Abstract
Study design:
Retrospective clinical study with 1-year follow-up.
Objectives:
Treatment of faecal incontinence by permanent sacral nerve stimulation (SNS) in patients suffering from cauda equina syndrome (CES).
Setting:
Klosterneuburg, Austria.
Background and purpose:
A flaccid paresis of the sphincter ani muscle and the pelvic floor combined with faecal incontinence can occur in patients suffering from CES as a result of a trauma in the region of the lumbar spine. If the incontinence cannot be managed by the use of laxatives or anal tampons, the patient is restricted in his/her quality of life. Thus, it was our aim to improve sphincter function and anal sensitivity to achieve voluntary rectal defaecation.
Materials and methods:
The functional integrity of at least one sacral root (S2–S4) was determined through percutaneous nerve evaluation (PNE). Following this analysis, a period of external temporary SNS was performed to evaluate the functional effect. If there was a decrease in the number of episodes of faecal incontinence during this evaluation period, a neurostimulation device (InterStim; Medtronic) was implanted.
Patients:
A total of 11 patients suffering from flaccid paresis of the anal sphincter muscle and faecal incontinence caused by CES underwent PNE, which was successful in 8 patients. Two of these patients were eliminated from the procedure at the end of the temporary SNS period, one patient refused the permanent implantation. Therefore, five patients proceeded to permanent implantation, which led to an improved continence in all the cases.
Discussion:
In the synopsis of the preoperative proctologic and neurological findings, successful electric stimulation of the sacral roots can be expected in incomplete CES. In the case of flaccid paresis of the anal sphincter muscles caused by an incomplete CES, permanent SNS offers a promising option for the treatment of faecal incontinence.
Similar content being viewed by others
Introduction
Cauda equina syndrome (CES) is a lesion of the peripheral nervous system. The neurological deficits can be classified by the ASIA (American Spinal Cord Injury Association) Classification.1 Clinically, it involves flaccid paresis of the striated muscles distal to the lesion, the pelvic floor and the bladder. Loss of function and tonus of the external sphincter as well as paraesthesia can also be seen.
Faecal incontinence due to spinal cord injury is sometimes overcome by attempting to fill the rectum at a defined time using laxatives. The spastic sphincter can retain the stool because of its tonus. With the help of local manipulation and suppositories, the reflex activity of the rectum can be provoked. Many paraplegic patients can thus reach manageable voluntary defaecation and avoid episodes of incontinence.
If the patient suffers from flaccid paresis and missing sphincter tonus, the stool cannot be retained in the rectum and passes without control. If other devices like tampons cannot seal the anal canal completely, severe incontinence results.
Some patients try to manage the situation by reducing their food intake. Both faecal incontinence and chronic constipation impair the quality of life of the patient.
Purpose: The aim of the procedure was to improve faecal continence by permanent electric sacral nerve stimulation (SNS). This is achieved by the patient recognizing the urgent need to defaecate earlier and by the prolonged muscular-controlled retention of the faeces.
Materials and methods
The InterStim therapy system (Medtronic Inc., Minneapolis, MN, USA; Figure 1)2 was used for SNS.
Percutaneous nerve evaluation
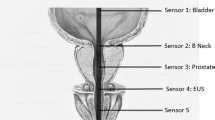
First, we performed a percutaneous nerve evaluation (PNE) of the sacral roots S2–S4 to assess their functional integrity. This was carried out under general anaesthesia without the use of relaxants. A spinal isolated needle (Foramen Needle, 20 gauge, 9 cm length, no. 041828, Medtronic) was inserted into the sacral foramina and connected to an external neurostimulator (Test stimulator, no. 3625-G, Medtronic). The sacral roots S2–S4 on both sides were then tested. Stimulation settings used in this procedure were pulse width of 210 μs and frequency of 25 Hz. The amplitude (2–8 V) was increased until a muscular response at the external sphincter as well as at the musculus levator ani could be recognized.
In all cases of adequate muscular response one to three needles were substituted by a temporary stimulator electrode (Test lead, no. 3057USC, Medtronic). The electrodes were connected to an external stimulator. As soon as the patient could communicate postoperatively, the stimulator was activated to an amplitude at which the patient reported the first sensory effect in the anal and perianal regions.
