Abstract
Purpose:
In cervical spinal cord injury (CSCI), afferent and efferent circuits that influence the basal production of melatonin and cortisol may be disrupted and hence disrupt the basal functions of human physiology. Therefore, the aim of this study was to assess circadian changes, if any, in serum cortisol and melatonin in patients with CSCI.
Methods:
Serum levels of cortisol and melatonin were measured at 6-h intervals of the day (0600, 1200, 1800 and 0000 hours) in 22 CSCI patients, as well as 22 healthy controls.
Results:
Significantly higher melatonin levels were observed in the patient group in morning hours, whereas a significantly lower level of melatonin was found during the night time in the patient group than in the control group. Moreover, significantly higher values were obtained in the evening and night time serum cortisol levels among the patients compared with controls. Further, when the mean values of cortisol throughout the day were tested among patient and control groups similar circadian rhythm was found. The only difference being that serum cortisol declined much more in controls in evening and night samples as compared with CSCI patients.
Conclusion:
We conclude that circadian variations exist in the circulating levels of serum cortisol and melatonin in patients with CSCI. Low levels of melatonin secretion during night may contribute to the pervasive sleep disruption and increased pain perception.
Similar content being viewed by others
Introduction
Circadian rhythm is characterized by the dynamics of 24 h cycle. The disturbance in secretion of hormones that are sensitive to light and dark cycle results in abnormal circadian rhythm. Melatonin is the primary hormone of the pineal gland, and it regulates the sleep and wake cycle.1 Melatonin has been documented to be a potent neuroprotective agent in traumatic brain injury and spinal cord injury.2 Normally, melatonin levels begin to rise in the mid-to-late evening, remain high for most of the night and then decline in the early morning hours.3 Melatonin is produced at night from 5-hydroxytryptophan, and it displays dynamic circadian rhythm.4 During the day, the intense light blocks the production of melatonin; however, during night, the decline in the intensity of light is registered in the retina, which then sends the information to the pineal gland.5 On arrival of the signal from the retina, melatonin begins to rise in blood and then in all other body tissues, between 0000 hours to 0400 hours melatonin rises to 70 pg ml–1 and it remains elevated during the normal hours of sleep at night.6 This increase in melatonin levels provides a convenient signal to all body cells about the onset of night, which is a signal for sleep. Moreover, cortisol is produced in the adrenal gland, and it normally follows a circadian pattern of secretion; it peaks in the morning just after waking (10–20 μg dl–1) followed by a gradual decrease at 1600 hours (3–10 μg dl–1) and during night time (>5 μg dl–1).7, 8 Melatonin and cortisol both are counter regulatory hormone. When melatonin level rises at night, cortisol level drops to its lowest, and when cortisol level rises to its highest in morning, melatonin level drops to its lowest. Abnormal levels of melatonin and cortisol rhythm result in problems with sleep. However, sleep deprivation is common and can be a serious problem negatively affecting health. Sleep has been reported to allow the removal of free radicals accumulated in the brain during wakefulness, whereas poor sleep can induce oxidative damage and disturb the removal of free radicals in the brain. Long-term sleep deprivation is harmful to health and can lead to multisystemic and multiorgan dysfunction, causing maladaptive emotional regulation, exaggerated neural reactivity and negative metabolic, psychological, physiological or even behavioral reactivity.
In cervical spinal cord injury (CSCI), afferent and efferent circuits that influence the basal production of melatonin and cortisol may be disrupted and hence disrupt the basal functions of human physiology. Further, cervical spinal cord lesions disrupt the circadian rhythm in human melatonin excretion. Levels of serum cortisol, aldosterone and growth hormone showed rhythmic variations in subjects with SCI. Reduced sleep efficiency in CSCI causes low night time melatonin secretion. Thus, the decrease in night time melatonin in CSCI may help explain the sleep disturbances, raising the possibility that melatonin replacement therapy could help normalize sleep in these patients.9 Rhythm of serum melatonin with acute SCI was observed at the cervical and thoracic regions in rats, and the study revealed that the complete injuries at the lower cervical spinal cord could not produce enough melatonin secretion, which may contribute to impaired sleep at night, fatigue during the day, and immense pain perception; on the contrary, complete injury at the upper thoracic spinal cord showed normal melatonin secretion.10 Rhythms of serum melatonin in patients with spinal lesions at the cervical region were seen, and it was observed that there were low levels of serum melatonin with no observable diurnal rhythm in the patient with CSCI (C4-5). This study suggests that the cervical region of the spinal cord is part of the neural pathway essential for the diurnal rhythm of pineal melatonin secretion in humans.11
The similarity in symptoms between patients with CSCI and individuals with misaligned circadian phase, as well as the observation of cortisol and melatonin levels being tightly regulated by the circadian pacemaker, raises the possibility that there may be a disturbed circadian pattern of these hormones in patients with CSCI. Disrupted hormone levels after SCI have been shown in previous studies, and in this study we are confirming previous finding in an Indian population by characterizing the results over more time intervals. Therefore, on the basis of above reports, we planned to study the circadian pattern of serum cortisol and melatonin in patients with CSCI and in healthy subjects. The goal of this study was to examine the 24-h pattern of cortisol and melatonin secretion in CSCI patients and controls. To test this hypothesis, cortisol and melatonin were measured at different time intervals during the day and night in patients with CSCI.
