Abstract
Objective:
Magnetic resonance imaging (MRI) is useful in diagnosing spontaneous spinal epidural hematoma (SSEH). The purpose of the present study is to determine whether apparent diffusion coefficient (ADC) values could determine severity of spinal cord damage and predict functional recovery in SSEH.
Methods:
The study involved four consecutive patients with SSEH (two men and two women: aged 21–76 years). Using axial slices, ADC values were determined in four separate regions of the spinal cord. These areas were classified into the following three groups based on findings in T2-weighted images: normal T2 intensity; persistent T2 abnormality; and temporary T2 abnormality. ADC values among different groups were compared. The relationship between preoperative ADC values and neurological grades were also evaluated.
Results:
ADC values in normal T2 areas were 0.89±0.10 × 10−3 mm2 s−1, whereas those for the persistent T2 abnormality group were significantly lower (0.63±0.14 × 10−3). In a patient who was Frankel A on admission and in the follow-up, the ADC value was as low as 0.41 × 10−3. Functional recovery was also limited in the spinal cord segments with lower ADC values. In the temporary T2 abnormality group, ADC values were significantly higher (1.05±0.10 × 10−3).
Conclusions:
In SSEH, if MRI demonstrated T2-hyperintensity with lower ADC values, patients may suffer from irreversible spinal cord damages. ADC values of the spinal cord can be added as a new factor that reliably indicated the severity of spinal cord damage and predicted functional recovery.
Similar content being viewed by others
Introduction
Spontaneous spinal epidural hematoma (SSEH) is a rare clinical entity with an estimated incidence of 0.1 patients per 100 000 per year.1 Patients typically present with sudden onset of acute back or neck pain. Accumulation of hematoma in the epidural space may compress the spinal cord or nerve roots and potentially cause neurological damage to patients.
If SSEH is suspected, conventional magnetic resonance images (MRIs) are obtained to confirm the diagnosis, and to evaluate the location and size of hematomas as well as the severity of spinal cord compression.2, 3 Conventional MRI can estimate when the bleeding occurred, based on the signal intensities of hematomas in T1- and T2-weighted images.1 Such information is valuable since it would determine whether emergent surgical evacuation of the hematoma is indicated to control the bleeding and decompress the spinal cord.
MRI can also detect spinal cord parenchymal injury by generating intramedullary hyperintense signals on a T2-weighted image.3 However, hyperintense signals do not differentiate between reversible and irreversible spinal cord damage. Thus, it may be difficult to predict a functional prognosis based on conventional MRIs in cases of SSEH.
Diffusion-weighted images (DWI) provide additional information regarding the microscopic motion of water protons.4 Apparent diffusion coefficient (ADC) values calculated through DWI have been used to differentiate between cytotoxic and vasogenic edema5 in the brain through markedly decreased and increased ADC values, respectively. Nonetheless, both of these pathological changes generate hyperintensities on T2-weighted images.
We hypothesized that cases with predominant vasogenic edema in a compressed spinal cord have high ADC values on admission and may show better functional recovery, if treated appropriately. On the other hand, cases with irreversible damages would have lower ADC values, indicating cytotoxic insults. The purpose of the present study was to investigate whether ADC values in spinal cord DWIs can determine the degree of damage in the spinal cord parenchyma and could be used to predict a prognosis for SSEH.
Patients and methods
Patient population
Four consecutive patients (two men and two women; aged, 21–76 years; mean age, 55.6 years) were admitted to our hospital for the treatment of SSEH between January 2009 and December 2009. All patients were evaluated by MRI soon after admission. Informed consent was obtained from all patients regarding their participation in the present study. Diffusion-weighted MRI was a part of the clinical imaging protocol at our institution (Sendai Medical Center), and institutional review board approval was not obtained.
Patient neurological status was assessed on admission and in last follow up using the Frankel classification (A–E). Patients were followed up for a duration of 30–45 months (mean, 37.3 months).
