Abstract
Objectives:
To develop an International Spinal Cord Injury (SCI) Musculoskeletal Basic Data Set as part of the International SCI Data Sets to facilitate consistent collection and reporting of basic musculoskeletal findings in the SCI population.
Setting:
International.
Methods:
A first draft of an SCI Musculoskeletal Basic Data Set was developed by an international working group. This was reviewed by many different organizations, societies and individuals over 9 months. Revised versions were created successively.
Results:
The final version of the International SCI Musculoskeletal Basic Data Set contains questions on neuro-musculoskeletal history before spinal cord lesion; presence of spasticity/spasms; treatment for spasticity within the last 4 weeks; fracture(s) since the spinal cord lesion; heterotopic ossification; contracture; the location of degenerative neuromuscular and skeletal changes due to overuse after SCI; SCI-related neuromuscular scoliosis; the method(s) used to determine the presence of neuromuscular scoliosis; surgical treatment of the scoliosis; other musculoskeletal problems; if any of the musculoskeletal challenges above interfere with activities of daily living. Instructions for data collection and the data collection form are freely available on the International Spinal Cord Society (ISCoS) website (www.iscos.org.uk).
Conclusion:
The International SCI Musculoskeletal Basic Data Set will facilitate consistent collection and reporting of basic musculoskeletal findings in the SCI population.
Similar content being viewed by others
Introduction
Individuals with spinal cord injury (SCI) are highly susceptible to premature musculoskeletal complications.1 An SCI potentially accelerates age-related declines in the function of body structures compared with able bodied individuals. In addition, individuals sustaining an SCI at older age may have other pre-existing co-morbidities that affect their functional capability. Therefore, individuals with SCI are at increased risk of upper limb extremity pain,2, 3 rapid bone loss in the lower extremities,4, 5 and fractures.6 SCI also results in spasticity and contractures in approximately two-thirds of individuals.7 There is the additional risk of progressive scoliosis for those injured at a young age. Information on these and other musculoskeletal complications are important because they help guide management and provide accurate estimates of the magnitude of the problem.
A spinal cord lesion may be traumatic or non-traumatic in etiology. Lesions to the spinal cord, conus medullaris and cauda equina are included in the term.
The purpose of the International Spinal Cord Injury (SCI) Musculoskeletal Basic Data Set for individuals with spinal cord lesions is to standardize the collection and reporting of a minimal amount of information on musculoskeletal status in accordance with the purpose and vision of the International SCI Data Sets.8 This will also enable evaluation and comparison of results from various published studies, and help guide future research.
The data in this International SCI Musculoskeletal Basic SCI Data Set will be used in connection with data in the International SCI Core Data Set,9 which includes information on date of birth and injury, gender, cause of spinal cord lesion and neurologic status. In addition, the Core Data Set contains information on the history of the injury and initial management including presence of vertebral injury, need for spinal surgery, associated injuries and place of residence upon discharge after initial inpatient care.
It is important that data can be collected in a uniform manner from a scientific perspective as well as for improved patient care. For this reason, each variable and its accompanying response category has been defined to promote the collection and reporting of comparable minimal data. In addition, a standardized format has been specified to enable the compilation of data from multiple investigators and locations. Various formats and coding schemes may be equally effective and could be used in individual studies or by agreement of the collaborating investigators. However, recommendations for variable names and database structure are available at the websites of the International Spinal Cord Society (ISCoS) (www.iscos.org.uk), and the National Institute of Neurological Disorders and Stroke (NINDS) the Common Data Elements Project website (www.CommonDataElements.ninds.nih.gov).10, 11 The International SCI Musculoskeletal Basic Data Set was produced under the auspices of ISCoS and the American Spinal Injury Association (ASIA).
Materials and methods
An initial version of the International SCI Musculoskeletal Basic Data Set was prepared by a working group consisting of the authors. All members of this group have clinical and research experience related to the topic of the data set. Attention was directed at ensuring the Data Set was basic and could be used by clinicians in various settings and countries at minimal cost or need of advanced technical equipment. The data collected would provide the minimal amount of standardized information necessary for a basic musculoskeletal evaluation of a person with SCI. To ensure consistency in the data collection and to facilitate interpretation, detailed information is provided in a syllabus for each specific variable and each response category.
The process for developing this version of the International SCI Musculoskeletal Basic Data Set followed the steps given below:
-
1
The working group of the International SCI Musculoskeletal Basic Data Set initially met over a 2-day workshop on 10–11th May 2010 in Copenhagen, Denmark.
-
2
Afterwards, the first draft of the data set was finalized during extensive E-mail contact among group members.