Materials and methods
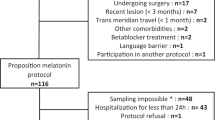
Twenty-four hour serum cortisol and melatonin patterns were determined in 22 CSCI patients and a control group comprising 22 healthy individuals without CSCI who were non-alcoholic, non-smokers, non-diabetic without any kind of cardiac, respiratory or endocrinal disease; 2 weeks post injury participants were recruited. All the patients were in their acute injury phases. Patients of CSCI were recruited from the spinal unit in the Department of Physical Medicine and Rehabilitation, Consultant Incharge recruited the patients, at the King George’s Medical University, Lucknow, India. The level of SCI was determined by neurological examination of complete (Frankel A) cervical injuries. Besides the neurological damage to their spinal cords, each subject was physically and mentally healthy, as determined by history and physical examination, psychological questionnaires and interview with a psychologist, electrocardiogram, blood and urine chemistries and chest radiographs. In no case was brain damage or extended loss of consciousness associated with the SCI was found. All subjects were drug free at the time of study, including prescription, nonprescription and over the counter medications, as well as caffeine, nicotine and alcohol, as confirmed by urine toxicology on the first day of the inpatient study. During the week before the study, subjects maintained a regular sleep/wake schedule, as confirmed by patient self-report. These SCI subjects were otherwise healthy as established by medical history and physical and psychological examination. Furthermore, all subjects were male; control subjects were recruited from relatives of the patients and other normal persons of Lucknow.
All the patients and controls were screened to exclude any recent chronobiological disruptions, such as shift work or travel across various time zones. Before enrolling in the study, written informed consent was obtained from both the subject groups using documents approved by the Institutional Ethics Committee, and all applicable institutional and governmental regulations concerning the ethical use of human volunteers were followed during this study. All data were coded to remove any identifiable information. Both the group of patients and controls were admitted to the Department of Physical Medicine and Rehabilitation, where the subjects completed a structured questionnaire, (developed inhouse and are not validated instruments) which assessed the biographical information, medical, personal and family history. Sleep status was measured from the questionnaire itself asking the patients they have sound sleep or disturbed sleep. Intravenous blood (5 ml) was collected at 0600, 1200, 1800 and 0000 hours. Blood samples were centrifuged at 3000 r.p.m. for 10 min; all the samples collected in the day were spun within half an hour of collection, and serum was separated and was aliquoted into labeled storage tubes and frozen at −40 °C until assayed. Serum cortisol and melatonin levels were assayed by standard capture enzyme-linked immunosorbent assay. The assays were performed according to the manufacturer's instructions, and absorbance was measured at λ=450 nm on a Microplate ELISA reader (BIO-RAD, i-MARK (IBL, Hamburg, Germany)).
Statistical analysis
Statistical analysis was performed using SPSS statistical software (16.0 versions, SPSS Inc., Chicago, IL, USA). Quantitative variables of CSCI patients and controls were presented as the mean±s.d. and are compared by an independent t-test. However, dichotomous (categorical) variables were compared by the chi-square (χ2) test. The circadian pattern in the hormonal profiles (cortisol and melatonin) was analyzed by the analysis of variance test. A value of P<0.05 was considered statistically significant, and P<0.01 is considered highly significant.
Results
Clinical parameters
Clinical assessments of CSCI patients and controls were reported, and parameters like muscle twitching, lack of energy, morning stiffness, morning fatigue, headache, frequent awakening, weight loss and sleep status were measured; these clinical parameters were more commonly seen among patients with CSCI than in controls. Further, there was no missing data from any participant (Table 1).
