Abstract
Study design:
Multi-center pilot study.
Objectives:
To investigate the use of an upper limb robotic rehabilitation device (Armeo Spring, Hocoma AG, Switzerland) in a subacute cervical spinal cord injury (SCI) population.
Setting:
Two Canadian inpatient rehabilitation centers.
Methods:
Twelve subjects (motor level C4–C6, ASIA Impairment Scale A–D) completed the training, which consisted of 16.1±4.6 sessions over 5.2±1.4 weeks. Two types of outcomes were recorded: (1) feasibility of incorporating the device into an inpatient rehabilitation program (compliance with training schedule, reduction in therapist time required and subject questionnaires) and (2) efficacy of the robotic rehabilitation for improving functional outcomes (Graded and Redefined Assessment of Strength, Sensibility and Prehension (GRASSP), action research arm test, grip dynamometry and range of motion).
Results:
By the end of the training period, the robot-assisted training was shown to require active therapist involvement for 25±11% (mean±s.d.) of the total session time. In the group of all subjects and in a subgroup composed of motor-incomplete subjects, no statistically significant differences were found between intervention and control limbs for any of the outcome measures. In a subgroup of subjects with partial hand function at baseline, the GRASSP-Sensibility component showed a statistically significant increase (6.0±1.6 (mean±s.e.m.) point increase between baseline and discharge for the intervention limbs versus 1.9±0.9 points for the control limbs).
Conclusion:
The pilot results suggest that individuals with some preserved hand function after SCI may be better candidates for rehabilitation training using the Armeo Spring device.
Similar content being viewed by others
Introduction
Activity-based rehabilitation is currently one of the principal strategies to promote functional recovery after a neurological injury, such as spinal cord injury (SCI) or stroke. In recent years, robotic devices have been proposed to facilitate or supplement rehabilitation. Examples for the upper limbs include: the MIT-Manus,1 the MIME,2 the ARM guide,3 the Bi-Manu-Track,4 the GENTLE/s,5 the T-WREX6, 7 and the ARMin.8
The main motivation for robotic rehabilitation devices is to assist in the repetitive labor-intensive manual therapy delivered by therapists as they move a patient's limbs during exercises. Without creating additional time demands on the therapists, robotic devices could perform the repetitive mechanical aspects of therapy, thereby increasing the amount of therapy for each patient and/or increasing the number of patients undergoing therapy simultaneously.9, 10, 11 Virtual reality (VR) interfaces may also increase patient engagement. By delivering more rehabilitation and increasing patient motivation, robotic rehabilitation might improve functional outcomes,2, 7, 8, 12 although current evidence from stroke patients suggests that improvements are because of the intensity of the therapy, regardless of delivery by robotic or conventional means.13, 14
Given the need for novel upper limb therapies after cervical SCI,15 the evaluation of robotic rehabilitation in this population is warranted. But to the best of the authors’ knowledge, no studies have examined the use of upper limb rehabilitation robotics in a subacute SCI population. We therefore present a multi-center pilot evaluation of a commercial upper limb rehabilitation device, the Armeo Spring (Hocoma AG, Switzerland). Our goals were to establish the feasibility of using the Armeo Spring in an SCI inpatient setting as well as to gather preliminary data on the device's efficacy. The Armeo Spring and its academic predecessor the T-WREX have previously been used with stroke survivors6, 7 and in a population with multiple sclerosis,16 but its usefulness in patients with cervical SCI is not known. Portions of these results have been presented in conference format.17
Methods
Study design
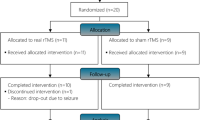
A total of 15 subjects with subacute cervical SCI were recruited from the inpatient populations at two rehabilitation centers. The inclusion criteria were to have sustained a traumatic cervical SCI with a motor level between C4 and C8 (as defined by the International Standards for the Neurological Classification of Spinal Cord Injury, and evaluated by the clinical staff). Subjects with a history of neuromuscular disease, severe upper limb spasticity, severe shoulder pain, unable to sit upright for 30 min or unable to understand and follow instructions were excluded. Subject demographics are provided in Table 1.
