Abstract
Study design:
To develop a method for the study of spinal cord injury (SCI) that can visualize the blood vessels and is compatible with hematoxylin and eosin (HE) staining and immunohistochemical techniques.
Objective:
Visualization of the vascular changes is important for the study of SCI. The original ferric tannate method can stain the spinal cord vasculature to its terminals, but the diffuse tannate precipitates spoil the delicacy of the picture. More importantly, it is incompatible with HE staining and immunohistochemical techniques, which is crucial for the study of SCI. We thus aimed to develop a modified ferric tannate method that could meet the requirement for the study of SCI.
Setting:
This study was carried out in China.
Methods:
The original ferric tannate method involves a two-step procedure: intravascular perfusion of tannic acid, followed by soaking the tissue sections in a solution of ferric chloride. In the modified method both chemicals were delivered through perfusion.
Results:
In the original method, diffuse ferric tannate precipitates blurred the profile of the vessels. More importantly, it was incompatible with either HE or immunostaining methods. Our modified method stained the blood vessels with clean background and was compatible with both HE staining and immunohistochemical techniques.
Conclusion:
The modified method is far superior to the original method and meets the requirement for the study of SCI.
Similar content being viewed by others
Introduction
The high oxygen demand of the CNS is met by its abundant blood supply. Changes in its blood circulation, either as the cause or the result of a disease, may lead to serious outcomes. It has been reported that during spinal cord injury (SCI) ischemia leads the wave front of the expending secondary injury.1, 2, 3 Our preliminary study on spinal cord contusion, using the NYU (New York University) impactor, showed a pale ischemic zone on hematoxylin and eosin-stained (HE) sections neighboring the primary injury. In this ischemic zone the neurons were disappearing and the staining intensity of their Nissl bodies was fading. Therefore, to study the changes in blood circulation during the developing secondary injury and devise a measure to ameliorate it, visualization of the blood vessel is undoubtedly important.
The ferric tannate method is a classical blood vessel-staining technique, which has been successfully used in the study of visceral vasculature.4, 5, 6 Owing to the high background of the staining, in particular its incompatibility with HE staining and immunohistochemical techniques, which are crucial in the study of SCI, we aimed to develop a modified method that could meet the requirement for SCI study.
Materials and methods
Animals
In total, 18 adult male Sprague–Dawley rats (200–220 g) were used, 6 for the original method and 12 for our modified method. The latter included a normal group (n=6) and a spinal cord contusion group (n=6). A NYU impactor was used to induce contusion in the spinal cord.7
The original method
The animals were killed by an overdose of sodium pentobarbital and perfused intracardially with normal saline (37 °C), followed by 300 ml of a mixture of 4% paraformaldehyde, 2% tannic acid (Sigma-Aldrich, St Louis, MO, USA) and 0.5% glutaraldehyde in 0.01 M PBS (phosphate-buffered saline) (37 °C). After perfusion, the laminae above and below T8 were removed, and a spinal cord segment of size 2 cm was dissected out and immersed in a 25% sucrose solution at 4 °C until the spinal cord sank. Sagittal sections of thickness 30 μm were cut on a cryostat and mounted on the slides. Sections were let dry and then immersed in 3% ferric chloride solution (37 °C) for 30–40 min. The tannic acid and ferric chloride solutions should be filtered before use.
The modified method
Instead of immersing the sections in ferric chloride, which results in a diffused deposition of ferric tannate, in our modified method, the ferric chloride is delivered through perfusion. The animals were perfused with tannic acid and fixative as in the original method, followed by thorough rinsing of the vascular system with 80–100 ml normal saline. Then 300 ml of 3% ferric chloride solution (37 °C) was perfused, following which all the ferric tannate precipitates would remain inside the vessels. Xylene was avoided in mounting the immunostained sections.
Spinal cord injury model
In the SCI model group, the animal was anesthetized with an intraperitoneal injection of 1% sodium pentobarbital (50 mg kg−1). A 30–40-mm dorsal midline longitudinal incision of the skin was made to expose the T6–T11 vertebrae. Bilateral laminectomy was carried out at T8. In accordance with the instructions accompanying the usage of a NYU impactor, a 10-g metal rod was dropped from a height of 12.5 mm to produce a mild contusion in the spinal cord. The animals were killed 8 h after the operation.