MRI and data processing
Patients were imaged using a 1.5-T MR imaging system, Magnetom Avanto (Siemens Medical Solutions, Erlangen, Germany). All patients underwent MR scanning on admission. If surgical treatments were indicated, MRIs were routinely performed 2 and 6 weeks postoperatively. For those who were treated conservatively, MRIs were obtained 2 weeks and 6 months after the onset. Conventional T1- and T2-weighted images were obtained with sagittal and axial planes; T2*-weighted images were also obtained. Referring to sagittal T1- and T2-weighted images, 15 axial slices, 8 mm apart, were placed to cover hematoma, compressed spinal cord and T2 hyperintensity areas in the spinal cord. Positions of the axial slices were identical in conventional MRI and DWI.
Diffusion-weighted imaging was performed using a fat-suppressed, spin-echo, echo-planar imaging sequence. Fifteen axial scans were performed with the following scanning parameters: repetition time, 4000 ms; echo time, 78 ms; echo space, 0.74 ms; imaging matrix, 128 × 128; field of view, 20 × 20 cm; average number, 8; slice thickness, 5 mm, with three orthogonal directional motion probing gradients (b=1000 s mm−2), followed by automatic generation of isotropic DWIs. Images without motion-probing gradients (b=0 s mm−2) were also obtained.
After ADC maps were generated, ADC values were determined using the OsiriX version 3.7.1 (http://www.osirix-viewer.com/) by placing four oval-shaped regions-of-interest (ROI; area, ∼0.05 cm2) side-by-side on axial section images of the spinal cord, with two located closer to the surface of the cord and the other two in the middle (Figures 1 and 2). These lateral and medial ROIs mainly covered the white and gray matter, respectively. To confirm positions of ROIs, we referred T2-weighted axial images obtained in the same slice positions.
Apparent diffusion coefficient (ADC) values in a spinal cord with normal intensity in T2-weighted images on admission (normal T2), T2-hyperintense lesions which persisted in follow-up MRIs (persistent T2 abnormality), and T2-hyperintense areas that resolved in follow ups (temporary T2 abnormality). ADC values in the persistent T2 abnormality group were 0.63±0.14 × 10−3 mm2 s−1, that is, significantly lower than those of the normal T2 group. In the temporary T2 abnormality group, ADC values (1.05±0.10 × 10−3 mm2 s−1) were significantly higher than those of the other groups. Data represent mean±s.d. *P<0.001.
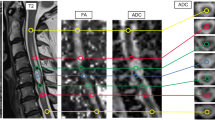
Pre and postoperative MRI and preoperative ADC values in case 1. (a–e) Sagittal (a) and axial (b and c) T2-weighted images showing a hyperintense lesion and compressed spinal cord. The position of axial slices in b and c are indicated by an arrowhead and arrow, respectively, in a. T2* (d) and DWI (e) were from the same slice as c. The locations of the hematoma and T2-hyperintense lesions in the spinal cord were mainly on the right side (c–e). (f) Graph showing ADC values calculated from axial DWI of the four different regions in the preoperative MRI (e). The gray area indicates disc levels where T2-hyperintense lesions were present. In particular, ADC values were lower on the right side from C4 to C6 levels. (g–i) Sagittal (g) and axial (h and i) T2-weighted images 6 weeks after the operation. Levels of axial slices (h and i) are indicated by an arrowhead and an arrow in g which correspond to (b and c), respectively. Note that a majority of T2-hyperintense lesions were resolved in postoperative MRIs (g and h), except the right side of the spinal cord at C5 level (i) in which preoperative ADC values were as low as 0.66 × 10−3 mm2 s−1. Right (Rt.) Lateral, Rt. Medial, Left (Lt.) Medial and Lt. Lateral. Numbers on the X-axis indicate vertebral disc levels.
Statistical analysis
The ROIs, in which ADCs were calculated, were classified into the following three groups based on the findings of MRI on admission and in follow ups: normal T2, areas without T2 abnormality on admission; persistent T2 abnormality, areas with T2-hyperintensity that persisted in follow ups; temporary T2 abnormality, areas with T2-hyperintensity that resolved in follow ups. For instance, in Case 1, T2-hyperintense lesions detected at C2/3 disc level (Figure 2b) had resolved in the follow-up MRI (Figure 2h). These areas were classified as the temporary T2 abnormality group. On the other hand, the T2 abnormality that was evident on the right side of the spinal cord at the C5 level on pre and postoperative MRIs (Figures 2c and i) was deemed persistent T2 abnormality.
