Abstract
Study design:
A prospective, non-randomized clinical series trial.
Objective:
To evaluate the effect of autogenous undifferentiated stem cell infusion for the treatment of patients with chronic spinal cord injury (SCI) on somatosensory evoked potentials (SSEPs).
Setting:
A public tertiary hospital in São Paulo, Brazil.
Methods:
Thirty-nine consecutive patients with diagnosed complete cervical and thoracic SCI for at least 2 years and with no cortical response in the SSEP study of the lower limbs were included in the trial. The trial patients underwent peripheral blood stem cell mobilization and collection. The stem cell concentrate was cryopreserved and reinfused through arteriography into the donor patient. The patients were followed up for 2.5 years and submitted to SSEP studies to evaluate the improvement in SSEPs after undifferentiated cell infusion.
Results:
Twenty-six (66.7%) patients showed recovery of somatosensory evoked response to peripheral stimuli after 2.5 years of follow-up.
Conclusion:
The 2.5-year trial protocol proved to be safe and improved SSEPs in patients with complete SCI.
Sponsorship:
None.
Similar content being viewed by others
Introduction
The annual incidence of spinal cord injuries (SCIs) in many countries is difficult to determine as reported incidences vary considerably, ranging from 10.4 to 83 per million inhabitants per year. SCIs mainly affect younger people.1
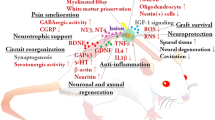
Neurological dysfunction due to SCI arises from the primary mechanical injury itself, as well as from secondary injury, which is an endogenous process involving complex biochemical changes with edema, inflammation, ischemia, reperfusion, growth factors, calcium metabolism and lipidic peroxidase that may foster or hinder axonal reconnection.2, 3
Neurons outside the central nervous system (CNS) in the immature spinal cord and in special culture media can regenerate.4 Although it has been recognized for decades that CNS is resistant to regeneration, recent studies have shown that these cells can also potentially recover and multiply.5
Spinal cord injury treatments consist of biological, surgical and pharmacological approaches, as well as physical procedures such as spinal stabilization.6 Steroids can regulate the endogenous response to acute primary SCI and reduce tissue damage, resulting in better functional recovery. These drugs help prevent the pathophysiological mechanisms causing secondary injuries. Tirilazad and gangliosides have also been studied.7
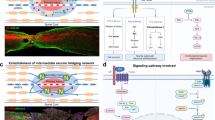
The biological therapeutic approach to SCI includes factors that stimulate neuronal regeneration, such as tissue growth factors and stem cells (totipotent, pluripotent or multipotent; autologous or allogenic). Transplantation of these cells has shown that they can differentiate and be integrated into the neural tissue.8 Embryonic stem cells (ESCs) are promising as a potentially regenerative therapy for neurological conditions such as Parkinson's disease and SCI.9
Neural stem cells taken from the embryonic spinal cord are an appealing source of cells for transplantation because they are involved in spinal cord progeny.10 The mechanisms by which they repair a CNS injury are still controversial, but they certainly involve functional reconstruction of neuronal circuits, synapses and connections with host cells, production of neurochemical substances such as neurotransmissors, growth factors and antibodies, and myelination of axons.11
Stem cell-based therapies are moving closer to clinical application. A solid human embryonic spinal cord tissue transplanted into the cavity of an adult injured spinal cord has been shown to yield beneficial morphological effects in both acute and chronic SCI.12
Donor-derived cells expressing neuronal proteins (neuronal phenotypes) were developed in the CNS after intravascular delivery of genetically marked adult mouse bone marrow into lethally irradiated normal adult hosts, resulting in a population of donor-derived cells in the brain with distinct characteristics from the bone marrow. The generation of neuronal phenotypes in the adult brain, 1–6 months post-bone marrow transplant, shows a remarkable plasticity of adult tissues with potential clinical applications, even after lethal irradiation.8 Thus, adult cells can be reprogrammed to express genes that are typical of differentiated cells, and the differentiated status is reversible.
Sasaki et al.13 have shown that transplantation of bone marrow cells in a demyelinated adult rat spinal cord results in extensive remyelination, showing that bone marrow cells could differentiate into a myelinating phenotype in vivo.
The objective of this study was to evaluate the effect of autogenous undifferentiated stem cell infusion for the treatment of patients with chronic SCI on somatosensory evoked potentials (SSEPs).
