Abstract
Here we present a test-retest dataset of functional magnetic resonance imaging (fMRI) data acquired at rest. 22 participants were scanned during two sessions spaced one week apart. Each session includes two 1.5 mm isotropic whole-brain scans and one 0.75 mm isotropic scan of the prefrontal cortex, giving a total of six time-points. Additionally, the dataset includes measures of mood, sustained attention, blood pressure, respiration, pulse, and the content of self-generated thoughts (mind wandering). This data enables the investigation of sources of both intra- and inter-session variability not only limited to physiological changes, but also including alterations in cognitive and affective states, at high spatial resolution. The dataset is accompanied by a detailed experimental protocol and source code of all stimuli used.
Design Type(s) | Test-retest Reliability |
Measurement Type(s) | nuclear magnetic resonance assay |
Technology Type(s) | MRI Scanner |
Factor Type(s) | |
Sample Characteristic(s) | Homo sapiens • brain |
Machine-accessible metadata file describing the reported data (ISA-Tab format)
Similar content being viewed by others
Background & Summary
In contrast to the focused scope of task-based functional studies using functional magnetic resonance imaging (fMRI), data acquired independent of specific task demands has become the basis for post hoc investigation of diverse functional systems. Rather than constraining the question of functional organization to a single paradigm, the observation that ongoing intrinsic activity replicates networks observed during task has made resting-state fMRI (rs-fMRI) data a highly versatile resource.
To date the vast majority of rs-fMRI research has been conducted using data acquired with a resolution of approximately 3–4 mm3. This voxel size provides a judicious compromise between signal-to-noise ratio and spatial resolution when using a standard 3 Tesla (3 T) MRI scanner. However, as the thickness of gray matter in the cerebral cortex can range from 1.5 to 3 mm, partial sampling of tissue becomes a substantial source of noise when using the standard voxel size. Although traditionally attenuated using techniques such as spatial smoothing, such approaches are not only considered increasing problematic for investigating brain function, but also reduce the resolution with which neuroanatomical questions can be investigated.
7 Tesla (7 T) ultra-high field MRI scanners, though not as common as their 3 T counterparts, offer notably enhanced spatial resolution and are becoming increasingly incorporated into academic and clinical research. The major advantage of using a field strength of 7 T scanner is the resulting high signal-to-noise ratio (SNR) which can be directly translated into a high spatial resolution. Initial studies have shown that using 7 T allows for the more precise delineation of resting state networks than with 3 T, especially if using small voxels with edge lengths in the range between 1 and 1.5 mm1–3. It was further shown that spontaneous neuronal activity is one of the major contributors to the measured fMRI signal fluctuations at 7 T, increasing almost twofold relative to earlier experiments under similar conditions at 3 T4. Hence, using the increased BOLD sensitivity of ultra-high fields for rs-fMRI, a number of studies with different neuroscientific questions were conducted at 7 T5,6. However, also an increased propensity for artifacts at 7 T compared to 3 T was reported for rs-fMRI7,8. It is therefore essential to investigate the reliability of rs-fMRI at 7 T. Thus, in this article, we present the first extensive 7 T test-retest dataset in order to address this issue.
A growing body of research continues to establish the reliability of various analytic approaches to rs-fMRI data9–12. While these studies have established rs-fMRI as a viable method for scientific inquiry, there are still methodological questions to be answered. To understand reliability within the context of rs-fMRI paradigms, it is important not only to assess the between-session variance, but also to attribute that variance to distinct confounding factors13,14. More specifically, these factors might be hardware-specific, physiological, or mental state-related in nature. It has been shown, for example, that participant motion increases between-session variance13,15. The following dataset allows researchers to address these questions, by including information about participant motion (derived from imaging data), breathing, pulse, and blood pressure. Additionally, to assess the influence of changing the position of the head of the participant within the coil, the participants were taken out, and then returned to the scanner within the same scanning session.
This dynamic aspect of spontaneous brain fluctuations has recently been the focus of intense methodological development16–18. Differences in mental state, mood, and content of spontaneously generated thoughts during the scanning session may translate into increased between-session variance that is independent of physiological factors. To address these uncertainties, the dataset includes assessments of the participants’ mood and sustained attention immediately prior to each scanning session. The content of self-generated thoughts immediately after each rs-fMRI run was also probed using an in-scanner adaptation of the New York Cognition Questionnaire19. This data allows researchers to relate dynamic changes in brain states to externally measured differences in mental states.
