Abstract
We engaged in cooperative research with fishers and stakeholders to characterize the fine-scale, spatio-temporal characteristics of spawning behavior in an aggregating marine fish (Cynoscion othonopterus: Sciaenidae) and coincident activities of its commercial fishery in the Upper Gulf of California. Approximately 1.5–1.8 million fish are harvested annually from spawning aggregations of C. othonopterus during 21–25 days of fishing and within an area of 1,149 km2 of a biosphere reserve. Spawning and fishing are synchronized on a semi-lunar cycle, with peaks in both occurring 5 to 2 days before the new and full moon and fishing intensity and catch are highest at the spawning grounds within a no-take reserve. Results of this study demonstrate the benefits of combining GPS data loggers, fisheries data, biological surveys and cooperative research with fishers to produce spatio-temporally explicit information relevant to the science and management of fish spawning aggregations and the spatial planning of marine reserves.
Similar content being viewed by others
Introduction
Large breeding migrations are widespread among animals and represent a critical yet vulnerable ecological process1. In certain marine fishes, individuals travel vast distances to breed within immense spawning aggregations that occur at specific times and locations over brief periods and may represent the only opportunities to reproduce2. Spawning aggregations of marine fishes support some of the most productive and valuable fisheries worldwide, as fishers capitalize on the predictable nature of aggregations to harvest large numbers of fish with minimal effort3,4. Many aggregations have declined or collapsed due to overfishing and several aggregating species are at risk of extinction5. Therefore, the design and implementation of effective management and conservation policies for fish spawning aggregations are necessary for maintaining socioeconomic livelihoods and protecting marine biodiversity.
Effective management of fish spawning aggregations requires a detailed understanding of the dynamics of spawning behavior and associated fishing activities in both time and space4. For coastal fishes inhabiting subtropical and tropical regions, aggregations are often ephemeral with individuals gathering at a specific site over several days during specific tidal or lunar phases (e.g. new or full moons) in each of a few consecutive months of the year2. Fish disperse and emigrate away from the aggregation site at the conclusion of spawning but regroup at the time of the subsequent aggregation period. Fishing activities may become synchronized with the spawning cycles of fish, such that effort and catch are maximized during aggregation periods4. Since reproductive patterns of aggregate-spawning fishes and their fisheries often operate and interact on fine spatial and temporal scales, this requires that research investigations of spawning and fishing activities, assessments of fishing impacts and possible actions to mitigate such impacts be conducted equivalently6. Following an ecosystem approach to fisheries management, interactions between fishing and spawning aggregations with critical habitats, vulnerable or protected species and spatial management zones must also be considered6,7.
Advances in global positing systems (GPS) technology make it possible to acquire highly accurate data in rapid succession and in continuous time, providing new opportunities to aid the research, management and conservation efforts of fish spawning aggregations. For example, GPS data loggers can be deployed on fishing vessels to acquire fine-scale information on fishery dynamics and behavior8,9. These data can be combined with biological surveys and fisheries data to reveal heterogeneity of catch and fishing effort at spatial and temporal scales relevant to the behavior and ecology of target species, infer and map the distribution and abundance of target species in relation to specific coastal ecosystems or managed areas, quantify interactions of fishing activities with essential habitats of vulnerable or protected species and understand fisher behavior in response to temporal or area closures7,10,11. Moreover, the quality of research on fish spawning aggregations and their fisheries can be enhanced through cooperative research with fishers and stakeholders who can provide access to valuable information that may be otherwise unavailable, such as insights on the timing and locations of spawning, detailed records of landings and effort and techniques used to harvest fish from aggregations12,13.
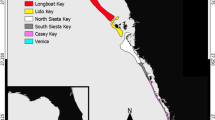
Here, we present information on the Gulf corvina (Cynoscion othonopterus), a large marine fish endemic to the Upper Gulf of California14 whose migration to its only known spawning grounds in the Colorado River Delta supports one of the most productive artisanal fisheries in Mexico (Figure 1; see Species Background). Gulf corvina aggregate to spawn in the shallow estuaries and channels of the Delta from late February to May, with spawning reported to occur during the peak spring tides (i.e. new and full moons)15,16. The commercial fishery for C. othonopterus targets its spawning aggregations, in which small fishing boats (pangas) use gill nets to harvest large volumes of fish as they move into the river mouth to reproduce15,17. The corvina fishery currently ranks only below shrimp in terms of the most important fisheries of the northern Gulf, with reported annual landings and ex-vessel revenues ranging from 2,200 to 5,900 tons and 2 to 3 million USD, respectively, over the past decade18,19.
Serious concerns exist regarding the sustainability of the corvina fishery and its environmental impact. C. othonopterus is listed as “vulnerable” by the American Fisheries Society and the International Union for the Conservation of Nature due to the loss of estuarine spawning and nursery habitat from diversions of Colorado River water, its susceptibility to overfishing when aggregated to spawn, its limited geographic range and reports of a prior population collapse20,21. The fishery is centered within the Upper Gulf of California and Colorado River Delta Biosphere Reserve, which includes a no-take zone to protect the spawning and nursery habitat of C. othonopterus and its endangered relative, the totoaba (Totoaba macdonaldi) and a refuge area for the world's smallest and most endangered marine mammal, the vaquita porpoise (Phocoena sinus)23,24. As a result of these issues, the corvina fishery is managed under a suite of regulations that include gear, effort and catch limits, a seasonal and area closure and an annual harvest quota24.
