Abstract
The treatment of women’s health-related conditions, including reproductive tract and pregnancy disorders, faces distinct challenges and microenvironments, such as the vaginal mucosa and the placenta, which may be overcome by nanotechnology- and biomaterials-based drug delivery technologies. Here, we discuss the design and optimization of therapeutic nanoparticle and biomaterial systems to deliver drugs for the treatment of conditions in non-pregnant and pregnant women, including vaginal infections, reproductive tract disorders, pregnancy conditions and fetal congenital disorders. We highlight how drug delivery systems can be engineered for specific administration routes such as vaginal, in utero and intraperitoneal administration, to penetrate biological barriers such as the vaginal mucosa, and to provide sustained and efficient drug delivery at the site of interest. Although these delivery systems have mainly been explored in preclinical settings thus far, implementing women-specific design considerations in delivery technologies will allow their optimization and clinical translation for the treatment of women’s health conditions.
Key points
-
Several barriers, challenges and microenvironments that are unique to the female body must be considered in the design of delivery technologies for women’s health applications.
-
Biomaterial systems can be engineered to deliver drugs for the treatment of vaginal infections, gynaecological cancers, endometriosis, pregnancy disorders and congenital disorders in utero.
-
In addition to efficacy, the safety and toxicity of delivery systems are key design considerations for the treatment of women’s health-related conditions.
-
Appropriate and accessible preclinical models are essential for the clinical translation of delivery technologies for women’s health, but the physiological and pathophysiological differences amongst species must be considered.
-
The engineering of delivery systems for women’s health can build on delivery technologies that have shown clinical success in other disease applications.
Similar content being viewed by others
Introduction
Women’s health concerns the study and treatment of conditions that disproportionately affect women, including reproductive tract and pregnancy disorders. Although many delivery technologies have been explored and some clinically approved for various applications, including cancer, immunotherapy, vaccines and gene therapy, their application in women’s health remains limited1,2,3. This can be partially explained by the disparity between the preclinical funding and disease burden for women-specific disorders, compared with men-specific disorders4; for example, for almost 75% of disorders that affect primarily one gender, US federal funding favours men4. In addition, women’s health research must consider the complex social sensitivities in reproductive health and the mother and fetus as a single entity5. As a result, only 3.7% of all clinical trials from 2007 to 2020 focused on female gynaecology, demonstrating not only the preclinical challenges of women’s health research, but also the limited clinical translation of therapies6 (Box 1). This Review uses the terms ‘women’ and ‘woman’ throughout; we recognize that not all those who experience menstrual cycles, pregnancy and endometriosis identify as women.
Engineered biomaterial and drug delivery systems have many advantages over traditional drug formulations, including improved cargo stability, increased transport across cellular barriers, controlled release, and local administration and retention1,2,7,8,9. In 1991, Norplant was the first FDA-approved drug delivery system for women’s health10. Norplant is a subdermal contraceptive implant consisting of six silicone capsules encapsulating the synthetic hormone levonorgestrel. This technology was followed by the approval of the transdermal contraceptive patch Ortho Evra and the contraceptive vaginal ring NuvaRing in 200111,12.
Drug delivery platforms must consider the barriers, microenvironments and challenges specific to women’s reproductive health; for example, the vaginal mucosa can be exploited for potent local delivery to the reproductive tract through the ‘first uterine pass effect’ (direct vagina-to-uterus transport)13. In addition, the vaginal microbiota and high mucin content contribute to a complex microenvironment that must be considered when designing delivery technologies for vaginal administration14,15. Similarly, preferential delivery to the reproductive tract is essential for local or systemically administered platforms designed to treat endometriosis or gynaecological cancers. Moreover, thorough, long-term studies evaluating impacts on fertility are equally important. In pregnancy, the placenta is a complex barrier that can be exploited for selective delivery to the mother or fetus with carefully designed delivery systems16. Although the consideration of mother and fetus as a single entity during pregnancy is a complex social construct, the administration of drugs and biomaterials during pregnancy is suggested to have long-term developmental effects on the fetus that are still not completely understood17 (Box 1).
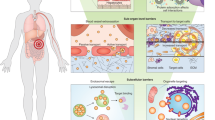
In this Review, we discuss engineered biomaterials and delivery technologies for reproductive health challenges in non-pregnant (Fig. 1a) and pregnant (Fig. 1b) women, with an emphasis on design considerations that exploit the biological barriers and microenvironments unique to women’s health. We highlight polymer, lipid and inorganic nanoparticles, as well as natural and synthetic biomaterial platforms, such as scaffolds and hydrogels that can deliver small-molecule drugs, such as chemotherapeutics and antibiotics, nucleic acids, such as small interfering RNA (siRNA) and messenger RNA (mRNA), and biologics, such as proteins and antibodies, for the treatment of vaginal infections, reproductive tract disorders, pregnancy-related conditions, and in utero therapies for fetal congenital disorders. Understanding women-specific biomaterial design considerations is key to the optimization and clinical translation of delivery technologies for the treatment of women’s reproductive health conditions.
a, Reproductive health challenges in non-pregnant women include endometriosis, gynaecological cancers and vaginal infections, including bacterial, fungal and sexually transmitted infections. b, Reproductive health challenges in pregnant women include pregnancy disorders, such as pre-eclampsia and fetal growth restriction, and fetal congenital disorders. Various delivery technologies and cargoes can be applied for the treatment of these conditions. ECM, extracellular matrix; miRNA, microRNA; mRNA, messenger RNA; NP, nanoparticle; pDNA, plasmid DNA; siRNA, small interfering RNA.
Delivery technologies in non-pregnant women
Vaginal infections
The vagina is a flexible, muscular canal that extends from the outside of the female genital area to the cervix (Fig. 2a). The vaginal lumen is lined with two distinct layers of mucus: the luminal mucus layer, which is rapidly cleared, and the adherent mucus layer, which is slowly cleared. Cervicovaginal mucus is composed primarily of water and contains only 5–8% of solid material, predominantly mucin glycoproteins, proteoglycans and lipids14. The primary purpose of cervicovaginal mucus is to trap and clear pathogens, preventing them from entering the body and infecting the underlying epithelium14.
a, The vagina extends from the cervix to the external genitalia, and its lumen is lined with two layers of cervicovaginal mucus: the luminal mucus layer, which is rapidly cleared, and the adherent mucus layer, which is more slowly cleared. b, Clinically used therapies for vaginal infections include oral medications, vaginal suppositories, creams, tablets and prophylactic vaginal rings for sexually transmitted infection. Delivery technologies such as nanoparticles (NPs), gels, nanofibres and films can improve the delivery and efficacy of clinically used therapeutics. c, Technologies for vaginal drug delivery should consider the vaginal mucus layers, as these may limit the penetration of the delivery platform into the underlying epithelium, which is often the target for vaginally administered therapeutics. Functionalizing nanoparticles with hydrophilic polymers, such as poly(ethylene glycol) (PEG), creates a water sheath around the nanoparticles that can aid in mucus penetration. Delivery technologies can be designed to be mucoadhesive, for example by formulation or functionalization with mucoadhesive molecules to improve adhesion to mucin. Nanoparticles can also be encapsulated in gel formulations to improve retention in the vaginal lumen.
Vaginal infections include bacterial infections such as chlamydia and gonorrhoea, fungal infections such as vulvovaginal candidiasis (the common yeast infection Candida albicans), and sexually transmitted viral infections such as human immunodeficiency virus (HIV), human papilloma virus (HPV) and herpes virus. Clinically approved treatments for these infections include oral antibiotics, antivirals and antifungals, as well as vaginal suppositories, creams, tablets and rings that act as drug depots (Fig. 2b). Oral treatments for vaginal infections have several limitations, including the high doses required to achieve therapeutically relevant levels of antibiotic or antifungal in the vaginal tissue.
Alternatively, local administration of suppositories and creams exploits several advantages of vaginal delivery, including avoiding first-pass clearance and dilution effects, which are commonly observed with systemically administered drugs18. Upon vaginal administration, the first uterine pass effect may allow preferential drug accumulation in the uterus and vagina through counter-current drug exchange between the blood in vaginal veins and the uterine artery19. The effect was first observed in a human ex vivo uterine perfusion model, in which high concentrations of vaginally administered, radioactively labelled progesterone accumulate in the uterine endometrium and myometrium20,21. In addition, higher plasma levels of vaginally administered progesterone are observed in the uterine artery, as compared with the radial artery, in post-menopausal women22. However, differences in the anatomy and physiology of the vagina amongst species raise concerns about whether the first uterine pass effect applies to all animal models23,24 (Box 2).
Vaginal delivery systems can also take advantage of the rich blood supply and high vaginal surface area, caused by the abundance of folds, which aid in drug absorption18. However, current clinically used vaginal suppositories and creams have limited efficacy owing to self-cleaning of the vagina that results in low residence time25. Therefore, engineered biomaterial and nanoparticle drug delivery systems can improve the treatment of vaginal infections.
Bacterial and fungal infections
Some of the most common vaginal bacterial infections include Neisseria gonorrhoeae (gonorrhoea) and Chlamydia trachomatis (chlamydia)25,26,27. The World Health Organization (WHO) estimates that over 100 million cases of gonorrhoea and over 80 million cases of chlamydia occur per year28. Fortunately, these infections are curable with antibiotics26. Local antibiotic administration in the vagina is preferred over oral administration, because it enables higher local drug concentrations and minimizes effects on the gastrointestinal tract microbiome26. However, drugs are quickly cleared from the vagina because of self-cleaning and leakage caused by gravity and walking (Table 1).
Bioadhesive delivery systems adhere to the vaginal mucosa, in particular to the mucin glycoproteins in the cervicovaginal mucus layers29, and thus improve drug residence time. The anionic glycosylated regions of mucin mediate bioadhesion through covalent bonding and ionic interactions with positively charged macromolecules such as chitosan29,30 (Fig. 2c). Therefore, nanoparticle delivery systems made of natural polymeric materials, such as chitosan, have been explored for treating vaginal bacterial infections25,27. For example, in vitro antibacterial activity has been demonstrated against several strains of gonorrhoea, including those resistant to multiple antibiotics, with unloaded chitosan microparticles, as chitosan itself has antibacterial activity27. Chitosan nanoparticles can also be loaded into polymeric nanofibre formulations made from polyvinylpyrrolidone, or gel formulations made from cellulose derivatives, such as hydroxypropyl methylcellulose (HPMC), to generate combination therapies with dual mucoadhesive effects31 (Fig. 2c). In an ex vivo model using sheep vaginal tissue, polyvinylpyrrolidone nanofibres and HPMC gels loaded with drug-containing chitosan nanoparticles increase mucoadhesion compared with free drug formulations31. Alternatively, liposomes encapsulating anti-inflammatory small molecule drugs can be loaded into mucoadhesive chitosan hydrogels, and have been shown to inhibit in vitro nitric oxide production in lipopolysaccharide-induced macrophages32. Moreover, antibiotic-loaded silicone emulsions have demonstrated mucoadhesive properties with in situ residence in the vaginal tissue of rats for up to 24 h following vaginal administration, and liquid crystal lipid emulsions have improved the in vitro antibacterial activity of ciprofloxacin against Escherichia coli, compared with free antibiotics, in infected reproductive tract epithelial cells18,26.