-
3
The data set was reviewed by members of the Executive Committee of the International SCI Standards and Data Sets.
-
4
Comments from the Committee members were discussed in the working group and appropriate adjustments made to the Data Set.
-
5
Members of the ISCoS Executive and Scientific Committees and ASIA Board were also asked to review the Data Set.
-
6
Comments from the Committee/Board members were discussed in the working group and a response was made and further adjustments of the Data Set were performed.
-
7
Relevant and interested scientific and professional (International) organizations and societies (around 40) and individuals were also invited to review the Data Set. In addition the Data Set was posted on the ISCoS and ASIA websites for over 2 months to allow comments and suggestions.
-
8
Comments were discussed and responded to by the working group. Where appropriate, adjustments to the Data Set were made.
-
9
To conclude this part of the consultation, members of the ISCoS Executive and Scientific Committees and ASIA Board members received the Data Set for final review and approval.
-
10
The International SCI Musculoskeletal Basic Data Set was then further scrutinized by the team working on the NINDS, Common Data Elements Project, in cooperation with the Executive Committee of the International SCI Standards and Data Sets committees.10, 11
-
11
Finally, the data set was used to collect information on several cases to confirm its function in the clinical setting.
Results
The data sheet is included in the Appendix and the data sheet and syllabus are available on the ISCoS website (www.iscos.org.uk). Listed below are the variables included in the International SCI Musculoskeletal Basic Data Set:
Date of data collection
Collection of data on musculoskeletal conditions may be performed at any time following the spinal cord lesion. It is therefore imperative to collect the date of data collection to compute the time lapse since the initial spinal cord lesion. This will permit the information obtained to be related to other data collected on the same individual at various time points.
Neuro-musculoskeletal history before spinal cord lesion
This variable consists of three parts, documenting: pre-existing congenital deformities of the spine and spinal cord, pre-existing degenerative spine disorders and pre-existing systemic neurodegenerative disorders. The diagnosis and location of any disorders, and the date and type of any previous surgery for congenital deformities or disorder shall be specified. It is important that any neuro-musculoskeletal disorders preceding the onset of the spinal cord lesion are documented, because relevant pre-existing neuromuscular disorders may influence management.
Congenital disorders include malformations or other pathologies of bones, such as infantile idiopathic scoliosis,12, 13 congenital spinal canal stenosis,14 achondroplasia,15, 16 muscles (muscular dystrophy) or neural tissues (syringo-hydromyelia). These also include combined multi system/organ disorders such as myelomenigocele,17, 18 malformations of the craniocervical junction (that is, Arnold Chiari type, congenital stenosis, Klippel-Feil anomalies )13, 19, 20 or tethered cord.21, 22 Sometimes, these conditions are present in early childhood but only become symptomatic during adolescence or advancing age. This may be due to progression or unmasking of the condition.
Degenerative spine disorders are more prevalent with aging. The most common disorders are lumbar23, 24 and cervical25, 26 spinal canal stenosis, spondylosis and degenerative systemic disorders (like diffuse idiopathic skeletal hyperostosis and rheumatoid arthritis). These disorders typically develop slowly with the spinal cord often adapting to extensive morphological changes before notable impairment. The onset of impairments is usually slow and insidious and therefore not noticed by individuals for a considerable time. However, the degenerative changes can increase susceptibility to spinal cord lesions from relatively minor insults, for example, minor falls with mild extension/flexion trauma inducing a central cord syndrome.27
Systemic neurodegenerative disorders comprise conditions like multiple sclerosis, amyotrophic lateral sclerosis and others. They typically occur in adults and are characterized by an acute or chronic progressive course which can eventually present as tetra/paraplegia.
Presence of spasticity/spasms
The presence of spasticity/spasms in the upper and lower extremities is captured using the modified Ashworth scale,28 that is, at or above one on the modified Ashworth scale, or observation of spasms. A spinal cord lesion leaves about half of all individuals with involuntary spasms (muscle jerks), altered motor control and/or spasticity.29 This altered control can be expressed in a variety of ways.30 The common definition of spasticity is based on the finding of increased resistance to passive stretch. Although modified Ashworth scale captures only a few aspects of a rather multidimensional and phenomenologically diverse symptomatology, the clinically important key elements are measured.31
Abnormal motor control is manifested as negative signs (paresis or paralysis) or positive signs, often termed as ‘spasticity’. This is a broader definition than focusing on exaggerated responses to passive movement, specifically velocity-dependent responses, which are present only about a third of the time in individuals with ‘spastic’ SCI.32 More commonly, the positive signs frequently and prominently include loss of coordination of voluntary movement and spasms, or involuntary movement. One way of characterizing the multidimensional nature of ‘spasticity’ is with a battery of tests, but additional validation of these tests is required before advocating their widespread use.33 (Modified) Ashworth28, 34 and Tardieu scales35 have proven useful to some extent, but rely on subjective evaluation of specific characteristics of spasticity and thus are limited in their applicability and are population dependent. They are commonly used for research purposes.36 It is noted that there may be mechanical changes in muscle fiber, collagen tissue and tendon properties secondary to ‘spasticity’,37 which may confound the assessment.38
Treatment for spasticity within the last 4 weeks
This variable documents if the person with SCI has received any kind of treatment for spasticity within the last 4 weeks. A ‘yes’ is indicated if any kind of treatment has been used for spasticity over the last 4 weeks regardless of whether it was prescribed or not. This may include physical, pharmacological, surgical or other. Four weeks has been chosen to give recent status.