Materials and methods
Study patients
This study was reviewed and approved by the Research Ethics Committee of the Hospital das Clínicas of the São Paulo School of Medicine (protocol #768/01) and the National Health Committee (CONEP, protocol #7524). All applicable institutional and governmental regulations regarding the ethical use of human volunteers/animals were followed during the course of this research trial. All patients signed an informed consent, agreeing to participate in the study.
Thirty-nine patients with complete traumatic cervical or thoracic SCI14 for at least 2 years were selected for this trial, from among those who attended the Raquimedular Trauma Treatment Service of a public tertiary university hospital in São Paulo, Brazil, between 2002 and 2004.
All the patients had SSEP studies with no cortical response on tibial nerves. Those patients who had cervical injuries and were quadriplegic were included if they had reduced or absent cortical response to median nerve stimulation.
All patients aged 18 years or more who were included in the study had normal blood cell counts and no coagulation disorders, but had shown no neurological improvement in the previous year. Those who did not show an artery with blood flow to the SCI area, as evidenced in the spinal cord angiography, were excluded.
The trial patients were followed up for 2.5 years after receiving an autologous graft of concentrated cryopreserved stem cells as described below. During this period, they were submitted to SSEP studies every 6 months, to evaluate neurological recovery.
Mobilization of stem cells and cryopreservation
Mobilization of bone marrow stem cells was obtained through subcutaneous administration of granulocyte colony-stimulating factor (25 g Granulokine with 300 μg filgrastim) at a dosage of 10 μg kg–1 day–1 for five consecutive days.15 On the sixth day after the first administration of the granulocyte colony-stimulating factor, the mobilized cells were collected in the hospital blood center by apheresis of peripheral blood, using a double-lumen hemodialysis catheter (12 Fr). Collections were carried out at least once a day until 2.5 × 106 CD 34+ cells kg–1 of body weight had been obtained.16 In 34 patients, only one session was needed in order to collect the desired amount of CD 34+ cells. In five patients, two sessions were needed. This protocol enabled 2.5 × 106 CD 34+ cells kg–1 to be obtained from all the patients. Positive CD34 cells were detected by flow cytometry.17, 18
Dimethylsulfoxide was used for stem cell cryopreservation (10% final concentration). The temperature was reduced by 1 °C min–1 up to −40 °C, and then by 10 °C min–1 up to −80 °C. The cells were then transferred to a freezer at −120 °C.19 The stem cells were stored for 1 week before infusion.
Stem cell infusion
The study patients were admitted to the Vascular Interventional Radiology Department for medullary angiography. They underwent general anesthesia, followed by right common femoral artery puncture and insertion of a Cobra 2 catheter (a radiopaque, stranded, tubular, polyurethane 4 Fr and 65 cm long, Cobra 2-shaped guiding catheter for hemodynamic catheterization).
For the quadriplegic patients, catheters were inserted into the femoral artery and guided up to the vertebral artery, to check for the presence of an artery branch supplying blood to the anterior spinal artery at the level of the injury. Heparin was administered to minimize the risk of stroke induced by this procedure.
For the paraplegic patients, the catheter was selectively inserted into the intercostal arteries, to check for the presence of an artery branch supplying blood to the anterior spinal artery at the level of the injury.
Immediately before infusion, the cells were thawed in a warm water bath at 37 °C. After they had completely thawed, they were infused into the patient's peripheral bloodstream at the rate of 10 ml min–1, through a regular transfusion tubing. The infusion was not radiographically monitored, in order to prevent radiation of the transplanted cells. During cell infusion, a sample of the infusion solution was collected and examined under the microscope to ensure the stem cells were viable.
Follow-up
On the day after infusion, the artery puncture site was examined, and the patient was discharged from hospital if there was no sign of hematoma or active bleeding.
A series of SSEP studies of the lower and upper limbs was conducted for the paraplegic and quadriplegic patients every 3 months, for the first 6 months and twice a year for 2.5 years. The studies were conducted and evaluated by the same physical medicine provider.
In the SSEP study of the upper limbs, the median nerves in the wrists were stimulated and recorded at the FZ position—ERB’S point, P3′/P4′–A1/A2. In the SSEP study of the lower limbs, the tibial nerves were stimulated at the ankles and recorded from the scalp at the FZ–PZ positions.
A positive response was defined as cortical response to peripheral stimulation of the lower limbs in both paraplegic and quadriplegic patients. For those quadriplegic patients with cervical injuries who had already shown some cortical response to the upper limb stimulation before treatment, a positive response was defined as a 10% reduction of latency time or normalization of SSEP.
