bromobutane41,43,44; (2) benzil (BZL) vs. m- nitroaniline (MNA); (3) o- vs. m- methoxymethylbenzene (MMB); (4) methyl m-toluate (MMT) vs. methyl o-toluate (MOT); (5) o- vs. m- dibromobenzene (DBB); (6) chloroform (CFM) vs. toluene8,34; (7) diethylether (DEE) vs. dichloromethane (DCM)8,37; (8) methanol vs. triethylamine29,33 (9) 2-ethyl-1-hexanol (2E1H) vs. 2-ethyl-1-hexylamine (2EHA)25 (10) chloroform (CFM) vs. diethylether (DEE)8,36 (11) chloroform (CFM) vs. triethylamine (TEA)8,35 (12) 1,2- propanediamine (12PDA) vs. propylene glycol (PG); (13) 1,2- propanediamine (12PDA) vs. 1,3- propanediol (13PDO)23,32. The correlation for the systems of (3), (4) and (5) is given in the insert.
Fig. 6 includes a typical glass forming binary system of 2-ethyl-1-hexanol (2E1H) and 2-ethyl-1-hexylamine (2EHA) and the accurate Tg data show a large and positive deviation with ΔTg/Tg reaching 5.6%39. The enthalpy of mixing in the system is not available. Yet, the value could be roughly evaluated from the mixtures composed of the homologous liquids with similar molecular structures and chemistry. For example, the ΔHmix maximum at 298 K reaches −2905 J/mol for the system of butyl amine vs. 1-butanol26, which is comparable with the value of −2755 J/mol for the system of amylamine vs. 1-butanol27. It is seen that the absolute values of the negative ΔHmix for mixtures of monoalcohols and monoamines decrease with the carbon number in the backbones of either alcohols or amines and also decrease with the enhanced steric hindrance due to the introduction of branched chains26,27,28,40. Approximately, the ΔHmix maximum for the mixtures of 2E1H vs. 2EHA is evaluated to be ~−2200 J/mol. The values of ΔHmix and ΔTg/Tg (5.6%39) for the 2E1H - 2EHA mixtures appear to coincide well with the correlation shown in Fig. 6.
The mixing systems of positive ΔHmix are much less available. The system of 1-butanol vs. 1-bromobutane is reported to have a quite large and positive ΔHmix with the maximum reaching 1044 J/mol at 298 K41. The composition dependence of the kinetic glass transition temperature, Tg-kin, defined by the temperature where the α-relaxation time approaches 100 s39,42, can be obtained based on the studies of the dielectric relaxation dynamics for the pure components and the mixtures investigated by Goresy et al43 and by Murthy et al44. When combining the data from the two groups, a negative Tg deviation with the maximum of ΔTg/Tg to be −3.2% is yielded. The result is comparable with the predicted one by the correlation in Fig. 6.
For ideally mixing systems with ΔHmix = 0, a common observation is the negative Tg deviation from the ideal mixing rule, as shown in Figs. 5 and 6 for the MMT vs. MOT mixtures. This is the typical feature driven by mixing entropy29,45. According to the Adam-Gibbs' configurational entropy (Sc) theory46, τ(T) = τ0 exp(ΔμCAG/TSc), where τ0 is the pre-exponent having the phonon time, Δμ is potential barrier hindering molecular rearrangement and CAG is a constant47,48,49, if the intermolecular interaction keeps unchanged (ΔHmix = 0), the variation in Δμ is negligible and the increase in Sc during mixing at a fixed temperature would correspond to a decrease in τ(T), which makes the relaxation time curves move toward low temperature regions in activation plots (log τ(T) vs. 1/T) and finally leads to a decrease in Tg-kin (or Tτ = 100 s). Furthermore, for the small and comparably-valued ΔHmix of opposite sign, the case with positive ΔHmix (for example, the o- vs. m- MMB system in the inset of Fig. 6) would correspond to relatively larger Tg shift (negative) than the shift (positive) in the case with negative ΔHmix (for example, the o- vs. m- DBB system).