Evaluation period
For each electrode an average evaluation period of 6 days was performed. The stimulation parameters were unipolar monophase impulse, 210 μs pulse width, 25 Hz frequency and 2–8 V amplitude. The amplitude was controlled by the sensory perception of the patient. The patient had to record all the regular defaecations and the episodes of incontinence during the period of evaluation in a diary that was compared with the baseline values.
Permanent stimulation
Patients who showed an improvement of the functional outcome during the evaluation period proceeded to permanent implantation.
Under general anaesthesia with the patient in a prone position, the permanent electrode was inserted through the use of an introduction set (Quadripolar lead for electrical stimulation, no. 393, Medtronic). The exact location was controlled by means of electric stimulation. An incision was made next to the needle. The permanent neurostimulator (InterStim generator, no. 3023, Medtronic) was implanted into a subcutaneous pocket in the lower abdomen or in the buttock. With the help of a tunnelling tool and the use of an extension lead, the electrode was subcutaneously connected to the neurostimulator. The system was programmed using a telemetric device (N'VISION, no. 8840, Medtronic). The patient received a programming unit (Patient Programmer 3031A, Medtronic) and could steer the stimulation within a predetermined amplitude range or turn the device on and off.
Patients
The patients were selected on the basis of defined criteria. The neurological statement described a CES for at least 12 months. For inclusion, faecal incontinence had to be documented by a personal protocol and conservative treatment with laxatives and mechanical devices had to have failed. Clinical assessment and anal manometry also were required to provide evidence of reduced tonus of the sphincters.
All patients underwent neurological, urological, proctologic and internal examination, including colonoscopy. The patients were asked to write an exact incontinence protocol before and during the evaluation period and the Wexner Score (Cleveland Clinic Continence Score)3 was also recorded.
Percutaneous nerve evaluation was performed in 11 patients (6 men, 5 women; aged 46.6 years on average). The median history of incontinence following spinal trauma was 5.8 years (17–432 months). The neurological findings documented a CES in all cases. ASIA Classification was A and B each in one patient and D in all other patients (Table 1).
Results
During PNE (Table 2), three patients had no muscular response of the anal sphincters and no temporary SNS during the evaluation period.
With one patient, only one sacral root could be evaluated effectively, whereas with seven patients, two ore more sacral roots could be stimulated successfully. Therefore, we implanted two testing leads in seven cases, whereas in one case, only one electrode was used. An evaluation period of 6 days on average was performed for each electrode, and improvement of incontinence could be observed among all patients (Table 3). However, two patients had to be excluded from the procedure at the end of the evaluation period because of extremely low compliance, and one patient refused the implantation of the permanent stimulation device; however, his incontinence had already existed for 36 years.
Finally, the implantation of the permanent SNS system was carried out in five patients (three men, and two women). In all cases one tined lead electrode was implanted. The postoperative processes took place without any complications and all patients were followed up for a minimum of 1 year.
After the implantation of the permanent SNS system, the Wexner Score (Table 3) documented an improvement of faecal continence for all patients. Scores were reduced from a median of 15 preoperatively (9 min–19 max) to 5 (2 min–9 max) postoperatively. The postoperative limitation of the patients' continence was caused by gas incontinence. One patient described an intermittent incomplete continence for liquid stool.
We tried to document the quality of life by the use of the ‘Faecal Incontinence Quality of Life Scale’,4 but we had to recognize its limited applicability to paraplegic patients. Therefore, we interviewed the patients about their subjective perceptions. All patients experienced a considerable increase in their quality of life.
In one patient, the intensity and frequency of the stimulation could even be reduced without any loss of continence 1 year after the implantation, as an increase of the tonus of the sphincter muscle was documented.
Complications
One patient showed a minimal leakage of cerebrospinal fluid following the PNE procedure. This was presumably caused by malposition of the needle due to altered anatomy of the sacrum stemming from a previous fracture of the pelvis. After the removal of the needle, there were no further symptoms. No other complications were observed during the evaluation or following the implant procedure.
One patient described acute faecal incontinence 14 months after the procedure. The patient had inadvertently deactivated the neurostimulator by the use of her own remote control. As soon as we reactivated the system she was continent again.