Candida albicans is responsible for vulvovaginal candidiasis, also known as the vaginal yeast infection. In their lifetime, 70–75% of women experience vulvovaginal candidiasis, most often treated with oral antifungal agents33,34,35. Oral treatments, however, are not ideal for vaginal infections, and antifungal agents, such as voriconazole and ketoconazole, have poor aqueous solubility, necessitating their encapsulation in delivery devices for local drug release and retention in the vagina33,35. Drug efficacy can be improved by using biomaterial-based delivery systems; for example, film and gel polymeric delivery systems formulated with chitosan show better inhibition of C. albicans, compared with free drug formulations, in vitro33,34. Similarly, a polymeric gel emulsion of miconazole improves antifungal activity against C. albicans infection in rats, compared with a clinically approved vaginal suppository formulation of the same drug36. Biomaterials allow control and tuning of the release of antifungal agents; for example, by altering the concentration of sodium alginate in polymer gel formulations, in vitro antifungal drug release in simulated vaginal fluid can be modulated37. Polymeric nanoparticle formulations can greatly improve the sustainability of drug release, compared with free drugs37,38. Polymeric gel and liposome delivery systems can also increase ex vivo or in vivo antifungal drug retention in vaginal tissues35,39; for example, vaginally administered liposomes preferentially accumulate in the reproductive tract of rats with similar cytokine levels to those of the control group, and show higher retention in porcine vaginal tissue ex vivo, compared with the free drug39. Importantly, using the rabbit vaginal irritation assay, the only FDA-approved animal model for testing vaginal irritation, the liposomes do not cause any histological changes in the vaginal tissue39. Of note, safety and toxicity of vaginally administered therapeutics have to be evaluated, because biomaterial-induced irritation or inflammation can make the vagina more susceptible to additional infections39. Together, delivery devices can improve the solubility, antifungal activity, sustained release and vaginal retention of antifungal agents, compared with free drug formulations.
Sexually transmitted viral infections
Unlike for bacterial and fungal infections, for which antibiotics and small molecules have mainly been explored, various cargos have been investigated for the prophylactic treatment of viral infections, including antivirals40,41,42,43,44, nucleic acids45,46,47,48, biologics49 and small molecules50,51. Delivering nucleic acids, such as siRNA, is particularly challenging for vaginal infections owing to the dependence on charge-based electrostatic interactions for many delivery systems and the acidic pH (3.5–4.0) of the vagina45,52. In vivo siRNA-mediated knockdown of enhanced green fluorescent protein (EGFP) has been achieved in the murine vaginal mucosa by using poly(lactic-co-glycolic acid) (PLGA) nanoparticles47; here, spermidine was used as a low-molecular-weight cationic agent to complex the negatively charged siRNA and improve cargo loading in the PLGA nanoparticle by >40-fold47,48. However, nucleic acid delivery systems typically contain a cationic or ionizable component to enable endosomal escape of the nucleic acid at acidic pH2,53. To avoid premature cargo release in the acidic environment of the vagina, a pH-responsive film can protect solid lipid nanoparticles encapsulating siRNA45. In simulated vaginal fluid, the nanoparticle-loaded film demonstrates little to no release of solid lipid nanoparticles at acidic pH, but shows increased cargo release with increasing pH45. This system could be applied for anti-HIV prophylaxis, ensuring drug release only on exposure to seminal fluid during heterosexual intercourse, whereby vaginal pH increases toward neutral.
Drug penetration through cervicovaginal mucus layers into the underlying epithelium can be improved by functionalization of polymeric nanoparticles with hydrophilic polymers, such as poly(ethylene glycol) (PEG)54,55,56 (Fig. 2c). Such mucus-penetrating particles show higher vaginal retention and coverage in mice, compared with non-functionalized nanoparticles56. Here, oestradiol was used to induce the oestrus phase in mice before vaginal administration of nanoparticles to better mimic the cervicovaginal mucus of humans56 (Box 2). These mucus-penetrating particles also better protect mice against a vaginal herpes simplex virus 2 challenge, compared with the soluble drug56. In addition, unlike mucus-penetrating nanoparticles, vaginal administration of non-functionalized nanoparticles causes neutrophil infiltration into the vaginal lumen, indicating better biocompatibility of PEG-functionalized particles56. PEG-functionalization also improves the ex vivo penetration in sheep vaginal epithelium of liposomes that deliver drugs against HPV infection57, and the in vivo retention in murine vaginal tissue of combination therapies that contain PEGylated nanoparticles and mucoadhesive polymeric fibres58. Alternatively, hyperbranched polyglycerols can improve the bioadhesion of polymeric particles encapsulating an antiretroviral small molecule59, resulting in nearly 5 times as much retention of the nanoparticles, compared with the non-adhesive control nanoparticles, in the murine reproductive tract 24 hours after vaginal administration59. Therefore, biomaterials can address several delivery challenges for vaginal infection, including mucus adhesion and penetration, and vaginal retention and stability in the acidic vaginal microenvironment.
Reproductive tract disorders
The female reproductive tract consists of the vulva, vagina, cervix, uterus, fallopian tubes and ovaries. Endometriosis and gynaecological cancers are two major disorders affecting the female reproductive tract. Endometriosis involves the formation of benign lesions outside the reproductive tract, sharing many physiological traits with malignant gynaecological cancers. For example, both endometriotic and cancerous lesions are invasive and can induce inflammation and angiogenesis60, which can cause high rates of relapse.
Endometriosis
Endometriosis is a hormone-dependent gynaecological disorder, defined as the presence of endometrial tissue outside the uterus. Endometriosis affects about 10–15% of women at the reproductive age61,62, and numerous endometriosis-related complications can occur, including chronic pelvic pain and infertility; however, there are currently no curative treatments for endometriosis62. Clinical treatment typically includes hormonal therapies to suppress oestrogen synthesis, small-molecule drugs to manage pain, and surgical intervention by laparoscopy to remove endometriotic lesions60. However, hormonal therapy can have long-term and systemic adverse side effects, and endometriotic lesions recur in 40–50% of women who have undergone surgery62.
Polymeric implantable drug delivery systems can mediate sustained drug release from the peritoneum into the uterus and to endometriotic lesions, with sustained in vitro release of small-molecule drugs for up to 30 days62,63. In addition to locally administered systems, selective and active targeting approaches have been explored for the treatment of endometriotic lesions. Selective drug delivery, for example by distinct cargo selection and design, to ectopic endometriotic lesions rather than healthy endometrial tissue is essential to minimize negative effects on female fertility64 (Table 1). A polymer/nucleic acid conjugate comprising miR-200c, a microRNA (miRNA) significantly downregulated in ectopic endometriotic lesions compared with normal endometrial tissue, reduces ectopic endometrial cyst volume in an autotransplantation rat model of endometriosis65. miR-200c interacts with the untranslated regions of mRNA to trigger translational repression or mRNA degradation, thereby suppressing the proliferation and migration of endometrial cells, and reducing the volume of the cyst. Similarly, micelle systems and polymeric or peptide conjugates can deliver small-molecule drugs or siRNA against targets that are upregulated in endometriotic lesions66,67,68; for example, overexpression of low-density lipoprotein (LDL) in endometriotic lesions can be targeted by lipid nanoparticles that resemble the native structure of LDL69. These particles can deliver small-molecule drugs specifically to endometrial tissues69. Alternatively, active targeting enables preferential localization of delivery technologies in endometriotic tissue; for example, silver nanoparticles functionalized with peptides that recognize extracellular matrix (ECM) receptors upregulated in endometriotic lesions enable the targeted delivery of antimitotic drugs60. Similarly, peptide–siRNA conjugates can target receptors that are critical to the development of endometriosis, thereby inhibiting the migration and invasion of endometrial cells outside the uterus60,64. Iron oxide nanoparticles coated with peptides that bind to vascular endothelial growth factor receptor 2 selectively accumulate in endometriotic lesions, generating sufficient ablation temperatures on exposure to an alternating magnetic field to completely eradicate endometriotic lesions in a mouse xenograft model of endometriosis70 (Box 2).
Inorganic nanoparticle platforms can also be used as imaging contrast and photothermal therapy agents for the identification and treatment of endometriotic lesions71,72; for example, hyaluronic-acid-modified iron oxide nanoparticles aided in the visualization of endometriotic tissue using magnetic resonance imaging, two hours after intravenous nanoparticle administration, in an autotransplantation rat model of endometriosis71. Nanoparticles can further improve surgical resection of lesions; for example, PEG-poly(caprolactone) (PCL) polymeric nanoparticles encapsulating silicon naphthalocyanine allow real-time near-infrared fluorescence imaging and photothermal therapy of endometriotic lesions72, demonstrating demarcation, and, on exposure to near-infrared light, complete eradication of endometriotic lesions transplanted from rhesus macaques into immunodeficient mice. The effectiveness and clinical relevance of such nanoparticle platforms depend on the specificity of markers to the ectopic endometriotic lesions of interest.
Gynaecological cancers
Gynaecological cancers include ovarian, cervical, uterine, vaginal and vulval cancer. Bioengineering research has thus far mainly focused on developing therapeutics for ovarian and cervical cancer. With the highest mortality rate of all gynaecological cancers, ovarian cancer also has a high rate of recurrence and a five-year survival rate of less than 50%73,74,75, partly owing to the asymptomatic nature of stage I and stage II ovarian cancer, resulting in 75% of diagnoses occurring at late stages76. Cervical cancer is more common in developing regions, where there is limited access to prophylactic vaccines against human papilloma virus (HPV) infection, which is highly associated with the development of cervical cancer77,78,79. However, owing to the availability of the Papanicolaou (Pap) test, early detection of cervical cancer is more feasible and accessible than for ovarian cancer80 (Box 1). Chemotherapeutic regimens for ovarian cancer often involve intravenous co-administration of a free platinum-based drug, such as cisplatin, and a taxane chemotherapeutic, such as paclitaxel; these chemotherapeutic regimens typically supplement surgical resection of the ovary or tumour75. Similarly, clinical treatment of cervical cancer involves surgery or radiotherapy for early-stage cancer, with the addition of chemotherapy for late-stage disease78.
Targeted delivery can improve the localization and efficacy of drugs against reproductive cancerous tissues and minimize their off-target toxicity that may impact fertility. For example, nanoparticles can be functionalized with antibody or peptide motifs for selective localization to a specific tissue or cell type9. For the treatment of ovarian and cervical cancer, delivery systems can be functionalized with epidermal growth factor receptor (EGFR) antibodies, human epidermal growth factor receptor 2 (HER2) antibodies, arginine–glycine–aspartic acid (RGD) peptides, luteinizing-hormone-releasing hormone receptor (LHRH) peptides, cell-penetrating peptides and single-stranded DNA aptamers73,74,75,78,79,81,82,83; for example, PCL nanoparticles modified with LHRH peptides increase drug localization within the tumour, compared with the free drug and non-targeted nanoparticles, in a mouse xenograft model for ovarian cancer74. Alternatively, through layer-by-layer assembly84,85,86,87, the core or surface of nanoparticles can be modified with hyaluronic acid, which binds to CD44 receptors on ovarian cancer cells84,86.
To address the recurrence of ovarian and cervical cancer, delivery systems can be designed to achieve sustained release of encapsulated drugs (Table 1); for example, chemotherapeutics can be encapsulated in injectable or implantable hydrogel formulations to reduce side effects and tumour size with a single local hydrogel injection, compared with multiple injections of free drug88,89,90. In a mouse xenograft model for ovarian cancer, tumour burden can be reduced with a single intratumoural injection of a platinum-based, thermosensitive polymeric gel system loaded with paclitaxel, compared with multiple systemic injections of free chemotherapeutic88. Local injection or implantation, such as intraperitoneal administration, can further minimize the dilution effect of therapeutic cargo as well as off-target side effects that may result from systemic administration91; for example, an alginate microparticle capsule encapsulating cells that stably express interleukin-2 (IL-2) can be intraperitoneally injected for ovarian cancer cytokine immunotherapy91,92, achieving therapeutically relevant levels of IL-2 in the peritoneal fluid for 15 days post-capsule administration and near complete eradication of tumours in a murine xenograft model of advanced ovarian cancer. Alternatively, drugs can be intravaginally delivered for cervical cancer treatment, which requires distinct design considerations for the delivery system, including drug retention in the vagina and penetration through the mucus to the underlying vaginal epithelium; in particular, nanoparticle surface functionalization and drug encapsulation in a hydrogel formulation can improve mucoadhesion, mucus penetration and retention for cervical cancer applications80,93,94.
