Abstract
Curcumin is a principal ingredient of traditional Chinese medicine, Curcuma Longa, which possesses a variety of pharmacological activities including pain relief. Preclinical studies have demonstrated that curcumin has antinociceptive effects for inflammatory and neuropathic pain. This study examined the effects of curcumin in a rat model of postoperative pain. A surgical incision on the right hind paw induced a sustained mechanical hyperalgesia that lasted for 5 days. Acute curcumin treatment (10–40 mg/kg, p.o) significantly and dose dependently reversed mechanical hyperalgesia. In addition, repeated curcumin treatment significantly facilitated the recovery from surgery. In contrast, repeated treatment with curcumin before surgery did not impact the postoperative pain threshold and recovery rate. All the doses of curcumin did not significantly alter the spontaneous locomotor activity. Combined, these results suggested that curcumin could alleviate postoperative pain and promote recovery from the surgery, although there was no significant preventive value. This study extends previous findings and supports the application of curcumin alone or as an adjunct therapy for the management of peri-operative pain.
Similar content being viewed by others
Introduction
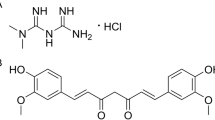
Curcumin, (1E, 6E)-1, 7-Bis (4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione, is the principal active ingredient of the traditional Chinese herb Curcuma Longa, commonly known as turmeric, which is also extensively used in South Asian cuisine. Scientific investigations of curcumin have seen a booming trend in the past decade, with several prominent therapeutic potentials being linked to curcumin. For example, curcumin has been found to possess anti-depressant like effects, anti-diabetic effects and anti-inflammatory activities1,2,3. Curcumin has also been reported to have beneficial effects on digestive disorders4. These exciting findings are of apparent significance given the easy availability and demonstrated long history of human use of curcumin-contained plants and herbs.
Recently, emerging studies suggest that curcumin also has pain relieving activities. For example, daily curcumin treatment reverts streptozotocin-induced diabetic neuropathy5,6,7,8. Acute curcumin treatment attenuates formalin-induced defensive behaviors, visceral pain as measured by acetic acid induced writhing response, capsaicin-induced thermal hyperalgesia and reserpine-induced fibromyalgia-like behaviors9,10,11,12,13,14. Regarding peripheral neuropathic pain, it seems that repeated curcumin treatment is necessary to revert the preexisting hyperalgesia. For instance, in a chronic constriction injury (CCI) model of neuropathic pain, acute curcumin treatment shows no effect on mechanical and thermal hyperalgesia, but repeated curcumin treatment progressively and completely reversed CCI-induced hypersensitivity15,16,17. Interestingly, a recent study suggests that pre- and peri-surgery curcumin treatment prevents the development of mechanical hyperalgesia in the rat model of CCI-induced neuropathic pain18, suggesting that curcumin might also have preventive value for injury-induced neuropathic pain. Combined, these previous studies strongly suggest that curcumin may have therapeutic value against some forms of pain.
This study was designed to examine the potential therapeutic effects of curcumin in a rat model of postoperative pain. Postoperative pain has well defined and predicable course of pain and thus this study examined the preventive, reverse and acute therapeutic activities of curcumin.
Methods
Animals
Male Sprague-Dawley rats with initial weights of 250–300 g (Laboratory Animal Center, Nantong University, China) were habituated to the temperature, humidity and lighting (12 h light/dark cycle, lights on at 7:00 AM) controlled housing facility and group housed for at least 3 days before behavioral studies began. The animals had free access to regular rodent chow and water except during the test sessions. All animal experimental protocols were approved by the Institutional Animal Care and Use Committee, Nantong University. Animals were maintained in accordance with the Guide for the Care and Use of Laboratory Animals (8th edition, Institute of Laboratory Animal Resources on Life Sciences, National Research Council, National Academy of Sciences, Washington DC). All testing was performed in accordance with the recommendations of the International Association for the Study of Pain. All efforts were made to minimize animal suffering and to decrease the number of animals used.
Drugs
Curcumin was purchased from Welch Materials Inc. (Shanghai, China) and was suspended in 0.5% carboxymethylcellulose. Drug was administered orally through gavage in a volume of 2 ml/kg of body weight.