We engaged in cooperative research with fishers and stakeholders to characterize the fine-scale, spatio-temporal dynamics of spawning in C. othonopterus and coincident activities of its commercial fishery. Our specific objectives were to combine the use of GPS data loggers, fisheries data and biological surveys to: (1) estimate daily patterns of spawning and fishing in relation to the lunar cycle; (2) calculate the number and proportion of corvinas harvested from their annual spawning aggregations; and (3) compare the spatial distribution of aggregations and fishing activities to the known spawning grounds of corvina and management zones within the biosphere reserve. Results from this study, principally the high-resolution maps showing the heterogeneity in the spatial distribution of spawning aggregations and fishing intensity, highlight the substantial benefits of GPS technology to the science and management of fish spawning aggregations and spatial planning of marine reserves.
Results
Temporal Patterns of Spawning and Fishing
Mean total length (TL) of corvina sampled in 2009-10 (n = 4,955) was 711.10 mm ± 0.52 s.e.m. (range = 482–1013 mm TL) and mean body mass was 3.04 kg ± 0.02 s.e.m. (range = 1.20–11.95 kg). Definitive evidence of spawning via the collection of female C. othonopterus with hydrated oocytes (Figure 2a) within ovaries was obtained from 5 to 2 days before the peak new and full moons (hereafter dbm) in both 2009 and 2010 (Figure 2b) and the highest percentages of spawning females were collected 3 dbm during both years. Landings of Gulf corvina were recorded 8 to 1 dbm and peak landings occurred from 5 to 3 dbm (Figure 3a). Catch per unit effort (CPUE) increased steadily from 8 dbm to a peak at 2 dbm (mean = 979.83 kg×boat trip−1 ± 123.73 s.e.m.) in 2009 and 3 dbm (mean = 886.39 kg×boat trip−1 ± 142.03 s.e.m.) in 2010, but decreased thereafter (Figure 3b). In both years, records of landings ended by the day of the peak moon and did not recommence until 8 days before the next peak moon.
Semi-lunar rhythm in spawning of the Gulf corvina.
(a) Photo of a female corvina in spawning condition with enlarged ovaries full of hydrated oocytes. Inset shows photomicrograph of hydrated oocytes (scale bar = 0.5 mm), which indicate that spawning is imminent. (b) Mean daily percentage of spawning females (± s.e.m.) in relation to lunar day (day before peak moon, dbm) from fish sampled during the peak spawning period of March and April.
Semi-lunar trends in catch rates of the Gulf corvina fishery.
(a) Mean daily catch (± s.e.m.) of the corvina fishery in relation to lunar day (day before peak moon, dbm) from commercial buyer records in March and April. (b) Mean catch-per-unit effort (CPUE ± s.e.m.) of the corvina fishery in relation to dbm from commercial buyer records in March and April.
The mean eviscerated mass (evm) of corvina sampled (n = 1,896) from the fishery in 2009 was 2.45 kg ± 0.01 s.e.m. When compared to official (CONAPESCA) landings reports of C. othonopterus during their reproductive season (February-May), we estimate that 1.8 million and 1.5 million corvinas were harvested from their spawning aggregations in 2009 and 2010, respectively. Landings for C. othonopterus were reported in El Golfo de Santa Clara on 21 days from February to May in 2009 and 25 days during the same period in 2010. Based on a comparison of mean daily catch with respect to dbm and days in which spawning occurred (5 to 2 dbm), we estimate that 94.4% (2009) and 90.1% (2010) of fish harvested were captured during the spawning period.
Spatio-temporal Distributions of Spawning Aggregations
Maps showing the daily locations of C. othonopterus spawning aggregations (Figure 4) revealed a predictable, coherent pattern involving an initial coalescence of aggregations along the eastern portion of the reserve followed by a northwestward shift in their distribution towards the reported spawning grounds (the “Y”; see Figure 1 and Species Background), the area comprised of the two main channels surrounding Isla Montague and the lower portion of the Colorado River. Aggregations of Gulf corvina were first observed in the Delta region 8 dbm in both 2009 and 2010, at which time they were dispersed across the southeastern area of the biosphere reserve. The first sightings of aggregations inside the no-take zone occurred 7 to 6 dbm in 2009 and 2010, respectively and the percentage of sightings inside the no-take zone increased sharply between 7 and 5 dbm. During the spawning period of 5 to 2 dbm, aggregations were distributed from ca. 5 km off the southern end of Isla Montague, throughout the two shallow channels surrounding the island, to 10–15 km upstream in the river. During this time, 76.7 to 100% (mean = 89.1% ± 3.56 s.e.m.) of the sightings of corvina aggregations were made inside the no-take zone and 35.1 to 100% (mean = 70.9% ± 10.4 s.e.m.) were observed at the spawning grounds. Sightings of aggregations ceased by the day of the peak moon and did not recommence until 8 days before the next peak moon.
Fishing Area and Fishing Intensity
Spatial patterns of the C. othonopterus fishery were consistent between years. The total area of the fishery equaled 1008.1 km 2 in 2009 and 999.8 km 2in 2010 (pooled = 1148.7 km 2), which comprised 18.2% of the available marine habitat inside the biosphere reserve (Figure 5; Supplementary Fig. S1). The total area of the corvina fishery was split nearly equally between the no-take (mean = 47.6% ± 1.5 s.e.m.) and the fishing zone (mean = 52.0% ± 1.5 s.e.m.). While the fishery utilized nearly half the total area of marine habitat inside the no-take zone (mean = 48.0% ± 1.7 s.e.m.), it operated within only a small portion of the available area inside the fishing zone (mean = 13.4% ± 0.3 s.e.m.). In both 2009 and 2010, fishing activities for C. othonopterus occupied the entire spawning grounds but were absent inside the vaquita refuge.