In addition, new cargos are being explored to avoid drug resistance for recurrent gynaecological cancers. For example, siRNA, photosensitizers for photothermal therapy, microRNA inhibitors, and small-molecule drugs, such as poly(ADP-ribose) polymerase inhibitors, curcumin and histone deacetylase inhibitors77,86,95,96,97,98, can be delivered in combination with chemotherapeutics; however, new animal models for recurrent ovarian and cervical cancer would be needed to investigate their efficacy and to evaluate repeated dosing and drug resistance.
Delivery technologies in pregnant women
Pregnancy-related conditions
Conditions that develop during pregnancy are complex and difficult to treat owing to the synergistic effects on the mother, fetus and placenta. The placenta is an organ and biological barrier unique to pregnancy that develops in the uterus in early gestation and mediates exchange of nutrients, oxygen and waste between maternal and fetal circulation16 (Fig. 3a). On the maternal side of the placenta is the decidua, which is a thick mucosal membrane that contains placental immune cells and regulates cell invasion into the uterus99,100. In a process called decidualization, the decidua forms following implantation of the blastocyst into the uterine wall and induces remodelling of uterine tissue. The maternal spiral arteries extend from the uterine wall through the decidua and supply blood to chorionic villi — the site of blood exchange between maternal and fetal circulation101. In healthy pregnancies, extravillous trophoblasts, one of the main cell types in the placenta, mediate spiral artery remodelling by invading the decidua and uterine wall to create a high-blood-flow environment99,102. Insufficient spiral artery remodelling and placentation can result in placental insufficiency disorders, such as pre-eclampsia99 (Fig. 3b).
a, The placenta is an organ of both maternal and fetal origin that develops during early gestation to supply nutrients and oxygen to the fetus by mediating exchange between maternal and fetal circulation. The decidua is the first layer of the placenta located on the maternal side, which forms upon implantation of the blastocyst in the uterine wall through a process called decidualization. Remodelling of the uterine tissue allows maternal spiral arteries to extend into the placenta and to supply blood to the chorionic villi. These villi are the site of oxygen and nutrient transport to the fetus, and they separate the maternal and fetal blood spaces by means of layers of villous trophoblasts (the syncytiotrophoblast and cytotrophoblasts) as well as fetal endothelial cells. b, In healthy pregnancies, extravillous trophoblasts invade the decidua and uterine myometrium, remodelling the spiral arteries to create a high-blood-flow environment to meet the oxygen and nutrient demands of the fetus. In pre-eclampsia and fetal growth restriction, inefficient invasion of extravillous trophoblasts into the uterus and decidua results in lower blood flow in the placenta, which can lead to maternal hypertension and lower fetal birth weights. On a cellular level, dysregulation in angiogenic factors, such as vascular endothelial growth factor (VEGF) and placenta growth factor (PlGF), also plays a role in placental insufficiency disorders. Upregulation of soluble fms-like tyrosine kinase 1 (sFlt-1), the soluble version of the VEGF and PlGF receptor, is observed in the serum of pre-eclamptic women, which decreases the bioavailability of VEGF and PlGF to mediate vasodilation in the placenta. c, Rodents, hamsters, rabbits, guinea pigs, non-human primates and humans have haemochorial placentas, which are defined by the presence of trophoblast-lined chorionic villi that are directly exposed to maternal blood. However, the nature of trophoblast cell layer(s) differs amongst species, which can affect the utility of these animal models as representations of human conditions during pregnancy. Panel a adapted from ref. 103, Springer Nature Limited.
The placenta can be exploited as a biological barrier for selective therapeutic delivery to the mother, fetus or placenta during pregnancy (Table 1). However, to treat maternal, fetal or placental disorders with drug delivery systems, their behaviour and placental transport during pregnancy must be well characterized.
Understanding drug and nanoparticle biodistribution in pregnancy
Besides mediating nutrient and oxygen transport between the mother and fetus, the placenta also minimizes the transport of toxins from maternal to fetal circulation101. This is achieved through a complex layer of cells, including the syncytiotrophoblast, cytotrophoblasts and fetal endothelial cells that separate maternal and fetal blood spaces in the chorionic villi103 (Fig. 3a). As gestation proceeds, cytotrophoblasts progressively fuse into the syncytiotrophoblast, which is a continuous, epithelial syncytial structure that serves as the endocrine portion of the placenta by producing vascular endothelial growth factor (VEGF) and placental growth factor (PlGF) to mediate vasodilation and angiogenesis104,105,106. Although this layer of trophoblasts and fetal endothelial cells functions as the barrier between maternal and fetal blood spaces, many small-molecule drugs can transport across the placenta, and some can induce fetal malformations16,107,108. For this reason, pregnant women are often advised to restrict their intake of small molecules, including antiepileptic drugs, depression and anti-anxiety medications, antibiotics and chemotherapeutics, owing to potential harmful effects on fetal development. However, discontinuing these medications can also have detrimental effects on pre-existing maternal disorders when unmanaged during pregnancy109. To address this challenge, drugs can be conjugated to polymers to increase their size and limit placental transport107,110; for example, a PEG–drug conjugate shows less placental uptake than the free drug in an ex vivo human placental explant model107, demonstrating the potential of delivery systems to reduce placental transport and increase drug localization in maternal circulation.
Reducing placental transport is not the only consideration for minimizing fetotoxic effects. Indirect effects can also occur, including maternal-mediated fetotoxicity and placental-mediated fetotoxicity, where delivery of foreign materials to maternal organs or the placenta can induce oxidative stress and consequently inflammation, which can impair fetal development17 (Table 1). Therefore, in addition to evaluating direct fetotoxicity (that is, the presence of the drug or delivery technology in fetal tissues), these indirect fetotoxic effects must be evaluated to ensure the safety of the platform before clinical translation.
The pharmacokinetics and biodistribution of drugs during pregnancy can be modulated by encapsulating drugs in nanoparticles. Here, the physicochemical properties of the nanoparticles, such as size, charge and surface modification, have a substantial impact on their biodistribution during pregnancy111,112,113,114,115,116; for example, 15-nm gold nanoparticles have higher accumulation in placental tissue than 150-nm gold nanoshells113, and increasing the molecular weight of elastin-like polypeptide drug conjugates from 25 kDa to 86 kDa can also improve accumulation in the placenta110. Regarding charge, cationic polymeric nanoparticles have higher accumulation and penetration in placental cells than do more anionic delivery systems114,115, and surface modifications, such as PEGylation, carboxylation or addition of targeting moieties, can improve cellular uptake in or transport across the placenta111,112,116.
In addition, gestational age has a substantial impact on the ability to deliver drugs to the placenta, transport across the placenta to the fetus, and consequently fetal survival113,114,117, which may be explained by the physiological changes in placental development that occur during gestation. For example, in humans, the surface area of chorionic villi and the volume of the intervillous space increase throughout gestation to increase the efficiency of exchange between maternal and fetal circulation118. For similar reasons, the thickness of the syncytiotrophoblast and fetal endothelium decreases during the last two trimesters. Therefore, differences in placental structure and development between species are a major consideration for selecting the most appropriate preclinical model to study delivery technologies for pregnancy applications. Humans have haemochorial placentas, defined by the presence of trophoblast-lined chorionic villi that are directly exposed to maternal blood119 (Fig. 3a), limiting the use of large species, such as pigs and sheep (with epitheliochorial placentas), and dogs (with endotheliochorial placentas), as appropriate preclinical models for pregnancy applications119,120. By contrast, rodents, guinea pigs, rabbits and non-human primates have haemochorial placentas and are thus common preclinical models for studying placental development, delivery and transport119 (Fig. 3c). In particular, humans, non-human primates and guinea pigs have haemomonochorial placentas with a single layer of syncytiotrophoblast in the chorionic villi. Conversely, rabbits have haemodichorial placentas with one complete layer of cytotrophoblast cells in addition to the syncytiotrophoblast layer, whereas rats, mice and hamsters have haemotrichorial placentas with three distinct trophoblast layers119. These differences in placentation must be considered when evaluating therapeutics for pregnancy, particularly when studying placental transport; the presence of additional cell layers in rodent placentas is likely to affect the extrapolation of results from these models to humans (Box 2).
Preterm birth
Preterm birth is defined as the premature delivery of the fetus, before 37 weeks of gestation19,121,122. Globally, an estimated 15 million babies are born prematurely every year, and preterm birth is the leading cause of neonatal mortality and morbidity19,122. Like many conditions that develop during pregnancy, preterm birth is a multifactorial disorder, with potential causes including cervical insufficiency, intrauterine inflammation and premature onset of uterine contractions123,124.
Cervical insufficiency resulting in a shortened cervix has been associated with an increased risk of preterm birth and is often treated with cervical cerclage, whereby the cervix is supported and closed with a stitch placed vaginally or abdominally121,123. However, cerclage therapy has the risk of cervical tissue damage or laceration, in particular if the cerclage is present during labour121,125. As cervical cerclage does not address the underlying pathogenesis of cervical insufficiency, that is, the impaired mechanical properties of the fibrous connective tissue of the cervix, preterm birth can still occur following cerclage therapy121,125. To address tissue softening in cervical insufficiency, silk-fibroin-based hydrogels can be injected directly into the cervix to increase cervical tissue volume121,123,125. Such hydrogels promote the viability and proliferation of cervical fibroblasts in vitro, and lead to a substantial increase in the cross-sectional area of the cervix in pregnant rabbits121,125. In addition to increasing tissue volume, the silk hydrogel may also improve resistance against stresses that act to dilate the cervix and induce preterm birth125.
Progesterone is the only FDA-approved therapeutic agent for treating preterm birth126, and can either be used alone or to supplement cerclage therapy. Since its approval in 1956, intramuscular injection of synthetic progestin had been administered to millions of pregnant women in the United States, but progestin was removed from the market in 1973 owing to a lack of evidence in preventing preterm birth127. In 2011, the FDA approved Makena for the treatment of preterm birth after a randomized, double-blind clinical trial with 463 women demonstrated a reduction in the risk of recurrent preterm birth with synthetic progestin128. However, in 2020, the FDA proposed to withdraw approval for Makena after a second clinical trial with 1,708 women demonstrated no significant differences in neonatal outcomes compared with the placebo group126,128. In 2022, an increased risk of cancer was reported in the offspring of women treated with intramuscular synthetic progestogen, further raising concerns over the safety of the therapeutic127. Therefore, delivery technologies are required that enable selective progestogen delivery to the uterus and cervix with minimal negative effects on fetal health.
As an alternative to systemically administered Makena, Crinone is a commercially available, vaginal progesterone gel for the treatment of reproductive disorders, which has been used off-label as a prophylactic drug for preterm birth126. As a vaginal gel, Crinone can take advantage of the first uterine pass effect, resulting in selective drug accumulation in the uterus126. To further improve drug accumulation in the uterus and prevent preterm birth, polymeric nanoparticles can be loaded in gel systems to deliver progesterone or histone deacetylase inhibitors19,126. These platforms show more effective prevention of preterm birth in progesterone antagonist or lipopolysaccharide-induced murine models of preterm birth, compared with Crinone19,126. In addition, the expression of inflammatory cytokines, such as interleukin (IL)-6, IL-1β, and cyclooxygenase (COX)-2, increases in the uterine myometrium in mice treated with the Crinone vaginal gel, compared with the nanoparticle-in-gel formulation, indicating potentially better safety profiles19. Therefore, biomaterial delivery systems may prolong drug release, increase retention and improve stability, compared with free drugs.