In addition to the correlation between ΔHmix and the Tg shifts in glass forming mixtures, recent studies explored the liquid fragility in various mixtures with varying ΔHmix, which defines how rapidly the liquid dynamic properties (viscosity or relaxation time) change with temperature at Tg and found a general trend of negative deviations from the ideal mixing rule25, somehow regardless of the enthalpy of mixing. Combining the impacts of ΔHmix on Tg and fragility in mixtures, it is evident that large and negative enthalpy of mixing would correspond to the high viscosity within supercooled liquid regions50, which further favors kinetically glass formation. This rationalizes the empirical criterion that the large and negative enthalpy of mixing is a crucial consideration in order to prepare multicomponent bulk metallic glasses51,52.
Finally, the present results of the quantitative correlation between ΔHmix and the composition dependence of Tg in mixtures can provide a guidance to estimating the Tg for materials which are extremely difficult to be vitrified, such as benzene. For the mixtures containing benzene, although a complete dataset of both mixing thermodynamics and glass transition are not available, considering the fact that for most mixtures the composition dependences of Tg generally show a similar trend as the composition dependence of viscosity29,53, the latter can consequently be used to roughly estimate the former. ΔHmix and viscosity for benzene vs. decane mixtures have been reported, showing a positive ΔHmix54 but negative deviation in the composition dependence of viscosity55, which could imply the negative deviation in the composition dependence of Tg. This agrees with the observation in the present work for the correlation between the mixing thermodynamics and glass transitions in various mixtures. It is therefore speculated that, provided that the enthalpy of mixing is given for the glass forming miscible mixtures containing benzene, the reliable extrapolation towards the Tg of pure benzene can be ensured based on the accessible Tg values of the mixtures. Such endeavor will be conducted in further study.
Methods
Chemicals
o- and m- isomeric systems of methoxymethylbenzene (MMB) and dibromobenzene (DBB) were selected in this study with the former system having small and positive ΔHmix and the latter system small and negative ΔHmix22. The system of 1, 2- propanediamine (12PDA) vs. propylene glycol (PG) was selected for the considerably large and negative ΔHmix generated during mixing23. For comparison, the results for the system of benzil (BZL) vs. m-nitroaniline (MNA) featured by the remarkable large and positive ΔHmix in our recent studies were used21. o- MMB (Alfa Aesar 99%), m- MMB (Alfa Aesar 99%), o- DBB (Alfa Aesar 98%), m- DBB (Alfa Aesar 97+%), 12PDA (Sigma Aldrich 99%), PG (Sigma Aldrich 99%) were used as received.
Thermodynamic measurements
A Perkin-Elmer differential scanning calorimeter (Diamond DSC) was used for the calorimetric determination of the heat capacity curves and the glass transition temperatures, Tg. A sapphire sample (31.4 mg) was used for the Cp standard. The sample was initially quenched to low temperatures at the accessible highest cooling rate in the calorimeter to guarantee the complete vitrification and a subsequent heating-cooling-reheating cycle was performed around the glass transition at a fixed rate of 20 K/min. The calorimetric quantities for the systems are determined from the reheating Cp curves. The sapphire and baseline heat curves were obtained following the same procedure. The DSC was calibrated using indium and cyclohexane prior to the measurements56.
References
Angell, C. A. & Ueno, K. Materials science: Soft is strong. Nature (London) 462, 45–46 (2009).
Capaccioli, S., Paluch, M., Prevosto, D., Wang, L.-M. & Ngai, K. L. Many-body nature of relaxation processes in glass-forming systems. J. Phys. Chem. Lett. 3, 735–743 (2012).
Ediger, M. D. & Harrowell, P. Perspective: Supercooled liquids and glasses. J. Chem. Phys. 137, 080901 (2012).