Additionally, reliability of an analysis is not only restricted to experimental properties of the data. The MRI field has developed a variety of data processing improvements focusing on data cleaning (removal of confounds), normalization (transforming brains of each individual into a common space), and feature selection (deciding which aspect of spontaneous activity the analysis should focus on). Those processing steps can have a significant influence on the ultimate reliability of the results. Access to high resolution and quality test-retest data will allow authors of tools and methods to test their performance in terms of reliability. High resolution data is especially important for methods dealing with interindividual differences and investigating cortical layer-dependent processes. Additionally reliability metrics such as the Intra-class Correlation Coefficient20 make the assumption about the spatial correspondence of each voxel between subjects. High resolution datasets like this one can reveal to what extent this assumption is just an approximation.
We present a dataset consisting of high resolution brain scans—including 0.7 mm3 MP2RAGE anatomical scans—collected from 22 individuals at two time points. This dataset is optimal for addressing methodological questions requiring reliability assessment, and also enables the investigation of different contributing confounds to between-session variance. In addition to the crucial need for methods validation specific to high resolution data, the relation of mental state (using measures of mood, sustained attention, and the content of self-generated thought) to intrinsic brain dynamics is a foundational question of neuroscience for which we hope this dataset will provide insight. Additionally it complements other publicly available 7 T fMRI datasets21 and is also part of a multi-site collection of test-retest datasets collected by the Consortium for Reliability and Reproducibility22.
Methods
Participants
22 participants (10 women) were selected from a database of people having previously taken part in 7 T experiments at the Max Planck Institute for Human Brain and Cognitive Sciences, Leipzig, Germany. All participants were native German speakers. Their age ranged from 21 to 30 with mean 25.1. All of the participants have previously taken part in MRI experiments at the 7 T facility (from 5 to 51 times, mean 23) and were therefore accustomed with the procedure. All subjects had given written informed consent and the study was approved by the Ethics Committee of the University of Leipzig.
Testing procedure
Each participant was invited to the institute twice exactly one week apart (therefore visits were matched in terms of time of the day and day of the week). A similar testing protocol was applied to the participants on both visits. Participants were instructed to refrain from drinking caffeinated products starting two hours before each visit. They were reminded of this requirement via a text message sent on the day of the experiment. The data acquired is summarized in Figure 1, and the order of tasks/measurements was the following (details of each test are described in the next section):
First visit:
-
1
At the behavioural testing room:
-
a. Briefing and consent signature
-
b. Conjunctive Continuous Performance Task (CCPT)
-
c. Mini New York Cognition Questionnaire (mini NYC-Q)
-
d. Positive and Negative Affect Schedule—Expanded Form Questionnaire (PANAS-X)
-
e. Hydration, caffeine, sleep questionnaire
-
f. 5 min of rest
-
g. Blood pressure and pulse measurements
-
-
2
At the scanner
-
h. Instructions for the in-scanner version of CCPT
-
i. Localizer scan
-
j. Structural scan
-
k. Field map scan
-
l. Whole-brain resting state scan (15 min)
-
m. In-scanner mini NYC-Q
-
n. Field map scan
-
o. Whole-brain resting state scan (15 min)
-
p. In-scanner mini NYC-Q
-
q. Prefrontal cortex submillimeter scan (10 min)
-
r. In scanner mini NYC-Q
-
Second visit:
-
1
At the behavioural testing room—same as the first visit
-
2
At the scanner—differences from the first visit in bold:
-
a
Instructions for the in scanner version of CCPT
-
b
Localizer scan
-
c
Field map scan
-
d
Whole-brain resting state scan (15 min)
-
e
In scanner mini NYC-Q
-
f
Take participant out of the scanner, ask them to sit in an upright position and put them back into the scanner
-
g
Localizer scan
-
h
Field map scan
-
i
Whole-brain resting state scan (15 min)
-
j
In-scanner mini NYC-Q
-
k
Prefrontal cortex submillimeter scan (10 min)
-
l
In-scanner mini NYC-Q
-
a
Behavioural tests
Conjunctive continuous performance task
The Conjunctive Continuous Performance Task looks at the ability to sustain attention over a period of approximately 20 min23. The task involves looking at series of shapes (triangles, squares, circles, and stars) in different colors (red, green, blue, yellow). The participant is asked to react by pressing the spacebar of a computer keyboard if and only if they see a red square (which appears 30% of the time). Each shape is displayed for 100 ms and is separated from the next by a randomly-selected inter-stimulus interval (ISI) of 1,000, 1,500, 2,000, or 2,500. There was no feedback. The test consisted of 320 trials preceded by 15 trials of a practice run (which did include immediate feedback). After the practice run and before the test proper the examiner made sure that each participant understood the task. All the instructions on the screen were presented in German. The task was implemented in OpenSesame24 using PsychoPy backend25,26—the code is available at https://github.com/NeuroanatomyAndConnectivity/ConjunctiveContinuousPerformanceTask For more details of the task please see the original paper23.