Systemically administered delivery systems encapsulating small-molecule drugs are also being explored for the treatment of other conditions associated with preterm birth, including intrauterine inflammation and premature contractions. For example, dendrimer nanoparticles encapsulating N-acetyl-L-cysteine, an antioxidant small molecule with anti-inflammatory properties, can reduce the rate of preterm birth and T-cell immune infiltration to the placenta in a lipopolysaccharide-induced murine model of preterm birth124. Alternatively, the tocolytic agent indomethacin can be delivered in targeted liposomes to inhibit uterine contractions122,129. Indomethacin is a small molecule and member of the non-steroidal anti-inflammatory drug family, and can freely cross the placenta; however, indomethacin has also been associated with negative fetal development side effects and adverse effects on maternal cardiovascular health122,129. Encapsulating the drug in targeted liposomal nanoparticles, typically about 200 nm in size, can minimize drug transport across the placenta and decrease systemic drug dosing. The liposomes can be further functionalized with an antibody against the oxytocin receptor, which is highly expressed in the uterus of pregnant women122,129, to improve nanoparticle localization in the mouse uterus, compared to non-targeted systems, and to reduce accumulation in the fetus, compared with the free drug control122,129. To promote clinical translation of these liposomes, however, their stability in vivo needs to be improved to limit premature drug release before uterine localization and to prevent free drug transport across the placenta that may harm the fetus.
Pre-eclampsia and fetal growth restriction
Pre-eclampsia and fetal growth restriction (FGR) are multifactorial placental disorders that affect 5–8% and 7–15% of all pregnancies worldwide, respectively130. Pre-eclampsia is defined as the onset of hypertension and proteinuria after 20 weeks of gestation, posing a risk of seizures, pulmonary embolism, renal failure, liver dysfunction, and maternal and fetal death131. Clinical treatments for pre-eclampsia, such as antihypertensive drugs to manage blood pressure and anticonvulsants to prevent seizures131, only address the associated symptoms rather than placental dysfunction, which is often the underlying cause. The only curative treatment option for pre-eclampsia is the induction of labour and early delivery of the placenta and fetus, which often resolves maternal hypertension within a few days132. However, early induction of labour can cause fetal morbidity and mortality, especially if FGR occurs concurrently with pre-eclampsia. In FGR, the fetus fails to achieve their full growth potential in utero, which can lead to impaired respiratory development and higher chance of infection, as well as long-term risks of cardiovascular disease and type 2 diabetes132,133.
Preclinical therapeutics that address placental dysfunction, underlying these disorders, are often administered systemically, aiming to mitigate impaired vascularization by targeting placental trophoblasts and endothelial cells134,135. Intravenous administration is often preferred for therapeutic delivery to the placenta owing to the cardiovascular changes that occur during pregnancy, including a 45% increase in total cardiac output by 24 weeks of gestation, compared with non-pregnant individuals136. Of the total cardiac output, 20–25% represents blood flow to the uterus and placenta, demonstrating the potential for high localization of intravenously administered delivery systems in the placenta137.
Nanoparticles made of polyamidoamine (PAMAM) and encapsulating soluble fms-like tyrosine kinase 1 (sFlt-1) siRNA can exploit passive placental targeting during pregnancy for treating pre-eclampsia138, by knocking down sFlt-1, which is an antiangiogenic factor upregulated during pre-eclampsia and secreted by the placenta138. sFlt-1 is the soluble receptor for VEGF and PlGF and acts by binding to the receptor binding domains of these growth factors, preventing their ability to interact with trophoblast or endothelial cell surface receptors and resulting in vascular dysfunction104 (Fig. 3b). The PAMAM nanoparticles can reduce maternal blood pressure, proteinuria and serum sFlt-1 levels in an induced tumour necrosis factor alpha (TNF-α) rat model of pre-eclampsia138, without gross morphological changes in the histological analysis of the major organs. Of note, as pre-eclampsia is believed to occur spontaneously only in humans and non-human primates, preclinical models generated with the administration of exogenous agents can often only model portions of the disorder (that is, exacerbated maternal immune response)139 (Box 2). Alternatively, gene therapies for angiogenic factors, such as insulin-like growth factor (IGF), can be delivered to treat pre-eclampsia and FGR133,140; for example, a diblock copolymer delivery system encapsulating IGF plasmid DNA that possesses the additional design feature of trophoblast-specific promoters133,140 achieves trophoblast-specific gene expression in immortalized trophoblast cell lines, with the potential to reduce off-target effects compared with other systemically administered nanoparticle technologies.
To improve nanoparticle uptake in the placenta in the treatment of pre-eclampsia and FGR, delivery systems can be modified with placenta-specific targeting moieties; for example, peptide-decorated nanoparticles can deliver cargos such as miRNA inhibitors, nitric oxide donor, epidermal growth factor and IGF132,141,142,143. In addition to new synthetic peptide sequences, peptides targeting chondroitin sulfate A, a protein present on the surface of placental trophoblasts, can be used to localize nanoparticles in the placenta131,144; for example, chondroitin-sulfate-A-targeted polymeric nanoparticles encapsulating sFlt-1 siRNA decrease the circulating levels of sFlt-1 in a genetically engineered mouse model of pregnancy-associated hypertension, compared with non-targeted nanoparticles, and decrease the rate of maternal and neonatal mortality144. These preclinical studies demonstrate that nanoparticles and biomaterials can deliver a variety of therapeutics to treat placental disorders, such as pre-eclampsia and FGR, and may allow the design of combination therapies by combining multiple delivery systems
In utero therapies for congenital disorders
Advances in prenatal diagnostics, such as genetic testing through detection of cell-free fetal DNA in maternal circulation and high-resolution ultrasound, have enabled the early diagnosis of genetic disorders and birth defects in utero145,146, including haemoglobinopathies such as β-thalassaemia, congenital lung disorders such as cystic fibrosis and diaphragmatic hernia, and spinal defects such as spina bifida145,147,148. Prenatal diagnostics can identify these disorders early in gestation; however, in utero treatment options for haemoglobinopathies and congenital lung disorders remain elusive. In the prenatal treatment of myelomeningocele, which is the most common form of spina bifida, the Management of Myelomeningocele Study demonstrated the benefits of prenatal surgical intervention, compared with postnatal surgery149. However, both open surgical repair and fetoscopic surgery may lead to maternal complications and increased occurrence of premature rupture of membranes148,149. Therefore, biomaterials and delivery systems are being explored for in utero intervention.
Gene therapies
In utero gene therapy may enable protein replacement and gene editing therapy to treat congenital disorders, such as β-thalassaemia or cystic fibrosis, before birth150. Compared with postnatal treatment options, in utero therapies have the advantage that the tolerogenic fetal immune system and small fetal size allow treatment with foreign materials with minimal fetal immune response and maximal dosing per fetal weight145,151. Thus far, viral platforms have mainly been explored for in utero gene therapy owing to their well-studied, efficient gene transduction151,152. However, viral platforms can potentially lead to ectopic integration into offsite regions of the genome and may be immunogenic for the mother147,153. Alternatively, non-viral delivery technologies may enable in utero gene therapy.
Only a few nanoparticle delivery technologies have been developed for in utero gene therapy thus far (Fig. 4a, Table 1). Nanoparticles encapsulating therapeutics can be intravenously administered to the fetus by injection into the vitelline vein, which drains directly into the fetal liver, the site of hematopoeisis146,147. This administration route simulates umbilical vein blood transfusions, which have been performed safely in humans since the 1980s147. Both polymeric and lipid nanoparticles (LNPs) can deliver nucleic acids to the fetal liver through intravenous administration; for example, peptide nucleic acids and donor DNAs can be delivered by intravenously injected PLGA nanoparticles to correct a mutation in the β-globin gene in a mouse model of β-thalassaemia147. Importantly, both polymeric and lipid-based delivery platforms have demonstrated minimal toxicity for the dam, as assessed by cytokine analyses. In addition, they have no effects on fetal survival. Non-hepatic delivery to the fetal lung, intestines and brain may also be achievable145,146,147.
a, In utero gene therapy allows treatment of congenital disorders before the onset of irreversible disease pathology through protein replacement therapy or gene editing therapeutics. These nucleic acid cargos are encapsulated in nanoparticle delivery systems and can be administered through the vitelline vein or by intra-amniotic injections in utero. Vitelline vein injections are comparable to umbilical vein transfusions performed clinically and provide direct access to the fetal liver to treat congenital disorders, such thalassaemia and anaemia, by targeting hematopoietic stem cells (HSCs). Alternatively, intra-amniotic injections take advantage of fetal breathing and swallowing of the amniotic fluid to deliver gene therapeutics to the fetal lungs and treat disorders such as cystic fibrosis and surfactant protein syndromes. b, Delivery technologies can also be applied to treat congenital structural abnormalities, including spina bifida, in utero. For example, cell-seeded polymeric and extracellular matrix (ECM) scaffolds, natural and synthetic polymer patches, and drug-loaded alginate microparticles can be used to protect and repair spinal defects associated with these congenital disorders. RBC, red blood cell.
Alternatively, intra-amniotic administration of gene therapeutics mimics amniocentesis procedures, which are used clinically to sample amniotic fluid during gestation for genetic testing147 (Fig. 4a). This route of administration provides the opportunity to target organs, such as the fetal lung and intestine, that are challenging to reach postnatally through systemic administration routes145,147,150. Intra-amniotic administration takes advantage of fetal breathing and swallowing of the amniotic fluid, but requires consideration of the stability and dilution of delivery platforms in the large volume of amniotic fluid150,151. The stability of LNPs in amniotic fluid can be assessed through ex utero screening of different LNP formulations in a series of amniotic fluids, with the aim to achieve mRNA-mediated in utero protein replacement therapy150. mRNA LNP formulations that are stable ex utero in amniotic fluid mediate higher in utero protein expression compared with unstable LNP formulations150. Thus, the material composition of nanoparticles should be optimized to prevent aggregation and degradation in the protein-rich environment of amniotic fluid. Moreover, cell-level delivery of polymeric nanoparticles has been explored in fetal lungs, including fetal lung epithelial cells, which are the target population for treating cystic fibrosis and surfactant protein syndromes in utero145. Polymeric nanoparticles (250 nm) of polyamine-co-ester (PACE) administered intravenously to gestational-age E15 fetal mice enabled accumulation in up to 50% and 44% of lung epithelial and endothelial cells, respectively145.
Transplacental delivery of non-viral delivery platforms provides an alternative to vitelline vein or intra-amniotic administration to reach the fetus in utero154,155. For example, delivery technologies encapsulating nucleic acids can be systemically administered to the pregnant dam through tail vein injections; here, delivery to the fetus relies on transport across the placenta. Plasmid DNA–lipid conjugates can cross the placenta and deliver the plasmid to fetal hearts in a transgenic mouse model, which can be quantified by knockout of EGFP155. Similarly, transplacental delivery of antibody-targeted liposomes encapsulating reporter plasmid DNA leads to luciferase expression in neonatal brains, 48 hours following tail vein administration to pregnant dams154. Although encouraging, neither maternal and fetal toxicity nor long-term neonatal survival have been assessed here, which would be important in evaluating an active targeting platform that crosses the blood–brain barrier.
Spina bifida
Spina bifida is a hereditary congenital disorder that would be likely to benefit from in utero prenatal treatment to minimize the onset of irreversible disease pathology. Open spina bifida, or myelomeningocele (MMC), is defined as the presence of a protruding and non-neurulated spinal cord through the back, caused by failed fusion of the vertebrae148,156. In utero, chemical and mechanical interactions of the exposed spinal cord with the amniotic fluid and surrounding membrane lead to neural toxicity148. Therefore, prenatal intervention strategies and therapies are being explored for MMC.
In the clinic, MMC is treated prenatally by open or fetoscopic surgical repair of the defect; however, these treatment strategies may cause premature labour148,157 (Fig. 4b) and are only offered at a few fetal surgery centres worldwide148,149. Alternatively, less-invasive intra-amniotic administration of therapeutics using a amniocentesis-related procedure can promote protection and healing of an MMC defect148,157; for example, alginate microparticles loaded with basic fibroblast growth factor can be intra-amniotically administered to improve soft tissue coverage in a retinoic-acid-induced rat model of MMC157. However, rigorous testing in large animal models will be required to demonstrate the safety of this platform.