Babu, R. J., Brostow, W., Kalogerous, I. M. & Sathigari, S. Glass transitions in binary drug polymer system. Mater. Lett. 63, 2666–2668 (2009).
Painter, P. C., Graf, J. F. & Coleman, M. M. Effect of hydrogen bonding on the enthalpy of mixing and the composition dependence of the glass transition temperature in polymer blends. Macromolecules 24, 5630–5638 (1991).
Shamblin, S. L., Taylor, L. S. & Zografi, G. Mixing behavior of colyophilized binary systems. J. Pharm. Sci. 87, 694–701 (1998).
Schneider, H. A. & Di Marzio, E. A. The glass temperature of polymer blends: Comparison of both the free volume and the entropy predictions with data. Polymer 33, 3453–3461 (1992).
Lesikar, A. V. Effect of association complexes on the glass transition in organic halide mixtures. J. Phys. Chem. 80, 1005–1011 (1976).
Murthy, S. S. N. & Tyagi, M. Dielectric study of the miscibility of binary liquids, one being an alcohol. J. Solution. Chem. 31, 33–58 (2002).
Sjöström, J., Mattsson, J., Bergman, R. & Swenson, J. Hydrogen bond induced nonmonotonic composition behavior of the glass transition in aqueous binary mixtures. J. Phys. Chem. B 115, 10013–10017 (2011).
Wang, L.-M., Tian, Y. J., Liu, R. P. & Ngai, K. L. Anomalous component dynamics of a binary mixture of associating glass-forming liquids. J. Phys. Chem. B 115, 719–724 (2011).
Gupta, P. K. & Mauro, J. C. Composition dependence of glass transition temperature and fragility. I. A topological model incorporating temperature-dependent constraints. J. Chem. Phys. 130, 094503 (2009).
Pinal, R. Entropy of mixing and the glass transition of amorphous mixtures. Entropy 10, 207–223 (2008).
Kalogeras, I. M. & Brostow, W. Glass transition temperatures in binary polymer blends. J. Polym. Sci., Part B: Polym. Phys. 47, 80–95 (2009).
Couchman, P. R. & Karasz, F. E. A classical thermodynamic discussion of the effect of the composition on glass-transition temperatures. Macromolecules 11, 117–119 (1978).
Gordon, J. M. & Taylor, J. S. Ideal copolymers and the second-order transitions of synthetic rubbers. I. Non-crystalline copolymers. J. Appl. Chem. 2, 493–500 (1952).
Corti, H. R., Nores-Pondal, F. J. & Angell, C. A. Heat capacity and glass transition in P2O5-H2O solutions: Support for Mishima's conjecture on solvent water at low temperature. Phys. Chem. Chem. Phys. 13, 19741–19748 (2011).
Takeda, K., Yamamuro, O. & Suga, H. Thermal study of glass transitions in binary systems of simple hydrocarbons. J. Therm. Anal. 38, 1847–1860 (1992).
Karavas, E., Georgarakis, E. & Bikiaris, D. Application of PVP/HPMC miscible blends with enhanced mucoadhesive properties for adjusting drug release in predictable pulsatile chronotherapeutics. Eur. J. Pharm. Biopharm. 64, 115–126 (2006).
Gong, H. X. et al. Unusual dielectric strength of debye relaxation in monohydroxy alcohols upon mixing. J. Phys. Chem. B 116, 11482–11487 (2012).
Tu, W. K. et al. Glass transition and mixing thermodynamics of a binary eutectic system. Phys. Chem. Chem. Phys. 16, 3586–3592 (2014).
Kimura, T., Takahashi, Y., Kamiyama, T. & Fujisawa, M. Enthalpy of mixing of o- and m- isomers at 298.15 K. J. Therm. Anal. Calorim. 88, 587–595 (2007)
Kimura, T., Ozaki, T., Nakai, Y., Takeda, K. & Takagi, S. Excess enthalpies of binary mixtures of propanediamine + propanediol at 298.15 K. J. Therm. Anal. 54, 285–296 (1998)
Wang, L.-M., Liu, R. P., Tian, Y. J. & Richert, R. Structural relaxation dynamics in binary glass-forming molecular liquids with ideal and complex mixing behavior. J. Phys. Chem. B 114, 3618–3622 (2010).