Counterstaining
1. Routine HE staining.
2. Immunohistochemical method: Monoclonal antibodies against glial fibrillary acidic protein (1:3000, Sigma-Aldrich), RECA1 for vascular endothelial cells (1:1000, Abcam, Cambridge, MA, USA) and occludin for tight junctions between the endothelial cells (1:400, Invitrogen, Carlsbad, CA, USA) were used. For glial fibrillary acidic protein immunostaining, the ferric tannate-stained sections were successively rinsed with 0.01 M PBS, placed in 0.3% methanol–H2O2 to quench endogenous peroxidase activity and treated with 0.3% Triton X-100 in 0.01 M PBS for 20 min. After washing with 0.01 M PBS, the antibody was applied overnight at 4 °C, followed by incubation with a biotinylated rabbit anti-mouse IgG (1:400, Sigma-Aldrich) for 2 h at room temperature. The avidin–biotin peroxidase complex (1:400, Sigma-Aldrich) was then applied for 1 h and developed using the glucose oxidase–diaminobenzidine (DAB)–nickel method.8 For RECA1 and occludin immunohistochemistry, the ferric tannate-stained sections were rinsed with 0.01 M PBS and treated with 0.3% Triton X-100 in 0.01 M PBS for 20 min. After washing with 0.01 M PBS, the primary antibody was applied overnight at 4 °C, and visualized with FITC (fluorescein isothiocyanate)-conjugated donkey anti-mouse IgG (1:400, Sigma-Aldrich) for 2 h at room temperature. Omission of the primary antibodies served as controls. We certify that institutional and governmental regulations regarding the ethical use of animals were followed.
Results
Staining of the normal spinal cord
1. The original method: Many blood vessels were stained black. The background was covered by a diffuse deposit of ferric tannate, which blurred the vascular profile (Figure 1a).
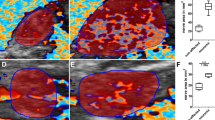
Comparison between the original and modified ferric tannate methods. (a) Original method: The section is covered with ferric tannate precipitate, blurring the vessel profile. (b) Modified method: The section is clean and the blood vessels are delicately displayed. (c, d) The modified method counterstained with hematoxylin and eosin, respectively. Bar=200 μm. GM, gram matter; WM, white matter. A full colour version of this figure is available at the Spinal Cord Journal online.
2. The modified method: The background was clean, which allowed the display of the vessels in a much better delicacy. The walls of the blood vessels appeared as clear-cut lines. Many vessels of small caliber could be seen (Figure 1b).
3. Double staining with HE: The original method resulted in poor staining (not shown), whereas the modified method stained well with either of the techniques (Figure 1c, d).
4. Immunohistochemical staining: The original method did not show immunostaining; the sections appeared virtually blank (not shown), whereas the modified method was fully compatible with immunohistochemistry. The glial fibrillary acidic protein immunoreactive astrocytes and their processes could be identified in great detail. With triple staining of the modified ferric tannate, glial fibrillary acidic protein immunohistochemical, and HE techniques, the relationship among the blood vessels, astrocytes and neurons could be clearly deciphered (Figures 2a and b). The anti-RECA1 could clearly stain the blood vessels of a normal spinal cord (Figure 2c), but failed to stain the ferric tannate-stained vessels (data not shown), whereas in the lesion site, where blood circulation was destroyed and ferric tannate could not reach, the vascular endothelium could be stained with anti-RECA1 (Figure 2d). Similar to anti-RECA1, anti-occludin could not stain the tight junction of the ferric tannate-stained vessels.
Immunohistochemistry. Triple staining with modified ferric tannate, anti-GFAP (glial fibrillary acidic protein) antibody and hematoxylin (a) or eosin (b). Note the details of the GFAP-immunoreactive astrocytes and their processes. RECA1 immunostaining (green) of the blood vessels of normal spinal cord (c) and vessels in the lesion area (d). Scale bars in (a) and (b)=20 μm; in (c) and (d)=100 μm. A full colour version of this figure is available at the Spinal Cord Journal online.