Areas of high fishing intensity (i.e. > 1 SD above mean; see Methods) jointly encompassed 96.3 km2 in 2009 and 65.8 km2 in 2010 (pooled = 120.5 km2) and were located primarily off the southern end of Isla Montague to the boundary of the no-take zone and inside the two channels surrounding the island (Figure 5). The spawning grounds comprised 39.5% of the total area of high fishing intensity (pooled), whereas the no-take zone encompassed 88.3% (pooled). When mean daily catch with respect to dbm was compared to the mean percent of aggregations inside the spawning grounds or inside the no-take zone with respect to dbm, we estimate that 53.8% (2009) to 67.1% (2010) of the fish harvested were captured from the spawning grounds and 86.1% (2010) to 90.3 % (2009) of the fish harvested were captured from inside the no-take zone.
Discussion
Collectively, our results show that commercial fishing activities for C. othonopterus in 2009 and 2010 were synchronized with its spawning cycle, which in turn followed a semi-lunar rhythm around the weeks preceding the new and full moons (i.e. rising spring tides) of March and April. Fishing commenced 8 to 7 dbm, when aggregations of corvina were observed at the southeast portion of the biosphere reserve. Catch rates increased as the distribution of aggregations shifted northwestward and fishing intensity, landings and CPUE peaked from 5 to 2 dbm, when actively spawning fish were sighted and harvested in the estuarine channels surrounding Isla Montague and upstream in the river. Fishing and spawning ceased abruptly by the day of the peak moon, when C. othonopterus are thought by fishers to migrate out of the river and the Delta with the outgoing spring tides15,16 and did not recommence until the week prior to the next peak moon.
Previous studies have documented the migration patterns of individual fish in relation to spawning aggregation sites25,26 or described interactions between aggregations and fisheries on coarse spatial and temporal scales27,28, including several reports on C. othonopterus15,16. However, this study generated the first fine-scale maps showing both daily changes in the spatial distribution of a spawning aggregation and the distribution and intensity of fishing activities that target the aggregation. When combined with detailed fisheries and biological data, the results provide a complete characterization of the interactions between an aggregate-spawning fish and its fishery, revealing the magnitude by which commercial fisheries can impact fish spawning aggregations over highly restricted time periods and areas. In the case of the Gulf corvina, commercial fishers exclusively target its spawning migration, harvesting several thousand tons of fish during the brief periods when fish are densely gathered and spawning at their only known spawning site inside the shallow waters of the Colorado River Delta. These results constitute the foundation of biological and fisheries information necessary to create models to predict fishing impacts on reproductive output and population growth in C. othonopterus, set harvest control rules that incorporate the interactions between spawning and fishing in both time and space and evaluate the effects of current or proposed management policies on fishing activities and the economic viability of the fishery.
The synchronization of fishing and spawning activities along with their predictability in time and space constitute information that is robust and pertinent to the design and management of the biosphere reserve. An objective of the no-take zone is to protect the spawning and nursery habitat of the Gulf corvina, an endemic marine fish that has proven to be highly vulnerable to overfishing and collapse20. Spawning activities of C. othonopterus occur exclusively inside the no-take zone, confirming that the area itself is appropriately located for protecting this critical habitat for reproduction. Unfortunately, fishing intensity is also greatest at this location when fish are spawning, which illustrates the direct conflict that exists between the current behavior of the artisanal fishing fleet and the intended purpose of the biosphere reserve.
The current strategy to restrict fishing inside the no-take zone involves the patrol of one or more government surveillance vessels along its southern entrance during the peak fishing days of the corvina season in March and April. To a certain extent, this is a practical approach to maximize enforcement in light of the limited resources allotted to do so. However, the large size of the no-take zone coupled with the extremely high ratio of fishing vessels to enforcement vessels severely limits its effectiveness, as evidenced by pervasive fishing activity inside the restricted area throughout the corvina fishing season and during periods when enforcement vessels are present. Moreover, strong tidal fluctuations of 10–12 m during spring tide periods, dangerous currents, shallow waters and shifting sand bars in the area pose significant challenges for enforcement and threaten the safety of both enforcement officers and fishers.
Sufficient protection of Gulf corvina spawning aggregations is critical given evidence of a prior collapse in its population and fishery15, the well-known collapse of totoaba populations in response to overfishing of its spawning aggregations in the same region29 and the history of collapse in fisheries elsewhere that have targeted the spawning migrations of large-bodied sciaenid (i.e. Teleostei: Sciaenidae) fishes at the mouths of major estuaries30. Because fishing and spawning activities related to C. othonopterus are predictable in time and space and peak simultaneously, simple fishing closures and sales bans during peak spawning days or during certain migration periods (e.g. first spawning migration of the year) may improve protection of spawning fish while also reducing monitoring costs and streamlining enforcement efforts. Of course, any proposed changes to management must also consider the importance of the corvina fishery to the economies and the subsistence needs of the surrounding communities in the Upper Gulf of California (i.e. El Golfo de Santa Clara and El Zanjón), because it will be difficult to establish or maintain an effective enforcement strategy without the proper support and compliance of fishers and stakeholders.
The restriction of fishing activities during spawning would likely enhance the reproductive output of C. othonopterus spawning aggregations by increasing spawning activity and mate encounter rates while reducing mortality, disturbance and stress on spawning fish4. However, fishes that form spawning aggregations often show sex and size-related differences in the temporal movement patterns into, out from and around aggregation sites and spatial distributions during and outside aggregation periods31,32. As a result, fishing closures during certain times can significantly alter population structure (e.g. changes in sex ratios, size distributions, age at sexual maturity)4,33,34 in ways that may actually counteract the expected benefits from such measures. More information on temporal variation in the size and age structure of aggregating corvina would help clarify this issue.