Fracture(s) since the spinal cord lesion
This variable documents whether the person with SCI has had any type of fracture since the spinal cord lesion, the date the fracture occurred, and whether or not the fracture was a fragility fracture. The detailed location is specified as seen in the Appendix. The date of the fracture should be recorded, but if the precise date is unavailable, the month and/or year should be recorded and the date left blank. Only fractures not previously documented need to be recorded. Therefore, if information for the data set is being collected for the first time after SCI, all previous fractures since SCI should be recorded. Thereafter, only fractures that have occurred since the last recording of information for the data set should be recorded.
It is important to distinguish between incident and fragility fractures. Fragility fractures result from low force injuries insufficient to fracture normal bone.39, 40 Common etiologies of fragility fractures after SCI include leg torsion during transfers or rolling in bed, or falling to the floor from a wheelchair or commode on a flexed knee. Compression fractures of vertebral bodies should be considered as fragility fractures, in the absence of reported trauma. In comparison, incident fractures are caused by injuries sufficient to fracture normal bone (that is, motor vehicle accident).
Heterotopic Ossification
This variable documents the diagnosis and location of Heterotopic Ossification (HO) (see Appendix). It is a diagnosis based on signs and symptoms and confirmed with positive imaging. Method used to document HO is specified as X-ray, CT scan, triple phase bone scan, or other specified method.
HO refers to the abnormal formation of bone in soft tissues typically around joints such as the hips, knees, shoulders and elbows. The initial signs and symptoms are often related to inflammation with swelling, restricted range of motion, hyperemia, and, if perceived, pain. HO is associated with elevated serum alkaline phosphatase and confirmed with plain X-ray or CT indicating detectable calcified bone formation or triple phase bone scan before calcification. These are reliable and sensitive indicators of the formation process. HO can also be confirmed with MRI and ultrasound although these are less commonly used.
The incidence of HO varies in SCI populations from 10 to 53%.41 It commonly develops within the first 2–3 weeks after SCI and is most common at the hip (70–97%) and knee.42, 43 Ultrasound can be used as a screening tool if there is a high index of suspicion, but should then be confirmed by one of the tests listed above.44
Contracture
This variable documents the presence of joint contracture(s) at various locations (see Appendix). Contractures are a common complication of SCI45, 46, 47, 48 and are characterized by a loss in passive joint range of motion.49, 50 It is important to identify contractures to implement appropriate treatments and monitor change.51 Passive joint range of motion can be measured quantitatively with a goniometer52 however, for the purposes of this data set the committee recommends the use of visual and physical assessment to determine the loss in joint range of motion. Only obvious loss in joint range of motion that can be readily seen or easily felt should be recorded as a contracture. More subtle loss in joint range of motion should only be recorded as a contracture if it warrants intervention or has clear and marked deleterious implications on function, hygiene, skin management or any other aspect of quality of life.53 For example, subtle loss of passive elbow extension in a person with C6 tetraplegia would be recorded as a contracture if it clearly prevents the person from transferring. An equivalent subtle loss of passive elbow extension would not be recorded as a contracture in a person with C4 tetraplegia if it had no obvious and marked deleterious implications.