Figure 1 shows the Armeo Spring. The user's arm is placed into an exoskeleton that provides adjustable antigravity weight support for the arm through a system of springs (no actuators). The orthosis allows movement along five degrees of freedom: three degrees of freedom at the shoulder to allow shoulder flexion–extension, abduction–adduction and internal/external rotation, one degrees of freedom at the elbow to allow flexion–extension and one degrees of freedom at the radioulnar joint to allow pronation–supination of the wrist. The orthosis also includes a handgrip pressure sensor. Exoskeleton movements are relayed to a computer and control the cursor during a number of different VR tasks. The range of motion (ROM) needed to control the VR tasks is adjustable. The tasks train different aspects of upper limb function, including reaching movements in various directions, pronation/supination at the radio–ulnar joint and handgrip strength.
Subjects were scheduled for three to five 1-hour training sessions per week for 6 weeks. Compliance with the training schedule was one of the outcome measures of the study. Training was unilateral. The choice of which arm to train was made in consultation with each subject and in accordance with their rehabilitation goals. Both the arms continued to receive conventional, occupational and physical therapy exercises as per the standard of care in the rehabilitation centers. The arms not receiving robotic rehabilitation served as the control group for the efficacy outcomes (see below). The antigravity weight support, and the type and difficulty of VR activities were tailored to each subject's capabilities at the initial session and adjusted throughout the training to ensure that the exercises remained challenging. The adjustments were made by the investigators delivering the training sessions (PT and MAM) and primarily took the form of increasing the level of difficulty of the games, the number of repetitions and introducing more difficult games into the training program when subjects recovered sufficient function. The study protocol was approved by the research ethics committee at both sites.
Feasibility outcomes
Feasibility outcomes focused on integrating robotic training into the rehabilitation program of SCI inpatients, given that the number of activities that patients can be involved in is limited by scheduling consideration both for the clinical staff and for the subjects themselves.
Compliance
The total number of sessions attended, total length of training (in weeks) and average number of sessions per week were tallied for each subject.
Therapist time
Although each training session was fully supervised, the investigator delivering the training recorded the amount of time that was spent actively assisting the subject (that is, assisting with inserting and removing the arm from the device, inputting commands to the computer and adjusting the settings and so on), as opposed to monitoring the training and guiding the subject's activities verbally. The ratio of active assistance time to the total length of the session was used to measure direct therapist involvement.
Subject questionnaires
At the end of the training period, the subjects provided a feedback about their experience with the Armeo Spring in the form of a questionnaire (see Table 3). The questionnaire used a 7-point Likert scale (1: ‘Disagree strongly’, 7: ‘Agree strongly’).
Efficacy outcomes
These outcomes collected data on the efficacy of the Armeo Spring in improving the upper limb function. Assessments were conducted before training (baseline), at the end of the training block (discharge), 2 weeks after the end of the training (2-week follow-up) and 6 weeks after the end of the training (6-week follow-up). Both of the subject's arms were assessed, with the group of limbs receiving conventional rehabilitation, but no robotic rehabilitation, serving as the control for the group of limbs receiving both conventional and robotic rehabilitation (interventional group).
Graded and redefined assessment of strength, sensibility and prehension (GRASSP)
The GRASSP is a clinical measure of upper limb impairment after tetraplegia.18 It includes manual muscle testing of 10 upper limb muscles (Strength), sensory testing of the fingertips using monofilaments (Sensibility), ability to form different types of grasps (Qualitative Prehension) and performance on a set of functional tasks (quantitative prehension).
Action research arm test
The action research arm test uses a series of functional tasks to evaluate grasp, grip and pinch function, as well as gross movement of the upper limb.19
Dynamometry
The maximal grip strength for each hand was measured using a Baseline hand dynamometer (Fabrication Enterprises Inc., White Plains, NY, USA). The average of three trials was used for each assessment.
