Abstract
Study design:
In this study, we explored how galvanic vestibular stimulation can modify the soleus H-reflex (Hoffman reflex), that is, the excitability of the spinal cord circuits, in healthy humans.
Objectives:
Our aim was to demonstrate H-reflex amplitude modulation caused by changing the duration and the intensity of the anodal galvanic vestibular stimulation. Therefore, we measured H-reflex before and after applied vestibular stimulation.
Settings:
This study was conducted in Rehabilitation Clinic, Belgrade, Serbia.
Methods:
The measurements were performed on 5 male volunteers aged 22–30 years. Anodal galvanic stimulation was applied on the right mastoid in prone position. H-reflex was elicited by nervus tibialis stimulation and measured from the right soleus muscle. In three subjects, trains of weak and strong galvanic stimuli (1, 5 and 9) were applied. In two subjects, only a train of 9 strong stimuli was applied.
Results:
A statistically significant decrease of the H-reflex amplitude after anodal galvanic stimulation was demonstrated in all subjects. The percentage of H-reflex amplitude diminution was between 6 and 18 in subjects with weak and strong stimuli and 5 and 6 in subjects with only 9 strong stimuli.
Conclusion:
We intend to use this paradigm of stimulation to explore whether the vestibulospinal function exists after spinal cord injury (SCI). If it exists, it can be used to influence the preserved spinal cord circuits after SCI.
Sponsorship:
One of the authors (Nadica Miljković) was partly supported by the Ministry of education, science and technological development, Republic of Serbia, grant OS175016.
Similar content being viewed by others
Introduction
It has been long known that a small direct current, also called galvanic current, applied between mastoids produces an inclination of the standing person.1 Several researchers have studied the electromyographic (EMG) responses occurring in response to galvanic stimulation of postural muscles in healthy subjects. They used a 1- or 4- to 6 -s duration, 1–4-mA intensity stimulus.2, 3 If averaging of the EMG activity was necessary, they used stimuli of shorter duration.4, 5, 6 There is general agreement that anodal stimulation causes hypotonia and cathodal stimulation causes hypertonia of the ipsilateral postural muscles. The effect of galvanic current is also influenced by head and body posture,7 as well as postural task.8 In squirrel monkeys, it was demonstrated that the galvanic stimulation acted directly on the vestibular afferents (from semicircular canals, as well as otoliths): cathodal current increased the firing rate (causing hypertonia) and anodal current decreased the firing rate (causing hypotonia of the muscles).9 More recently,10, 11, 12, 13 the electrical-induced analog of the monosynaptic reflex called the H-reflex has been used in assessment of the modulation of the monosynaptic activity at the spinal cord level.
If a short-lasting electrical stimulus is applied to the tibial nerve within the popliteal fossa, motor and sensory fibers are stimulated simultaneously. The stimulation of motor fibers results in a muscle twitch of the soleus muscle, which can be demonstrated by means of bipolar superficial electrodes placed over the corresponding muscle belly. If recorded by means of EMG, this muscle twitch is called the M wave, and it appears approximately 10 s after the electrical stimulation. The simultaneous stimulation of sensory fibers proceeds to the spinal cord to the α-motoneuron cells of the anterior horn of the S6 segment (by monosynaptic connection). This stimulation results in a second muscle twitch, and it can be demonstrated with the same EMG recording after approximately 40 ms. It is called H wave or H-reflex (Hoffman reflex).
As it is known, spasticity can be one of the difficult-to-manage clinical problems in patients after spinal cord injury (SCI). Thus, in one of our previous preliminary measurements, we tried to decrease hypertonia of the postural lower limb muscles by applying anodal galvanic stimulation in patients with spasticity after SCI. We examined the effect of anodal galvanic vestibular stimulation by clinical Ashworth scale. In some of the patients, we succeeded to lower the spasticity of the ipsilateral limb by 1–2 grades. The reduction lasted for 1–5 min.14
After SCI, the spinal pathways remain intact to a greater or lesser extent. Even in individuals with severe SCIs, some spared trans-lesional systems, descending long-tract fibers or propriospinal fibers, are nearly always present.15, 16 It has been shown that the remaining altered spinal cord circuits below the injury can be activated and modified by transcutaneous spinal cord stimulation.17 We suppose that in patients in whom the vestibulospinal pathways remained preserved, the altered spinal cord circuits below the injury could be conditioned by activation of the vestibulospinal pathways. To confirm this assumption, additional experimental work must be done.