Staining of contused spinal cord
To further show the advantages of the modified method, sections of the contused spinal cord were stained by the modified method in combination with hematoxylin staining. The center of injury was full of red blood corpuscles. Next to it was a pale ischemic zone, in which most of the neurons and blood vessels disappeared. In the few remaining neurons, the Nissl bodies were dispersed and weakly stained. The terminal vessels of some remaining blood vessels were found collapsed (Figure 3a). An intriguing feature was that in the area next to the pale ischemic zone, the neurons appeared normal, yet there were a substantial amount of collapsed terminal vessels (Figure 3b), indicating that ischemia had already been occurring in this apparently normal region.
Modified ferric tannate method counterstained with hematoxylin. (a) High magnification of the ischemic zone surrounding the bleeding. Most of the neurons and vessels have disappeared. Note the collapsed terminal vessels (arrows). Bar=50 μm. (b) Lower magnification, showing the bleeding, pale ischemic zone, and its neighboring area with normally stained neurons and their Nissl bodies. Note the collapsed terminal vessels in the pale ischemic zone and, in particular, in the apparent normal region (arrows). Bar=100 μm. A full colour version of this figure is available at the Spinal Cord Journal online.
Discussion
In the original ferric tannate method, the tannic acid is perfused intravascularly, followed by immersing the section in a solution of ferric chloride. The section is blurred by the diffuse ferric tannate precipitations. Furthermore, it takes up HE staining poorly and is completely incompatible with immunohistochemistry. In our modified method, both the tannic acid and ferric chloride are perfused intravascularly. Thus, diffuse precipitation is avoided. Furthermore, it is perfectly compatible with HE staining and immunohistochemistry. Its compatibility with immunohistochemistry is an important feature of the modified technique, because it is often necessary to study the relationship between the blood vessels and other elements in the tissue, such as neurons, nerve fibers, glial cells, extracellular matrix and various other chemical components, which play important roles in the pathology of SCI, and the great majority of them can be immunostained.
There are other methods that can stain the blood vessels. Those that can stain vascular endothelium are particularly important, as it can display the vasculature down to the capillaries. It has been reported that the endothelial cells can be immunostained well with antibodies against SMI71, RECA-1 and PECAM-1.9, 10, 11, 12 The endothelium-staining methods have an advantage over the perfusion methods, such as the ferric tannate method, in that the latter cannot stain the blood vessels distal to any blockade, which often happens during SCI. The sites of obstruction and the resulted ischemia are very important aspects for the study of SCI. Therefore, a combination of these two techniques can be most helpful, particularly if the modified ferric tannate method can be counter immunostained with antibodies against endothelial cells. We have tried to counterstain the ferric tannate-stained sections with an antibody against the endothelial cell (anti-RECA1). We found that the ferric tannate-stained vessels cannot be stained with anti-RECA1. Apparently, the ferric tannate precipitation on the endothelium interferes with the immunostaining. In areas devoid of ferric tannate staining, such as the lesion sites, anti-RECA1 can stain the vessels well.
An intriguing discovery with this method is that there are two ischemic zones with collapsed vessels near the developing secondary SCI, namely, a zone adjacent to the injury, where most of the neurons are degenerating or have disappeared, and a farther zone, in which most of the neurons appear normal. There seems to be little chance to reverse the process of the severely degenerating neurons in the adjacent zone. However, it is possible to rescue most of the neurons in the farther zone, which is clinically very important. This technique provides a tool to study the therapeutic effects of different measures, such as to improve blood flow or encourage angiogenesis, which is of significant value in the study of SCI.