Matrix and scaffold biomaterials have also been investigated to improve soft tissue coverage and repair during fetoscopic surgery. For example, natural biomaterials, such as cryopreserved human umbilical cord and acellular dermal matrix patches, and synthetic polymer patches have been evaluated in vivo to cover and repair open spina bifida defects158,159,160. In a surgically induced sheep model of spina bifida, a human umbilical cord patch can better repair the defect than a biocellulose film158, and compared with an acellular dermal matrix patch in a retinoic-acid-induced rat model of spina bifida, the human umbilical cord patch can reduce acute inflammation and improve cellular growth at the defect site159. In addition, polymeric gel systems can be chemically functionalized to adhere in the amniotic fluid environment and protect an MMC defect161,162. Scaffold materials, such as gelatin, collagen, chitosan and ECM, can further encapsulate cells or therapeutics for additional regenerative effects156,163,164,165,166; for example, treatment of surgically induced MMC defects in time-dated ewes with small-intestine-derived ECM seeded with placental mesenchymal stromal cells improves neuron cell density and motor function, compared with defects treated with ECM scaffolds alone164. Similarly, hybrid gelatin–collagen scaffolds encapsulating basic fibroblast growth factor cause the formation of granulation tissue to preserve the spinal cord in a retinoic-acid-induced rat model of MMC163. Toxicity to both mother and fetus will need to be assessed for these platforms.
Outlook
Clinically translatable therapeutics for women’s health applications may greatly benefit from delivery technologies that have already been explored and tested in other applications, such as cancer and vaccines. However, the behaviour of delivery systems during pregnancy and their capacity to treat pregnancy-related conditions remain poorly understood. Indeed, a consensus on the ideal therapeutic target or type of cargo has not yet been reached for the treatment of many women’s health-related conditions (Table 1). Therefore, new combinations of delivery systems and cargos should be explored to identify formulations that can treat women’s health-related disorders most effectively.
Importantly, the most suitable administration route has to be identified for targeting a specific organ, including consideration of the microenvironment and its impact on the delivery technology. Intravaginal delivery allows delivery to the vaginal epithelium but requires platforms to penetrate the thick vaginal mucosa. In utero delivery, including intra-amniotic administration, provides the opportunity to target the fetal skin or lungs before birth; however, the amniotic fluid has not yet been well explored and has distinct protein and pH conditions that may affect the stability and efficacy of delivery systems. In addition, the pharmacokinetics, biodistribution, placental transport, and maternal and fetal safety of delivery systems need to be established, which are affected by their size, charge and composition (Box 1).
Delivery technologies for anticancer therapy and cancer immunotherapy have been studied for decades, with the first liposomal drug delivery platform for the chemotherapeutic drug doxorubicin gaining FDA approval in 19951,167. Lessons can be learned from the cancer drug delivery field to apply these technologies in women’s health applications; for example, endometrial lesions and placental cells share many physiological features with cancer60,132. Tumour-homing peptides can also be applied to target extravillous trophoblasts in the placenta, which behave similarly to metastatic cancer cells by invading the wall of the uterus and remodelling the uterine spiral arteries during pregnancy132. Recognizing and exploiting these similarities can be a pivotal step towards the development of delivery technologies for women’s health applications, addressing many of the challenges that limit their clinical translation (Box 1).
Key barriers to the clinical translation of delivery systems for women’s health applications also include systemic and societal challenges, such as the exclusion of pregnant women from clinical trials, the limited funding designated explicitly for women’s health research, and the racial disparities for many women-specific disorders (Box 1). Therefore, in addition to designing, optimizing and evaluating delivery technologies, community members and advocates must play a role in addressing these systemic barriers.
Perhaps one of the most successful clinical demonstrations of a delivery technology in the field of women’s health research is the growing evidence of the efficacy of LNP-mediated mRNA vaccines for COVID-19 in pregnant and lactating women168,169, with demonstration of potent maternal immune responses and the potential of transplacental antibody transfer that benefits the fetus. Therefore, LNPs may also enable the delivery of other drugs and cargo during pregnancy. However, thorough and long-term studies evaluating the safety of the COVID-19 vaccines in pregnant women, particularly in regard to chronic effects from prenatal exposure to foreign biomaterials, will be essential to better understand direct and indirect effects on fetal development. Preclinical research and collaboration between bioengineers, clinicians, patients, regulatory officials and advocates will be required to develop and translate therapeutic delivery technologies for women’s health.
References
Riley, R. S., June, C. H., Langer, R. & Mitchell, M. J. Delivery technologies for cancer immunotherapy. Nat. Rev. Drug Discov. 18, 175–196 (2019).
Kauffman, K. J., Webber, M. J. & Anderson, D. G. Materials for non-viral intracellular delivery of messenger RNA therapeutics. J. Control. Rel. 240, 227–234 (2016).
Pardi, N., Hogan, M. J., Porter, F. W. & Weissman, D. mRNA vaccines — a new era in vaccinology. Nat. Rev. Drug Discov. 17, 261–279 (2018).
Mirin, A. A. Gender disparity in the funding of diseases by the U.S. National Institutes of Health. J. Womens Health 30, 956–963 (2021).
Nijjar, S. et al. Participation in clinical trials improves outcomes in women’s health: a systematic review and meta-analysis. BJOG 124, 863–871 (2017).
Steinberg, J. R. et al. Early discontinuation, results reporting, and publication of gynecology clinical trials from 2007 to 2020. Obstet. Gynecol. 139, 821–831 (2022).
Sultana, A., Zare, M., Thomas, V., Kumar, T. S. S. & Ramakrishna, S. Nano-based drug delivery systems: conventional drug delivery routes, recent developments and future prospects. Med. Drug Discov. 15, 100134 (2022).
Swingle, K. L., Hamilton, A. G. & Mitchell, M. J. Lipid nanoparticle-mediated delivery of mRNA therapeutics and vaccines. Trends Mol. Med. 27, 616–617 (2021).
Mitchell, M. J. et al. Engineering precision nanoparticles for drug delivery. Nat. Rev. Drug Discov. https://doi.org/10.1038/s41573-020-0090-8 (2020).
Diaz, S. et al. A five-year clinical trial of levonorgestrel silastic implants (NorplantTM). Contraception 25, 447–456 (1982).
Smallwood, G. H. et al. Efficacy and safety of a transdermal contraceptive system. Obstet. Gynecol. 98, 799–805 (2001).
Roumen, F. J. M. E., Apter, D., Mulders, T. M. T. & Dieben, T. O. M. Efficacy, tolerability and acceptability of a novel contraceptive vaginal ring releasing etonogestrel and ethinyl oestradiol. Hum. Reprod. 16, 469–475 (2001).
De Ziegler, D., Bulletti, C., De Monstier, B. & Jääskeläinen, A.-S. The first uterine pass effect. Ann. N. Y. Acad. Sci. 828, 291–299 (1997).
Adnane, M., Meade, K. G. & O’Farrelly, C. Cervico-vaginal mucus (CVM) — an accessible source of immunologically informative biomolecules. Vet. Res. Commun. 42, 255–263 (2018).
Gajer, P. et al. Temporal dynamics of the human vaginal microbiota. Sci. Transl. Med. 4, 132ra52–132ra52 (2012).
Figueroa-Espada, C. G., Hofbauer, S., Mitchell, M. J. & Riley, R. S. Exploiting the placenta for nanoparticle-mediated drug delivery during pregnancy. Adv. Drug Deliv. Rev. 160, 244–261 (2020).
Dugershaw, B. B., Aengenheister, L., Hansen, S. S. K., Hougaard, K. S. & Buerki-Thurnherr, T. Recent insights on indirect mechanisms in developmental toxicity of nanomaterials. Part. Fibre Toxicol. 17, 31 (2020).
Campaña-Seoane, M. et al. Vaginal residence and pharmacokinetic preclinical study of topical vaginal mucoadhesive W/S emulsions containing ciprofloxacin. Int. J. Pharm. 554, 276–283 (2019).
Hoang, T. et al. Development of a mucoinert progesterone nanosuspension for safer and more effective prevention of preterm birth. J. Control. Rel. 295, 74–86 (2019).
Bulletti, C. et al. Vaginal drug delivery: the first uterine pass effect. Ann. N. Y. Acad. Sci. 828, 285–290 (1997).
Bulletti, C. et al. Targeted drug delivery in gynaecology: the first uterine pass effect. Hum. Reprod. 12, 1073–1079 (1997).
Cicinelli, E. et al. Plasma concentrations of progesterone are higher in the uterine artery than in the radial artery after vaginal administration of micronized progesterone in an oil-based solution to postmenopausal women. Fertil. Steril. 69, 471–473 (1998).
McCracken, J. M. et al. Animal models and alternatives in vaginal research: a comparative review. Reprod. Sci. 28, 1759–1773 (2021).
Vermani, K. & Garg, S. The scope and potential of vaginal drug delivery. Pharm. Sci. Technol. Today 3, 359–364 (2000).
Maestrelli, F., Jug, M., Cirri, M., Kosalec, I. & Mura, P. Characterization and microbiological evaluation of chitosan-alginate microspheres for cefixime vaginal administration. Carbohydr. Polym. 192, 176–183 (2018).
Pisano, S. et al. Liquid crystal delivery of ciprofloxacin to treat infections of the female reproductive tract. Biomed. Microdevices 21, 36 (2019).
Alqahtani, F. et al. Antibacterial activity of chitosan nanoparticles against pathogenic N. gonorrhoea. Int. J. Nanomed. 15, 7877–7887 (2020).
Rowley, J. et al. Chlamydia, gonorrhoea, trichomoniasis and syphilis: global prevalence and incidence estimates, 2016. Bull. World Health Organ. 97, 548–562P (2019).
Palmeira-de-Oliveira, R., Palmeira-de-Oliveira, A. & Martinez-de-Oliveira, J. New strategies for local treatment of vaginal infections. Adv. Drug Deliv. Rev. 92, 105–122 (2015).
Shapiro, R. L. et al. In vitro and ex vivo models for evaluating vaginal drug delivery systems. Adv. Drug Deliv. Rev. 191, 114543 (2022).
Tuğcu-Demiröz, F. et al. Development and characterization of chitosan nanoparticles loaded nanofiber hybrid system for vaginal controlled release of benzydamine. Eur. J. Pharm. Sci. 161, 105801 (2021).
Jøraholmen, M. W., Basnet, P., Tostrup, M. J., Moueffaq, S. & Škalko-Basnet, N. Localized therapy of vaginal infections and inflammation: liposomes-in-hydrogel delivery system for polyphenols. Pharmaceutics 11, 53 (2019).
Abd Ellah, N. H. et al. Efficacy of ketoconazole gel-flakes in treatment of vaginal candidiasis: formulation, in vitro and clinical evaluation. Int. J. Pharm. 567, 118472 (2019).
Calvo, N. L. et al. Chitosan-hydroxypropyl methylcellulose tioconazole films: a promising alternative dosage form for the treatment of vaginal candidiasis. Int. J. Pharm. 556, 181–191 (2019).
Deshkar, S. S. & Palve, V. K. Formulation and development of thermosensitive cyclodextrin-based in situ gel of voriconazole for vaginal delivery. J. Drug Deliv. Sci. Technol. 49, 277–285 (2019).
Salah, S., Awad, G. E. A. & Makhlouf, A. I. A. Improved vaginal retention and enhanced antifungal activity of miconazole microsponges gel: formulation development and in vivo therapeutic efficacy in rats. Eur. J. Pharm. Sci. 114, 255–266 (2018).
Querobino, S. M. et al. Sodium alginate in oil-poloxamer organogels for intravaginal drug delivery: influence on structural parameters, drug release mechanisms, cytotoxicity and in vitro antifungal activity. Mater. Sci. Eng. C. 99, 1350–1361 (2019).
Martínez-Pérez, B. et al. Controlled-release biodegradable nanoparticles: from preparation to vaginal applications. Eur. J. Pharm. Sci. 115, 185–195 (2018).
Li, W.-Z. et al. Propylene glycol-embodying deformable liposomes as a novel drug delivery carrier for vaginal fibrauretine delivery applications. J. Control. Rel. 226, 107–114 (2016).