Mini New York cognition questionnaire
The mini version of the New York Cognition Questionnaire is an adaptation of the full version of New York Cognition Questionnaire19. The aim of the mini version was to shorten the questionnaire based on factors extracted from the full version (see19 for details). The questionnaire consists of series of statements, each starting with ‘During the last measurement’:
-
1
I thought about something positive
-
2
I thought about something negative
-
3
my thoughts involved future events
-
4
my thoughts involved past events
-
5
my thoughts involved myself
-
6
my thoughts involved other people
-
7
my thoughts involved my surroundings
-
8
I was fully awake
-
9
my thoughts were in the form of images
-
10
my thoughts were in the form of words
-
11
my thoughts were more specific than vague
-
12
my thoughts were intrusive
Participants had to rate each statement on a visual analog scale ranging from ‘Completely did not describe my experience’ (score 0, left hand side) to ‘Completely described my experience’ (score 100, right hand side). All questions and instructions were presented in German. The questionnaire was implemented in OpenSesame24 using PsychoPy backend25,26 as a slider operated with three buttons (left, right, next question). The slider range was from 0 to 100 with increments of 5. The initial position of the slider was 50. Question order was randomized at each presentation. The questionnaire was presented immediately after the CCPT task, and after each rs-fMRI scan (a special version of the task operated with a response box was used inside the scanner). The code of the questionnaire is available at https://github.com/NeuroanatomyAndConnectivity/NYC-Q.
Positive and negative affect schedule—expanded form questionnaire
Positive and Negative Affect Schedule—Expanded Form Questionnaire is a questionnaire probing the mood of participants27. It consists of 60 items which are broken down into several subscales such as General Positive and General Negative Affect. For each item (an adjective such as ‘happy’ or ‘alone’) participants had to rate ‘to what extent have they felt this way during the past week.’ This was on a scale from ‘very slightly or not at all’ (1) to ‘extremely’ (9). We have used a German version of the questionnaire first introduced by Gruhn et al.28 The questionnaire was presented using the LimeSurvey software (http://www.limesurvey.org/).
Hydration, caffeine, sleep questionnaire
To assess sleep patterns, hydration, and caffeine intake, participants were asked to reply to a series of additional questions.
-
1
On average, how many hours do you sleep every night?
-
2
How many hours did you sleep last night?
-
3
How well rested do you feel right now? 1 (Extremely tired)—9 (Perfectly well rested)
-
4
How well did you sleep last night? 1 (I slept terribly)—9 (I slept very well)
-
5
How well hydrated do you feel right now? 1 (Completely dehydrated)—9 (Perfectly hydrated)
-
6
On average, how much water (and other liquids) do you drink every day (in litres)?
-
7
Comparing to other days did you drink more or less water today? 1 (I drank much less than usual)—5 (I drank the same amount as usual)—9 (I drank much more than usual)
-
8
On average, how much caffeinated drinks (coffee, cola, club mate etc.) do you drink every day (in litres—one cup=0.2 l)?
-
9
Comparing to other days did you drink more or less coffee and other caffeinated drinks (coffee, cola, club mate etc.) today? 1 (I drank much less than usual)—5 (I drank the same amount as usual)—9 (I drank much more than usual)
This questionnaire was also presented using LimeSurvey software (http://www.limesurvey.org/).
Blood pressure and pulse measurement
After filling the questionnaires participants were asked to relax for 5 min in order for their heart rate to stabilize. After this period, their systolic and diastolic blood pressure, as well as pulse, were measured for both arms. The measurement was performed using Omron M500 device (Omron Healthcare Co. Ltd.).
Magnetic resonance imaging
All experiments were performed on a 7 T whole-body MR scanner (MAGNETOM 7 T, Siemens Healthcare, Erlangen, Germany). A combined birdcage transmit and 24 channel phased array receive coil (NOVA Medical Inc, Wilmington MA, USA) was used for imaging. During the scan the participants’ pulse was monitored using a pulse oximeter. Breathing was measured using a pneumatic sensor. Both breathing and pulse signals were recorded using Biopac MP150 system (Biopac Systems Inc., USA) sampled at 5,000 Hz with the Biopac Acqknowledge 4.1 software (Biopac Systems Inc., USA). Participants filled the mini NYC-Q questionnaire using a four button response box held in both hands. The position of the slider was adjusted by the left and right buttons, pressing any of the two middle buttons advanced to the next question. Acquisition parameters of the relevant sequences are summarised below.