Information on possible interactions between the Gulf corvina fishery and current conservation strategies for the totoaba and the vaquita were also deduced from our results. Similar to C. othonopterus, totoaba form large spawning aggregations at the mouth of the Colorado River from February to April each year35. Thus, while the direct impacts of fishing activities for Gulf corvina on totoaba were not assessed, the considerable overlap in the timing and location of spawning of the two species indicates that strong interactions are likely to occur. This conclusion is supported by previous studies, which have reported both incidental bycatch of totoaba from the corvina fishery as well as targeted poaching of totoaba inside the biosphere reserve during the corvina fishing season15,35. Conversely, our results showed no spatial interaction between fishing activities for C. othonopterus and the vaquita refuge. Undoubtedly, vaquita are likely to use habitats outside the designated refuge and therefore a lack of spatial overlap of the corvina fishery with the vaquita refuge does not guarantee that incidental bycatch is absent. Yet, sightings of vaquita inside the corvina fishing area are rare and bycatch of vaquita in the corvina fishery has not been documented36,37, which suggests that impacts on vaquita from the corvina fishery are lower than those documented in other fisheries (e.g. shrimp). It is not known whether fishers with trackers preferentially avoided the vaquita refuge due to concerns over the penalties (e.g. fines, imprisonment, loss of permits), or because corvina aggregations simply do not occur there. However, interviews with fishers conducted prior to the enforcement of the vaquita refuge in 2007 indicate that some fishing for corvina did occur in that area during the spawning season22,38. Regardless, if the objective of the vaquita refuge is to protect vaquita populations from interactions with commercial fishing activities, it appears that this is occurring, at least to some degree, with respect to the corvina fishery.
All conclusions drawn from this study regarding the spatial and temporal patterns of spawning in C. othonopterus, including daily changes in the spatial distributions of aggregations, were derived from fisheries-dependent data acquired from the two main fishing communities involved in the corvina fishery in the Upper Gulf of California. The long history of fishers from this region targeting the spawning migrations of C. othonopterus, the magnitude of fishing effort relative to the size of the biosphere reserve (>500 pangas operating in 5,532 km2 of marine habitat), the consistent manner by which fishers locate and harvest fish and anecdotal evidence from prior reports of fishers15,16,17 suggest that the results obtained from this study accurately reflect the general spawning behavior of the adult corvina population and associated activities of its commercial fishery. However, additional information is needed from the town of San Felipe, which has accounted for ca. 10% of the annual landings of Gulf corvina since 1995 and may show differences in their temporal and spatial patterns of fishing. Also, fisheries-independent research (e.g. passive and active acoustic telemetry) on the movement patterns of individual fish, the spatial distribution of aggregations and the temporal spawning patterns would help verify the conclusions of this study and reveal possible spawning-related activities that occur outside the time period and area of the fishery. Similarly, the gill nets used in the commercial fishery select for the capture of fish larger than 50 cm TL (see Species Background and Results), which indicates a need to investigate the spawning patterns of small (30–50 cm TL) adults that escape the fishery. Finally, it is of paramount importance to understand the spatial distribution of adult corvina between spawning migrations and outside the spawning season and to estimate the total biomass of the stock, factors that are highly relevant to the management of the fishery yet have not been examined.
Methods
Species Background
The Gulf corvina is a large sciaenid fish (Teleostei: Sciaenidae) endemic to the upper Gulf of California, Mexico, with a geographic range that extends from the Colorado River Delta south to the Midriff Islands region14 (Figure 1). Individuals can reach 1,013 cm in total length, 12 kg in body mass and 9 years in age15,39. C. othonopterus is a schooling, mesocarnivorous predator that feeds mainly on anchoveta, shrimp and crabs15. Gulf corvina are synchronous, multiple batch spawners with indeterminate annual fecundity that reproduce through broadcast spawning (i.e. external fertilization of pelagic eggs)39. Schools of adult corvina migrate into the Colorado River Delta to spawn within larger aggregations from late February to May, with peak spawning occurring in March and April, coincident with the timing of the spring tides during the weeks prior to the new and full moons16,17. The spawning grounds are reported by fishers to be located at an area known as the “Y”, which is comprised of two main channels around Isla Montague and the lowermost portion of the Colorado River17. Between spawning migrations and after the spawning season, it is speculated that adult corvina migrate south to deeper waters, but their distributions during these periods have not been thoroughly investigated15,17.
From 1917 to 1940, C. othonopterus represented an important commercial and sport fishery in the Upper Gulf of California. However, the fishery disappeared between the 1960s and 1980s, when corvina migrations were believed to have ceased15,16. The commercial fishery resumed in the late 1980s and reported landings have ranged from 2,466 to 5,942 tons per year since 2000, with most fish landed at El Golfo de Santa Clara, Sonora40 (Figure 6a). The estimated number of pangas involved in the Gulf corvina fishery doubled from 250 to 500 between the 1990s and 2000s15,16,17,18,19,20. In El Golfo de Santa Clara in 2010, there were 73 licensed fishing cooperatives and 443 licensed fishing vessels active in the fishery (fisheries official, El Golfo de Santa Clara, pers. comm.).
The commercial fishery for C. othonopterus utilizes gill nets to harvest fish almost exclusively during the timing of their spring spawning migrations (February-May; Figure 6b), with effort concentrated at its known spawning grounds in the core zone of the biosphere reserve15,16,17,24,40,41. The fishery serves as a major supply of fish to population centers around Mexico (e.g. Mexico City, Guadalajara, Tijuana) during the 40 days of Lent and the important national holiday of Holy Week in Mexico15,18. While the Gulf corvina fishery is deemed as highly selective, bycatch of totoaba occurs due to considerable overlap of their spawning migration periods and spawning grounds17,35. Incidental mortality from entanglement in commercial gill nets is considered the biggest threat to the recovery of vaquita populations, although no records exist showing bycatch of vaquita from the corvina fishery37,42. Other reported bycatch includes several species of sharks and seabirds and bigeye croaker Micropogonias megalops16. Juvenile corvina are landed as bycatch in other finfish fisheries and both adults and juvenile corvina are caught as bycatch in the shrimp fishery during the fall and winter months15.