Cautela, M. P., Moshe, H., Sosnik, A., Sarmento, B. & das Neves, J. Composite films for vaginal delivery of tenofovir disoproxil fumarate and emtricitabine. Eur. J. Pharm. Biopharm. 138, 3–10 (2019).
Crakes, K. R. et al. Efficacy of silk fibroin biomaterial vehicle for in vivo mucosal delivery of Griffithsin and protection against HIV and SHIV infection ex vivo. J. Int. AIDS Soc. 23, e25628 (2020).
Cunha-Reis, C. et al. Nanoparticles-in-film for the combined vaginal delivery of anti-HIV microbicide drugs. J. Control. Rel. 243, 43–53 (2016).
Ramyadevi, D., Rajan, K. S., Vedhahari, B. N., Ruckmani, K. & Subramanian, N. Heterogeneous polymer composite nanoparticles loaded in situ gel for controlled release intra-vaginal therapy of genital herpes. Colloids Surf. B 146, 260–270 (2016).
Yang, H. et al. Design of poly(lactic-co-glycolic acid) (PLGA) nanoparticles for vaginal co-delivery of Griffithsin and Dapivirine and their synergistic effect for HIV prophylaxis. Pharmaceutics 11, 184 (2019).
Kim, S. et al. Design and development of pH-responsive polyurethane membranes for intravaginal release of nanomedicines. Acta Biomater. 82, 12–23 (2018).
Wang, J. et al. A thermosensitive gel based on w1/o/w2 multiple microemulsions for the vaginal delivery of small nucleic acid. Drug Deliv. 26, 168–178 (2019).
Woodrow, K. A. et al. Intravaginal gene silencing using biodegradable polymer nanoparticles densely loaded with small-interfering RNA. Nat. Mater. 8, 526–533 (2009). This article reports a new technique for small interfering RNA loading into polymeric nanoparticles formulated with FDA-approved materials for gene silencing in the mouse reproductive tract following intravaginal administration.
Steinbach, J. M., Weller, C. E., Booth, C. J. & Saltzman, W. M. Polymer nanoparticles encapsulating siRNA for treatment of HSV-2 genital infection. J. Control. Rel. 162, 102–110 (2012).
Ariza-Sáenz, M. et al. Design, characterization, and biopharmaceutical behavior of nanoparticles loaded with an HIV-1 fusion inhibitor peptide. Mol. Pharm. 15, 5005–5018 (2018).
Gong, T., Patel, S. K., Parniak, M. A., Ballou, B. & Rohan, L. C. Nanocrystal formulation improves vaginal delivery of CSIC for HIV prevention. AAPS PharmSciTech 20, 286 (2019).
Orłowski, P. et al. Antiviral activity of tannic acid modified silver nanoparticles: potential to activate immune response in herpes genitalis. Viruses 10, 524 (2018).
Hajj, K. A. et al. Branched-tail lipid nanoparticles potently deliver mRNA in vivo due to enhanced ionization at endosomal pH. Small 15, 1805097 (2019).
Heyes, J., Palmer, L., Bremner, K. & MacLachlan, I. Cationic lipid saturation influences intracellular delivery of encapsulated nucleic acids. J. Control. Rel. 107, 276–287 (2005).
Cu, Y. & Saltzman, W. M. Controlled surface modification with poly(ethylene)glycol enhances diffusion of PLGA nanoparticles in human cervical mucus. Mol. Pharm. 6, 173–181 (2009).
Cu, Y., Booth, C. J. & Saltzman, W. M. In vivo distribution of surface-modified PLGA nanoparticles following intravaginal delivery. J. Control. Rel. 156, 258–264 (2011).
Ensign, L. M. et al. Mucus-penetrating nanoparticles for vaginal drug delivery protect against herpes simplex virus. Sci. Transl. Med. 4, 138ra79–138ra79 (2012). This article reports the design of mucus-penetrating nanoparticles for uniform drug delivery to the vaginal epithelium, and more effective protection against herpes simplex virus 2 challenge in mice than the soluble drug.
Jøraholmen, M. W., Basnet, P., Acharya, G. & Škalko-Basnet, N. PEGylated liposomes for topical vaginal therapy improve delivery of interferon alpha. Eur. J. Pharm. Biopharm. 113, 132–139 (2017).
Krogstad, E. A. et al. Nanoparticle-releasing nanofiber composites for enhanced in vivo vaginal retention. Biomaterials 144, 1–16 (2017).
Mohideen, M. et al. Degradable bioadhesive nanoparticles for prolonged intravaginal delivery and retention of Elvitegravir. Biomaterials 144, 144–154 (2017).
Simón-Gracia, L. et al. Homing peptide-based targeting of tenascin-C and fibronectin in endometriosis. Nanomaterials 11, 3257 (2021).
Liu, Q. et al. Evaluation of PLGA containing anti-CTLA4 inhibited endometriosis progression by regulating CD4+CD25+Treg cells in peritoneal fluid of mouse endometriosis model. Eur. J. Pharm. Sci. 96, 542–550 (2017).
Boroumand, S., Hosseini, S., Pashandi, Z., Faridi-Majidi, R. & Salehi, M. Curcumin-loaded nanofibers for targeting endometriosis in the peritoneum of a mouse model. J. Mater. Sci. Mater Med. 31, 8 (2019).
Siddiqa, A. J. et al. Preparation of letrozole dispersed pHEMA/AAm-g-LDPE drug release system: in-vitro release kinetics for the treatment of endometriosis. Colloids Surf. B 179, 445–452 (2019).
Egorova, A. et al. Anti-angiogenic treatment of endometriosis via anti-VEGFA siRNA delivery by means of peptide-based carrier in a rat subcutaneous model. Gene Ther. 25, 548–555 (2018).
Liang, Z. et al. miR-200c suppresses endometriosis by targeting MALAT1 in vitro and in vivo. Stem Cell Res. Ther. 8, 251 (2017).
Yuan, M. et al. Effect of A-317491 delivered by glycolipid-like polymer micelles on endometriosis pain. Int. J. Nanomed. 12, 8171–8183 (2017).
Zhao, M.-D. et al. Hyaluronic acid reagent functional chitosan-PEI conjugate with AQP2-siRNA suppressed endometriotic lesion formation. Int. J. Nanomed. 11, 1323–1336 (2016).
Kiisholts, K. et al. Cell-penetrating peptide and siRNA-mediated therapeutic effects on endometriosis and cancer in vitro models. Pharmaceutics 13, 1618 (2021).
Bedin, A. et al. Nanotechnology for the treatment of deep endometriosis: uptake of lipid core nanoparticles by LDL receptors in endometriotic foci. Clinics 74, e989 (2019).
Park, Y. et al. Targeted nanoparticles with high heating efficiency for the treatment of endometriosis with systemically delivered magnetic hyperthermia. Small 18, 2107808 (2022).
Zhang, H. et al. Hyaluronic acid-modified magnetic iron oxide nanoparticles for MR imaging of surgically induced endometriosis model in rats. PLoS ONE 9, e94718 (2014).
Moses, A. S. et al. Nanoparticle-based platform for activatable fluorescence imaging and photothermal ablation of endometriosis. Small 16, 1906936 (2020).
Wang, C. et al. Silencing of MEF2D by siRNA loaded selenium nanoparticles for ovarian cancer therapy. Int. J. Nanomed. 15, 9759–9770 (2020).
Pan, Q. et al. Tumor-targeting polycaprolactone nanoparticles with codelivery of paclitaxel and IR780 for combinational therapy of drug-resistant ovarian cancer. ACS Biomater. Sci. Eng. 6, 2175–2185 (2020).
Domínguez-Ríos, R. et al. Cisplatin-loaded PLGA nanoparticles for HER2 targeted ovarian cancer therapy. Colloids Surf. B 178, 199–207 (2019).
Michy, T. et al. Verteporfin-loaded lipid nanoparticles improve ovarian cancer photodynamic therapy in vitro and in vivo. Cancers 11, 1760 (2019).
Zaman, M. S. et al. Curcumin nanoformulation for cervical cancer treatment. Sci. Rep. 6, 20051 (2016).
Liu, B. et al. Co-delivery of paclitaxel and TOS-cisplatin via TAT-targeted solid lipid nanoparticles with synergistic antitumor activity against cervical cancer. Int. J. Nanomed. 12, 955–968 (2017).
Xia, Y. et al. Functionalized selenium nanoparticles for targeted siRNA delivery silence Derlin1 and promote antitumor efficacy against cervical cancer. Drug Deliv. 27, 15–25 (2020).
Ci, L. et al. Enhanced delivery of imatinib into vaginal mucosa via a new positively charged nanocrystal-loaded in situ hydrogel formulation for treatment of cervical cancer. Pharmaceutics 11, 15 (2019).
Zhai, J. et al. Paclitaxel-loaded self-assembled lipid nanoparticles as targeted drug delivery systems for the treatment of aggressive ovarian cancer. ACS Appl. Mater. Interfaces 10, 25174–25185 (2018).
Yi, Y. et al. Targeted systemic delivery of siRNA to cervical cancer model using cyclic RGD-installed unimer polyion complex-assembled gold nanoparticles. J. Control. Release 244, 247–256 (2016).
Xu, G. et al. Robust aptamer–polydopamine-functionalized M-PLGA–TPGS nanoparticles for targeted delivery of docetaxel and enhanced cervical cancer therapy. Int. J. Nanomed. 11, 2953–2965 (2016).
Byeon, Y. et al. CD44-targeting PLGA nanoparticles incorporating paclitaxel and FAK siRNA overcome chemoresistance in epithelial ovarian cancer. Cancer Res. 78, 6247–6256 (2018).
Correa, S. et al. Tuning nanoparticle interactions with ovarian cancer through layer-by-layer modification of surface chemistry. ACS Nano 14, 2224–2237 (2020). This article reports the identification of layer-by-layer nanoparticle formulations with specificity for ovarian cancer and demonstrates in vivo tumour-homing in a murine model of metastatic ovarian cancer.
Mensah, L. B. et al. Layer-by-layer nanoparticles for novel delivery of cisplatin and PARP inhibitors for platinum-based drug resistance therapy in ovarian cancer. Bioeng. Transl. Med. 4, e10131 (2019).
Cheng, C., Meng, Y., Zhang, Z., Li, Y. & Zhang, Q. Tumoral acidic pH-responsive cis-diaminodichloroplatinum-incorporated Cy5.5-PEG-g-A-HA nanoparticles for targeting delivery of CDDP against cervical cancer. ACS Appl. Mater. Interfaces 10, 26882–26892 (2018).
Shen, W. et al. Sustained codelivery of cisplatin and paclitaxel via an injectable prodrug hydrogel for ovarian cancer treatment. ACS Appl. Mater. Interfaces 9, 40031–40046 (2017).
Sun, B. et al. Intraperitoneal chemotherapy of ovarian cancer by hydrogel depot of paclitaxel nanocrystals. J. Control. Rel. 235, 91–98 (2016).
Jamal, A. et al. Identification of anti-cancer potential of doxazocin: loading into chitosan based biodegradable hydrogels for on-site delivery to treat cervical cancer. Mater. Sci. Eng. C. 82, 102–109 (2018).
Nash, A. M. et al. Clinically translatable cytokine delivery platform for eradication of intraperitoneal tumors. Sci. Adv. 8, eabm1032 (2022). This article reports the development of an interleukin-2 cytokine factory with alginate microcapsules for reducing ovarian cancer tumour burden following intraperitoneal administration.
Nash, A. M. et al. Activation of adaptive and innate immune cells via localized Interleukin-2 cytokine factories eradicates mesothelioma tumors. Clin. Cancer Res. https://doi.org/10.1158/1078-0432.CCR-22-1493 (2022).
Wang, X. et al. Vaginal delivery of mucus-penetrating organic nanoparticles for photothermal therapy against cervical intraepithelial neoplasia in mice. J. Mater. Chem. B 7, 4528–4537 (2019).