Three analyses of the outcomes were performed. The first analysis included all subjects (population of intervention arms compared with population of control arms). The second analysis used only the subgroup of limbs with some hand function at baseline defined as baseline-manual muscle testing scores of two or more in at least one of the six hand function muscles assessed in the GRASSP test (extensor digitorum, opponens pollicis, flexor pollicis longus, flexor digitorum profundus at the middle finger, finger abductors at the little finger and first dorsal interossei). This determination was made separately for the control and intervention limbs (that is, a given subject can have either both or neither limbs included in this analysis, depending on the level of function in each of the two hands). This subgroup was motivated by the fact that subjects with some hand function were able to use the grip module on the Armeo Spring to access a greater number of training exercises. The third analysis included only subjects with motor incomplete injuries at baseline (ASIA Impairment Scale grade C or D).
In addition to standardized assessments, we analyzed the ROM measurements provided by the Armeo Spring. The device requires users to move through their comfortable ROM at the beginning of each session and adjusts the VR workspace accordingly. We additionally required that subjects reach as far as possible in every direction at the beginning of each session and analyzed this ‘maximal’ ROM information. The components of the recorded ROM are the right-to-left range, the close-to-far range (where close is trying to touch one's abdomen and far is straightening the arm forward) and the top-to-bottom range (where bottom is limited by the subject's knees). The total ROM volume is the product of these three values. The mean of the ROM volumes for the first three sessions was used as the baseline ROM and the mean of the last three sessions as the discharge ROM.
The efficacy outcomes and subject questionnaires were collected by the investigators who were not involved in delivering the rehabilitation sessions (JZ and NK).
Results
Of the 15 subjects recruited, 3 dropped out of the study before completing the training. One cited a busy schedule as his reason for dropping out, the second cited a combination of busy schedule and secondary health complications (unrelated to the Armeo training), whereas the third subject cited a lack of interest in the Armeo training.
Of the remaining 12 subjects, 10 chose to train the arm that was dominant before injury. One subject trained the arm that was non-dominant before injury because it was less affected by the injury. One subject trained the non-dominant arm because of pain while moving the dominant arm.
The distribution of the 12 subjects having completed training, between the two centers was 5 at one center and 7 at the other.
The number of limbs resulting from each of the subgroups analyzed is shown in Table 2 for each time point.
Feasibility outcomes
Compliance
The average number of training sessions conducted per subject was 16.1±4.6 (mean±s.d.), and ranged from 9 to 24. Training duration varied from 2.3 to 7.1 weeks with a mean of 5.2±1.4. Subject 5 was discharged earlier than expected and accounts for the minimum in both metrics (nine sessions delivered over 16 days). The average number of sessions per week was 3.2±0.8. The average length of the sessions was 43±10 min and ranged from 20 to 74 min.
Therapist time
The mean ratio of active assistance time to total session length was 0.41±0.27 (mean±s.d.) across all sessions for all subjects. Using only the last three training sessions of each subject, the ratio was 0.25±0.11. In contrast, using the first three training sessions, the ratio was 0.61±0.30. These two values were significantly different (P<0.01 using a one-way analysis of variance). More therapist effort was needed at the beginning of training, while the subject-specific rehabilitation program was developed.
Subject questionnaires
The results of the discharge questionnaire filled out by the subjects are provided in Table 3.
Efficacy outcomes
GRASSP
For the total subject population group (Figure 2), no significant differences were observed for any of the GRASSP test subscores at any time point (Kruskal–Wallis non-parametric analysis of variance, defining statistical significance as P⩽0.05).
For the subgroup with some hand function (Figure 3), significant differences were found between the intervention and control groups in the GRASSP Sensibility subscore; at discharge, the intervention limbs improved by 6.0±1.6 points (mean±s.e.m.) and the control limbs by 1.9±0.9 points (P=0.04); at the 2-week follow-up, the intervention limbs had improved by 10.0±2.0 points and the control limbs by 1.5±0.9 points (P=0.04). The Sensibility score comparison at the 6-week follow-up did not reveal a significant difference between the two groups (P=0.12). None of the other GRASSP subscores were statistically significant for between-group differences at any time point for this subgroup.