Since 2007, the Gulf corvina fishery has been regulated by an official standard (NOM-063-PESC-2005)24. Fishers are required to use gill nets of minimum mesh size of 14.6 cm and a maximum length of 293 m. There is a minimum size limit of 65 cm TL but with a tolerance of 35% of the catch under this length. Only one net is allowed per panga or fisherman and fishing is restricted to daylight hours. The National Fisheries Commission (CONAPESCA) recognizes the no-take zone of the biosphere reserve as a restricted area, but enforcement has been sporadic. The fishery is closed from May 1st to August 31st each year in order to protect a portion of spawning adults (some spawning occurs in May) and juveniles that inhabit the biosphere reserve during that period41. Fish can only be landed whole and at one of three locations: San Felipe, El Golfo de Santa Clara, or El Zanjón. The National Fisheries Institute (INAPESCA) is required to recommend a quota for each fishing season, which is published by the Fisheries Secretary in the Official Diary of the Federation24.
Spawning Periodicity
We surveyed a total of 4,955 C. othonopterus (2,897 fish in 2009; 2,058 fish in 2010) landed by the commercial fishery at El Zanjón and El Golfo de Santa Clara in March and April of 2009 and 2010. Fish were sampled opportunistically, throughout the day (0800-2130 h) and on a daily basis during all days the commercial fishery landed corvina. We randomly sampled 20–50 fish from a single net's catch and 4–10 nets per day. We recorded data on total length (1 mm) body mass (1 g), sex and reproductive condition. Whole gonads were dissected from each fish and preserved in a 10% formalin-seawater solution. Female C. othonopterus were easily distinguished from males by gonadal inspection, because the mesh size of gill nets selectively targets sexually mature (i.e. adult) fish15,40 and collections were made during the spawning season when fish were reproductively active.
Reproductive condition was identified by macroscopic inspections of gonads and verified by microscopic evaluations of gonad tissue sections prepared using standard histological procedures. Briefly, pieces of preserved gonad tissue were taken from the central portion of one lobe, embedded in paraffin, sectioned transversely at 6 µm, mounted, stained with Mayer's haemotoxylin-eosin and observed under a compound microscope. Classification of gonadal development stages followed Brown-Peterson et al. (2007)43. Emphasis was placed on identifying females with ovaries containing batches of hydrated or ovulated oocytes to specify days in which spawning occurred44. The percentage of spawning females collected was calculated separately for each net haul and the mean percentage of spawning females among net hauls was then calculated for each sampling day. Mean daily percentages were then organized by dbm across sampling days (% daily spawning by year) and years (% daily spawning – pooled across years) to characterize the spawning cycle in relation to the lunar cycle.
Trends in Fisheries Catch and Effort
We obtained detailed catch (kg) and catch per unit effort (CPUE: kg×trip−1) data for 2009 and 2010 from daily purchase records of the largest commercial buyer involved in the corvina fishery at El Golfo de Santa Clara. These included landings from a total of 313 fishers from 59 fishing cooperatives across both years, including landings from 208 fishers from 50 cooperatives in 2009 and 243 fishers from 53 cooperatives in 2010. We assumed these data were representative of CPUE, because the unit volume of fish purchased is equivalent to a single panga's catch and has been used to estimate CPUE in previous studies of this fishery16. Briefly, fish landed from a fishing trip of a single panga are transported directly to the fishing cooperative to be eviscerated and cleaned and then transported directly to the processing plant to be sold to the commercial buyer in the same unit volume [i.e. kg of eviscerated mass (evm) of fish]. Specific dates of landings were converted to dbm and mean CPUE and mean daily catch (kg×day−1) was averaged for both 2009 and 2010 and pooled across years by dbm to describe harvest trends in relation to lunar phase.
Gulf corvina are eviscerated prior to being weighed, sold, or recorded in official landings records (see above). We randomly sampled 1,896 C. othonopterus from the commercial fishery in 2009 at El Golfo de Santa Clara, recorded evm (1 g) for each fish and calculated the mean evm of harvested fish. We divided landings of corvina reported during the spawning season (February-May) in both 2009 and 201039 by the mean evm of harvested fish to estimate the reported number of corvina harvested from their spawning aggregations each year. We used official landings reports and commercial purchase records from El Golfo de Santa Clara to determine the total numbers of fishing days for corvina in both years. We calculated the percentage of harvested fish that were captured inside the no-take zone by comparing the mean daily catch by dbm (pooled across years) to the mean percentage of corvina aggregations observed inside the no-take zone by dbm (pooled across years).
Description of Cooperative Research
We engaged in cooperative research12 with commercial fishers from the two main communities involved in the Gulf corvina fishery, El Golfo de Santa Clara, Sonora and El Zanjón, Baja California, Mexico to deploy GPS data loggers (AMOD AGL3080) and record the movement patterns of pangas (8 m in length) from fishing trips for corvina in March and April of 2009 and 2010. Given widespread concerns about the potential use of these data for surveillance or enforcement purposes, we made a confidentiality agreement with individual fishers, fishing cooperatives and commercial buyers that no raw data or personal information on vessel movement or landings would be shared with anyone without their full consent. In order to build and maintain full transparency and trust with respect to this research, we engaged in frequent meetings and discussions with fishers and stakeholders to communicate ongoing results and their implications. All maps and results from this study were shared with fishers and stakeholders prior to their publication and dissemination.