Jalalvandi, E. & Shavandi, A. In situ-forming and pH-responsive hydrogel based on chitosan for vaginal delivery of therapeutic agents. J. Mater. Sci. Mater Med. 29, 158 (2018).
Risnayanti, C., Jang, Y.-S., Lee, J. & Ahn, H. J. PLGA nanoparticles co-delivering MDR1 and BCL2 siRNA for overcoming resistance of paclitaxel and cisplatin in recurrent or advanced ovarian cancer. Sci. Rep. 8, 7498 (2018).
Zhang, Q. et al. Photoactivatable prodrug-backboned polymeric nanoparticles for efficient light-controlled gene delivery and synergistic treatment of platinum-resistant ovarian cancer. Nano Lett. 20, 3039–3049 (2020).
Bertucci, A. et al. Tumor-targeting, microRNA-silencing porous silicon nanoparticles for ovarian cancer therapy. ACS Appl. Mater. Interfaces 11, 23926–23937 (2019).
Zhang, X.-F., Yan, Q., Shen, W. & Gurunathan, S. Trichostatin A enhances the apoptotic potential of palladium nanoparticles in human cervical cancer cells. Int. J. Mol. Sci. 17, 1354 (2016).
Dunk, C. et al. Failure of decidualization and maternal immune tolerance underlies uterovascular resistance in intra uterine growth restriction. Front. Endocrinol. 10, 160 (2019).
Kim, C. J., Romero, R., Chaemsaithong, P. & Kim, J.-S. Chronic inflammation of the placenta: definition, classification, pathogenesis, and clinical significance. Am. J. Obstet. Gynecol. 213, S53–S69 (2015).
Knöfler, M. et al. Human placenta and trophoblast development: key molecular mechanisms and model systems. Cell. Mol. Life Sci. 76, 3479–3496 (2019).
Thompson, L. P., Pence, L., Pinkas, G., Song, H. & Telugu, B. P. Placental hypoxia during early pregnancy causes maternal hypertension and placental insufficiency in the hypoxic guinea pig model. Biol. Reprod. 95, 128 (2016).
Aengenheister, L. et al. An advanced human in vitro co-culture model for translocation studies across the placental barrier. Sci. Rep. 8, 5388 (2018).
Levine, R. J. et al. Circulating angiogenic factors and the risk of preeclampsia. N. Engl. J. Med. 350, 672–683 (2004).
Umapathy, A., Chamley, L. W. & James, J. L. Reconciling the distinct roles of angiogenic/anti-angiogenic factors in the placenta and maternal circulation of normal and pathological pregnancies. Angiogenesis 23, 105–117 (2020).
Whigham, C.-A. et al. The untapped potential of placenta-enriched molecules for diagnostic and therapeutic development. Placenta 84, 28–31 (2019).
Dodd, A. et al. Conjugation to PEG as a strategy to limit the uptake of drugs by the placenta: potential applications for drug administration in pregnancy. Mol. Pharm. 19, 345–353 (2022).
Ali, S. et al. Formulation effects on paclitaxel transfer and uptake in the human placenta. Nanomedicine 33, 102354 (2021).
Joshi, M. D. Drug delivery during pregnancy: how can nanomedicine be used? Ther. Deliv. 8, 1023–1025 (2017).
Kuna, M., Waller, J. P., Logue, O. C. & Bidwell, G. L. Polymer size affects biodistribution and placental accumulation of the drug delivery biopolymer elastin-like polypeptide in a rodent pregnancy model. Placenta 72–73, 20–27 (2018).
Aengenheister, L. et al. Gold nanoparticle distribution in advanced in vitro and ex vivo human placental barrier models. J. Nanobiotechnol. 16, 79 (2018).
Muoth, C. et al. Impact of particle size and surface modification on gold nanoparticle penetration into human placental microtissues. Nanomedicine 12, 1119–1133 (2017).
Irvin-Choy, N. S., Nelson, K. M., Dang, M. N., Gleghorn, J. P. & Day, E. S. Gold nanoparticle biodistribution in pregnant mice following intravenous administration varies with gestational age. Nanomedicine 36, 102412 (2021).
Ho, D. et al. Maternal–placental–fetal biodistribution of multimodal polymeric nanoparticles in a pregnant rat model in mid and late gestation. Sci. Rep. 7, 2866 (2017).
Juch, H. et al. Dendritic polyglycerol nanoparticles show charge dependent bio-distribution in early human placental explants and reduce hCG secretion. Nanotoxicology 12, 90–103 (2018).
Zhang, B. et al. Placenta-specific drug delivery by trophoblast-targeted nanoparticles in mice. Theranostics 8, 2765–2781 (2018).
Yang, H., Du, L., Wu, G., Wu, Z. & Keelan, J. A. Murine exposure to gold nanoparticles during early pregnancy promotes abortion by inhibiting ectodermal differentiation. Mol. Med. 24, 62 (2018).
Georgiades, P., Ferguson-Smith, A. C. & Burton, G. J. Comparative developmental anatomy of the murine and human definitive placentae. Placenta 23, 3–19 (2002).
Hemberger, M., Hanna, C. W. & Dean, W. Mechanisms of early placental development in mouse and humans. Nat. Rev. Genet. 21, 27–43 (2020).
Furukawa, S., Kuroda, Y. & Sugiyama, A. A comparison of the histological structure of the placenta in experimental animals. J. Toxicol. Pathol. 27, 11–18 (2014).
Raia, N. R., Bakaysa, S. L., Ghezzi, C. E., House, M. D. & Kaplan, D. L. Ex vivo pregnant-like tissue model to assess injectable hydrogel for preterm birth prevention. J. Biomed. Mater. Res. B 108, 468–474 (2020).
Refuerzo, J. S. et al. Uterus-targeted liposomes for preterm labor management: studies in pregnant mice. Sci. Rep. 6, 34710 (2016).
Brown, J. E., Partlow, B. P., Berman, A. M., House, M. D. & Kaplan, D. L. Injectable silk-based biomaterials for cervical tissue augmentation: an in vitro study. Am. J. Obstet. Gynecol. 214, 118.e1–118.e9 (2016).
Lei, J. et al. Maternal dendrimer-based therapy for inflammation-induced preterm birth and perinatal brain injury. Sci. Rep. 7, 6106 (2017).
Zhang, Y., Raia, N., Peterson, A., Kaplan, D. L. & House, M. Injectable silk-based hydrogel as an alternative to cervical cerclage: a rabbit study. Tissue Eng. A 26, 379–386 (2020).
Zierden, H. C. et al. Enhanced drug delivery to the reproductive tract using nanomedicine reveals therapeutic options for prevention of preterm birth. Sci. Transl. Med. 13, eabc6245 (2021). This article reports the use of mucoinert nanosuspensions of histone deacetylase inhibitors for preventing preterm birth and promoting birth of live pups with neurotypical development in a murine model of intrauterine inflammation.
Murphy, C. C., Cirillo, P. M., Krigbaum, N. Y. & Cohn, B. A. In utero exposure to 17α-hydroxyprogesterone caproate and risk of cancer in offspring. Am. J. Obstet. Gynecol. 226, 132.e1–132.e14 (2022).
Chang, C. Y. et al. Withdrawing approval of Makena — a proposal from the FDA Center for Drug Evaluation and Research. N. Engl. J. Med. 383, e131 (2020).
Paul, J. W. et al. Drug delivery to the human and mouse uterus using immunoliposomes targeted to the oxytocin receptor. Am. J. Obstet. Gynecol. 216, 283.e1–283.e14 (2017).
Alfaifi, A. A. et al. Megalin-targeting liposomes for placental drug delivery. J. Control. Rel. 324, 366–378 (2020).
Li, L. et al. Nanoparticle-mediated simultaneous downregulation of placental Nrf2 and sFlt1 improves maternal and fetal outcomes in a preeclampsia mouse model. ACS Biomater. Sci. Eng. 6, 5866–5873 (2020).
King, A. et al. Tumor-homing peptides as tools for targeted delivery of payloads to the placenta. Sci. Adv. 2, e1600349 (2021). This article reports the design of tumour-homing peptide-targeted liposomes for in vivo accumulation in the placenta and demonstrates that delivery of insulin-like growth factor 2 can improve fetal weight distribution in a murine model of fetal growth restriction.
Ellah, N. A. et al. Development of non-viral, trophoblast-specific gene delivery for placental therapy. PLoS ONE 10, e0140879 (2015).
Young, R. E. et al. Lipid nanoparticle composition drives mRNA delivery to the placenta. Preprint at bioRxiv https://www.biorxiv.org/content/10.1101/2022.12.22.521490v1 (2022).
Swingle, K. L. et al. Ionizable lipid nanoparticles for in vivo mRNA delivery to the placenta during pregnancy. J. Am. Chem. Soc. https://doi.org/10.1021/jacs.2c12893 (2023).
Sanghavi, M. & Rutherford, J. D. Cardiovascular physiology of pregnancy. Circulation 130, 1003–1008 (2014).
Frederiksen, M. C. Physiologic changes in pregnancy and their effect on drug disposition. Semin. Perinatol. 25, 120–123 (2001).
Yu, J., Jia, J., Guo, X., Chen, R. & Feng, L. Modulating circulating sFlt1 in an animal model of preeclampsia using PAMAM nanoparticles for siRNA delivery. Placenta https://doi.org/10.1016/j.placenta.2017.07.360 (2017).
McCarthy, F. P., Kingdom, J. C., Kenny, L. C. & Walsh, S. K. Animal models of preeclampsia; uses and limitations. Placenta 32, 413–419 (2011).
Wilson, R. L. et al. Nanoparticle mediated increased insulin-like growth factor 1 expression enhances human placenta syncytium function. Placenta https://doi.org/10.1016/j.placenta.2020.02.006 (2020).
Beards, F., Jones, L. E., Charnock, J., Forbes, K. & Harris, L. K. Placental homing peptide-microRNA inhibitor conjugates for targeted enhancement of intrinsic placental growth signaling. Theranostics 7, 2940–2955 (2017).
Cureton, N. et al. Selective targeting of a novel vasodilator to the uterine vasculature to treat impaired uteroplacental perfusion in pregnancy. Theranostics 7, 3715–3731 (2017).
Renshall, L. J. et al. Targeted delivery of epidermal growth factor to the human placenta to treat fetal growth restriction. Pharmaceutics 13, 1778 (2021).
Li, L. et al. Trophoblast-targeted nanomedicine modulates placental sFLT1 for preeclampsia treatment. Front. Bioeng. Biotechnol. 8, 64 (2020).
Ullrich, S. J. et al. Nanoparticles for delivery of agents to fetal lungs. Acta Biomaterialia 123, 346–353 (2021).
Riley, R. S. et al. Ionizable lipid nanoparticles for in utero mRNA delivery. Sci. Adv. 7, eaba1028 (2021). This article reports the identification of ionizable lipid nanoparticle formulations for mRNA delivery to the fetal liver, lungs and intestines following vitelline vein administration in murine fetuses.
Ricciardi, A. S. et al. In utero nanoparticle delivery for site-specific genome editing. Nat. Commun. 9, 2481 (2018). This article reports safe and effective in utero gene editing in murine β-thalassemia fetuses following vitelline vein administration of polymeric nanoparticles encapsulating peptide nucleic acids and donor DNAs.
Freedman-Weiss, M. R. et al. Engineering alginate microparticles for optimized accumulation in fetal rat myelomeningocele. J. Pediatr. Surg. https://doi.org/10.1016/j.jpedsurg.2021.03.060 (2021).
Adzick, N. S. et al. A randomized trial of prenatal versus postnatal repair of myelomeningocele. N. Engl. J. Med. 364, 993–1004 (2011).
Swingle, K. L. et al. Amniotic fluid stabilized lipid nanoparticles for in utero intra-amniotic mRNA delivery. J. Control. Rel. 341, 616–633 (2022).