Structural scan
For structural images a 3D MP2RAGE29 sequence was used: 3D-acquisition with field of view 224×224×168 mm3 (H-F; A-P; R-L), imaging matrix 320×320×240, 0.7 mm3 isotropic voxel size, Time of Repetition (TR)=5.0 s, Time of Echo (TE)=2.45 ms, Time of Inversion (TI) 1/2=0.9 s/2.75 s, Flip Angle (FA) 1/2=5°/3°, Bandwidth (BW)=250 Hz/Px, Partial Fourier 6/8, and GRAPPA acceleration with iPAT factor of 2 (24 reference lines).
Field map
For estimating B0 inhomogeneities, a 2D gradient echo sequence was used. It was acquired in axial orientation with field of view 192×192 mm2 (R-L; A-P), imaging matrix 64×64, 35 slices with 3.0 mm thickness, 3.0 mm3 isotropic voxel size, TR=1.5 s, TE1/2=6.00 ms/7.02 ms (which gives delta TE=1.02 ms), FA=72°, and BW=256 Hz/Px.
Whole-brain rs-fMRI
Whole-brain rs-fMRI scans were acquired using a 2D sequence. It used axial orientation, field of view 192×192 mm2 (R-L; A-P), imaging matrix 128×128, 70 slices with 1.5 mm thickness, 1.5 mm3 isotropic voxel size, TR=3.0 s, TE=17 ms, FA=70°, BW=1,116 Hz/Px, Partial Fourier 6/8, GRAPPA acceleration with iPAT factor of 3 (36 reference lines), and 300 repetitions resulting in 15 min of scanning time. Before the scan subjects were instructed to stay awake, keep their eyes open and focus on a cross. In order to avoid a pronounced g-factor penalty30 when using a 24 channel receive coil, the acceleration factor was kept at a maximum of 3, preventing the acquisition of whole-brain data sets at submillimeter resolution. However, as 7 T provides the necessary SNR for such high spatial resolutions a second experiment was performed with only partial brain coverage but with an 0.75 mm isotropic resolution.
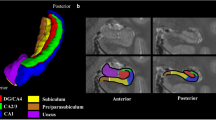
Prefrontal cortex rs-fMRI
The submillimeter rs-fMRI scan was acquired with a zoomed EPI31 2D acquisition sequence. It was acquired in axial orientation with skewed saturation pulse32 suppressing signal from posterior part of the brain (see Figure 2). The position of the field of view was motivated by the involvement of medial prefrontal cortex in the default mode network and mindwandering33. This location can also improve our understanding of functional anatomy of the prefrontal cortex which is understudied in comparison to primary sensory cortices. Field of view was 150×45 mm2 (R-L; A-P), imaging matrix=200×60, 40 slices with 0.75 mm thickness, 0.75 mm3 isotropic voxel size, TR=4.0 s, TE=26 ms, FA=70°, BW=1,042 Hz/Px, Partial Fourier 6/8. A total of 150 repetitions were acquired resulting in 10 min of scanning time. Before the scan subjects were instructed to stay awake, keep their eyes open and focus on a cross.
It has to be mentioned that for both rs-fMRI experiments only a single slice was excited at a time. Although simultaneous-multi-slice imaging enables a significant reduction in repetition time, a certain coil geometry is necessary for its successful application34. Preliminary tests with our 24 channel coil showed that the somewhat limited number of receive elements in head-feet direction prevented an artifact-free implementation of multi-band acquisition techniques as it would be possible with, e.g., a 32 channel coil. The lack of multislice acquisition resulted in relatively slow repetition time (3 and 4 s for full brain and prefrontal cortex respectively). This unfortunately limits the use of this dataset to investigating brain dynamics (and their physiological correlates) at lower frequencies.
Data Records
All data records listed in this section are available from the INDI/CoRR consortium facilitated through the COINS database35 (Data Citation 1). A README file with a detailed description of the content of all downloads is also available at this URL. The data is also available at http://openscience.cbs.mpg.de/7t_trt as a standard FTP download as well as BitTorrent facilitated by Academic Torrents to improve distribution36. Additionally the data has been deposited in our institute’s XNAT37 instance available publicly at http://xnat.cbs.mpg.de. We strongly encourage all users to subscribe to the http://groups.google.com/group/7t_trt mailing list for future updates and announcements.
All DICOM files were anonymized to remove any information that could identify the participants. files were converted to the NIfTI format using the dcmstack converter (https://github.com/moloney/dcmstack). All DICOM metadata were saved in the header of the NIFTI files in JSON format.