In this report, we explored how vestibulospinal stimulation, applied with different duration and intensities in healthy humans, can modify (condition) the H-reflex—that is, the excitability of the spinal cord circuits.
Materials and methods
H-reflex was obtained from five human subjects, all male, aged 21–32 years. None of them had a history of cochlear or vestibular disease. All subjects gave their informed consent to the study protocol, which conformed to the standard set by the Declaration of Helsinki and was approved by the Ethics Committee of the Rehabilitation Clinic ‘Miroslav Zotović’ in Belgrade (No 03932/2011). Three subjects underwent the long protocol of galvanic stimulation, and two subjects only underwent the short one.
To elicit the H-reflex, the stimulation was applied via J10R00 PULSE and 895340 (Axelgaard Co., Ltd, Fallbrook, CA, USA) neurostimulation electrodes. A smaller cathode electrode with a radius of 2.5 cm was placed over the popliteal fossa, and a large anode rectangular electrode (7.5 × 10 cm2) was placed over the patella. The stimulation electrodes were strapped to the leg by means of a plastic ball, to ensure that a constant stimulus was delivered to the nerve. We used ‘FES stimulator #2’ designed in the Center for Sensory-Motor Interaction, Aalborg University, Aalborg, Denmark. To obtain the M wave and H-reflex, we delivered a 500-μs square wave pulse of 16–30 mA intensity with a stimulation frequency of 0.1 Hz.
Surface EMG was recorded from the right soleus muscle with pregelled, disposable, surface Ag/AgCl Ambu Neuroline 720 electrodes (Ambu, Neuroline, Ballerup, Denmark) sized 20 × 30 mm2 and positioned over the soleus muscle according to the SENIAM protocol.18 We used a 25-cm-long ground electrode (GCB-geliMED KG, Bad Segeberg, Germany) and placed it between stimulation and bipolar recording electrodes. EMG signals were amplified with Grass RPS 107 amplifier (Grass Technologies, An Astro-Med, Inc., West Warwick, RI, USA). The parameters of the amplifier were as follows: band-pass filter 3–3000 Hz and gain 500. Signals were digitized with DAQCard-6062E (National Instruments Inc., Austin, TX, USA) with a resolution of 12 bits and with a sampling rate of 10 000 samples per second.
For galvanic stimulation of the vestibular system, we used custom-made galvanic stimulator (GS). The GS was built upon the known characteristics of the vestibular system, and it allows repetitive stimulation in the frequency range of 0.1–10 Hz. The pulse duration can be set in the interval from 0.5 to 9 s. The pulse amplitude of monopolar pulses can be set in the range of 1–10 mA. The stimulator is galvanically separated from the rest of the stimulation/acquisition system. The GS has two potentiometers: one for the current amplitude control and the second for frequency setting. The stimulator has an indicator light that blinks in the rhythm of the stimulation. The stimulation was applied via self-adhering, reusable BF-2 Bio-Flex electrodes (Bio-Medical Instruments, Warren, MI, USA). The number of stimuli of GS can be manually adjusted to be 1, 2, …, 9 or until the stop button is pressed.
Both stimulators and acquisition are controlled by the data acquisition software in the LabVIEW (National Instruments Inc., Austin, TX, USA) environment. The data acquisition uses the MrKick software (Center for Sensory-Motor Interaction, Aalborg, Denmark). The triggering of the stimulator for the peripheral pathways is controlled by the same software.
The subjects were lying in a quiet, darkened room in the prone position. The subjects were instructed to place their forehead on a marker on the bed in order to maintain the midline position. Their eyes were closed. Their right foot was slightly elevated by a thin pillow (10 cm high) in order to avoid discomfort from the foot strike over the edge of the bed during the stimulation of the tibial nerve.