Estimation of Fishing Area and Fishing Intensity
Data loggers were given to fishers as they departed the beach to embark on a fishing trip and were retrieved upon their return to shore. Sampling was opportunistic, such that we worked with all fishers and cooperatives willing to participate in the study45. We obtained complete tracks from 71 fishing trips in 2009 and 91 trips in 2010. These data included one or more tracks from 51 different pangas and 12 cooperatives involved in the fishery. Data loggers automatically recorded their velocity and position each second to an accuracy of 5–15 m (http://www.amod.com.tw) throughout the duration of each fishing trip and required no manipulation by fishers. Data from each track were uploaded to a computer and displayed using GPSBabel (OPEN source ware, see http://www.gpsbabel.org) to ensure files with aberrant data were excluded from the analysis. GPS data were then transformed to ASCII code outputs, imported into a Geographic Information System (GIS) format using ArcGIS™ 10 software and transformed into track maps that displayed the instantaneous (per second) locations and velocities of each panga throughout the duration of the fishing trip.
We estimated the total area of the corvina fishery (2009, 2010 and pooled) by simultaneously plotting all fishing trips on a regional map and creating a polygon around the outer boundaries using the aggregate points tool in ArcGIS™ 10 (see Supplementary Fig. S1). We then calculated the area of the polygon in order to estimate the percentage of the fishery that occurred inside management zones (vaquita refuge, no-take zone, biosphere reserve limits) and the percentage of each zone that was occupied by the fishery. We used a tracking effort-weighted kernel density analysis46 to map the spatial fishing intensity of pangas within the polygon that defines the corvina fishing area and identify high use areas47,48. Fishing intensity was estimated from the residence time of pangas by using the inverse velocity as the population field and search radius of 1 km49. Kernel density analysis for grid files and cell-based statistics were performed using ModelBuilder in ArcGIS™ 10. For visualization purposes, the grid size was set at 100 m and the analysis was performed in a Universal Transverse Mercator (UTM) projection. The distributions of high use areas across kernel categories were classified using one standard deviation (SD) method as the class size. Fishing intensity was divided into three classes: low (> 1 SD below the mean); medium (< 1 SD below or above the mean); and high (> 1 SD above the mean). Portions of each track located within 500 m of departure points at El Golfo de Santa Clara and El Zanjón were removed from the analysis, since they likely reflect periods of inactivity unrelated to fishing50.
Spatial Distribution and Migration of Corvina Aggregations
We used the known technique fishers use to capture fish with gill nets (called “encierre”) to identify the exact location of Gulf corvina aggregations from individual tracks (Figure 7). When an aggregation is sighted on the surface of the water, fishers throw the net directly onto the aggregation and then drive the panga in a circle around the aggregation while continuing to deploy the net. Immediately after encirclement, the net and entangled fish are retrieved while the panga drifts with the current. Therefore, each segment of a track that showed a small loop pattern followed by an extended, slow drift was selected as a location where gill nets were deployed and thus where aggregations were present. Standard query commands based on velocity attributes were used to identify the fishing location where aggregations were present50 and the location was selected by identifying the fishing activity pattern using ArcGIS™ 10. We acquired a total of 691 data points on the location of corvina aggregations from 162 commercial fishing trips for corvina in 2009 (n = 234 points) and 2010 (n = 457 points). Data points representing the locations of aggregations were organized by lunar day (dbm, or the # of days before the peak new or full moon) and plotted on a regional map to simulate the daily migration patterns of aggregations. Data from 2009 and 2010 were first analyzed separately and later pooled across sampling years to assess coherence and predictability of migration patterns in relation to year and dbm.
Locations of Gulf corvina spawning aggregations.
Map of a typical fishing trip for Gulf corvina, with inset showing methods used to identify the location of spawning aggregations. When an aggregation of corvina is sighted, fishers throw the net directly onto the aggregation and then drive the boat in a circle at high speed around the aggregation while continuing to deploy the net (called “encierre”). The boat then drifts with the current while the fishers retrieve the net and entangled fish.
References
Wilcove, D. S. & Wilkelski, M. Going, going, gone: is animal migration disappearing. PLoS Biol. 6, 1361–1364 (2008).
Domeier, M. L. & Colin, P. L. Tropical reef fish spawning aggregations: defined and reviewed. Bull. Mar. Sci. 60, 698–726 (1997).
Sadovy, Y., & Domeier, M. Are aggregation-fisheries sustainable? Reef fish fisheries as a case study. Coral Reefs 24, 254–262 (2005).
Sadovy de Mitcheson, Y. & Erisman, B. Fishery and biological implications of fishing spawning aggregations and the social and economic importance of aggregating fishes. In Reef Fish Spawning Aggregations: Biology, Research and Management, Sadovy, Y.J., Colin, P.L., editors, 225–284 (2011).
Sadovy de Mitcheson, Y., Cornish, A., Domeier, M., Colin, P. L., Russell, M. & Lindeman, K. A global baseline for spawning aggregations of reef fishes. Conserv. Biol. 22, 1233–1244 (2009).
Piet, G. J. & Quirijns, F. J. The importance of scale for fishing impact estimations. Can. J. Fish. Aquat. Sci. 66, 829–835 (2009).
McCluskey, S. & Lewison, R. Quantifying fishing effort: a synthesis of current methods and their applications. Fish and Fisheries 9, 188–200 (2008).
Bertrand, S. & Díaz, E. Patterns in the spatial distribution of Peruvian anchovy (Engraulis ringens) revealed by spatially explicit fishing data. Prog. Ocean. 79, 379–389 (2008).
Saitoh, S. et al. Some operational uses of satellite remote sensing and marine GIS for sustainable fisheries and aquaculture. ICES J. Mar. Sci. 68, 687–695 (2011).
Witt, M. J. & Godley, B. J. A step towards seascape scale conservation: using vessel monitoring systems (VMS) to map fishing activity. PLoS One 2, e1111 (2007).
Gerritsen, H. & Lordan, C. Integrating vessel monitoring systems (VMS) data with daily catch data from logbooks to explore the spatial distribution of catch and effort at high resolution. ICES J. Mar. Sci. 68, 245–252 (2010).