Gong, H. X. et al. Kinetic fragility of binary glass forming liquid mixtures. Eur. Phys. J. E 34, 86 (2011).
Pradhan, S. D. The chain length and isomeric effect of alcohol on the excess properties of amine-alcohol systems: Excess free energy of mixing, enthalpy of mixing and volume change on mixing. Proc. Indian Acad. Sci. (Chem. Sci.) 90, 261–273 (1981).
Siman, J. E. & Vera, J. H. Heats of mixing of amine-alcohol systems. An analytical group solution model approach. Can. J. Chem. Eng. 57, 355–362 (1979).
Fernandez, J., Paz Andrade, M. I., Pintos, M., Sarmiento, F. & Bravo, R. Excess enthalpies of (secondary amine + alcohol) at 298.15 K. J. Chem. Thermodyn. 15, 581–584 (1983).
Lesikar, A. V. On the glass transition in mixtures between the normal alcohols and various Lewis bases. J. Chem. Phys. 66, 4263–4276 (1977).
Davis, M. I., Molina, M. C. & Douheret, G. Excess molar enthalpies of 2-methoxy ethanol + water and 2-ethoxy ethanol + water at 25°C. Therm. Acta 317, 105–109 (1998).
Kalasy, A. K., Nageshwar, G. D. & Mene, P. S. Excess enthalpy of water + 2- ethoxy ethanol. J. Chem. Thermodyn. 7, 101–102 (1975).
Takeda, K., Murata, K. & Yamashita, S. Thermodynamic study of the glass transition in polyamine - polyalcohol mixtures: entropy-theoretical interpretation of anomalous glass transition behavior. J. Phys. Chem. B 103, 3457–3460 (1999).
Chand, A. & Fenby, D. V. Thermodynamic properties of alcohol-amine mixtures: Excess enthalpies of methanol-triethylamine and ethanol-triethylamine. J. Chem. Eng. Data 22, 289–290 (1977).
Abello, L. Excess heats of binary systems containing benzene hydrocarbons and chloroform or methylchloroform. I. Experimental results. J. Chim. Phys. Phys. - Chim. Biol. 70, 1355 (1973).
Hepler, L. G. & Fenby, D. V. Thermodynamic study of complex formation between triethylamine and chloroform. J. Chem. Thermodyn. 5, 471–475 (1973).
Becker, F. & Kiefer, M. Mischungswärmen und verbindungsbildung in binären flüssigen systemen III. Äther - chloroform-systeme. Z. Naturforsch. A26, 1040–1046 (1971).
Findlay, T. J. V. & Kavanagh, P. J. Calorimetry of some hydrogen-bonding mixtures 2. Excess enthalpies of some mixtures containing halomethane. J. Chem. Thermodyn. 6, 367–371 (1974).
Christensen, C., Gmehling, J., Rasmussen, P. & Weidlich, U. Heats of Mixing Data Collection, Binary Systems, Vol III, Part 1, DECHEMA Chemistry Data Ser. (DECHEMA, Frukfurt am Main, 1984).
Wang, L.-M. & Richert, R. Debye type dielectric relaxation and the glass transition of alcohols. J. Phys. Chem. B 109, 11091–11094 (2005).
Kimura, T. et al. Excess enthalpies of binary mixtures of butylamines + propanols at 298.15 K. J. Therm. Anal. Calorim. 113, 1467–1474 (2013).
Garriga, R., Martinez, S., Perez, P. & Gracia, M. Vapor pressures at several temperatures between 278.15 K and 323.15 K and excess functions at T = 298.15 K for 1-bromobutane with 1-butanol or 2-methyl-2-propanol. J. Chem. Eng. Data 47, 322–328 (2002).