Palanki, R., Peranteau, W. H. & Mitchell, M. J. Delivery technologies for in utero gene therapy. Adv. Drug Deliv. Rev. 169, 51–62 (2021).
Alapati, D. et al. In utero gene editing for monogenic lung disease. Sci. Transl. Med. 11, eaav8375 (2019).
Almeida-Porada, G. et al. In utero gene therapy consensus statement from the IFeTIS. Mol. Ther. 27, 705–707 (2019).
Cornford, E. M. et al. Non-invasive gene targeting to the fetal brain after intravenous administration and transplacental transfer of plasmid DNA using PEGylated immunoliposomes. J. Drug Target. 24, 58–67 (2016).
Nakamura, S. et al. Transplacental delivery of genome editing components causes mutations in embryonic cardiomyocytes of mid-gestational murine fetuses. IUBMB Life 71, 835–844 (2019).
Mazzone, L. et al. Bioengineering and in utero transplantation of fetal skin in the sheep model: A crucial step towards clinical application in human fetal spina bifida repair. J. Tissue Eng. Regen. Med. 14, 58–65 (2020).
Farrelly, J. S. et al. Alginate microparticles loaded with basic fibroblast growth factor induce tissue coverage in a rat model of myelomeningocele. J. Pediatr. Surg. 54, 80–85 (2019).
Papanna, R. et al. Cryopreserved human umbilical cord patch for in-utero spina bifida repair. Ultrasound Obstet. Gynecol. 47, 168–176 (2016).
Mann, L. K. et al. Cryopreserved human umbilical cord versus acellular dermal matrix patches for in utero fetal spina bifida repair in a pregnant rat model. J. Neurosurg. Spine 32, 321–331 (2019).
Oria, M., Tatu, R. R., Lin, C.-Y. & Peiro, J. L. In vivo evaluation of novel PLA/PCL polymeric patch in rats for potential spina bifida coverage. J. Surg. Res. 242, 62–69 (2019).
Bardill, J. R., Park, D. & Marwan, A. I. Improved coverage of mouse myelomeningocele with a mussel inspired reverse thermal gel. J. Surg. Res. 251, 262–274 (2020).
Bardill, J. et al. An injectable reverse thermal gel for minimally invasive coverage of mouse myelomeningocele. J. Surg. Res. 235, 227–236 (2019).
Watanabe, M. et al. Complete tissue coverage achieved by scaffold-based tissue engineering in the fetal sheep model of myelomeningocele. Biomaterials 76, 133–143 (2016).
Vanover, M. et al. High density placental mesenchymal stromal cells provide neuronal preservation and improve motor function following in utero treatment of ovine myelomeningocele. J. Pediatr. Surg. 54, 75–79 (2019).
Li, X. et al. Application potential of bone marrow mesenchymal stem cell (BMSCs) based tissue-engineering for spinal cord defect repair in rat fetuses with spina bifida aperta. J. Mater. Sci. Mater Med. 27, 77 (2016).
Dasargyri, A., Reichmann, E. & Moehrlen, U. Bio-engineering of fetal cartilage for in utero spina bifida repair. Pediatr. Surg. Int. 36, 25–31 (2020).
Barenholz, Y. Doxil® — the first FDA-approved nano-drug: lessons learned. J. Control. Rel. 160, 117–134 (2012).
Beharier, O. et al. Efficient maternal to neonatal transfer of antibodies against SARS-CoV-2 and BNT162b2 mRNA COVID-19 vaccine. J. Clin. Invest. 131, e150319 (2021).
Prahl, M. et al. Evaluation of transplacental transfer of mRNA vaccine products and functional antibodies during pregnancy and infancy. Nat. Commun. 13, 4422 (2022).
Yang, S., Traore, Y., Jimenez, C. & Ho, E. A. Autophagy induction and PDGFR-β knockdown by siRNA-encapsulated nanoparticles reduce Chlamydia trachomatis infection. Sci. Rep. 9, 1306 (2019).
Köllner, S. et al. Self-emulsifying drug delivery systems: design of a novel vaginal delivery system for curcumin. Eur. J. Pharm. Biopharm. 115, 268–275 (2017).
Luiz, M. T. et al. In vitro evaluation of folate-modified PLGA nanoparticles containing paclitaxel for ovarian cancer therapy. Mater. Sci. Eng. C. 105, 110038 (2019).
Lv, F. et al. Enhanced mucosal penetration and efficient inhibition efficacy against cervical cancer of PEGylated docetaxel nanocrystals by TAT modification. J. Control. Rel. 336, 572–582 (2021).
Hu, R. et al. Core–shell magnetic gold nanoparticles for magnetic field-enhanced radio-photothermal therapy in cervical cancer. Nanomaterials 7, 111 (2017).
You, L., Liu, X., Fang, Z., Xu, Q. & Zhang, Q. Synthesis of multifunctional Fe3O4@PLGA-PEG nano-niosomes as a targeting carrier for treatment of cervical cancer. Mater. Sci. Eng. C 94, 291–302 (2019).
Wei, L., Chen, J., Zhao, S., Ding, J. & Chen, X. Thermo-sensitive polypeptide hydrogel for locally sequential delivery of two-pronged antitumor drugs. Acta Biomater. 58, 44–53 (2017).
Xiao, C. et al. Biomimetic nanoparticles loading with gamabutolin-indomethacin for chemo/photothermal therapy of cervical cancer and anti-inflammation. J. Control. Rel. 339, 259–273 (2021).
Zhao, Z., Zhang, X., Li, C. & Chen, T. Designing luminescent ruthenium prodrug for precise cancer therapy and rapid clinical diagnosis. Biomaterials 192, 579–589 (2019).
Wang, L. & Jia, E. Ovarian cancer targeted hyaluronic acid-based nanoparticle system for paclitaxel delivery to overcome drug resistance. Drug Deliv. 23, 1810–1817 (2016).
Ma, X. et al. Gold nanoparticles cause size-dependent inhibition of embryonic development during murine pregnancy. Nano Res. https://doi.org/10.1007/s12274-018-1969-0 (2018).
Bolandparvaz, A. et al. Biodistribution and toxicity of epitope-functionalized dextran iron oxide nanoparticles in a pregnant murine model. J. Biomed. Mater. Res. Part A 108, 1186–1202 (2020).
Tang, H. et al. Uptake and transport of pullulan acetate nanoparticles in the BeWo b30 placental barrier cell model. Int. J. Nanomed. 13, 4073–4082 (2018).
Sezgin-Bayindir, Z., Elcin, A. E., Parmaksiz, M., Elcin, Y. M. & Yuksel, N. Investigations on clonazepam-loaded polymeric micelle-like nanoparticles for safe drug administration during pregnancy. J. Microencapsul. 35, 149–164 (2018).
Turanov, A. A. et al. RNAi modulation of placental sFLT1 for the treatment of preeclampsia. Nat. Biotechnol. https://doi.org/10.1038/nbt.4297 (2018). This article reports the development of cholesterol–siRNA conjugates for extrahepatic gene silencing in the placenta in a baboon model of pre-eclampsia.
Colafella, K. M. M. & Denton, K. M. Sex-specific differences in hypertension and associated cardiovascular disease. Nat. Rev. Nephrol. 14, 185–201 (2018).
Gerdts, E. & Regitz-Zagrosek, V. Sex differences in cardiometabolic disorders. Nat. Med. 25, 1657–1666 (2019).
Takahashi, T. et al. Sex differences in immune responses that underlie COVID-19 disease outcomes. Nature 588, 315–320 (2020).
Camunas-Soler, J. et al. Predictive RNA profiles for early and very early spontaneous preterm birth. Am. J. Obstet. Gynecol. 227, 72.e1–72.e16 (2022).
Rasmussen, M. et al. RNA profiles reveal signatures of future health and disease in pregnancy. Nature 601, 422–427 (2022).
Ngo, T. T. M. et al. Noninvasive blood tests for fetal development predict gestational age and preterm delivery. Science 360, 1133–1136 (2018).
Sewell, C. A. et al. Scientific, ethical, and legal considerations for the inclusion of pregnant people in clinical trials. Am. J. Obstet. Gynecol. https://doi.org/10.1016/j.ajog.2022.07.037 (2022).
Ren, Z., Bremer, A. A. & Pawlyk, A. C. Drug development research in pregnant and lactating women. Am. J. Obstet. Gynecol. 225, 33–42 (2021).
Mazer-Amirshahi, M., Samiee-Zafarghandy, S., Gray, G. & van den Anker, J. N. Trends in pregnancy labeling and data quality for US-approved pharmaceuticals. Am. J. Obstet. Gynecol. 211, 690.e1–690.e11 (2014).
Couzin-Frankel, J. The pregnancy gap. Science 375, 1216–1220 (2022).
Carter, E. B. & Mazzoni, S. E. A paradigm shift to address racial inequities in perinatal healthcare. Am. J. Obstet. Gynecol. 224, 359–361 (2021).
Phillippe, M. Telomeres, oxidative stress, and timing for spontaneous term and preterm labor. Am. J. Obstet. Gynecol. 227, 148–162 (2022).
Lacroix, G., Gouyer, V., Gottrand, F. & Desseyn, J.-L. The cervicovaginal mucus barrier. Int. J. Mol. Sci. 21, 8266 (2020).
France, M., Alizadeh, M., Brown, S., Ma, B. & Ravel, J. Towards a deeper understanding of the vaginal microbiota. Nat. Microbiol. 7, 367–378 (2022).
Saunders, P. T. K. in Animal Models for Endometriosis: Evolution, Utility and Clinical Relevance (ed. Sharpe-Timms, K. L.) 99–111 (Springer, 2020).
Laganà, A. S. et al. Translational animal models for endometriosis research: a long and windy road. Ann. Transl. Med. 6, 431 (2018).
Bobbs, A. S., Cole, J. M. & Dahl, K. D. C. Emerging and evolving ovarian cancer animal models. Cancer Growth Metastasis 8, 29–36 (2015).
Mitchell, L. E. et al. Spina bifida. Lancet 364, 1885–1895 (2004).
Ehlers, K., Stürje, H., Merker, H.-J. & Nau, H. Valproic acid-induced spina bifida: a mouse model. Teratology 45, 145–154 (1992).
Meuli, M. et al. Creation of myelomeningocele in utero: a model of functional damage from spinal cord exposure in fetal sheep. J. Pediatr. Surg. 30, 1028–1033 (1995).
Joyeux, L. et al. Validation of the fetal lamb model of spina bifida. Sci. Rep. 9, 9327 (2019).
Acknowledgements
M.J.M. acknowledges support from a US National Institutes of Health (NIH) Director’s New Innovator Award (DP2 TR002776), a Burroughs Wellcome Fund Career Award at the Scientific Interface (CASI), a US National Science Foundation CAREER Award (CBET-2145491), an American Cancer Society Research Scholar Grant (RSG-22-122-01-ET), and the National Institutes of Health (NCI R01 CA241661, NCI R37 CA244911 and NIDDK R01 DK123049). K.L.S. acknowledges support from the US National Science Foundation Graduate Research Fellowship.
Author information
Authors and Affiliations
Contributions
K.L.S. contributed to conceptualization, writing the original draft, reviewing and editing the text. A.S.R. and W.H.P. contributed to writing, reviewing and editing. M.J.M. contributed to conceptualization, writing, reviewing and editing, and was responsible for supervision and funding acquisition.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Bioengineering thanks Tina Buerki-Thurnherr, Thomas Rduch and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Swingle, K.L., Ricciardi, A.S., Peranteau, W.H. et al. Delivery technologies for women’s health applications. Nat Rev Bioeng 1, 408–425 (2023). https://doi.org/10.1038/s44222-023-00040-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44222-023-00040-w
This article is cited by
-
The Technical and Ethical Framework of Fetal Therapy: Past and Current Advances
Current Stem Cell Reports (2024)
-
Natural resveratrol analogs differentially target endometriotic cells into apoptosis pathways
Scientific Reports (2023)