Demographics
Location: MPG_1_PhenotypicData
File format: plain text, comma separated values
Basic demographic information (sex, age at the first scan in years, handedness, and number of 7 T scans previously taken) is available in a comma-separated value (CSV) file.
Questionnaires and blood pressure measurements
Location: MPG_1_questionnaires_and_blood_pressure.csv
File format: plain text, comma separated values
Participants’ responses to the PANAS-X, sleep, caffeine, hydration questionnaires as well as the pre scan blood pressure are available in a CSV file. Data are structured as two lines per participant (one line per visit per participant) with questionnaire items as columns. A description of all items is given in Supplementary Table 1.
CCPT responses
Location: MPG_1_CCPT.csv
File format: plain text, comma separated values
Responses to the CCPT task are available as a CSV file. Each response for each participant and each visit is described by the presented stimuli, response and response time. A description of all items is given in Supplementary Table 2.
Mini NYC-Q responses
Location: MPG_1_mini_NYCQ.csv
File format: plain text, comma separated values
Responses to the mini NYC-Q questionnaire are available as a CSV file. Each participant responded to every item eight times (four per visit, one after CCPT task, three after resting state scans). A description of all items is given in Supplementary Table 3.
Anatomical scans
Location: MPG_1_ <ID>.tar.gz/session_[1–2]/anat_1/anat_*.nii.gz
File format: NIfTI, gzip-compressedSequence
MRI data are available in NIFTI formats. All structural scans have been defaced to protect participants identity. The defacing procedure was performed after DICOM to NIFTI conversion and therefore we do not include DICOM files in this dataset. Different reconstruction images are included:
-
1
anat_inv1—first inversion time volume
-
2
anat_inv1_phs—phase image for the first inversion
-
3
anat_inv2—second inversion time volume
-
4
anat_inv2_phs—phase image for the second inversion
-
5
anat_uni—unified volume (T1 weighted)
-
6
anat—quantitative T1 map (note: T1 time estimates above 4,000 ms in the CSF are arbitrarily set to 0 ms when calculated on the scanner)
Fieldmaps
Location: MPG_1_ <ID>.tar.gz/session_1-2]/fieldmap_[1–2]/fieldmap_[magnitude|phase].nii.gz
File format: NIfTI, gzip-compressed
fMRI data are available in NIFTI format. The fieldmap_magnitude files consist of two volumes corresponding to the two readouts at different TEs. The fieldmap_phase is the phase difference image.
Whole-brain rs-fMRI
Location: MPG_1_ <ID>.tar.gz /<ID>/session_[1–2]/rest_[1–2]/rest.nii.gz
File format: NIfTI, gzip-compressed
Prefrontal cortex rs-fMRI
Location: MPG_1_ <ID>.tar.gz /<ID>/session_[1–2]/rest_1_prefrontal/rest.nii.gz
File format: NIfTI, gzip-compressed
fMRI data are available in NIFTI and DICOM formats. Even though the NIFTI headers include all of the DICOM meta information thanks to the use of dcmstack converter, some tools (such as AFNI38) use different formats for storing meta information in NIFTI files.
Physiological recordings
Location: MPG_1_physio.tar.bz2/physio/<ID>/session_[1–2]/physio_trig_resp_card_oxy_[prefrontal|full_brain_1|full_brain_2].txt and MPG_1_physio.tar.bz2/physio/<ID>/session_[1–2]/physio_original_[prefrontal|full_brain_1|full_brain_2].acq
File format plain text, gzip-compressed or ACQ
Physiological data were down-sampled to 100 Hz and truncated to start with the first MRI trigger pulse and to end one volume acquisition duration after the last trigger pulse. Data are provided in a four-column (MRI trigger, respiratory trace, cardiac trace, and oxygen saturation), space-delimited text file for each rs-fMRI scan. Additionally, the original BIOPAC ACQ files recorded at a sampling rate of 5,000 Hz are provided. They can be read using the bioread package https://github.com/njvack/bioread.
Technical Validation
CCPT
To describe the distribution and variance across visits of the CCPT results we have plotted reaction times and percentage of mistakes each participant made (see Figure 3). Reaction times were stable across time and most participants made very few mistakes suggesting that the task was easy for them.
Mini NYC-Q
To assess the variance of the self reported content of self generated thoughts we have plotted the evolution of the answers over the period of the two visits (see Figure 4). There is a high variance between as well as within participants, consistent with the nature of mind wandering.
PANAS-X
We have decomposed the 60 items of the PANAS-X questionnaire into 13 subscales27 by averaging corresponding items (see Figure 5). It should be noted that the skewed distributions of ‘Sadness’ and ‘Hostility’ are due to the high responses of the same participant.