The current intensity was increased until the maximal M wave response was obtained. After that, the current intensity of the electrical stimulus was diminished to produce the M wave equal to 10–15% of its maximal amplitude. This M wave amplitude was maintained throughout the experiment.
Electrodes for galvanic stimulation were adjusted to the size of the subject’s mastoid and fixed to the retroauricular skin. Anodal galvanic stimulation was applied to the right mastoid. The duration of single galvanic stimulus was 1 s. In three subjects, we applied two groups of stimulation. In the first group, the intensity of the stimulus was somewhat above the subject’s level of perception of the galvanic stimulus (supraliminal stimulus— weak stimulus). First, one stimulus was applied, and then 5 and then 9 stimuli were applied during one recording. We enhanced the galvanic stimulation until the maximal tolerable and then diminished the intensity by 30%. With this intensity (submaximal—strong stimulus), we repeated the same pattern of stimulation (long protocol). To avoid the effect of previous galvanic stimulation on the next recording, a pause of 5 min was introduced between each stimulus within a group and of 10 min between two groups.
In three subjects, only the strong stimulus was applied 9 times (short protocol) (Figure 1).
In all subjects, the H-reflex was recorded 1 min before the galvanic stimulation and 2 min after the galvanic stimulation.
The recorded evoked potentials before and after the galvanic current application were stored on a computer for off-line analysis. For studying the statistical significance of the H-wave change, statistical analyses were performed by Difference between two proportions, using the Statistica 6.0 Software (StatSoft, Inc., Tulsa, OK, USA, 2001). Furthermore, the percentage of diminution of the H-wave amplitude was calculated during the same off-line procedure.
Results
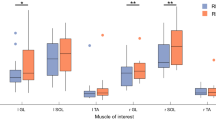
The M wave-averaged peak-to-peak values (amplitudes) and the H-reflex-averaged peak-to-peak values (amplitudes) were calculated before and after galvanic current application (Figure 2).
The M wave amplitude did not exceed 5% of the initial value in any of the measurements.
The H-reflex amplitude decreased generally with increasing current intensity and with increasing number of stimuli. In Table 1, the arithmetical means of the M and H amplitudes before and after galvanic current application, together with s.ds., only for subject number 2 are shown.
Statistical significance of the amplitude decrease was demonstrated in all subjects with 5 and 9 strong stimuli (P<0.01 or P<0.05). After weak stimulation, the change of the H-reflex amplitude was statistically significant in subject number 2 with 5 and 9 weak stimuli and in subject number 3 with 9 weak stimuli (Table 2). In measurements in which the statistically significant decrement of the amplitudes was shown, the percentage of the decrement was calculated. In subject number 1, the percentage was between 17 and 18; in subject number 2 it was between 12 and 18; in subject number 3 it was between 4 and 6; and in subjects 4 and 5 the percentage was between 5 and 6 (Table 2).
We can conclude that a dependence of the response (H wave amplitude decrement) to stimulus parameters exists. An inter-subject variability of the susceptibility on galvanic stimulation exists as well. It is shown that the threshold of the galvanic stimulus which causes a statistically significant decrement is presented as the percentage of H wave amplitude decrement. Two out of three subjects who underwent the long protocol had a better decrease of the H wave amplitude than the two subjects who underwent the short protocol. Therefore, we suppose that a temporal summation effect of the galvanic stimulation exists.
Discussion
For registration of H-reflex, we followed the procedure recommended by several authors.12, 19 The M wave and H wave recruitment curve was established at the beginning for each subject. The M wave amplitude was diminished by 10–15% of the maximal M wave amplitude, and it was maintained with this value throughout the measurement. The inter-stimuli interval of tibial nerve stimulation was 10 s, as recommended. Because of the influence of the head position on the vestibular apparatus and on H-reflex generation, all subjects maintained a midline head position.7 Visual inputs were eliminated by eye closure,20 and noise influence was eliminated by maintaining a quiet environment.19
Because of all the circumstances described, we presume that the H-reflex amplitude in the period after galvanic stimulation is because of the galvanic current influence on spinal cord circuitry on the measured level.