Four ROIs in each of the 15 axial slices per patient provided total 240 ROIs in four patients. ADC values in these three groups were compared using one-way analysis of variance followed by post-hoc Tukey’s test. A receiver operating characteristic analysis determined cutoff ADC value that provides the best combination of sensitivity and specificity to detect the persistent T2 abnormality. Data represent mean±s.d. Differences were deemed statistically significant for P<0.05.
Results
Patient characteristics including clinical grades and findings of the conventional MRIs are shown in Table 1. Two patients (Cases 1 and 2) underwent surgical evacuation of the hematoma owing to the severity of their clinical symptoms. The other two patients (Cases 3 and 4) were treated conservatively and showed full functional recovery. Three out of four patients sustained parenchymal injury as confirmed by the T2-hyperintense area on admission. In two patients, follow-up MRIs confirmed that a previously T2-hyperintense lesion was present. In a patient with complete spinal cord damage and no functional recovery (Case 2), ADC values were as low as 0.41 × 10−3 mm2 s−1 (Table 1). In two patients who were classified as Frankel E in follow ups, ADC values on admission were higher than 0.73 × 10−3 mm2 s−1.
We classified the areas in the spinal cord as those with a normal appearance in T2-weighted images on admission (normal T2; n=182), areas with T2-hyperintense lesions that persisted in follow-up MRIs (persistent T2 abnormality; n=30), and T2-hyperintense areas that resolved in the follow-up (temporary T2 abnormality; n=28). ADC values on admission were 0.89±0.09, 0.63±0.14 and 1.05±0.10 × 10−3 mm2 s−1, and they were significantly different (P<0.001; Figure 1). In particular, ADC values lower than 0.79 × 10−3 mm2 s−1 predicted persistent T2 abnormality in follow-up MRI with sensitivity and specificity as 89.3% and 89.8%, respectively.
Illustrative cases
Case 1
A 76-year-old woman with a chief complaint of neck pain and tetraparesis was transferred from another hospital. Her limb paresis was more evident on the right side. When initial symptoms occurred, she was riding a bicycle and thus fell down. She had been conservatively treated for 24 h for possible brainstem infarction before admission to our hospital. The patient had no medical history related to coagulopathy, and was classified as Frankel C on admission. In particular, motor strength in the upper extremities was 1/5 in a manual muscle test on the right side and 4/5 on the left side. Lower extremities were judged as 0/5 and 2/5 on the right and left side, respectively. An MRI disclosed spinal epidural hematoma from C3 to C7 with cord compression. T2-weighted hyperintense lesions were noted in the vertebral levels from C2 to C7 (Figure 2). At the levels of T2-hyperintensity, ADC values were higher on the left side and as low as 0.66 × 10−3 mm2 s−1 on the right side of the spinal cord. The patient underwent a right C4–6 hemilaminectomy and evacuation of the hematoma 41 h after the onset. The diagnosis of SSEH was confirmed. Postoperatively, the patient showed partial neurological recovery. At the final follow up, 45 months after the onset, the patient recovered to Frankel D. Her motor strength recovered to 5/5 and 4/5 in the upper and lower extremities, respectively on the left side. However, the motor strength on the right side, in which ADC values were lower on admission, was 2/5 in the upper and 3/5 in the lower limbs. A follow-up MRI revealed that the T2 abnormality was sustained on the right side of the cervical spinal cord from C4 to C6 (Figure 2).