Hu, L. N. & Yue, Y. Z. Secondary relaxation behavior in a strong glass. J. Phys. Chem. B 112, 9053–9057 (2008).
EI Goresy, T. & Bohmer, R. Diluting the hydrogen bonds in viscous solutions of n-butanol with n-bromobutane: A Dielectric Study. J. Chem. Phys. 128, 154520 (2008).
Murthy, S. S. N., Sobhanadri, J. & Gangasharan . The origin of β relaxation in organic glasses. J. Chem. Phys. 100, 4601–4606 (1994).
Mugica, A., Calahorra, M. E. & Cortazar, M. Compositional variation of glass-transition temperature in miscible polymer blends involving weak and strong specific interactions. Macromol. Chem. Phys. 203, 1088–1098 (2002).
Adam, G. & Gibbs, J. H. On the temperature dependence of cooperative relaxation properties in glass forming liquids. J. Chem. Phys. 43, 139–146 (1965).
Bohmer, R., Gainaru, C. & Richert, R. Structure and dynamics of monohydroxy alcohols - milestones towards their microscopic understanding, 100 years after Debye. Phys.Rep. 545, 125–195 (2014).
Martinez-Garcia, J. C., Rzoska, S. J., Drzozd-Rzoska, A., Martinez-Garcia, J. & Mauro, J. C. Divergent dynamics and the Kauzmann temperature in glass forming systems. Sci. Rep. 4, 5160 (2014).
Paluch, M., Casalini, R., Best, A. & Patkowski, A. Volume effects on the molecular mobility close to glass transition in supercooled phenylphthalein-dimethylether. II. J. Chem. Phys. 117, 7624–7630 (2002).
Wang, L.-M., Tian, Y. J. & Liu, R. P. Dependence of glass forming ability on liquid fragility: Thermodynamics versus kinetics, Appl. Phys. Lett. 97, 181909 (2010).
Inoue, A. Stabilization of metallic supercooled liquid and bulk amorphous alloys. Acta Mater. 48, 279–306 (2000).
Wang, W. H. Roles of minor additions in formation and properties of bulk metallic glasses. Prog. Mater. Sci. 52, 540–596 (2007).
Shan, Z. J. & Asfour, A.-F. A. Viscosities and densities of nine binary 1- alkanol systems at 293.15 K and 298.15 K. J. Chem. Eng. Data 44, 118–123 (1999).
Diaz Pena, M. & Menduina, C. Excess enthalpies at 298.15 K of binary mixtures of benzene and n-alkanes. J. Chem. Thermodyn. 6, 387–393 (1974).
Lal, K., Tripathi, N. & Dubey, G. P. Densities, viscosities and refractive indices of binary liquid mixtures of hexane, decane, hexadecane and squalene with benzene at 298.15 K. J. Chem. Eng. Data 45, 961–964 (2000).
Chen, Z. M., Zhao, Y. & Wang, L.-M. Enthalpy and dielectric relaxations in supercooled methyl m- toluate. J. Chem. Phys. 130, 204515 (2009).
Acknowledgements
This work was supported by National Science Foundation of China (NSFC) (Grant No. 51131002, 51121061, 51271160 and 51071138), Natural Science Foundation of Hebei Province (No. A2014203260) and Project for Researches in Universities of Hebei Province (No. ZD2010113).
Author information
Authors and Affiliations
Contributions
W.K.T., Y.X.W., P.Z. and L.-M.W. finished the experiments and wrote the main manuscript. X.L., S.H.J. and Y.J.T. provide experimental and writing guidance. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Tu, W., Wang, Y., Li, X. et al. Unveiling the Dependence of Glass Transitions on Mixing Thermodynamics in Miscible Systems. Sci Rep 5, 8500 (2015). https://doi.org/10.1038/srep08500
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep08500
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.