The location of degenerative neuromuscular and skeletal changes due to overuse after SCI
The location is to be given as indicated in the Appendix. This variable requires the assessor to distinguish musculoskeletal changes induced by overuse from independently occurring neuropathic and visceral pain. The most common symptoms of overuse are pain and discomfort. Overuse injuries commonly occur at the musculotendinous junction but can also occur at the cartilage, bone and bursa.54 Shoulder pain is present in approximately 30–70% of persons with SCI. Its severity and presence is influenced by age, duration of injury, neurological level (more in people with tetraplegics), severity of injury, wheelchair use, sitting posture, flexibility, stability of the shoulder joint and overall body mass index. Shoulder pain is more common in people with SCI of older age, and for women.55, 56, 57 Manual wheelchair users often experience pain that limits activities of daily living (ADL) such as transfers, propulsion and overhead reaching. Shoulder pain may be due to propelling a wheelchair over many years.58 It is however also seen in individuals heavily reliant on crutches or canes to ambulate.59 Overuse injuries of the elbow tend to result in muscle/tendon strains or nerve entrapments.60 Wrist overuse injuries often lead to carpal tunnel syndrome. Lower extremities may also be affected in individuals with SCI. Knee problems have been described, due to, for example, trauma, and tears of ligaments.61 In case, the degenerative neuromuscular or skeletal changes due to overuse in the neck, upper or lower back is located in the midline without lateralization both right and left is to be marked.
SCI-related neuromuscular scoliosis
This variable documents any appreciable observable lateral deviation in the normally straight vertical line of the spine due to the sequelae of SCI, that is, if there is an observable deviation of the head over the trunk and pelvis during unsupported sitting or standing due to scoliosis as a co-morbidity of SCI. If the head is aligned over the pelvis during unsupported sitting (not balanced with arms/hands; lateral supports; chest straps; and so on) or while standing erect then there is no scoliosis. This variable does not include problems with the alignment of the spine due to problems other than SCI, such as idiopathic scoliosis, pre-injury neuromuscular scoliosis, as in Marfan’s Syndrome, Larson’s Syndrome, Down’s Syndrome, Klippel Feil Syndrome, degenerative disk disease, osteoporosis in the aging spine, and so on.
There is a wide range of normal variation in sagittal profiles and it is possible that each individual has specific requirements for cervical/lumbar lordosis and thoracic kyphosis as a result of pelvic orientation. Scoliosis is defined as a 10 degree curvature of the spine.62 Scoliosis is a known musculoskeletal complication of SCI, particularly when SCI occurs at a younger age.63, 64, 65, 66, 67, 68, 69
Method of assessment, when a scoliosis is present
This variable indicates the method(s) used to determine the presence of neuromuscular scoliosis, for example, observation in sitting; observation in standing; plain radiographs in sitting; or plain radiographs in standing.
The clinical and physical examination is pivotal to the diagnosis of scoliosis and is evidenced by the observed lateral deviation of the head, trunk and pelvis over the spine and shoulder asymmetry. For the evaluation of neuromuscular scoliosis, it is important to temporarily remove any modification to a wheelchair or seating system that is providing external support to maintain head and spine alignment (for example, lateral supports, chest harness).69, 70, 71 Likewise, any type of support to the trunk in the form of a brace or binder needs to be removed to determine if scoliosis is present.
A diagnosis of scoliosis requires a plain radiograph that shows a Cobb Angle of at least 10 degrees.62, 72 While the Cobb Angle of 10 degrees is used as the definitive diagnoses for idiopathic scoliosis, there is evidence that strong inter-rater reliability of the Cobb Angle in SCI also falls within 10 degrees and hence has been adopted as the radiographic diagnosis of neuromuscular scoliosis.73
Surgical treatment of scoliosis
This variable documents if and when scoliosis has been surgically treated. Nearly, all children injured with SCI before reaching skeletal maturity will develop scoliosis and 75% will require some type of surgical intervention to stop the progression of the curve.63, 64 When the SCI occurs in adolescence, 78% of children injured at 14 years, 57% of children injured at 15 years, and 50% of children injured at 16 years develop scoliosis that requires either conservative treatment (modifications to wheelchair, bracing) or surgical intervention (spinal fusion).63, 68
Other musculoskeletal problems
This variable documents the presence of any other specified musculoskeletal problems not described above. This variable requires the assessor to specify any other type of musculoskeletal problem not captured in the other variables. This could among other issues include gibbus formation in relation to Pott’s paraplegia.74, 75
Do any of the musculoskeletal challenges above interfere with your activities of daily living?
This variable documents if any of the musculoskeletal challenges above interfere with daily activities, such as transfers, walking, dressing, showers, and so on. and is recorded as No, not at all; Yes, a little; Yes, a lot. This variable requires the assessor to directly ask the individual with a spinal cord lesion the following question ‘Do any of the musculoskeletal challenges above interfere with your activities of daily living (transfers, walking, dressing, showers, and so on)?’ This variable captures the individual’s perceptions about the deleterious implications of any of the musculoskeletal challenges above on daily life. The perspective of the individual is important and this variable enables individuals to focus on the impact of musculoskeletal changes on activities relevant to them, whether it is due to spasticity,76 heterotopic ossification, contracture(s), neuromuscular or skeletal overuse, scoliosis68, 69 or other musculoskeletal problems.