Case 2
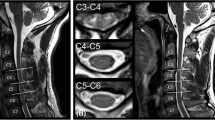
A 21-year-old man who experienced back pain and hypesthesia in his bilateral lower extremities was transferred to our hospital. On admission, 14 h after the onset of his symptoms, he had complete paraplegia below the level of T4. The patient was classified as Frankel A on admission. An MRI revealed spinal epidural hematoma in the vertebral levels of T3–5. A hyperintense signal in the T2-weighted image was noticed in the spinal cord centered at T4 where the cord compression was most severe (Figure 3). ADC values were calculated from all 15 axial slices. At the T4 level, ADC values were as low as 0.42 × 10−3 mm2 s−1 (Figure 3). No parenchymal hemorrhage was detected on T2*-weighted images. After discussing the relative risks and benefits of surgical intervention with the patient, we immediately performed a T3 hemilaminectomy on the right side to evacuate the hematoma and decompress the dural sac and spinal cord 16 h after the initial symptoms emerged. No vascular or neoplastic lesions were encountered during the operation. Considering that there was no history of trauma or coagulopathy, SSEH was diagnosed. The postoperative course was uneventful. The patient did not show any functional recovery. Postoperative MRIs performed at 6 weeks after the operation showed a T2-hyperintense lesion at the T4 level, where cord compression was most severe and ADC values were minimum on the preoperative MRI. The patient was transferred to a local rehabilitation center. During the last follow up, 40 months after the operation, he was classified as Frankel A.
MRI and ADC values in Case 2. (a) Preoperative sagittal T2-weighted image, showing epidural hematoma (*) and cord compression with abnormal-T2 hyperintense lesions from T3 to T4 vertebral levels (arrowheads). (b–d) Axial T1-weighted image (b), T2-weighted image (c), and DWI (d) on admission at the T3/4 level as indicated by an arrow in a. (e) Magnified image of d focusing on the spinal cord with oval-shaped ROIs placed to measure ADC values. (f) Graph showing ADC values calculated from axial DWI at the four different regions as indicated in e. The gray area indicates disc levels where T2-hyperintensity lesions were present in the spinal cord, and ADC values were as low as 0.41 × 10−3 mm2 s−1. Abbreviations are as indicated in Figure 2.
Discussion
In SSEH, T2-hyperintense lesions in the spinal cord indicate parenchymal damage. This study demonstrated that abnormalities in T2-weighted MRIs persist if the area had lower ADC values on admission. Furthermore, patients with lower ADC values sustained severe neurological deficits and showed limited functional recovery. On the other hand, if ADC values were higher, T2-hyperintense lesions resolved in follow-up MRIs. In our series, the neurological outcome was favorable in patients with higher ADC values. Although the number of cases in this study is limited, there is a tendency that low ADC values in the spinal cord parenchyma may indicate severe and irreversible damages in SSEH.
Correlation between pathological changes and ADC values
Another limitation of the present study is that histological evaluation was not possible. Thus, we can only speculate mechanisms explaining the correlation between low ADC values and persistent T2 abnormality, that is, irreversible spinal cord damage. When the spinal cord is compressed owing to epidural hematoma, blood flow in the spinal cord decreases as demonstrated in a rat spinal cord compression model.6 Such ischemic insult breaks the integrity of the blood–spinal cord barrier, followed by excessive accumulation of water in the extracellular space. In this situation, ADC values are higher, reflecting increased water diffusion. If the compression persists, ischemia and nutritional deficits cause progressive degeneration.7 When it progressed and cells lost their functions, intracellular edema occurs owing to sodium influx.8 Water mobility is restricted because of a net influx of water from the extracellular to intracellular space. This decreased water diffusion in the predominant cytotoxic edema may reflect as lower ADC values. In animal models, when the degree of cord compression was more severe and the duration of compression was longer, histological damage progressed to apoptotic cell death or loss of the myelin sheath.6, 9 In experimental traumatic brain injury, ADC values were lower when cytotoxic edemas were observed in earlier phases after injury. This was followed by an increase in ADC values corresponding to demyelination and decreased tissue cellularity in the long term.10, 11
Spinal cord DWI
A DWI of the spinal cord is technically demanding because of the small volume of the spinal cord. Physiological motion, including cerebrospinal fluid flow and cardiac and respiratory motion, also influenced the quality of images.12 However, spinal MRIs with DWI parameters have become available after modifications in parameters.13 In experimental spinal cord injury using rodents, the diffusivity, parameters obtained from diffusion tensor imaging, decreased at the site injury within 6 h. Importantly, decrement of axial diffusion predicted injury severity and correlated with histological changes.14 Recently, diffusion tensor imaging can discriminate microstructural differences between the gray and white matter of the spinal cord with 9.4T magnet.15 It would be intriguing if MRI parameters could discriminate different histological changes in the gray and white matter in human studies.