Melatonin
Significantly higher melatonin levels were observed among the patients group (patients 25.1±6.7; controls 13.7±6.9 P<0.01) in morning hours, whereas significantly lower levels of melatonin were found in the night time in the patients group (patients 49.3±19.5; controls 65.6±10.6 P<0.01) than in the control group. However, no statistically significant difference was found in the afternoon and evening serum melatonin levels among patient and control groups. Further, when the mean values of melatonin in the morning, afternoon, evening and night time samples among patient and control groups were tested with analysis of variance, a significant difference in the circadian rhythm of melatonin among the patients and controls was found (Figure 1).
Cortisol
Significant difference in the evening (patients 12.4±6.8; controls 10.9±4.7; P<0.05) and night time (patients 18.3±10.9; controls 8.1±4.0; P<0.01) serum cortisol levels was observed among the patient and control groups. However, serum cortisol was not found significantly different between patients and control groups in the morning hours and afternoon time. Further, when the mean values of melatonin in the morning, afternoon, evening and night time samples among patients and control groups were tested with analysis of variance, a significant difference in the circadian rhythm of cortisol among the patients and controls was found (Figure 2).
Discussion
The present study was designed to examine the hypothesis that patients with CSCI exhibit alterations in circadian rhythm of cortisol and melatonin secretion. Neuro-endocrine hormone secretion is characterized by circadian rhythmicity, for example, cortisol12 and melatonin are secreted in periodic bursts,10 and these bursts cannot be seen unless sampling is performed rapidly enough to capture the basic structure of the burst. In this study, we found a significant difference in the evening and mid-night level of serum cortisol in patients compared with the control group, which revealed a disturbed circadian pattern in serum cortisol level in patients with CSCI. However, study by Kalpakjian et al.,13 reported no changes in cortisol pattern in both the patient and control group. Moreover, a significantly higher melatonin levels were observed in morning hours, whereas a significantly lower levels of melatonin was found in the night time in the patient group than in the control group. However, no statistically significant difference was found in the afternoon and evening serum melatonin levels among patient and control groups.
Cortisol has neuroprotective effects, and activation of cortisol secretion starts immediately after the SCI injury and remained high compared with the control.14 In a study reported by Llompart et al.,15 circadian variability of cortisol evaluated by serum and cerebral microdialysis samples was found to be lost in traumatic brain injury patients. Previous studies have reported conflicting data on cortisol amplitude in chronic SCI, with some claiming low, others claiming normal and yet others claiming high circulating concentrations of cortisol.16, 17, 18 However, those studies generally relied on one or two time points in the determination of amplitude. Under such circumstances, the daily rhythm of cortisol concentrations and its inherent pulsatility may have confounded the results. Our study therefore have measured the 6 hourly blood levels of cortisol in CSCI subjects, which exhibit alterations in circadian rhythm of serum cortisol secretion. This increase in nocturnal serum cortisol in the patient group suggests disturbed circadian patterns that may explain in part the patient complaint of disturbed sleep.
Our findings of the altered level of serum melatonin at night agree with Scheer et al.9 report of disturbed level of night time melatonin secretion, which results in chronically reduced sleep quality. Thus, the decreased night time melatonin secretion may explain the excessive daytime sleepiness and disturbed sleep observed in patients with CSCI.19, 20, 21 Furthermore, the increased latency to rapid eye movement sleep in subjects with CSCI suggests that the decreased night time melatonin secretion may partially underlie the previously observed reduction in rapid eye movement sleep propensity.20 Melatonin has been proposed to facilitate sleep by inhibiting the circadian drive for waking that emanates from the suprachiasmatic nucleus,22, 23, 24 by binding to the high-affinity melatonin receptors in the suprachiasmatic nucleus.25 Also, sleep propensity, sleepiness and rapid eye movement sleep expression are mainly under circadian control.26 Study by Shekleton et al., reported reduced evening melatonin production that may indicate disruption to circadian regulation of melatonin synthesis.27 However, our findings are in concurrence with the report of Gezici et al.10 who found that injuries at the lower cervical spinal cord may disturb the normal melatonin secretion, and on the contrary injury at the upper thoracic spinal cord showed normal melatonin secretion. Thus, the decrease in night time melatonin secretion in CSCI may help explain the sleep disturbances. Therefore, poor subjective sleep quality was associated with higher ratings of pain intensity, anxiety and depression. It is possible that disturbed melatonin secretion at night serves as a modulator of these different aspects.28
In a study by Jones et al., the correlations between evening changes in melatonin, core and skin temperature between thoracic and CSCI and able-bodied participants were compared. They reported that the correlation between evening changes in melatonin and thermoregulation is of a similar magnitude in paraplegic and controls.29 However, others reported that in cervical spinal cord lesions normal melatonin secretion is disturbed; this disruption of normal melatonin secretion may be caused by decentralization of the pineal gland due to a lesion within the cervical spinal cord interrupting descending sympathetic fibers.11 SCI remains a devastating complication of thoracic and thoraco-abdominal aortic operations, but the melatonin-treated group had better neurologic function than the non-treated group.30 Potent protective effects of melatonin on experimental SCI showed that injection of melatonin reduced thiobarbituric acid reactive substances content and myeloperoxidase activity, facilitating recovery of the damaged spinal cord.31 Moreover, melatonin supplementation and/or cortisol inhibitors could be investigated in future as interventions that may help facilitate normal sleep patterns in acute CSCI patients, and why might this be important? healing? better wake/sleep cycles? Quality of life?.