Discussion
The data collected in the International SCI Musculoskeletal Basic Data Set will be available in conjunction with the data in the International SCI Core Data Set, which among other items, includes information on date of birth and injury, gender, the cause of SCI and neurological status.9 To make this basic data set as useful as possible in a clinical setting, we have kept the number of items to a minimum. The working group believes that the items included cover the most clinically relevant information about possible musculoskeletal conditions in individuals with a spinal cord lesion. The working group recognizes that information in the International SCI Musculoskeletal Basic Data Set could be extended by other clinically important information, whenever appropriate.
To facilitate the use of the International SCI Data Sets, this International SCI Musculoskeletal Basic Data Set and its data collection (form included in the Appendix) have been developed in a similar format to the previous International SCI Basic Data Sets. Additional work and research is now required to validate and translate this data set into use. In this respect, it is additionally advised to adhere to the recommendations given by the Executive Committee for the International SCI Standards and Data Sets.77 The authors invite all those who are interested to participate in this open and ongoing process.
References
Hitzig SL, Eng JJ, Miller WC, Sakakibara BM . SCIRE Research Team. An evidence-based review of aging of the body systems following spinal cord injury. Spinal Cord 2011; 49: 684–701.
Ballinger DA, Rintala DH, Hart KA . The relation of shoulder pain and range-of-motion problems to functional limitations, disability, and perceived health of men with spinal cord injury: A multifaceted longitudinal study. Arch Phys Med Rehabil 2000; 81: 1575–1581.
Weitzenkamp DA, Jones RH, Whiteneck GG, Young DA . Aging with spinal cord injury: Cross-sectional and longitudinal effects. Spinal Cord 2001; 39: 201–309.
Biering-Sørensen F, Bohr HH, Schaadt OP . Longitudinal study of bone mineral content in the lumbar spine, the forearm and the lower extremities after spinal cord injury. Eur J Clin Invest 1990; 20: 330–335.
Garland DE, Stewart CA, Adkins RH, Hu SS, Rosen C, Liotta FJ et al. Osteoporosis after spinal cord injury. J Orthop Res 1992; 10: 371–378.
Vestergaard P, Krogh K, Rejnmark L, Mosekilde L . Fracture rates and risk factors for fractures in patients with spinal cord injury. Spinal Cord 1998; 36: 790–796.
Sköld C, Levi R, Seiger A . Spasticity after traumatic spinal cord injury: nature, severity, and location. Arch Phys Med Rehabil 1999; 80: 1548–1557.
Biering-Sørensen F, Charlifue S, DeVivo M, Noonan V, Post M, Stripling T et al. International Spinal Cord Injury Data Sets. Spinal Cord 2006; 44: 530–534.
DeVivo M, Biering-Sørensen F, Charlifue S, Noonan V, Post M, Stripling T et al. International Spinal Cord Injury Core Data Set. Spinal Cord 2006; 44: 535–540.
Biering-Sørensen F, Charlifue S, Devivo MJ, Grinnon ST, Kleitman N, Lu Y et al. Incorporation of the International Spinal Cord Injury Data Set elements into the National Institute of Neurological Disorders and Stroke Common Data Elements. Spinal Cord 2010; 49: 60–64.
Biering-Sørensen F, Charlifue S, Devivo MJ, Grinnon ST, Kleitman N, Lu Y et al. Using the spinal cord injury common data elements. Top Spinal Cord Inj Rehabil 2012; 18: 23–27.
Dobbs MB, Lenke LG, Szymanski DA, Morcuende JA, Weinstein SL, Bridwell KH et al. Prevalence of neural axis abnormalities in patients with infantile idiopathic scoliosis. J Bone Joint Surgery Am 2002; 84-A: 2230–2234.
Pahys JM, Samdani AF, Betz RR . Intraspinal anomalies in infantile idiopathic scoliosis: prevalence and role of magnetic resonance imaging. Spine (Phila Pa 1976) 2009; 34: E434–E438.
Kotil K, Kalayci M, Bilge T . Management of cercicomedullary compression in patients with congenital and acquired osseous-ligamentous pathologies. J Clin Neurosci 2007; 14: 540–549.
Laederich MB, Horton WA . Achondroplasia: pathogenesis and implications for future treatment. Curr Opin Pediatr 2010; 22: 516–523.
Benglis DM, Sandberg DI . Acute neurological deficit after minor trauma in an infant with achondroplasia and cervicomedullary compression. Case report and review of the literature. J Neurosurg 2007; 107: 152–155.