For the motor incomplete subgroup, or ASIA Impairment Scale-C and ASIA Impairment Scale-D subjects (Figure 4), no significant differences were found on any subscore, at any time point.
Action research arm test
The changes in action research arm test scores are shown in Figure 5. No statistically significant group differences were found at any time point, for any stratification.
Change in action research arm test score from baseline for the intervention and control limb groups. The top figure shows the data from all subjects, the middle figure shows the data from limbs with partial hand function at baseline and the bottom figure shows the data from subjects with motor incomplete injuries. Error bars reflect the s.e.m.
Grip dynamometry
The changes in the dynamometer readings are shown in Figure 6. No statistically significant group differences were found at any time point, for any stratification.
Change in grip dynamometer readings from baseline for the intervention and control limb groups. The top figure shows the data from all subjects, the middle figure shows the data from arms with partial hand function at baseline and the bottom figure shows the data from subjects with motor incomplete injuries. Error bars reflect the s.e.m.
Range of motion
For each subject, the discharge ROM value was expressed as a percentage of the baseline ROM value. The mean and s.e.m of the discharge ROM were 101.9±14; 7%, compared with 100±0% at baseline (n=12 at both time points), a nonsignificant difference (P=0.89 using a one-way analysis of variance).
Discussion
Feasibility
Our results show that incorporating upper limb robotic training into the rehabilitation program of subacute SCI inpatients is feasible, and that the device allowed more rehabilitation exercises to be performed with progressively less hands-on involvement by the therapist.
In addition to therapist schedules, it is also important to look at patient schedules; additional training time is limited by other rehabilitation activities, subject fatigue, ongoing medical complications and discharge dates. Our target for frequency of training (3–5 sessions/week) was met with 3.2±0.8 sessions per week, though two of the subjects who dropped out of the study cited scheduling as a factor. The duration of training was slightly lower than desired, primarily because of discharge date constraints. The amount of time that any novel intervention requires for making a measurable impact must be compared with limitations on scheduling the necessary sessions and length of stay.
Subjects found the Armeo Spring easy to use and helpful for tracking their progress and reported a moderately beneficial impact on their motivation to perform their exercises. Despite these benefits, subjects did not express any preference for using the Armeo Spring compared with conventional therapy.
Efficacy
Despite the increased therapy hours, this pilot study showed few functional benefits in the limbs receiving Armeo training compared with the limbs not receiving robotic training. Nevertheless, our small sample size prevents us from drawing any definitive conclusions. We observed a statistically significant improvement only in the GRASSP Sensibility scores of subjects with partial hand function at baseline. The control and intervention groups did not have significantly different Sensibility scores at baseline (P=0.12 on a Kruskal–Wallis test) and neither group was subject to ceiling effects for the scale (one subject did reach the maximum Sensibility score of 24 at discharge, but for both limbs). Nevertheless, the Sensibility difference between the two groups was larger at baseline (13.5±2.3 for the intervention group vs 18.8±1.4 for the control group) than at discharge (19.5±1.5 for the intervention group vs 20.7±0.8 for the control group). The observed change in Sensibility score should therefore be confirmed in a larger study to ensure that it was not because of differences between our control and intervention limbs. The Armeo Spring does not directly address tactile sensation, but the training of the subjects with partial hand function generally focused heavily on exercises that include grip function and this emphasis on the hand might have been reflected in the GRASSP Sensibility score, which is based on testing at the fingers only (no upper arm). These observations suggest that individuals able to incorporate grip exercises in their training may be most likely to benefit from Armeo Spring training.