As expected, our measurements demonstrated a general diminution of the H-reflex amplitude after anodal galvanic stimulation. Stronger influence on H-reflex formation was obtained by stronger galvanic current, and this was demonstrated in all three subjects who underwent the long protocol with 5 and 9 strong stimuli application.
With weak galvanic current stimulation, the susceptibility was better in subject number 2 compared with subject numbers 1 and 3. A variability in the degree of H-reflex amplitude decrement between subjects, as well as a variability of the percentage of amplitude decrement, was present. This could be because of the individual threshold of the vestibular nerve to galvanic current excitation, as well as to individual supraspinal influence on H-reflex generation.20 In addition, we observed that the same electrical intensity of the galvanic stimulus was experienced differently in different subjects. Before the beginning of the measurements, we augmented the galvanic stimulation to the level of maximal tolerance (and then diminished the stimulus intensity for 30%: this was the strong stimulus). The same electrical intensity of maximal tolerance, which was defined as very unpleasant by some subjects, was described as not unpleasant at all by others. Neither strong nor weak stimulus was painful for the subject.
Thompson et al.21 demonstrated that the H-reflex size can be decreased or increased voluntarily. However, to accomplish this task, the examinees had biofeedback information on the monitor with H-reflex presentation, and multiple sessions4, 5, 6 were needed. Our subjects were blinded to the H-reflex amplitude during the measurement, and they did not receive instructions to influence the H-reflex. Therefore, we assume that our subjects did not influence the H-reflex amplitude voluntarily.
Kennedy and Inglis22 applied a monaural galvanic stimulation of 4 mA intensity, 2 s duration, on subject’s mastoid. Cathodal (negative) and anodal (positive) stimulations were used. Ten subjects participated in the study. The galvanic stimulus was chosen so that the subjects could barely perceive the stimulus. An interval of 100 ms was interposed between the galvanic and the tibial nerve stimulation; the galvanic current proceeded the tibial nerve stimulation by 100 ms. Measurement of the H-reflex revealed an inhibition of the H-reflex amplitude after anodal stimulation (mean value −3.3%). Cathodal stimulation produced a mean increase of 3.2% of the H-reflex amplitude.7
The results obtained by Kennedy and Inglis22 are not contradictory to the results of our measurements. During our measurements, there was no H-reflex amplitude change with a single weak stimulus, probably because we did not interpose the time interval needed that the galvanic stimulus reaches the measured spinal level at the same time as the tibial nerve stimulus. To decrease the amplitude of the H wave, we needed more repetitions. The effect of galvanic stimulation was >3%, when we increased the stimulus intensity and the number of stimulations.
We demonstrated that galvanic current stimulation can be a tool for modification of the spinal cord circuits. The influence was more successful as the current amplitude and the number of stimuli increased.
Our next task will be to explore in which patients after SCI the vestibulospinal function exists after SCI. If it exists, the question erases how much the vestibular stimulation can influence the spasticity of the patient’s lower limb muscles.
Data Archiving
There were no data to deposit.
References
Coats AC, Stoltz MS . The recorded body sway response to galvanic stimulation of the labyrint: a preliminary study. Laryngoscope 1969; 79: 85–103.
Tokita T, Ito Y, Miyata H, Koizumi H . Labyrinthine control of upright standing posture in humans. Progr Brain Res 1988; 76: 291–295.
Iles JF, Pisini JV . Vestibular-evoked postural reactions in man and modulation of transmission in spinal reflex pathways. J Physiol 1992; 455: 407–424.
Watson SRD, Colebatch JG . Vestibular-evoked electromyographic responses in soleus: a comparison between click and galvanic stimulation. Exp Brain Res 1998; 119: 504–510.
Britton TC, Day BL, Brown P, Rothwell JC, Thompson PD, Marsden CD . Postural electromyographic responses in the arm and leg following galvanic vestibular stimulation in man. Exp Brain Res 1993; 94: 143–151.
Fitzpatrick R, Burke D, Gandevia SC . Task dependent reflex responses and movement illusions evoked by galvanic vestibular stimulation in standing humans. J Physiol (Lond) 1994; 478: 363–372.
Kennedy PM, Inglis TJ . Interaction effects of galvanic vestibular stimulation and head position on the soleus H reflex in human. Clin Neurophysiol 2002; 113: 1709–1714.