Guille JT, Sarwark JF, Sherk HH, Kumar SJ . Congenital and developmental deformities of the spine in children with myelomeningocele. J Am Acad Orthop Surg 2006; 14: 294–302.
Rajpal S, Tubbs RS, George T, Oakes WJ, Fuchs HE, Hadley MN et al. Tethered cord due to spina bifida occulta presenting in adulthood: a tricenter review of 61 patients. J Neurosurg Spine 2007; 6: 210–215.
Fernández AA, Guerrero AI, Martinez MI, Vázquez ME, Fernández JB, Chesa i Octavio E et al. Malformations of the craniocervical junction (Chiari type I and syringomyelia: classification, diagnosis and treatment. BMC Musculoskelet Disord 2009; 10: S1.
Nakamura M, Ishii K, Watanabe K, Tsuji T, Matsumoto M, Toyama Y et al. Clinical significance and prognosis of idiopathic syringomyelia. J Spinal Disord Tech 2009; 22: 372–375.
Kramer JL, Dvorak M, Curt A . Thoracic disc herniation in a patient with tethered cord and lumbar syringomyelia and diastematomyelia: magnetic resonance imaging and neurophysiological findings. Spine (Phila Pa 1976) 2009; 34: E484–E487.
Iskandar BJ, Fulmer BB, Hadely MN, Oakes WJ . Congenital tethered spinal cord syndrome in adults. Neurosurg Focus 2001; 10: e7.
Abbas J, Hamoud K, Masharawi YM, May H, Hay O, Medlej B et al. Ligamentum flavus thickness in normal and stenotic lumbar spines. Spine (Phila Pa 1976) 2010; 35: 1225–1230.
Yasar B, Simsek S, Er U, Yigitkanli K, Eksioglu E, Altug T et al. Functional and clinical evaluation for the surgical treatment of degenerative stenosis of the lubar spinal canal. J Neurosurg Spine 2009; 11: 347–352.
Tracy JA, Bartleson JD . Cervical spondylotic myelopathy. Neurologist 2010; 16: 176–187.
Fehlings MG, Arvin B . Surgical management of cervical degenerative disease: the evidence related to indications, impact and outcome. J Neurosurg Spine 2009; 11: 97–100.
Matz PG, Anderson PA, Holly LT, Groff MW, Heary RF, Kaiser MG et al. The natural history of cercical spondylotic myelopathy. J Neurosurg Spine 2009; 11: 104–111.
Bohannon RW, Smith MB . Interrater reliability of a Modified Ashworth Scale of muscle spasticity. Phys Ther 1987; 67: 206–207.
Hsieh JTC, Wolfe DL, Miller WC, Curt A and the SCIRE Research Team. Spasticity outcome measures in spinal cord injury: psychometric properties and clinical utility. Spinal Cord 2008; 46: 86–95.
Priebe MM, Sherwood AM, Thornby JI, Kharas NF, Markowski J . Clinical assessment of spasticity in spinal cord injury: a multidimensional problem. Arch Phys Med Rehabil 1996; 77: 713–716.
Pandyan AD, Gregoric M, Barnes MP, Wood D, van Wijck F, Burridge J et al. Spasticity: clinical perceptions, neurological realities and meaningful measurement. Disabil Rehabil 2005; 27: 2–6.
Lance JW . Symposium synopsis. In: Feldman RG, Young RR, Koella WP (eds). Spasticity: Disordered Motor Control. Yearbook Medical: Chicago, IL. 1980 pp 17–24.
Sherwood AM, McKay WB . Assessment of spasticity and upper motor neuron dysfunction. In:. Wiley Encyclopedia of Biomedical Engineering, Metin Akay (ed). John Wiley & Sons, Inc., Hoboken, NJ, USA, Vol. 5, 2006 pp 3306–3315.
Ashworth B . Preliminary trial of carisoprodol in multiple sclerosis. Practitioner 1964; 192: 540–542.
Tardieu GA . La recherche d'une technique de mesure de la spasticitė ‘imprime’ avec le periodique. Rev Neurol 1954; 91: 143–144.
Biering-Sørensen F, Nielsen JB, Klinge K . Spasticity-assessment: a review. Spinal Cord. 2006; 44: 708–722.
Dietz V, Sinkjaer T . Spastic movement disorder: impaired reflex function and altered muscle mechanics. Lancet Neurol 2007; 6: 725–733.
Sherwood AM, Graves DE, Priebe MM . Altered motor control and spasticity after spinal cord injury: subjective and objective assessment. J Rehabil Res Dev 2000; 37: 41–52.
Jiang SD, Dai LY, Jiang LS . Osteoporosis after spinal cord injury. Osteoporos Int 2006; 17: 180–192.