Incisional surgery
Incisional surgery was performed as previously described19, with minor modifications. Briefly, rats were anaesthetized with ketamine (60 mg/kg)/xylazine (15 mg/kg) mixture and the plantar surface of the right hind paw was sterilely prepared. A 1 cm longitudinal incision was cut with a number 11 scalpel, through skin and fascia of the plantar aspect of the paw, starting 0.5 cm from the proximal end of the heel and extending toward the toes. The plantaris muscle was elevated and incised longitudinally. After bleeding was stopped through gentle pressure, the skin was opposed with two single sutures using 5–0 nylon. The animals were allowed to recover in their home cages.
Von Frey filament test
The mechanical hyperalgesia was measured by assessing paw withdrawal thresholds to mechanical stimuli using a series of von Frey filaments (Stoelting, USA). Ten von Frey filaments, with approximately equal logarithmic incremental bending forces (equivalent to 1, 1.4, 2, 4, 6, 8, 10, 15, 26, 60 g force, respectively), were used. Rats were placed in custom-made individual transparent Perspex cubicles with a wire mesh floor and allowed to acclimate for at least 15 min before testing began. The filaments were applied to the plantar surface of each hind paw in a series of ascending forces. Each filament was tested three times per paw and the mechanical threshold was defined as the minimal force that caused at least two withdrawals observed out of three consecutive trials20.
Locomotor activity
The locomotor activity of rats were measured using commercially available apparatus (YLS-1B, Shandong Academy of Medical Sciences, China), which consists of a controller unit and 4 circular black acrylic locomotion units (30 cm in diameter and 30 cm in height). The photo beam sensor located in the center of the cover of the locomotion units records the spontaneous locomotor activity. Locomotor activity was measured during a 60-min test period with each count indicating one beam break by the animal.
Experimental design
For the measurement of mechanical hyperthermia, the paw withdrawal threshold was measured 2 h after the surgery and daily thereafter for 7 days. Rats also received daily baseline measures prior to surgery for 3 days to habituate to the procedure and experimenters. For acute curcumin treatment, oral dosing of different doses of curcumin (10–40 mg/kg) was administered 1 day after surgery and then paw withdrawal threshold was measured every 30 min or until the drug effect dissipated. For chronic treatment study, a dose was administered 20 min prior to surgery and twice daily for 7 days. For prevention study, twice daily curcumin treatment was given for 7 days and then surgery was conducted and the paw withdrawal threshold was measured daily for 7 days.
Data analyses
For anti-hyperalgesia studies, paw withdrawal threshold (gram) was plotted as a function of time (min or days). For the locomotor activity study, total locomotion counts per 5 min blocks were plotted as a function of time. Data were analyzed using two-way repeated measures analysis of variance (ANOVA) using time as within group factor and treatment dose as between group factor, which was followed by post hoc Bonferroni analysis. Total locomotion counts within 60 min were also analyzed using one way ANOVA followed by post hoc Bonferroni analysis.
Results
Before the incisional surgery, the rats responded to von Frey filament stimulation in a similar manner on both hind paws (Fig. 1). Two hours after the surgery, the paw withdrawal threshold on right hind paw was drastically decreased, which gradually recovered to the pre-surgery level 6 days after the surgery. Two way ANOVA analysis revealed significant main effects of time (F [7, 98] = 17.82, P < 0.0001), surgery (F [1, 14] = 125.4, P < 0.0001) and time × surgery interaction F [7, 98] = 10.81, P < 0.0001). Post hoc analyses indicated that the paw withdrawal threshold remained significantly lower on the right hind paw as compared to the left hind paw for 5 days (P < 0.05). In contrast, the paw withdrawal threshold on the left paw remained relatively stable throughout the experimental period, which was not significantly different from rats that did not receive the incisional surgery (Fig. 1).
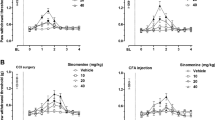
Acute curcumin administration dose-dependently increased the paw withdrawal threshold on the right hind paw (Fig. 2). Repeated testing within the 3 hr test period did not alter the baseline paw withdrawal threshold. Two way ANOVA analysis revealed significant main effects of time (F [6, 42] = 79.71, P < 0.0001), curcumin treatment (F [3, 21] = 5.48, P < 0.01) and time × treatment interaction F [18, 126] = 3.07, P < 0.001). Post hoc analyses indicated that the paw withdrawal threshold was significantly increased between time 30 and 150 min after 20 mg/kg curcumin administration and between 30 to 180 min after 40 mg/kg curcumin treatment (P < 0.05). The dose of 40 mg/kg oral curcumin administration produced anti-hyperalgesic effect that lasted for at least 3 hours (Fig. 2).