Capaday C, Stein RB . Amplitude modulation of the soleus H-reflex in the human during walking and standing. J Neurosci 1986; 6: 1308–1313.
Goldberg JM, Fernandez C, Smith CE . Response of vestibular nerve Afferents in the squirrel monkey to externally applied galvanic currents. Brain Res 1982; 252: 156–160.
Schubert M, Beck S, Taube W, Amtage F, Faist M, Gruber M . Balance training and ballistic strength training are associated with task specific corticospinal adaptations. Eur J Neurosci 2008; 27: 2007–2018.
Costa J, Guszmain J, Valldeoriola F, Rumia J, Tolosa E, Casanova-Molla J et al. Modulaton of the soleus H reflex by electrical subcortical stimuli in humans. Exp Brain Res 2011; 2012: 439–448.
Palmieri RM, Ingersoll ChD, Hoffman MA . The Hoffmann reflex: methodological considerations and applications for use in sports medicine and athletic training research. J Athl Train 2004; 39: 268–277.
Chen Y-Sh, Zhou Sh . Soleus H-reflex and its relation to the static postural control. Gait Posture 2011; 33: 169–178.
Ribarić-Jankes K, Čobeljić R, Švirtlih L. The influence of unipolar galvanic vestibular stimulation on lower limb spasticity in spinal cord injured patients. In: J Zidar (ed.), Proceedings of the International Symposium on Spinal Cord Motor Control: ‘From Denervated Muscle to Neurocontrol’; 7–9 September 2006; Ljubljana. Society of Clinical Neurophysiology, Ljubljana, Slovenia, p. 101.
McKay WB, Lim HK, Priebe MM, Stokic DS, Sherwood AM . Clinical neurophysiological assessment of residual motor control in post-spinal cord injury paralysis. Neurorehabil Neural Repair 2004; 18: 144–153.
Kakulas BA . Neuropathology: the foundation for new treatments in spinal cord injury. Spinal Cord 2004; 42: 549–563.
Hofstoetter US, McKay WB, Tansey KE, Mayr W, Kern H, Minassian K . Modification of spasticity by transcutaneous spinal cord stimulation in individuals with incomplete spinal cord injury. J Spinal Cord Med 2014; 37: 202–211.
Hermens HJ, Freriks B, Disselhorst-Klug C, Rau G . Development of recommendations for SEMG sensors and sensor placement procedures. J Electromyogr Kinesiol 2000; 10: 361–374.
Kameyama O, Hayes KC, Wolfe D . Methodological considerations contributing to variability of the quadriceps H-reflex. Am J Phys Med Rehab 1989; 68: 2.
Wolpaw JR . Spinal cord plasticity in acquisition and maintenance of motor skills. Acta Physiol (Oxf) 2007; 189: 155–169.
Thompson AK, Chen XY, Wolpaw JR . Acquisition of a simple motor skill: task-dependent adaptation plus long-term change in the human soleus H-reflex. J Neurosci 2009; 29: 5784–5792.
Kennedy PM, Inglis TJ . Modulation of the soleus H-reflex in prone human subjects using galvanic vestibular stimulation. Clin Neurophysiol 2001; 112: 2159–2163.
Acknowledgements
We thank Professor Milan R Dimitrijević whose work inspired our work and who supervised the measurements. Professor Dejan Popovic kindly provided the necessary instrumentation. We also thank Dobrivoje Stokić, MD, who taught us how to perform the H-reflex measurements. In preparing this article, biologist Zoran Gačić and Ljubomir Damjanović, MSc, provided valuable technical help. We also thank technician Mihajlo Tančić who volunteered to help us in taking the measurements. Nadica Miljković was partly supported by the Ministry of Education, science and technological development, Republic of Serbia, grant OS175016.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Čobeljić, R., Miljković, N., Ribarić-Jankes, K. et al. A paradigm of galvanic vestibular stimulation diminishes the soleus muscle H-reflex in healthy volunteers. Spinal Cord 54, 150–153 (2016). https://doi.org/10.1038/sc.2015.135
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2015.135