Blood pressure and pulse
As with other measures we also looked at the distribution of blood pressure and pulse measurements across participants and between visits (see Figure 6). Measurements from both arms were averaged together. All participants showed diastolic blood pressure to be within a healthy range (60–79 mm Hg). Some participants, however, experienced systolic blood pressure within the prehypertension range (120–139 mm Hg). The distribution of pulse measurements is rightly skewed due to one outlier (different than the outlier observed in PANAS-X).
Magnetic resonance imaging
To assess the quality of the whole-brain rs-fMRI scans we have calculated a series of quality metrics (see Figure 7). Many of these metrics overlap with the ones used by the Consortium for Reliability and Reproducibility (CoRR) (http://fcon_1000.projects.nitrc.org/indi/CoRR) which enables easier comparison with other studies in the consortium. The following metrics were calculated:
-
Framewise Displacement (FD)—a measure of volume to volume movement in millimeters. Calculated based on parameters estimated using AFNI 3dvolreg with -Fourier -twopass -zpad 4 arguments. Lower values mean less motion.
-
Temporal Signal to Noise Ratio (tSNR)—a voxelwise measure of signal strength. It is calculated by dividing the mean across time with standard deviation across time for each voxel individually. A median of all voxels within a brain mask is used to characterise each scan. Mask for this as well as all other metrics was derived using AFNI 3dAutomask command. Higher tSNR values mean better signal.
-
Entropy Focus Criterion—Shannon’s entropy is used to summarize the principal directions distribution39,40. Higher values indicate the distribution is more uniform (i.e., less noisy).
-
Foreground to Background Energy Ratio—ratio of average absolute value intensities within and outside of the brain mask. Higher values mean more clear signal.
-
Ghost to Signal Ratio—A measure of the mean signal in the ‘ghost’ image (signal present outside the brain due to acquisition in a particular phase encoding direction) relative to mean signal within the brain41. Lower values indicate fewer ghost artefacts.
-
Percentage of outliers—The mean fraction of outlier voxels found in each volume using AFNI 3dTout command in AFNI. Fewer outliers means better quality.
-
Median Distance Index—The mean distance (1—spearman’s rho) between each time-point’s volume and the median volume using AFNI 3dTqual command. Smaller values mean more homogeneous timeseries.
-
Image Smoothness—Smoothness of the image expressed as a millimeter Full Width Half Maximum. Smoothness was estimated using AFNI 3dFWHMx command.
Due to a mistake, one of the participant’s whole-brain rs-fMRI scans was acquired using a different sequence. This resulted in a change in the resolution from 1.5 to 3.0 mm. We kept the data of this participant in the dataset due to usefulness of all the other measurements, but we do not recommend mixing resolutions in fMRI analysis. This and other data acquisition anomalies are listed in Table 1.
Usage Notes
All data are made available under the terms of the Creative Commons Zero (CC0; PDDL; http://creativecommons.org/publicdomain/zero/1.0/). In short, this means that anybody is free to download and use this dataset for any purpose as well as to produce and re-share derived data artifacts. While not legally enforced, we hope that all users of the data will acknowledge the original authors by citing this publication and will follow good scientific practice (i.e. not try to re-identify the subjects).
Data are shared in documented standard formats, such as DICOM, NIfTI or plain text files, to enable further processing in arbitrary analysis environments with no imposed dependencies on proprietary tools. Many standard software packages may be currently challenged by the increase in data resolution and the changes in anatomical contrasts brought by the MP2RAGE sequence. Dedicated 7 T MP2RAGE methods for image segmentation and analysis are freely available within our CBS Tools software package (www.nitrc.org/projects/cbs-tools/) and have been tested and validated on similar data42. These tools are modular can be interfaced with other software packages43.
Additional information
How to cite this article: Gorgolewski, K. J. et al. A high resolution 7-Tesla resting-state fMRI test-retest dataset with cognitive and physiological measures. Sci. Data 2:140054 doi: 10.1038/sdata.2014.54 (2015).
References
References
Hale, J. R. et al. Comparison of functional connectivity in default mode and sensorimotor networks at 3 and 7T. MAGMA 23, 339–349 (2010).
De Martino, F. et al. Whole brain high-resolution functional imaging at ultra high magnetic fields: an application to the analysis of resting state networks. Neuroimage 57, 1031–1044 (2011).
Newton, A. T., Rogers, B. P., Gore, J. C. & Morgan, V. L. Improving measurement of functional connectivity through decreasing partial volume effects at 7 T. Neuroimage 59, 2511–2517 (2012).