Limitations
The small number of subjects in our pilot study likely resulted in insufficient statistical power to detect changes and may account for the limited functional improvements observed. Stratifying the population not only proved beneficial in identifying the effects and possible trends for robotic training of upper limb function, but also reduced the number of subjects in each comparison. Another possible factor is the amount of robotic rehabilitation delivered (dose and duration), which may have been insufficient or too variable between subjects to produce measurable changes.
Our study did not include an independent control group of subjects undergoing no Armeo training, but an equivalent amount of additional conventional therapy in the intervention arm. This study was a pilot project aiming to establish that this type of therapy can be effectively incorporated into the rehabilitation program of individuals with subacute SCI and to collect preliminary efficacy data that will inform the design of future studies. In this context, the use of the untrained arms as a control group is justified and ensures that the control limbs were matched to the intervention limbs for the amount of conventional therapy received and demographic considerations. Asymmetries in the amount of recovery between the two limbs of a given subject are expected to average out over the population. Note that differences between control and intervention limbs were also examined on a subject-by-subject basis rather than as a population (results not shown), but no additional trends were identified using this method. An additional consideration is that unilateral training is known to produce a small strength increase in the contralateral homologous muscle (‘cross-education’20). In the population investigated here, however, any cross-education occurring in the untrained arm as a result of the Armeo training is likely negligible compared with the combined effects of conventional rehabilitation and spontaneous recovery after SCI.21
Conclusion
We provided the first report on the use of upper limb robotic rehabilitation in a subacute cervical SCI population. The device accomplished its goal of increasing the amount of rehabilitation training while reducing the amount of active therapist time required. This small study did not identify that robotic rehabilitation translated into defined functional benefits, but a larger study is needed before definitive conclusions can be reached. Our results suggest that a more homogeneous subject population should be focused on, particularly subjects with cervical SCI who are able to perform some minimal hand movements.
References
Krebs HI, Hogan N, Aisen ML, Volpe BT . Robot-aided neurorehabilitation. IEEE Trans Rehab Eng 1998; 1: 75–87.
Lum PS, Burgar CG, Shor PC, Majmundar M, Van der Loos M . Robot-assisted movement training compared with conventional therapy techniques for the rehabilitation of upper-limb motor function after stroke. Arch Phys Med Rehabil 2002; 83: 952–959.
Reinkensmeyer DJ, Kahn LE, Averbuch M, McKenna-Cole A, Schmit BD, Rymer WZ . Understanding and treating arm movement impairment after chronic brain injury: progress with the ARM guide. J Rehabil Res Dev 2000; 37: 653–662.
Hesse S, Schulte-Tigges G, Konrad M, Bardeleben A, Werner C . Robot-assisted arm trainer for the passive and active practice of bilateral forearm and wrist movements in hemiparetic subjects. Arch Phys Med Rehabil 2003; 84: 915–920.
Loureiro R, Amirabdollahian F, Topping M, Driesse B, Harwin W . Upper limb robot mediated stroke therapy - GENTLE/s approach. Auton Robots 2003; 15: 35–51.
Sanchez RJ, Liu J, Rao S, Shah P, Smith R, Rahman T et al. Automating arm movement training following severe stroke: functional exercises with quantitative feedback in a gravity-reduced environment. IEEE Trans Neural Sys Rehab Eng 2006; 14: 378–389.
Housman SJ, Scott KM, Reinkensmeyer DJ . A randomized controlled trial of gravity-supported, computer-enhanced arm exercise for individuals with severe hemiparesis. Neurorehab Neural Repair 2009; 23: 505–514.
Staubli P, Nef T, Klamroth-Marganska V, Riener R . Effects of intensive arm training with the rehabilitation robot ARMin II in chronic stroke patients: four single-cases. J Neuroeng Rehabil 2009; 6: 46.
Reinkensmeyer DJ, Emken JL, Cramer SC . Robotics, motor learning, and neurologic recovery. Annu Rev Biomed Eng 2004; 6: 497–525.