Repeated curcumin treatment facilitated the recovery of postoperative pain (Fig. 3). Two way ANOVA analysis revealed significant main effects of time (F [7, 49] = 198.1, P < 0.0001) and curcumin treatment (F [3, 21] = 10.41, P < 0.001). Post hoc analyses indicated that the paw withdrawal threshold was significantly increased at 2 hr, 3 and 4 days post-surgery after repeated 40 mg/kg curcumin treatment (P < 0.05). Repeated treatment with 10 or 20 mg/kg curcumin did not significantly improve the mechanical hyperalgesia (Fig. 3). Thus, in rats that received repeated 40 mg/kg curcumin treatment, their response to the von Frey filament stimulation returned to pre-surgery level at day 4 after surgery; while in rats that received vehicle treatment it typically took 6 days to recover, clearly demonstrating that curcumin could facilitate the recovery of surgical pain (Fig. 3). However, repeated curcumin treatment before incisional surgery did not alter the animals' paw withdrawal threshold nor did it facilitate the recovery from surgical pain (Fig. 4). Two way ANOVA did not identify statistical significance among rats with or without curcumin treatment.
In the test to measure the impact of acute curcumin treatment on the spontaneous locomotor activity, it was found that curcumin at the doses studied (10–40 mg/kg) did not significantly alter the general locomotor activity in rats (Fig. 5). One way and two way ANOVA did not reveal any significant statistical differences.
Discussion
The primary finding of this study was that acute curcumin had significant anti-hyperalgesic effects and repeated curcumin facilitated the recovery of postoperative pain. Although pre-surgery treatment did not seem to prevent or reduce postoperative pain, the positive findings on acute anti-hyperalgesia and pain recovery suggest that curcumin may be useful as an adjuvant treatment for pain management during peri-operative period.
Postoperative pain is perceived by patients as one of the most obnoxious aspects of surgical pain. Despite the ever-increasing understanding of the neurobiology and molecular biology of postoperative pain, the knowledge have not yet been translated to the successful development of novel analgesics for pain management including postoperative pain21. Consequently a large proportion of patients remain experiencing moderate to severe pain after surgery and the postoperative pain is not adequately controlled22,23. Basic studies have developed several rodent models of postoperative pain including the incision model as used in this study19. In this study, we found that the incision induced mechanical hyperalgesia lasted for 6 days, which is consistent with the literature19,24. Acute curcumin treatment was found to markedly alleviate mechanical hyperalgesia in this model of postoperative pain and the effect was clearly dose- and time-dependent. It should be noted that the observed antinociceptive effect is unlikely due to nonspecific behavioral suppression of curcumin because these doses of curcumin apparently did not alter the general locomotor activity. In previous studies, it has been shown that acute curcumin treatment attenuates formalin-induced defensive behaviors, reduces visceral pain in acetic acid induced writhing test and alleviates capsaicin-induced thermal hyperalgesia and reserpine-induced fibromyalgia-like behaviors9,10,11,12,13,14. Thus, acute curcumin demonstrates antinociceptive activity in a broad spectrum of pain conditions, supporting the potential utility of curcumin as an emerging analgesic for pain management.
Curcumin has been reported to revert the development and progression of neuropathic pain during repeated drug treatment15,16,17. In these studies, it was found that acute curcumin has little effect on peripheral neuropathic pain and the effect becomes evident only when the animals receive repeated curcumin treatment. We examined the potential effects of curcumin on the recovery of postoperative pain using a similar dosing regimen. It was found that the dose of 40 mg/kg curcumin significantly expedited the recovery of postoperative pain, as measured by the response to von Frey filament. Thus, at day 4 post-surgery, the paw withdrawal threshold of rats that received repeated 40 mg/kg curcumin was similar to that of pre-surgery, while rats that only received vehicle treatment had a much longer recovery period (6 days). This has apparent clinical significance. Shortening the recovery period will greatly reduce the medical health cost of surgery that relates both to the treatment of primary disorder and to the adverse effects.