Discussion
Although faecal incontinence has long been a taboo subject in our society, modern medicine has dealt with this problem more frequently and modern methods of treatment have been developed.5, 6 Permanent SNS has proven to be suitable for patients suffering from the so-called idiopathic faecal incontinence as well as in cases of neurological pathogenesis.
With respect to the treatment of paraplegic patients' faecal incontinence, laxatives and mechanical devices are primarily used to create manageable and controllable defaecation. For most of the patients suffering from spastic paraplegia due to a spinal cord injury, this procedure operates quite well although arbitrarily controllable defaecation is lost because of the disruption of the corticosacral track and constipation results, intact reflex defaecation remains.
It is much more complicated to control defaecation in CES patients with flaccid paresis: on the one hand, deliberate control of defaecation is disturbed because of the discontinuity of the sacral nerves, and on the other hand, reflex defaecation is not possible because the reflex circuit is destroyed.
Additionally, mechanical retention of the stool by the sphincter is impossible as the tonus is flaccid. This kind of faecal incontinence appears to be an indication for PNE of sacral roots S2–S4.
If the sacral part of the spinal cord or all the responsible sacral nerves are completely transected, the stimulation will not be effective. However, experience has shown that if at least one intact sacral nerve can be located and stimulated, faecal continence can be enhanced. In addition, it is quite difficult to distinguish between complete and incomplete CES. We could find positive reactions to the stimulation even though the clinical tests of the bulbocavernosus and anal reflexes were negative. We also had to recognize that ASIA Classification cannot give any information about the expected reactions to the stimulation. Therefore, the indication for the testing of the SNS should be considered more frequently in light of the current results.
All patients experiencing permanent SNS described an increase in perianal sensitivity and an improved deliberate retention of faeces. Within some months of permanent stimulation, augmentation of the sphincter tonus could also be noticed. Some factors seem to contribute to the effect of the permanent SNS in case of incomplete CES. On the one hand, the stimulation of the efferent tracts could cause a direct impact on the external sphincter. On the other hand, the stimulation of the afferent tracts intensifies the patient's sensitivity, and thus his ability to control defaecation via the corticosacral tracts. Additionally, the stimulation of the reflex circuits and reflex defaecation could be important factors. At the moment, the different tasks and functions of each component can only be discussed and require further research. Owing to clinical successes, permanent SNS seems to be an option for the treatment of faecal incontinence in patients suffering from flaccid paresis due to incomplete CES.
References
Ditunno JF, Young W, Donovan WH, Creasey G . The international standards booklet for neurological and functional classification of spinal cord injury. American Spinal Injury Association. Paraplegia 1994; 32: 70–80.
Matzel KE, Stadelmaler U, Hohenfellner M, Gall FP . Electrical stimulation of sacral spinal nerves for treatment of faecal incontinence. Lancet 1995; 346: 1124–1127.
Jorge JM, Wexner SD . Etiology and management of fecal incontinence. Dis Colon Rectum 1993; 36: 77–97.
Rockwood TH, Church JM, Fleshman JW, Kane RL, Mavrantonis C, Thorson AG et al. Fecal Incontinence Quality of Life Scale: quality of life instrument for patients with fecal incontinence. Dis Colon Rectum 2000; 43: 9–16.
Gourcerol G, Galla S, Michot F, Denis Ph, Leroi A . Sacral nerve stimulation in fecal incontinence: are there factors associated with success? Dis Colon Rectum 2007; 50: 3–12.
Person B, Wexner SD . Advances in the surgical treatment of fecal incontinence. Surg Innov 2005; 12: 7–21.
Acknowledgements
We thank the AUVA (Austrian Social Insurance for Occupational Risks) for financial support of this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gstaltner, K., Rosen, H., Hufgard, J. et al. Sacral nerve stimulation as an option for the treatment of faecal incontinence in patients suffering from cauda equina syndrome. Spinal Cord 46, 644–647 (2008). https://doi.org/10.1038/sc.2008.6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2008.6
Keywords
This article is cited by
-
Guideline for the management of neurogenic bowel dysfunction in spinal cord injury/disease
Spinal Cord (2022)
-
Leitlinie: Neurogene Darmfunktionsstörung bei Querschnittlähmung (Langfassung)
coloproctology (2020)
-
Stuhlinkontinenz
coloproctology (2017)