Craven BC, Robertson LA, McGillivray CF, Adachi JD . Detection and treatment of sublesional osteoporosis among patients with chronic spinal cord injury: proposed paradigms. Top Spinal Cord Rehabil 2009; 14: 1–22.
Stover SL, Hataway CJ, Zeiger HE . Heterotopic ossification in spinal cord injured patients. Arch Phys Med Rehabil 1975; 56: 199–204.
van Kuijk AA, Geurts AC, van Kuppevelt HJ . Neurogenic heterotopic ossification in spinal cord injury. Spinal Cord 2002; 40: 313–326.
Banovac K, Williams JM, Patrick LD, Haniff YM . Prevention of heterotopic ossification after spinal cord injury with indomethacin. Spinal Cord 2001; 39: 370–374.
Citak M, Backhaus M, Kälicke T, Ucher I, Aach M, Meindl R et al. Treatment of heterotopic ossification after spinal cord injury – clinical outcome after single dose radiation therapy. Z Orthop Unfall 2011; 149: 90–93.
Fergusson D, Hutton B, Drodge A . The epidemiology of major joint contractures: a systematic review of the literature. Clin Orthop Relat Res 2006; 14: 22–29.
Vogel L, Krajci K, Anderson C . Adults with pediatric-onset spinal cord injury: part 2: musculoskeletal and neurological complications. J Spinal Cord Med 2002; 25: 117–123.
Eriks-Hoogland I, de Groot S, Post M, van der Woude L . Passive shoulder range of motion in people with spinal cord injury during and one year after rehabilitation. In:. Proceedings on the International Spinal Cord Society Annual Scientific Meeting, 2008 Durban:58.
Diong J, Harvey LA, Kwah LK, Eyles J, Ling MJ, Ben M et al. Incidence and prediction of contracture after spinal cord injury: a prospective cohort study. Spinal Cord 2012; 50: 579–584.
Farmer SE, James M . Contractures in orthopaedic and neurological conditions: a review of causes and treatment. Disabil Rehabil 2001; 23: 549–558.
Lieber R . Skeletal Muscle Structure, Function, and Plasticity. Lippincott Williams & Wilk, Philadelphia, USA. 2009.
Harvey LA, Herbert RD . Muscle stretching for treatment and prevention of contracture in people with spinal cord injury. Spinal Cord 2002; 40: 1–9.
van de Pol RJ, van Trijffel E, Lucas C . Inter-rater reliability for measurement of passive physiological range of motion of upper extremity joints is better if instruments are used: a systematic review. Aust J Physiother 2010; 56: 7–17.
Center NSCIS . The Model Spinal Cord Injury Systems′ Data Collection Syllabus for the National Spinal Cord Injury Database. University of Alabama at Birmingham: Birmingham, AL. 1990, 110–230.
Apple D, Cody R, Allen A . Overuse syndrome of the upper limb in people with spinal cord injury. In: Apple DF (ed) Physical Fitness: a guide for individuals with spinal cord injury. J Rehabil Res Dev 1996, 97–108 Chapter 5.
Dyson-Hudson T, Kirshblum S . Shoulder pain in chronic spinal cord injury, Part I: epidemiology, etiology and pathomechanics. J Spinal Cord Inj Med 2004; 27: 4–17.
Lal S . Premature degenerative shoulder changes in spinal cord injury patients. Spinal Cord 1998; 36: 186–189.
van Drongelen S, de Groot S, Veeger HE, Angenot EL, Dallmeijer AJ, Post MW et al. Upper extremity musculoskeletal pain during and after rehabilitation in wheelchair-using persons with a spinal cord injury. Spinal Cord 2006; 44: 152–159.
Pentland WE, Twomey LT . Upper limb function in persons with long term paraplegia and implications for independence: Part I. Paraplegia 1994; 32: 211–218.
Jain NB, Higgins LD, Katz JN, Garshick E . Association of shoulder pain with the use of mobiity devices in persons with chronic spinal cord injury. PM R 2010; 2: 896–900.
Boninger M, Cooper R, Fay B . Musculoskeletal pain and overuse injuries. In: Lin V, Cardinas DD, Koontz A (eds). Spinal Cord Medicine: Principles and Practice 2003, 527–534.
Mukand J, Sniger W, Kaufman J, Biener-Bergman S . Common causes of knee efusions in spinal cord injury. A random study. Am J Phys Med Rehabil 1998; 77: 113–117.
Terminology committee of the Scoliosis Research Society: A glossary of scoliosis terms. Spine 1976; 1: 57–58.
Brown JC, Swank SM, Matta J, Barras DM . Late spinal deformity in quadriplegic children. J Pediatr Orthop 1984; 4: 456–461.