Curcumin has also been found to have preventive value against the development of mechanical hyperalgesia in the rat model of CCI-induced neuropathic pain18. Animals that receive continual curcumin treatment before and after CCI surgery develop a much smaller magnitude of mechanical hyperalgesia12. In this study, we treated rats with curcumin repeatedly for 7 days before the incisional surgery and then measured the mechanical hypersensitivity daily after the surgery. No significant effect of curcumin treatment was found. Thus, curcumin may have little value as a prophylactic medication for painful surgical procedures.
The primary mechanisms of curcumin underlying the antinociceptive actions in postoperative pain are unclear. Indeed, numerous potential biochemical and pharmacological mechanisms have been linked to the pharmacological actions of curcumin such as monoamine system15, nitrogen oxidase signaling25, GABAa receptors26 and interleukins27, yet most of these biomarker changes seem to be secondary effects. Two mechanisms that involve the antinociceptive actions of curcumin seem more relevant15. Spinal β2 adrenoceptors and 5-HT1A receptors have been shown to involve curcumin antinociception in a mouse model of neuropathic pain, two receptor systems that have long been recognized as involving spinal pain processing15. These mechanisms may also be the underlying targets of curcumin for post-operative pain and future studies will examine this possibility.
In conclusion, the present study firstly demonstrated the anti-hyperalgesic effects of curcumin in a rat model of postoperative pain and that curcumin facilitated the recovery of surgical pain. This extends previous findings to the potential therapeutic value of curcumin on postoperative pain and further suggests in-depth assessment of the therapeutic value of curcumin as a novel analgesic for a broad spectrum of painful conditions.
References
Aggarwal, B. B., Gupta, S. C. & Sung, B. Curcumin: an orally bioavailable blocker of TNF and other pro-inflammatory biomarkers. Br J Pharmacol 169, 1672–1692 (2013).
Kulkarni, S., Dhir, A. & Akula, K. K. Potentials of curcumin as an antidepressant. ScientificWorldJournal 9, 1233–1241 (2009).
Zhang, D. W., Fu, M., Gao, S. H. & Liu, J. L. Curcumin and Diabetes: A Systematic Review. Evid Based Complement Alternat Med 2013, 636053 (2013).
Dulbecco, P. & Savarino, V. Therapeutic potential of curcumin in digestive diseases. World J Gastroenterol 19, 9256–9270 (2013).
Sharma, S., Kulkarni, S. K., Agrewala, J. N. & Chopra, K. Curcumin attenuates thermal hyperalgesia in a diabetic mouse model of neuropathic pain. Eur J Pharmacol 536, 256–261 (2006).
Li, Y. et al. Curcumin attenuates diabetic neuropathic pain by downregulating TNF-alpha in a rat model. Int J Med Sci 10, 377–381 (2013).
Banafshe, H. R. et al. Effect of curcumin on diabetic peripheral neuropathic pain: possible involvement of opioid system. Eur J Pharmacol 723, 202–206 (2014).
Zhao, W. C. et al. Curcumin ameliorated diabetic neuropathy partially by inhibition of NADPH oxidase mediating oxidative stress in the spinal cord. Neurosci Lett 560, 81–85 (2014).
Tajik, H., Tamaddonfard, E. & Hamzeh-Gooshchi, N. Interaction between curcumin and opioid system in the formalin test of rats. Pak J Biol Sci 10, 2583–2586 (2007).
Tajik, H., Tamaddonfard, E. & Hamzeh-Gooshchi, N. The effect of curcumin (active substance of turmeric) on the acetic acid-induced visceral nociception in rats. Pak J Biol Sci 11, 312–314 (2008).
Mittal, N., Joshi, R., Hota, D. & Chakrabarti, A. Evaluation of antihyperalgesic effect of curcumin on formalin-induced orofacial pain in rat. Phytother Res 23, 507–512 (2009).
Yeon, K. Y. et al. Curcumin produces an antihyperalgesic effect via antagonism of TRPV1. J Dent Res 89, 170–174 (2010).
Arora, V., Kuhad, A., Tiwari, V. & Chopra, K. Curcumin ameliorates reserpine-induced pain-depression dyad: behavioural, biochemical, neurochemical and molecular evidences. Psychoneuroendocrinology 36, 1570–1581 (2011).
Han, Y. K. et al. Analgesic effects of intrathecal curcumin in the rat formalin test. Korean J Pain 25, 1–6 (2012).