In conclusion, our study suggests that using the rigorously controlled, constant routine protocol to examine circadian variation in the endocrine function, we demonstrated that patients with CSCI suffer with disturbed cortisol and melatonin secretion. Therefore, the results of the present study indicate that disturbed sleep in patients with CSCI may in part be due to the disturbed nocturnal melatonin secretion. Furthermore, increase in nocturnal serum cortisol in the patients group suggests dysregulated circadian patterns. However, the findings of the present study may be a very small step put forward. Further studies are necessary to confirm, evaluate and replicate this study in a larger sample size with different ethnicities.
Data Archiving
There were no data to deposit.
References
Fatima G, Sharma VP, Shukla R . Circadian rhythm in patients with spinal cord injuries. Ann Neurosci 2009; 16: 31–33.
Naseem M, Parvez S . Role of melatonin in traumatic brain injury and spinal cord injury. Sci World J 2014; 2014: 54–67.
Mehdi AA, Fatima G, Das SK, Verma NS . Abnormality of circadian rhythm in serum melatonin and other biochemical parameters in fibromyalgia syndrome. IJBB 2011; 48: 82–87.
Sun X, Deng J, Liu T, Borjigin J . Circadian 5-HT production regulated by adrenergic signaling. Proc Natl Acad Sci 2002; 99: 4686–4691.
Reiter RJ . The melatonin rhythm: both a clock and a calendar. Experientia 1993; 49: 654–1664.
Lynch HJ, Brzezinski A, Deng MH, Lieberman H. Advances in Pineal Research. In: Fraschini F, Reiter RJ (eds) vol. 2. Libbey, London, 1987, pp 181–189..
Bauer ME . Stress, glucocorticoids and ageing of the immune system. Stress 2005; 8: 69–83.
Wallerius S, Rosmond R, Ljung T . Rise in morning saliva cortisol is associated with abdominal obesity in men: a preliminary report. J Endocrinol Invest 2003; 26: 616–619.
Scheer FA, Zeitzer JM, Ayas NT, Brown R, Czeisler CA, Shea SA . Reduced sleep efficiency in cervical spinal cord injury; association with abolished night time melatonin secretion. J Spinal cord 2006; 44: 78–81.
Gezici AR, Karaka ÅŸ . Rhythms of serum melatonin in rats with acute spinal cord injury at the cervical and thoracic regions. Spinal Cord 2010; 48: 10–14.
Kneisley LW, Moskowitz MA, Lynch HG . Cervical spinal cord lesions disrupt the rhythm in human melatonin excretion. J Neural Transm Suppl 1978; 13: 311–323.
Mazzoccoli G, Carughi S, Sperandeo M . Neuro-endocrine correlations of hypothalamicpitutary-thyroid axis in healthy humans. J Biol Regul Homeost Agents 2011; 25: 249–257.
Kalpakjian CZ, Farrell DJ, Albright KJ, Chiodo A, Young EA . Association of daily stressors and salivary cortisol in spinal cord injury. Rehabil Psychol 2009; 54: 288–298.
Gezici AR, Karakas A, Ergün R, Gündüz B . Serum cortisol level following acute cervical and thoracic lesions. J Neurol Neurosurg 2009; 43: 352–357.