Bianciardi, M. et al. Sources of functional magnetic resonance imaging signal fluctuations in the human brain at rest: a 7 T study. Magn. Reson. Imaging 27, 1019–1029 (2009).
Lenglet, C. et al. Comprehensive in vivo mapping of the human basal ganglia and thalamic connectome in individuals using 7T MRI. PLoS ONE 7, e29153 (2012).
Raemaekers, M. et al. Patterns of resting state connectivity in human primary visual cortical areas: a 7T fMRI study. Neuroimage 84, 911–921 (2014).
Yang, X., Holmes, M. J., Newton, A. T., Morgan, V. L. & Landman, B. A. A comparison of distributional considerations with statistical analysis of resting state fMRI at 3T and 7T. Proc. Soc. Photo. Opt. Instrum. Eng. 8314, 831416 (2012).
Yang, X., Kang, H., Newton, A. T. & Landman, B. A. Evaluation of statistical inference on empirical resting state fMRI. IEEE Trans. Biomed. Eng. 61, 1091–1099 (2014).
Shehzad, Z. et al. The resting brain: unconstrained yet reliable. Cereb. Cortex 19, 2209–2229 (2009).
Zhang, H. et al. Test-retest assessment of independent component analysis-derived resting-state functional connectivity based on functional near-infrared spectroscopy. Neuroimage 55, 607–615 (2011).
Birn, R. M. et al. The effect of scan length on the reliability of resting-state fMRI connectivity estimates. Neuroimage 83C, 550–558 (2013).
Braun, U. et al. Test-retest reliability of resting-state connectivity network characteristics using fMRI and graph theoretical measures. Neuroimage 59, 1404–1412 (2012).
Gorgolewski, K. J., Storkey, A. J., Bastin, M. E., Whittle, I. & Pernet, C. Single subject fMRI test-retest reliability metrics and confounding factors. Neuroimage 69, 231–243 (2013).
Bennett, C. M. & Miller, M. B. How reliable are the results from functional magnetic resonance imaging? Ann. N. Y. Acad. Sci. 1191, 133–155 (2010).
Yan, C.-G. et al. A comprehensive assessment of regional variation in the impact of head micromovements on functional connectomics. Neuroimage 76, 183–201 (2013).
Kucyi, A. & Davis, K. D. Dynamic functional connectivity of the default mode network tracks daydreaming. Neuroimage 100, 471–480 (2014).
Allen, E. A. et al. Tracking whole-brain connectivity dynamics in the resting state. Cereb. Cortex 24, 1–14 (2012).
Zalesky, A., Fornito, A., Cocchi, L., Gollo, L. L. & Breakspear, M. Time-resolved resting-state brain networks. Proc. Natl Acad. Sci. USA 111, 10341–10346 (2014).
Gorgolewski, K. J. et al. A correspondence between individual differences in the brain’s intrinsic functional architecture and the content and form of self-generated thoughts. PLoS ONE 9, e97176 (2014).
Shrout, P. E. & Fleiss, J. L. Intraclass correlations: uses in assessing rater reliability. Psychol. Bull. 86, 420–428 (1979).
Hanke, M. et al. A high-resolution 7-Tesla fMRI dataset from complex natural stimulation with an audio movie. Sci. Data 1, 140003 (2014).
Zuo, X. N. et al. An open science resource for establishing reliability and reproducibility in functional connectomics. Sci. Data 1, 140049 (2014).
Shalev, L., Ben-Simon, A., Mevorach, C., Cohen, Y. & Tsal, Y. Conjunctive Continuous Performance Task (CCPT)--a pure measure of sustained attention. Neuropsychologia 49, 2584–2591 (2011).
Mathôt, S., Schreij, D. & Theeuwes, J. OpenSesame: an open-source, graphical experiment builder for the social sciences. Behav. Res. Methods 44, 314–324 (2012).
Peirce, J. W. PsychoPy—Psychophysics software in Python. J. Neurosci. Methods 162, 8–13 (2007).
Peirce, J. W. Generating Stimuli for Neuroscience Using PsychoPy. Front. Neuroinform. 2, 10 (2008).
Watson, D. & Clark, L. A. The PANAS-X manual for the positive and negative affect schedule—expanded form http://ir.uiowa.edu/psychology_pubs/11/ (1999).
Grühn, D., Kotter-Grühn, D. & Röcke, C. Discrete affects across the adult lifespan: Evidence for multidimensionality and multidirectionality of affective experiences in young, middle-aged and older adults. J. Res. Pers. 44, 492–500 (2010).