Recently, we evaluated ADC values in patients with sustained acute spinal cord injury by applying the same parameters used in the present study.16 We demonstrated a correlation between the severity of spinal cord lesions and ADC values in spinal cord trauma. If lesions had low ADC values, they remained as T2-hyperintense lesions in the long term and eventually evolved into cystic cavities, whereas T2-hyperintense lesions with higher ADC values were more likely to resolve after surgical interventions. These observations suggest that a spinal cord DWI is a reliable method for evaluating pathological changes in the spinal cord parenchyma. Other investigators have also confirmed the usefulness of ADC values obtained from spinal cord DWIs in acute and chronic spinal cord injury12, 17 and cervical spondylosis.18
Prognostic factors for SSEH
Factors affecting the prognosis of SSEH have been listed in various studies. They include size or location of the hematoma, speed of hematoma accumulation, neurological grade on admission and timing of the surgery.2, 19, 20 For better functional recovery, these studies commonly propose prompt surgical evacuation of the epidural hematoma before complete or irreversible damage occurs in the spinal cord parenchyma. Moreover, in case of the occurrence of complete dysfunction, functional recovery can be expected if decompression is performed early.2, 19 In one of these studies, 65.9% of patients who received surgical treatment within 12 h of the onset of symptoms recovered completely, while the complete recovery rate decreased to 29.1% if surgical decompression was performed from 13 h to 1week.3
To date, we have relied on neurological examination and the duration of the symptoms to estimate the irreversibility of spinal cord damage in SSEH. Our observations have added other parameters; ADC values of the spinal cord can be used to estimate severity of the spinal cord damage in SSEH. In particular, T2-hyperintense lesions in the spinal cord with higher ADC values may correspond to reversible vasogenic edema, whereas lesions with lower ADC values may indicate irreversible cytotoxic damage. Although further studies are warranted, we suggest that patient classification based on preoperative ADC values helps in deciding surgical indications and predicting the prognosis of patients suffering from SSEH.
Conclusions
ADC values are used to differentiate between cytotoxic and vasogenic edema in neuroimaging. Our results indicate that lower ADC values in SSEH may represent irreversible histological changes. Patients with lower ADC values had poor preoperative neurological grades and showed limited functional recovery. In contrast, lesions with high ADC values, representing vasogenic edema, were resolved radiographically. It is hoped that a useful algorithm for treating SSEH depending on ADC values of the spinal cord can be established in the near future by having larger sample sizes with longer follow ups.
Data Archiving
There were no data to deposit.
References
Holtas S, Heiling M, Lonntoft M . Spontaneous spinal epidural hematoma: findings at MR imaging and clinical correlation. Radiology 1996; 199: 409–413.
Liu Z, Jiao Q, Xu J, Wang X, Li S, You C . Spontaneous spinal epidural hematoma: analysis of 23 cases. Surg Neurol 2008; 69: 253–260.
Kreppel D, Antoniadis G, Seeling W . Spinal hematoma: a literature survey with meta-analysis of 613 patients. Neurosurg Rev 2003; 26: 1–49.
Moseley ME, Kucharczyk J, Mintorovitch J, Cohen Y, Kurhanewicz J, Derugin N et al. Diffusion-weighted MR imaging of acute stroke: correlation with T2-weighted and magnetic susceptibility-enhanced MR imaging in cats. Am J Neuroradiol 1990; 11: 423–429.
Schaefer PW, Buonanno FS, Gonzalez RG, Schwamm LH . Diffusion-weighted imaging discriminates between cytotoxic and vasogenic edema in a patient with eclampsia. Stroke 1997; 28: 1082–1085.