Llompart-Pou JA, Pérez G, Raurich JM, Riesco M, Brell M, Ibáñez J et al. Loss of cortisol circadian rhythm in patients with traumatic brain injury: a microdialysis evaluation. Neurocrit Care 2010; 13: 211–216.
Grant JMF, Yeo JD . Studies on the levels of 17 hydroxy-corticoids in 24-hour specimens of urine from five quadriplegic patients and two paraplegic patients admitted to the Royal North Shore Hospital, Sydney. Paraplegia 1968; 6: 29–31.
Eisenstein AB, Wenneker AS, Londe AM . Effect of spinal cord transection on adrenocortical function. Proc Soc Exp Biol Med 1962; 109: 947–950.
Osborn W, Schoenberg HM, Murphy JJ, Erdman WJ, Young D . Adrenal function in patients with lesions high in the spinal cord. J Urol 1962; 88: 1–4.
Adey WR, Bors E, Porter RW . EEG sleep patterns after high cervical lesions in man. Arch Neurol 1968; 19: 377–383.
McEvoy RD, Mykytyn I, Sajkov D, Flavell H, Marshall R, Antic R et al. Sleep apnoea in patients with quadriplegia. Thorax 1995; 50: 613–619.
Biering-Sorensen F, Biering-Sorensen M . Sleep disturances in the spinalcord injured: an epidemiological questionnaire investigation, including a normal population. Spinal Cord 2001; 39: 505–513.
Lavie P . Melatonin: role in gating nocturnal rise in sleep propensity. J Biol Rhythms 1997; 12: 657–665.
Sack RL, Hughes RJ, Edgar DM, Lewy AJ . Sleep-promoting effects of melatonin: at what dose, in whom, under what conditions, and by what mechanisms? Sleep 1997; 20: 908–915.
Barinaga M . How jet-lag hormone does double duty in the brain. Science 1998; 277: 480.
Reppert SM, Weaver DR, Rivkees SA, Stopa EG . Putative melatonin receptors in a human biological clock. Science 1988; 242: 78–81.
Dijk DJ, Czeisler CA . Contribution of the circadian pacemaker and the sleep homeostat to sleep propensity, sleep structure, electroencephalographic slow waves, and sleep spindle activity in humans. J Neurosci 1995; 15: 3526–3538.
Shekleton JA, Parcell DL, Redman JR, Phipps-Nelson J, Ponsford JL, Rajaratnam SM . Sleep disturbance and melatonin levels following traumatic brain injury. Neurology 2010; 74: 1732–1738.
Norrbrink Budh C, Hultling C, Lundeberg T . Quality of sleep in individuals with spinal cord injury: a comparison between patients with and without pain. Spinal Cord 2005; 43: 85–95.
Jones H, Eijsvogels TM, Nyakayiru J, Verheggen RJ, Thompson A, Groothuis JT et al. Within-subject correlations between evening-related changes in body temperature and melatonin in the spinal cord injured. Chronobiol Int 2014; 31: 157–165.
Korkmaz A, Oyar EO, KardeAY O, OmeroA Ylu S . Effects of melatonin on ischemic spinal cord injury caused by aortic cross clamping in rabbits. Curr Neurovasc Res 2008; 5: 46–51.
Fujimoto T, Nakamura T, Ikeda T, Takagi K . Potent protective effects of melatonin on experimental spinal cord injury. J Spine 2000; 25: 769–775.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Fatima, G., Sharma, V. & Verma, N. Circadian variations in melatonin and cortisol in patients with cervical spinal cord injury. Spinal Cord 54, 364–367 (2016). https://doi.org/10.1038/sc.2015.176
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2015.176
This article is cited by
-
Melatonin secretion and sleep disorders in patients with spinal cord injuries
Spinal Cord (2024)
-
A comprehensive look at the psychoneuroimmunoendocrinology of spinal cord injury and its progression: mechanisms and clinical opportunities
Military Medical Research (2023)
-
Melatonin-mediated FKBP4 downregulation protects against stress-induced neuronal mitochondria dysfunctions by blocking nuclear translocation of GR
Cell Death & Disease (2023)
-
Endocrinological and inflammatory markers in individuals with spinal cord injury: A systematic review and meta-analysis
Reviews in Endocrine and Metabolic Disorders (2022)
-
The effects of a high-fat/high-carbohydrate meal on leukocyte populations in adults with chronic spinal cord injury
Spinal Cord Series and Cases (2021)