Johnson, T. R. & van Densen, W. L. T. Benefits and organization of cooperative research for fisheries management. ICES J. Mar. Sci. 64, 834–840 (2007).
Moreno-Báez, M., Orr, B. J., Cudney-Bueno, R. & Shaw, W. W. Using fishers' local knowledge to aid management at regional scales: spatial distribution of small-scale fisheries in the northern Gulf of California, Mexico. Bull. Mar. Sci. 86, 339–353 (2010).
Robertson, D. R. & Allen, G. R. Shorefishes of the Tropical Eastern Pacific online information system. Version 1.0. Smithsonian Tropical Research Institute, Balboa Panama. www.neotropicalfishes.org/sftep (2008).
Román-Rodríguez, M. J. 2000. Estudio poblacional del chano norteño, Micropogonias megalops y la curvina Golfina Cynoscion othonopterus (Gilbert) (Pisces: Sciaenidae), especies endémicas del alto Golfo California, Mexico. Instituto del Medio Ambiente y Desarrollo Sustentable del Estado de Sonora. Informe final SNIB-CONABIO proyecto No. L298. Mexico D.F.
Campoy, J. R. & Román Rodríguez, M. J. Observaciones sobre la biologia y pesqueria de la curvina golfina (Cynoscion othonopterus) en la reserva de la Biosfera Alto Golfo de California y Delta del Río Colorado. Documento de Trabajo, INE. IMADES, 1–13 (2002).
INAPESCA. Compilación biológico-pesquero de curvina golfina, Cynoscion othonopterus, en el alto Golfo de California. Doc. Téc. Instituto Nacional de Pesca-CRIP Ensenada y Guaymas, 1–12 (2005).
Paredes, G. A. et al. La Curvina Golfina: Biología, Pesquería y Su Gente. Biodiversitas 91, 1–15 (2010).
Rodríguez-Quiroz, G., Aragón-Noriega, E. A., Valenzuela-Quiñónez, W. & Esparza-Leal, H. M. Artisanal fisheries in the conservation zones of the Upper Gulf of California. Rev. Biol. Mar. Ocean. 45, 89–98 (2010).
Chao, L. Espinosa, H., Findley, L. & van der Heiden, A. Cynoscion othonopterus In:IUCN 2011. IUCN Red List of Threatened Species.Version 2011.2 www.iucnredlist.org. Downloaded on 19 November 2011.
Musick, J. A. et al. Marine, estuarine and diadromous fish stocks at risk of extinction in North America (exclusive of Pacific salmonids). Fisheries 25, 6–30 (2000).
Aragón-Noriega, E. A., Rodriguez-Quiroz, G., Cisneros-Mata, M. A. & Ortega-Rubio, A. Managing a protected marine area for the conservation of critically endangered vaquita (Phocoena sinus Norris, 1958) in the upper Gulf of California. Int. J. Sust. Dev. World Ecol. 17, 410–416.
Bobadilla, M., Alvarez-Borrego, S., Avila-Foucat, S., Lara-Valencia, F. & Espejel, I. Evolution of environmental policy instruments implemented for the protection of totoaba and the vaquita porpoise in the Upper Gulf of California. Environ. Sci. Pol. 14, 998–1007 (2011).
Diario Oficial de la Federación. NORMA Oficial Mexicana NOM-063-PESC-2005, pesca responsable de curvina golfina (Cynoscion othonopterus) en aguas de jurisdicción federal del Alto Golfo de California y Delta del Río Colorado. Especificaciones para su aprovechamiento. Pub. DOF, 16 de Agosto de 2007. (2007).
Starr, R. M., Sala, E., Ballesteros, E. & Zabala, M. Spatial dynamics of the Nassau grouper Epinephelus striatus in a Caribbean atoll. Mar. Ecol. Prog. Ser. 343, 239–249 (2007).
Walli, A. et al. Seasonal movements, aggregations and diving behavior of Atlantic bluefin tuna (Thunnus thynnus) revealed with archival tags. PLOS One 4, e6151 (2009).
Begg, G. A. & Marteinsdottir, G. Spatial partitioning of relative fishing mortality and spawning stock biomass of Icelandic cod. Fish. Res. 59, 343–362 (2003).
Erisman, B. et al. Seasonal, annual and long-term trends in commercial fisheries for aggregating reef fishes in the Gulf of California, Mexico. Fish. Res. 106, 279–288 (2010).
Lercari, D. & Chávez, E. A. Possible causes related to historic stock depletion of the totoaba, Totoaba macdonaldi (Perciformes: Sciaenidae), endemic to the Gulf of California. Fish. Res. 86, 136–142 (2007).
Sadovy, Y. & Cheung, W. L. Near extinction of a highly fecund fish: the one that nearly got away. Fish. Fish. 4, 86–99 (2003).
Robichaud, D. & Rose, G. A. Sex differences in cod residency on a spawning ground. Fish. Res. 60, 33–43 (2003).
Nemeth, R. S., Blondeau, J., Herzlieb, S. & Kadison, E. Spatial and temporal patterns of movement and migration at spawning aggregations of red hind, Epinephelus guttatus, in the U.S. Virgin Islands. Environ. Biol. Fish. 78, 365–381 (2007).
Rowe, S. & Hutchings, J. A. Mating systems and the conservation of commercially exploited marine fish. Trends Ecol. Evol. 18, 567–572.
Berkeley, S. A., Hixon, M. A., Larson, R. J. & Love, M. S. Fisheries sustainability via protection of age structure and spatial distribution of fish populations. Fisheries 29, 23–32 (2004).