Hamamoto Y, Ogata T, Morino T, Hino M, Yamamoto H . Real-time direct measurement of spinal cord blood flow at the site of compression: relationship between blood flow recovery and motor deficiency in spinal cord injury. Spine (Phila Pa 1976) 2007; 32: 1955–1962.
Tator CH, Fehlings MG . Review of the secondary injury theory of acute spinal cord trauma with emphasis on vascular mechanisms. J Neurosurg 1991; 75: 15–26.
Choi DW . Ionic dependence of glutamate neurotoxicity. J Neurosci 1987; 7: 369–379.
Carlson GD, Gorden CD, Oliff HS, Pillai JJ, LaManna JC . Sustained spinal cord compression: part I: time-dependent effect on long-term pathophysiology. J Bone Joint Surg Am 2003; 85-A: 86–94.
Van Putten HP, Bouwhuis MG, Muizelaar JP, Lyeth BG, Berman RF . Diffusion-weighted imaging of edema following traumatic brain injury in rats: effects of secondary hypoxia. J Neurotrauma 2005; 22: 857–872.
Mac Donald CL, Dikranian K, Bayly P, Holtzman D, Brody D . Diffusion tensor imaging reliably detects experimental traumatic axonal injury and indicates approximate time of injury. J Neurosci 2007; 27: 11869–11876.
Shanmuganathan K, Gullapalli RP, Zhuo J, Mirvis SE . Diffusion tensor MR imaging in cervical spine trauma. Am J Neuroradiol 2008; 29: 655–659.
Tsuchiya K, Katase S, Fujikawa A, Hachiya J, Kanazawa H, Yodo K . Diffusion-weighted MRI of the cervical spinal cord using a single-shot fast spin-echo technique: findings in normal subjects and in myelomalacia. Neuroradiology 2003; 45: 90–94.
Loy DN, Kim JH, Xie M, Schmidt RE, Trinkaus K, Song SK . Diffusion tensor imaging predicts hyperacute spinal cord injury severity. J Neurotrauma 2007; 24: 979–990.
Ellingson BM, Kurpad SN, Li SJ, Schmit BD . In vivo diffusion tensor imaging of the rat spinal cord at 9.4T. JMRI 2008; 27: 634–642.
Endo T, Suzuki S, Utsunomiya A, Uenohara H, Tominaga T . Prediction of neurological recovery using apparent diffusion coefficient in cases of incomplete spinal cord injury. Neurosurgery 2011; 68: 329–336.
Tsuchiya K, Fujikawa A, Honya K, Tateishi H, Nitatori T . Value of diffusion-weighted MR imaging in acute cervical cord injury as a predictor of outcome. Neuroradiology 2006; 48: 803–808.
Demir A, Ries M, Moonen CT, Vital JM, Dehais J, Arne P et al. Diffusion-weighted MR imaging with apparent diffusion coefficient and apparent diffusion tensor maps in cervical spondylotic myelopathy. Radiology 2003; 229: 37–43.
Liao CC, Hsieh PC, Lin TK, Lin CL, Lo YL, Lee SC . Surgical treatment of spontaneous spinal epidural hematoma: a 5-year experience. J Neurosurg Spine 2009; 11: 480–486.
Groen RJ, van Alphen HA . Operative treatment of spontaneous spinal epidural hematomas: a study of the factors determining postoperative outcome. Neurosurgery 1996; 39: 494–509.
Acknowledgements
We thank Kazuyuki Yoshikawa (Sendai Medical Center) for his technical assistance and contribution to this project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Endo, T., Suzuki, S., Inoue, T. et al. Prediction of neurological recovery in spontaneous spinal epidural hematoma using apparent diffusion coefficient values. Spinal Cord 52, 729–733 (2014). https://doi.org/10.1038/sc.2014.53
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2014.53
This article is cited by
-
Spontaneous spinal epidural haematomas in children
European Spine Journal (2019)
-
Cervical Spine Prospective Feasibility Study
Clinical Neuroradiology (2019)