Campbell J, Bonnett C . Spinal cord injury in children. Clin Orthop 1975; 112: 114–123.
Dearolf WW, Betz RR, Vogel LC et al. Scoliosis is pediatric spinal-cord injured patients. J Pediatr Orthop 1990; 10: 214–218.
Lancourt JE, Dickson JH, Carter RE . Paralytic spinal deformity following traumatic spinal cord injury in children and adolescents. J Bone Joint Surg Am 1981; 63: 47–53.
Mayfield JK, Erkkila JC, Winter RB . Spine deformity subsequent to acquired childhood spinal cord injury. J Bone Joint Surgery Am 1981; 63: 1401–1411.
Vogel LC, Betz RR, Mulcahey MJ . Pediatric spinal cord disorders in children and adolescents. In: Lin V (ed).. Spinal Cord Medicine Textbook. Demos: New York. 2003, 851–884.
Lubicky J, Betz R . Spinal deformity in children and adolescents with spinal cord injury. In: Betz RR, Mulcahey MJ (eds). The Child with Spinal Cord injury. American Academy of Orthopedic Surgeons: Rosemont, IL. 1996 pp 363–370 Chapter 32.
Lord J, Behrman B, Varzos N, Cooper D, Lieberman JS, Fowler WM . Scoliosis associated with Duchenne muscular dystrophy. Arch Phys Med Rehabil 1990; 71: 13–17.
Mulcahey MJ, Betz RR . Pediatric spinal cord injury rehabilitation, Ch 66. In:. The Pediatric Spine 3rd edn. Thieme: New York. 2008.
O’Brien MF . Spinal Deformity Study Group Radiographic Measurement Manual. Medtronic Sofamor Danek USA, Inc, Memphis, TN, USA. 2005.
Gupta MC, Wijesekera S, Sossan A, Martin L, Vogel LC, Boakes JL et al. Reliability of radiographic parameters in neuromuscular scoliosis. Spine 2007; 32: 691–695.
Benzagmout M, Boujraf S, Chakour K, Chaoui Mel F . Pott's disease in children. Surg Neurol Int 2011; 2: 1.
Moon MS, Moon JL, Moon YW, Kim SS, Kim SS, Sun DH et al. Pott's paraplegia in patients with severely deformed dorsal or dorsolumbar spines: treatment and prognosis. Spinal Cord 2003; 41: 164–171.
Lechner HE, Frotzler A, Eser P . Relationship between self- and clinically rated spasticity in spinal cord injury. Arch Phys Med Rehabil 2006; 87: 15–19.
Biering-Sørensen F, Alexander MS, Burns S, Charlifue S, Devivo M, Dietz V et al. Recommendations for translation and reliability testing of international spinal cord injury data sets. Spinal Cord 2011; 49: 357–360.
Acknowledgements
Medtronic A/S, Denmark has supported the development of this Data Set with an unconditional grant. We are thankful for comments and suggestions received from Lawrence Vogel, Susan Charlifue, Vanessa Noonan, Volker Dietz, Marcel Post, Govert J Snoek, Inge Eriks Hoogland, Giorgio Scivoletto, Douglas J Brown, Vivien Jørgensen, Anne Lannem, Brooke Wadsworth, Martha Somers, Michael G Fehlings, Jon Hassler, and the National Institute of Neurological Disorders and Stroke (NINDS) the Common Data Elements (CDE) Project Team.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Appendix
Appendix
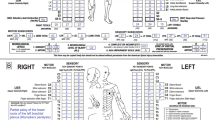
International spinal cord injury musculoskeletal basic data set form (Version 1.0)


Rights and permissions
About this article
Cite this article
Biering-Sørensen, F., Burns, A., Curt, A. et al. International spinal cord injury musculoskeletal basic data set. Spinal Cord 50, 797–802 (2012). https://doi.org/10.1038/sc.2012.102
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2012.102
Keywords
This article is cited by
-
Corticospinal Motor Circuit Plasticity After Spinal Cord Injury: Harnessing Neuroplasticity to Improve Functional Outcomes
Molecular Neurobiology (2021)
-
Scoliosis in paediatric onset spinal cord injuries
Spinal Cord (2020)
-
The data set development for the National Spinal Cord Injury Registry of Iran (NSCIR-IR): progress toward improving the quality of care
Spinal Cord Series and Cases (2020)
-
The International Spinal Cord Injury Pediatric Activity and Participation Basic Data Set
Spinal Cord Series and Cases (2019)
-
Electronic medical record: data collection and reporting for spinal cord injury
Spinal Cord Series and Cases (2018)