Cisneros-Mata, M. A., Montemayor-López, G. & Román-Rodríguez, M. J. Life history and conservation of Totoaba macdonaldi. Conserv. Biol. 9, 806–814 (1995).
Gerrodette, T. & Rojas-Bracho, L. Estimating the success of protected areas for the vaquita, Phocoena sinus. Mar. Mammal Sci. 27, E101–E125 (2011).
D'agrosa, C., Lennert-Cody, C. E. & Vidal, O. Vaquita bycatch in Mexico's artisanal gillnet fisheries: driving a small population to extinction. Conserv. Biol. 14, 1110–1119 (2000).
Moreno-Báez, M. et al. Integrating the spatial and temporal dimensions of fishing activities for management in the Northern Gulf of California, Mexico. Ocean Coast. Manage. 55, 111–127 (2012).
Gherard, K. E. Age, growth and batch fecundity of Gulf corvina, Cynoscion othonopterus. Masters Thesis, California State University Northridge. 1–40. (2011).
CONAPESCA. (Mexico). Subdelegación de Pesca en Sonora. 2010. En: Manual de capacitación y diseño de recursos sobre diferentes técnicas recomendadas para el manejo y procesamiento de la curvina golfina que permitirá dar el valor agregado requerido, que será diverso y muy relacionado a los hábitos de consumo de los diferentes mercados en los que se pretende colocar el recurso (producto 3). Consultores Ejecutivos de Sonora S.A.de C.V. Agosto de 2010, Guaymas, Sonora (2010).
Diario Oficial de la Federación. ACUERDO por el que se establece veda temporal para la captura de curvina golfina (Cynoscion othonopterus) en las aguas marinas y estuarinas de jurisdicción federal de la reserva de la Biosfera Alto Golfo de California y Delta del Río Colorado, durante el periodo del 1 de mayo al 31 de agosto de cada año. Pub. DOF, 25 de Agosto de 2005 (2005).
Vidal, O. Population biology and incidental mortality of the vaquita, Phocoena sinus. Rep. Int. Whal. Commn. 16, 247–272 (1995).
Brown-Peterson, N. et al. An improved and simplified terminology for reproductive classification in fishes. Gonadal Histology Workshop of the Join Meeting of Ichthyologists and Herpetologists, July 2007, St. Louis, Missouri, USA. (2007).
Erisman, B. E., Buckhorn, M. L. & Hastings, P. A. Spawning patterns in the leopard grouper, Mycteroperca rosacea, in comparison with other aggregating groupers. Mar. Biol. 151, 1849–1861 (2007).
Wagner, J. Who knows? On the importance of identifying experts when researching local ecological knowledge. Human Ecology 31, 463–489 (2003).
Silverman, B. W. Density Estimation for Statistics and Data Analysis. Chapman and Hall, London, England (1986).
Block, B. A. et al. Electronic tagging and population structure of Atlantic Bluefin tuna. Nature 434, 1121–1127 (2005).
Mills, C., Rogers, S. I., Tasker, M. L., Eastwood, P. D. & Piet, G. J. Developing the use of satellite fishing vessel monitoring system data in spatial management. ICES J. Mar. Sci. Annual Science Conference, 22–25 September, Vigo, Spain (2004).
Smith, A. T. & Dobson, F. S. A technique for evaluation of spatial data using asymmetrical weighted overlap values. Anim. Behav. 48, 1285–1292 (1994).
Lee, J., South, A. B. & Jennings, S. Developing reliable, repeatable and accessible methods to provide high-resolution estimates of fishing-effort distributions from vessel monitoring system (VMS) data. ICES J. Mar. Sci. 67, 1260–1271 (2010).
Acknowledgements
We thank fishers of Santa Clara and El Zanjón, Alto Golfo Sustentable, SIO Center for Marine Biodiversity and Conservation, NOS Noroeste Sustentable, the Ochoa family, G. Paredes, K. Gherard, C. Lopez, M. Calderon, J. Cota, T. Plomozo and S. Sánchez for their support and assistance. L.G. Alvarez and colleagues at Centro de Investigacíon Científica y de Educacíon Superior de Ensenada (CICESE) provided bathymetry data for maps. R. Warner, Y. Sadovy de Mitcheson and M. Craig provided comments on an earlier draft of the manuscript. This study was funded by the Walton Family Foundation and the David and Lucile Packard Foundation.
Author information
Authors and Affiliations
Contributions
B.E. and O.A. conceived the project, collected data, performed analyses and wrote the manuscript; C.G. and M.M. created the maps and performed spatial analyses; I.M. collected and analyzed biological and fisheries data; P.H. supervised the project and assisted in writing and editing the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplementary Information - Suppl. Fig. S1
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareALike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Erisman, B., Aburto-Oropeza, O., Gonzalez-Abraham, C. et al. Spatio-temporal dynamics of a fish spawning aggregation and its fishery in the Gulf of California. Sci Rep 2, 284 (2012). https://doi.org/10.1038/srep00284
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep00284
This article is cited by
-
Long-Term Passive Acoustic Monitoring to Support Adaptive Management in a Sciaenid Fishery (Tagus Estuary, Portugal)
Reviews in Fish Biology and Fisheries (2024)
-
Aggregation site fidelity and movement patterns of the protected marine predator giant sea bass (Stereolepis gigas)
Environmental Biology of Fishes (2021)
-
Emerging from the murk: threats, challenges and opportunities for the global swim bladder trade
Reviews in Fish Biology and Fisheries (2019)
-
Massive spawning behavior of the triplespine deepwater cardinalfish Sphyraenops bairdianus Poey, 1861 observed on an offshore sea mount in the West Mariana Ridge
Environmental Biology of Fishes (2018)
-
Ecological guidelines for designing networks of marine reserves in the unique biophysical environment of the Gulf of California
Reviews in Fish Biology and Fisheries (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.