Statistical analysis
Comparisons of frequencies between groups (samples) were carried out by applying Fisher's exact test in contingency tables. For the assessment of distribution normality, the Kolmogorov–Smirnov (K–S) test for continuous variables and Pearson's correlation coefficient (PCC%) were applied. The distribution of a measurement was considered normal if the K–S test did not show any significant difference (P⩾0.05) and/or if the PCC was below 30%. For these cases, parametric tests were used. For all other cases (K–S: P⩽0.05 and/or PCC⩾30%) non-parametric tests were used.
In-group comparisons were applied for the mean estimates, Student's t-test was applied to two non-related parametric samples (independent or non-paired) and the Mann–Whitney U-test was applied to two independent non-parametric samples. A 5% level of significance was set.
The statistical package StatSoft, Inc. (2001) Statistica (data analysis software system) v. 6.0 and GraphPad Software, Inc. (1996) Graphpad Prism v. 2.01 were used for the analyses.
Results
Patients
The trial sample comprised 11 females and 28 males; 30.8% of the patients were quadriplegic and 69.2% paraplegic. Three patients—two quadriplegic and one paraplegic—were lost to follow-up during the trial, and they were included in the analysis as having the worst outcome, that is, no response in the SSEP study. No patient died during the 2.5-year follow-up.
There was no complication, neoplasm or aggravated neurological condition requiring discontinuation or withdrawal from the trial. Four patients had complications: one was a case of pneumothorax caused by insertion of the catheter for stem cell collection, which was resolved with thorax drainage; the other three were cases of local allergic reaction occurring during the injection of contrast dye to check for the presence of intercostal arteries for stem cell infusion, which was resolved with intravenous administration of dexamethasone 4 mg. There was no statistically significant difference in terms of complications between the paraplegic and the quadriplegic patients (P=0.57).
The causes of SCI were motor vehicle accidents (61.5%), sports injuries (such as diving) (17.9%), motorcycle accidents (15.4%) and aircraft accidents (5.1%).
In relation to the injury site, 15.4% of the injuries were between the second and the fourth cervical vertebra; 20.5% between the fifth cervical vertebra and the first thoracic vertebra; and 64.1% between the second thoracic vertebra and the first lumbar vertebra.
SSEP improvement
Of the 39 patients studied, 26 (66.7%) showed a positive response in the SSEP study (Figure 1).
No statistically significant difference was found between the paraplegic and the quadriplegic patients in terms of improved response in the SSEP study (P=0.27). Moreover, no statistically significant difference was found in relation to the frequency of improvement in the lower limb response for paraplegic patients, when compared with upper limb improvement in the quadriplegic patients in the SSEP study (P=0.17). There was no difference in response when compared by sex (P=1.00), age at the time of injury (P=0.07), age at the time of blood collection (P=0.13) or stem cell infusion (P=0.13).
Regarding time elapsed between SCI and autogenous stem cell infusion, there was no statistically significant difference (P=0.57) between those who showed SSEP response and those who did not show any changes in SSEP patterns.
In the comparison of SCI level and SSEP response, no statistically significant correlation was seen (P=0.08). In addition, the external cause of SCI did not affect the SSEP results (Table 1).
In those patients who showed SSEP response, there was a mean time of 9 months between infusion and lower limb response. In the quadriplegic patients, there was a mean time of 10.4 months between infusion and improvement of the SSEP upper limb response (Tables 2, 3, 4).
Discussion
A positive response in SSEP studies was obtained for the majority of the patients studied. The positive cortical response to peripheral stimuli could be explained by the formation of new synapses between host neurons and neurons formed after stem cell transplantation,16 or by new myelination of glial cells derived from the transplanted cells.13, 20
In this study, no variable was correlated with a higher likelihood of achieving improved neural function in 2 years.