Prange GB, Jannink MJ, Groothuis-Oudshoorn CG, Hermens HJ, Ijzerman MJ . Systematic review of the effect of robot-aided therapy on recovery of the hemiparetic arm after stroke. J Rehabil Res Dev 2006; 43: 171–184.
Kwakkel G, Kollen BJ, Krebs HI . Effects of robot-assisted therapy on upper limb recovery after stroke: a systematic review. Neurorehab Neural Repair 2008; 22: 111–121.
Fasoli SE, Krebs HI, Stein J, Frontera WR, Hughes R, Hogan N . Robotic therapy for chronic motor impairments after stroke: follow-up results. Arch Phys Med Rehabil 2004; 85: 1106–1111.
Volpe BT, Lynch D, Rykman-Berland A, Ferraro M, Galgano M, Hogan N et al. Intensive sensorimotor arm training mediated by therapist or robot improves hemiparesis in patients with chronic stroke. Neurorehabil Neural Repair 2008; 22: 305–310.
Lo AC, Guarino PD, Richards LG, Haselkorn JK, Wittenberg GF, Federman DG et al. Robot-assisted therapy for long-term upper-limb impairment after stroke. N Engl J Med 2010; 362: 1772–1783.
Anderson KD . Targeting recovery: priorities of the spinal cord injured population. J Neurotrauma 2004; 21: 1371–1383.
Gijbels D, Lamers I, Kerkhofs L, Alders G, Knippenberg E, Feys P . The Armeo Spring as training tool to improve upper limb functionality in multiple sclerosis: a pilot study. J Neuroeng Rehabil 2011; 8: 5.
Zariffa J, Kapadia N, Kramer JLK, Taylor P, Alizadeh-Meghrazi M, Zivanovic V et al. Effect of a robotic rehabilitation device on upper limb function in a sub-acute cervical spinal cord injury population. Proc. IEEE 12th Int Conf Rehab Robotics, June 29–July 1 2011, Zurich, Switzerland.
Kalsi-Ryan S, Beaton D, Curt A, Duff S, Popovic MR, Rudhe C et al. The graded redefined assessment of strength sensibility and prehension (GRASSP) - reliability and validity. J Neurotrauma (e-pub ahead of print 5 September 2011).
Carroll D . A quantitative test of upper extremity function. J Chronic Diseases 1965; 18: 479–491.
Carroll TJ, Herbert RD, Munn J, Lee M, Gandevia SC . Contralateral effects of unilateral strength training: evidence and possible mechanisms. J Appl Physiol 2006; 101: 1514–1522.
Steeves JD, Kramer JK, Fawcett JW, Cragg J, Lammertse DP, Blight AR et al. Extent of spontaneous motor recovery after traumatic cervical sensorimotor complete spinal cord injury. Spinal Cord 2011; 49: 257–265.
Acknowledgements
This work was supported by the Rick Hansen Institute (grant number 2009-34). We are grateful to the study participants.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Zariffa, J., Kapadia, N., Kramer, J. et al. Feasibility and efficacy of upper limb robotic rehabilitation in a subacute cervical spinal cord injury population. Spinal Cord 50, 220–226 (2012). https://doi.org/10.1038/sc.2011.104
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2011.104
Keywords
This article is cited by
-
Upper Limb Recovery in Cervical Spinal Cord Injury After a Brain-Computer Interface Controlled Functional Electrical Stimulation Intervention
Journal of Medical and Biological Engineering (2023)
-
A suite of automated tools to quantify hand and wrist motor function after cervical spinal cord injury
Journal of NeuroEngineering and Rehabilitation (2019)
-
A single-subject study of robotic upper limb training in the subacute phase for four persons with cervical spinal cord injury
Spinal Cord Series and Cases (2019)
-
Clinical efficacy of upper limb robotic therapy in people with tetraplegia: a pilot randomized controlled trial
Spinal Cord (2019)
-
Rehabilitation robots for the treatment of sensorimotor deficits: a neurophysiological perspective
Journal of NeuroEngineering and Rehabilitation (2018)