Marques, J. P. et al. MP2RAGE, a self bias-field corrected sequence for improved segmentation and T1-mapping at high field. Neuroimage 49, 1271–1281 (2010).
Pruessmann, K. P., Weiger, M., Scheidegger, M. B. & Boesiger, P. SENSE: sensitivity encoding for fast MRI. Magn. Reson. Med. 42, 952–962 (1999).
Heidemann, R. M., Ivanov, D. & Trampel, R. Isotropic submillimeter fMRI in the human brain at 7 T: Combining reduced field of view imaging and partially parallel acquisitions. Magn. Reson. Med. 68, 1506–1516 (2012).
Pfeuffer, J. et al. Zoomed functional imaging in the human brain at 7 Tesla with simultaneous high spatial and high temporal resolution. Neuroimage 17, 272–286 (2002).
Andrews-Hanna, J. R., Reidler, J. S., Huang, C. & Buckner, R. L. Evidence for the default network’s role in spontaneous cognition. J. Neurophysiol. 104, 322–335 (2010).
Setsompop, K. et al. Improving diffusion MRI using simultaneous multi-slice echo planar imaging. Neuroimage 63, 569–580 (2012).
Scott, A. et al. COINS: An innovative informatics and neuroimaging tool suite built for large heterogeneous datasets. Front. Neuroinform 5, 33 (2011).
Langille, M. G. I. & Eisen, J. A. BioTorrents: a file sharing service for scientific data. PLoS ONE 5, e10071 (2010).
Marcus, D. S., Olsen, T. R., Ramaratnam, M. & Buckner, R. L. The extensible neuroimaging archive toolkit. Neuroinformatics 5, 11–33 (2007).
Cox, R. W. AFNI: software for analysis and visualization of functional magnetic resonance neuroimages. Comput. Biomed. Res. 29, 162–173 (1996).
Farzinfar, M. et al. Entropy based DTI quality control via regional orientation distribution. Proc. IEEE Int. Symp. Biomed. Imaging 22–26 (2012).
Atkinson, D., Hill, D. L., Stoyle, P. N., Summers, P. E. & Keevil, S. F. Automatic correction of motion artifacts in magnetic resonance images using an entropy focus criterion. IEEE Trans. Med. Imaging 16, 903–910 (1997).
Giannelli, M., Diciotti, S. & Tessa, C. Characterization of Nyquist ghost in EPI-fMRI acquisition sequences implemented on two clinical 1.5 T MR scanner systems: effect of readout bandwidth and echo spacing. J. Appl. Clin. Med. Phys. 11, 170–180 (2010).
Bazin, P.-L. et al. A computational framework for ultra-high resolution cortical segmentation at 7Tesla. Neuroimage 93, 201–209 (2014).
Gorgolewski, K. et al. Nipype: a flexible, lightweight and extensible neuroimaging data processing framework in python. Front. Neuroinform 5, 13 (2011).
Data Citations
Gorgolewski, K. J. Functional Connectomes Project International Neuroimaging Data-Sharing Initiative (2014) http://dx.doi.org/10.15387/fcp_indi.corr.mpg1
Acknowledgements
This work has been funded by the Max Planck Society.
Author information
Authors and Affiliations
Contributions
K.J.G. conceived and implemented the study and wrote the manuscript. D.W., E.W., N.M., T.B., K.J.G., C.J.G., and D.S.M. acquired the data. R.T. selected and fine-tuned MRI sequences. F.J.M.R. and J.S. helped with creation of the mini NYC-Q. R.C. helped with preparing the final data set version. T.B., P.-L.B., D.S.M., R.T., N.M. and C.G. helped in writing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interest.
ISA-Tab metadata
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0 Metadata associated with this Data Descriptor is available at http://www.nature.com/sdata/ and is released under the CC0 waiver to maximize reuse.
About this article
Cite this article
Gorgolewski, K., Mendes, N., Wilfling, D. et al. A high resolution 7-Tesla resting-state fMRI test-retest dataset with cognitive and physiological measures. Sci Data 2, 140054 (2015). https://doi.org/10.1038/sdata.2014.54
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/sdata.2014.54
This article is cited by
-
Prediction of stimulus-independent and task-unrelated thought from functional brain networks
Nature Communications (2021)
-
Distinct regions of the striatum underlying effort, movement initiation and effort discounting
Nature Human Behaviour (2020)
-
Maintenance and Representation of Mind Wandering during Resting-State fMRI
Scientific Reports (2017)
-
Towards a mechanistic understanding of the human subcortex
Nature Reviews Neuroscience (2017)
-
Test–retest reliability of brain morphology estimates
Brain Informatics (2017)