Many animal studies investigating progenitor cells for SCI treatment have evidenced that these cells, once transplanted, can differentiate into glial and neuronal cells and promote neurological recovery. This therapeutic strategy is very appealing because of the availability of autologous material for transplant, and the fact that these cells have tropism for the injured area in the spinal cord, and can be effective even through remote infusion either by intravascular or by intrathecal administration.21
There have been a number of clinical trials worldwide on the use of progenitor cells, but few have been published to date. Park et al.22 conducted a trial in 2005 that included six patients with complete SCI who were transplanted with progenitor cells drawn from their bone marrow, between 7 and 14 days post-injury. All the patients were administered colony-stimulating factors and showed neurological improvement.22
Syková et al.21 investigated 20 patients with complete SCI who received autologous progenitor cells through intravascular injection (intravenous peripherally, or through vertebral artery catheterization). They reported neurological improvement in all the patients with subacute injury receiving cells through the vertebral artery, and in only one patient with subacute injury receiving cells intravenously through a peripheral vein. These authors also reported improvement in only two patients with chronic injury who received cells through the vertebral artery. They concluded that intravenous injection is a safe approach, and emphasized that treatment should be provided 3–4 weeks post-SCI, and infusion applied as close as possible to the injured site for best results.21 The patients’ baseline neurological status was used in this study as control, and was evaluated in the SSEP studies, a common approach in clinical trials.23
The study sample was homogeneous as we chose to include only patients with closed traumatic SCI, given that this is the most widely studied and well-established condition.24, 25, 26 Grafting autologous rather than embryonic cells eliminates the need for immunosuppression, which would be contra-indicated in SCI patients, as they are more predisposed to respiratory and urinary infections and scarring. Confounders were controlled by selecting patients with SCI for at least 2 years, as it is well-known that neurological recovery in patients with complete injury largely occurs in the first 6–9 months post-injury, and that 12–18 months later there is virtually no further recovery.27 Hence, any change in the neurological status of these patients after inclusion in the trial could be attributed to the therapeutic approach under investigation. In addition, when stem cells are transplanted during acute injury, only a small proportion of cells survive, and some even differentiate into astrocytes because of the unfavorable microenvironment that develops immediately after SCI.28
This is a prospective, groundbreaking study though it is neither randomized nor blind. The physical medicine provider who conducted all the SSEP studies was aware that the patients under evaluation were participating in a stem cell trial protocol.
Modern science has been showing the mechanisms of cellular protection and neurological regeneration, but so far, only support therapy has been available to SCI patients. This study shows that further knowledge of differentiation and proliferation of stem cells, and their association with drugs and physical and surgical approaches, can offer excellent opportunities for SCI treatment.
Autogenous stem cell infusion in patients with complete chronic SCI resulted in SSEP response or improved latency for cortical response to peripheral stimuli. During a 2.5-year follow-up, this trial protocol for stem cell mobilization, collection and infusion proved to be a safe approach.
References
Wyndaele M, Wyndaele JJ . Incidence, prevalence and epidemiology of spinal cord injury: what learns a worldwide literature survey? Spinal Cord 2006; 44: 523–529.
Tator CH, Fehlings G . Review of the secondary injury theory of acute spinal cord trauma with emphasis on vascular mechanisms. J Neurosurg 1991; 75: 15–26.
Dusart I, Schwab ME . Secondary cell death and the inflammatory reaction after dorsal hemisection of the rat spinal cord. Eur J Neurosci 1994; 6: 712–724.
Zietlow R, Lane EL, Dunnett SB, Rosser AE . Human stem cells for CNS repair. Cell Tissue Res 2008; 331: 301–322.
Temple S, Alvarez-Buylla A . Stem cells in the adult mammalian central nervous system. Curr Opin Neurobiol 1999; 9: 135–141.
Legos JJ, Gopez JJ, Young WF . Non-surgical management of spinal cord injury. Expert Opin Investig Drugs 2002; 11: 469–482.
Geisler FH, Dorsey FC, Coleman WP . Past and current clinical studies with GM-1 ganglioside in acute spinal cord injury. Ann Emerg Med 1993; 22: 1041–1047.
Cízková D, Rosocha J, Vanickỳ I, Jergová S, Cízek M . Transplants of human mesenchymal stem cells improve functional recovery after spinal cord injury in the rat. Cell Mol Neurobiol 2006; 26: 1167–1180.
Li JY, Christophersen NS, Hall V, Soulet D, Brundin P . Critical issues of clinical human embryonic stem cell therapy for brain repair. Trends Neurosci 2008; 31: 146–153.
Lowry N, Goderie SK, Adamo M, Lederman P, Charniga C, Gill J et al. Multipotent embryonic spinal cord stem cells expanded by endothelial factors and Shh/RA promote functional recovery after spinal cord injury. Exp Neurol 2008; 209: 510–522.
Okano H . Stem cell biology of the central nervous system. J Neurosci Res 2002; 69: 698–707.
Akesson E, Holmberg L, Jönhagen ME, Kjaeldgaard A, Falci S, Sundström E et al. Solid human embryonic spinal cord xenografts in acute and chronic spinal cord cavities: a morphological and functional study. Exp Neurol 2001; 170: 305–316.
Sasaki M, Honmou O, Akiyama Y, Uede T, Hashi K, Kocsis JD . Transplantation of an acutely isolated bone marrow fraction repairs demyelinated adult rat spinal cord axons. Glia 2001; 35: 26–34.