Zhao, X. et al. Curcumin exerts antinociceptive effects in a mouse model of neuropathic pain: descending monoamine system and opioid receptors are differentially involved. Neuropharmacology 62, 843–854 (2012).
Ji, F. T., Liang, J. J., Liu, L., Cao, M. H. & Li, F. Curcumin exerts antinociceptive effects by inhibiting the activation of astrocytes in spinal dorsal horn and the intracellular extracellular signal-regulated kinase signaling pathway in rat model of chronic constriction injury. Chin Med J (Engl) 126, 1125–1131 (2013).
Zhu, X. et al. Curcumin Alleviates Neuropathic Pain by Inhibiting p300/CBP Histone Acetyltransferase Activity-Regulated Expression of BDNF and Cox-2 in a Rat Model. PLoS One 9, e91303 (2014).
Jeon, Y. et al. Curcumin could prevent the development of chronic neuropathic pain in rats with peripheral nerve injury. Curr Ther Res Clin Exp 74, 1–4 (2013).
Brennan, T. J., Vandermeulen, E. P. & Gebhart, G. F. Characterization of a rat model of incisional pain. Pain 64, 493–501 (1996).
Yalcin, I. et al. beta(2)-adrenoceptors are critical for antidepressant treatment of neuropathic pain. Ann Neurol 65, 218–225 (2009).
Woolf, C. J. Overcoming obstacles to developing new analgesics. Nat Med 16, 1241–1247 (2010).
Apfelbaum, J. L., Chen, C., Mehta, S. S. & Gan, T. J. Postoperative pain experience: results from a national survey suggest postoperative pain continues to be undermanaged. Anesth Analg 97, 534–540 (2003).
Phillips, C., Gelesko, S., Proffit, W. R. & White, R. P., Jr Recovery after third-molar surgery: the effects of age and sex. Am J Orthod Dentofacial Orthop 138, 700 e701–708 (2010).
Whiteside, G. T. et al. Pharmacological characterisation of a rat model of incisional pain. Br J Pharmacol 141, 85–91 (2004).
Brouet, I. & Ohshima, H. Curcumin, an anti-tumour promoter and anti-inflammatory agent, inhibits induction of nitric oxide synthase in activated macrophages. Biochem Biophys Res Commun 206, 533–540 (1995).
Zhao, X. et al. Chronic curcumin treatment normalizes depression-like behaviors in mice with mononeuropathy: involvement of supraspinal serotonergic system and GABA receptor. Psychopharmacology (Berl) Epub (2013).
Abe, Y., Hashimoto, S. & Horie, T. Curcumin inhibition of inflammatory cytokine production by human peripheral blood monocytes and alveolar macrophages. Pharmacol Res 39, 41–47 (1999).
Acknowledgements
This work was supported by grants from National Natural Science Foundation of China (81373390) and Natural Science Foundation of Jiangsu Province (BK20130396).
Author information
Authors and Affiliations
Contributions
Q.Z., Y.S., W.Z. and J.L. conceived and designed the project and prepared the manuscript. Q.Z., Y.S., X.Y. and Y.O. conducted the experiments. Q.Z. and J.L. analyzed the data. All authors read and approved the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License. The images in this article are included in the article's Creative Commons license, unless indicated otherwise in the image credit; if the image is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the image. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Zhu, Q., Sun, Y., Yun, X. et al. Antinociceptive effects of curcumin in a rat model of postoperative pain. Sci Rep 4, 4932 (2014). https://doi.org/10.1038/srep04932
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep04932
This article is cited by
-
Chronic nanocurcumin treatment ameliorates pain-related behavior, improves spatial memory, and reduces hippocampal levels of IL-1β and TNFα in the chronic constriction injury model of neuropathic pain
Psychopharmacology (2021)
-
Analgesic effects of intravenous curcumin in the rat formalin test
Journal of Inclusion Phenomena and Macrocyclic Chemistry (2021)
-
Curcumin ameliorates neuropathic pain by down-regulating spinal IL-1β via suppressing astroglial NALP1 inflammasome and JAK2-STAT3 signalling
Scientific Reports (2016)
-
Intraoperative Dexmedetomidine Promotes Postoperative Analgesia and Recovery in Patients after Abdominal Hysterectomy: a Double-Blind, Randomized Clinical Trial
Scientific Reports (2016)
-
Zingiberaceae extracts for pain: a systematic review and meta-analysis
Nutrition Journal (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.