Conclusion
The modified tannic acid–ferric chloride technique can stain the blood vessels up to the terminal vessels in high resolution. It can be counterstained with HE and immunohistochemal methods. Its compatibility with immunostaining is an important feature, because the majority of the elements related to the blood vessels can be immunostained. The ferric tannate method cannot stain the vasculature distal to the blockades. This can be supplemented by immunostaining with antibodies against the endothelium. In the case of SCI, the ferric tannate method is able to reveal two ischemia zones near the injury site, an adjacent zone with advanced neuronal degeneration and a farther region in which most of the neurons appear normal. The ferric tannate method is a valuable tool for the study of the pathological process of the SCI, as well as to study the therapeutic effects of the measures taken to rescue the neurons in the ischemic zones.
References
Balentine JD . Pathology of experimental spinal cord trauma. I. The necrotic lesion as a function of vascular injury. Lab Invest 1978; 39: 236–253.
Mautes AE, Weinzierl MR, Donovan F, Noble LJ . Vascular event after spinal cord injury contribution to secondary pathogenesis. Phys Ther 2000; 80: 673–687.
Tator CH . Review of experimental spinal cord injury with emphasis on the local and systemic circulatory effects. Neurochirurgie 1991; 37: 291–302.
Li CH, Pan LH, Li CY, Zhu CL, Xu WX . Localization of ANP-synthesizing cells in rat stomach. World J Gastroenterol 2006; 12: 5674–5679.
Li CH, Yang ZW, Yin ZR, Jin Z, Xing DG, Piao LH et al. Relationship between atrial natriuretic peptide-immunoreactive cells and microvessels in rat gastric mucosa. Acta Pharmacol Sin 2006; 27: 205–211.
Nakashima Y, Sueishi K . Alteration of elastic architecture in the lathyritic rat aorta implies the pathogenesis of aortic dissecting aneurysm. Am J Pathol 1992; 140: 959–969.
Kwo S, Young W, Decrescito V . Spinal cord sodium, potassium, calcium, and water concentration changes in rats after graded contusion injury. J Neurotrauma 1989; 6: 13–24.
Shu SY, Ju G, Fan LZ . The glucose oxidase-DAB-nickel method in peroxidase histochemistry of the nervous system. Neurosci Lett 1988; 85: 169–171.
Loy DN, Crawford CH, Darnall JB, Burke DA, Onifer SM, Whittemore SR . Temporal progression of angiogenesis and basal lamina deposition after contusive spinal cord injury in the adult rat. J Comp Neurol 2002; 445: 308–324.
Duijvestijn AM, van Goor H, Klatter F, Majoor GD, van Bussel E, van Breda Vriesman PJ . Antibodies defining rat endothelial cells: RECA-1, a pan-endothelial cell-specific monoclonal antibody. Lab Invest 1992; 66: 459–466.
DeLisser HM, Christofidou-Solomidou M, Strieter RM, Burdick MD, Robinson CS, Wexler RS et al. Involvement of endothelial PECAM-1/CD31 in angiogenesis. Am J Pathol 1997; 151: 671–677.
Ulger H, Karabulut AK, Pratten MK . Labelling of rat endothelial cells with antibodies to vWF, RECA-1, PECAM-1, ICAM-1, OX-43 and ZO-1. Anat Histol Embryol 2002; 31: 31–35.
Acknowledgements
This work was supported by the National Basic Research Program of China(2003CB515301) and Grant no. 06MA148 from the PLA Military Medical Research Fund. We are grateful to Ms Xi-Ying Jiao for his assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shen, XF., Zhao, Y., Zhang, YK. et al. A modified ferric tannate method for visualizing a blood vessel and its usage in the study of spinal cord injury. Spinal Cord 47, 852–856 (2009). https://doi.org/10.1038/sc.2009.30
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2009.30
Keywords
This article is cited by
-
Beneficial effects of early hemostasis on spinal cord injury in the rat
Spinal Cord (2016)
-
Effect of norepinephrine on spinal cord blood flow and parenchymal hemorrhage size in acute-phase experimental spinal cord injury
European Spine Journal (2014)
-
Protective effects of Batroxobin on spinal cord injury in rats
Neuroscience Bulletin (2013)
-
The Protective Effects of Inosine Against Chemical Hypoxia on Cultured Rat Oligodendrocytes
Cellular and Molecular Neurobiology (2011)