American Spinal Injury Association. Standard neurological classification of spinal cord injury. Available at http://www.asia-spinalinjury.org/publications/2006_Classif_worksheet.pdf (accessed 17 July 2008).
Stroncek DF, Clay ME, Petzoldt ML, Smith J, Jaszcz W, Oldham FB et al. Treatment of normal individuals with granulocyte-colony-stimulating factor: donor experiences and the effects on peripheral blood CD34+ cell counts and on the collection of peripheral blood stem cells. Transfusion 1996; 36: 601–610.
Ogawa Y, Sawamoto K, Miyata T, Miyao S, Watanabe M, Nakamura M et al. Transplantation of in vitro-expanded fetal neural progenitor cells results in neurogenesis and functional recovery after spinal cord contusion injury in adult rats. J Neurosci Res 2002; 69: 925–933.
Körbling M, Katz RL, Khanna A, Ruifrok AC, Rondon G, Albitar M et al. Hepatocytes and epithelial cells of donor origin in recipients of peripheral-blood stem cells. N Engl J Med 2002; 346: 738–746.
Huss R . Isolation of primary and immortalized CD34-hematopoietic and mesenchymal stem cells from various sources. Stem Cells 2000; 18: 1–9.
Stiff PK . Cryopreservation of hematopoietic stem cells. In: Warkentin PI, Lasky LC (eds). Marrow and Stem Cell Processing for Transplantation. American Association of Blood Banks: Bethesda, MD, 1995, pp 69–82.
Liu S, Qu Y, Stewart TJ, Howard MJ, Chakrabortty S, Holekamp TF et al. Embryonic stem cells differentiate into oligodendrocytes and myelinate in culture and after spinal cord transplantation. Proc Natl Acad Sci USA 2000; 97: 6126–6131.
Syková E, Jendelová P, Urdzíková L, Lesnỳ P, Hejcl A . Bone marrow stem cells and polymer hydrogels--two strategies for spinal cord injury repair. Cell Mol Neurobiol 2006; 26: 1113–1129.
Park HC, Shim YS, Ha Y, Yoon SH, Park SR, Choi BH et al. Treatment of complete spinal cord injury patients by autologous bone marrow cell transplantation and administration of granulocyte-macrophage colony stimulating factor. Tissue Eng 2005; 11: 913–922.
Tator CH . Review of treatment trials in human spinal cord injury: issues, difficulties, and recommendations. Neurosurgery 2006; 59: 957–982; discussion 982–987.
Burns AS, Lee BS, Ditunno Jr JF, Tessler A . Patient selection for clinical trials: the reliability of the early spinal cord injury examination. J Neurotrauma 2003; 20: 477–482.
Barros TE, Oliveira R, Barros EMK, Cristante AF, Marcon RM, Camargo A . Somatosensory evoked potential in the evaluation of the effects of 4-aminopyridine. In: American Spinal Injury Association 29th Annual Meeting; 4–6 April 2003; Miami, FL. J Spinal Cord Med 2003; 26 (Suppl 1): S33 (abstract).
Marino RJ, Barros T, Biering-Sorensen F, Burns SP, Donovan WH, Graves DE et al. International standards for neurological classification of spinal cord injury. J Spinal Cord Med 2003; 26 (Suppl 1): S50–S56.
Burns AS, Ditunno JF . Establishing prognosis and maximizing functional outcomes after spinal cord injury: a review of current and future directions in rehabilitation management. Spine 2001; 26 (24 Suppl): S137–S145.
Johansson CB, Momma S, Clarke DL, Risling M, Lendahl U, Frisén J . Identification of a neural stem cell in the adult mammalian central nervous system. Cell 1999; 96: 25–34.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cristante, A., Barros-Filho, T., Tatsui, N. et al. Stem cells in the treatment of chronic spinal cord injury: evaluation of somatosensitive evoked potentials in 39 patients. Spinal Cord 47, 733–738 (2009). https://doi.org/10.1038/sc.2009.24
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2009.24
Keywords
This article is cited by
-
A scoping review of trials for cell-based therapies in human spinal cord injury
Spinal Cord (2020)
-
Adult mesenchymal stem cells: is there a role for purine receptors in their osteogenic differentiation?
Purinergic Signalling (2020)
-
Translational considerations in injectable cell-based therapeutics for neurological applications: concepts, progress and challenges
npj Regenerative Medicine (2017)
-
Current status of cell-mediated regenerative therapies for human spinal cord injury
Neuroscience Bulletin (2014)
-
Evaluation of the effects of hyperbaric oxygen therapy for spinal cord lesion in correlation with the moment of intervention
Spinal Cord (2012)