Abstract
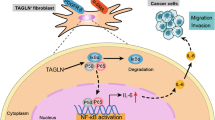
Primary tumor-derived factors act upon normal cells to generate a pre-metastatic niche, which promotes colonization of target organs by disseminated malignant cells. Here we report that tumor-derived factor-induced activation of the p38α kinase in lung fibroblasts plays a critical role in the formation of a pre-metastatic niche in the lungs and subsequent pulmonary metastases. Activation of p38α led to inactivation of type I interferon signaling and stimulation of expression of fibroblast activation protein. Fibroblast activation protein played a key role in remodeling of the extracellular matrix as well as inducing the expression of chemokines that enable lung infiltration by neutrophils. Increased activity of p38 in normal cells was associated with metastatic disease and poor prognosis in human patients with melanoma, whereas inactivation of p38 suppressed lung metastases. We discuss the p38α-driven mechanisms stimulating the metastatic processes and potential use of p38 inhibitors in adjuvant therapy of metastatic cancers.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
RNA-sequencing data that support the findings of this study have been deposited in the Gene Expression Omnibus (GEO) under accession code GSE133781. Source data for Figs. 1–8 and Extended Data Figs. 1–8 have been provided as Source data files. All other data supporting the findings of this study are available from the corresponding author upon reasonable request.
Change history
20 August 2020
The version of this article initially published included an incorrect Reporting Summary. The correct Reporting Summary is now provided.
References
Joyce, J. A. & Pollard, J. W. Microenvironmental regulation of metastasis. Nat. Rev. Cancer 9, 239–252 (2009).
Kaplan, R. N. et al. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 438, 820–827 (2005).
Peinado, H., Lavotshkin, S. & Lyden, D. The secreted factors responsible for pre-metastatic niche formation: old sayings and new thoughts. Semin. Cancer Biol. 21, 139–146 (2011).
Peinado, H. et al. Pre-metastatic niches: organ-specific homes for metastases. Nat. Rev. Cancer 17, 302–317 (2017).
Francia, G., Cruz-Munoz, W., Man, S., Xu, P. & Kerbel, R. S. Mouse models of advanced spontaneous metastasis for experimental therapeutics. Nat. Rev. Cancer 11, 135–141 (2011).
Woodard, P. K., Dehdashti, F. & Putman, C. E. Radiologic diagnosis of extrathoracic metastases to the lung. Oncology 12, 431–438 (1998).
Kim, S. et al. Carcinoma-produced factors activate myeloid cells through TLR2 to stimulate metastasis. Nature 457, 102–106 (2009).
Peinado, H. et al. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat. Med. 18, 883–891 (2012).
Liu, Y. et al. Tumor exosomal RNAs promote lung pre-metastatic niche formation by activating alveolar epithelial TLR3 to recruit neutrophils. Cancer Cell 30, 243–256 (2016).
Erler, J. T. et al. Hypoxia-induced lysyl oxidase is a critical mediator of bone marrow cell recruitment to form the premetastatic niche. Cancer Cell 15, 35–44 (2009).
Liu, Y. & Cao, X. Characteristics and significance of the pre-metastatic niche. Cancer Cell 30, 668–681 (2016).
Arthur, J. S. & Ley, S. C. Mitogen-activated protein kinases in innate immunity. Nat. Rev. Immunol. 13, 679–692 (2013).
Cuadrado, A. & Nebreda, A. R. Mechanisms and functions of p38 MAPK signalling. Biochem. J. 429, 403–417 (2010).
Hui, L., Bakiri, L., Stepniak, E. & Wagner, E. F. p38ɑ: a suppressor of cell proliferation and tumorigenesis. Cell Cycle 6, 2429–2433 (2007).
Wagner, E. F. & Nebreda, A. R. Signal integration by JNK and p38 MAPK pathways in cancer development. Nat. Rev. Cancer 9, 537–549 (2009).
Huangfu, W. C. et al. Inflammatory signaling compromises cell responses to interferon alpha. Oncogene 31, 161–172 (2012).
Katlinskaya, Y. V. et al. Suppression of type I interferon signaling overcomes oncogene-induced senescence and mediates melanoma development and progression. Cell Rep. 15, 171–180 (2016).
Ortiz, A. et al. An interferon-driven oxysterol-based defense against tumor-derived extracellular vesicles. Cancer Cell 35, 33–45 e36 (2019).
Fearon, D. T. The carcinoma-associated fibroblast expressing fibroblast activation protein and escape from immune surveillance. Cancer Immunol. Res. 2, 187–193 (2014).
Kraman, M. et al. Suppression of antitumor immunity by stromal cells expressing fibroblast activation protein-ɑ. Science 330, 827–830 (2010).
Pure, E. & Blomberg, R. Pro-tumorigenic roles of fibroblast activation protein in cancer: back to the basics. Oncogene 37, 4343–4357 (2018).
Fidler, I. J. & Nicolson, G. L. Organ selectivity for implantation survival and growth of B16 melanoma variant tumor lines. J. Natl Cancer Inst. 57, 1199–1202 (1976).
Bhattacharya, S. et al. Triggering ubiquitination of IFNAR1 protects tissues from inflammatory injury. EMBO Mol. Med. 6, 384–397 (2014).
Bhattacharya, S. et al. Role of p38 protein kinase in the ligand-independent ubiquitination and down-regulation of the IFNAR1 chain of type I interferon receptor. J. Biol. Chem. 286, 22069–22076 (2011).
Tcyganov, E., Mastio, J., Chen, E. & Gabrilovich, D. I. Plasticity of myeloid-derived suppressor cells in cancer. Curr. Opin. Immunol. 51, 76–82 (2018).
Zhou, J., Nefedova, Y., Lei, A. & Gabrilovich, D. Neutrophils and PMN-MDSC: their biological role and interaction with stromal cells. Semin. Immunol. 35, 19–28 (2018).
Andzinski, L. et al. Type I IFNs induce anti-tumor polarization of tumor associated neutrophils in mice and human. Int. J. Cancer 138, 1982–1993 (2016).
Kumar, V. et al. Cancer-associated fibroblasts neutralize the anti-tumor effect of CSF1 receptor blockade by inducing PMN-MDSC infiltration of tumors. Cancer Cell 32, 654–668 e655 (2017).
Li, J. et al. Tumor cell-intrinsic factors underlie heterogeneity of immune cell infiltration and response to immunotherapy. Immunity 49, 178–193 e177 (2018).
Kroetz, D. N. et al. Type I interferon induced epigenetic regulation of macrophages suppresses innate and adaptive immunity in acute respiratory viral infection. PLoS Pathog. 11, e1005338 (2015).
Schliehe, C. et al. The methyltransferase Setdb2 mediates virus-induced susceptibility to bacterial superinfection. Nat. Immunol. 16, 67–74 (2015).
Hoye, A. M. & Erler, J. T. Structural ECM components in the premetastatic and metastatic niche. Am. J. Physiol. Cell Physiol. 310, C955–C967 (2016).
Roberts, E. W. et al. Depletion of stromal cells expressing fibroblast activation protein-ɑ from skeletal muscle and bone marrow results in cachexia and anemia. J. Exp. Med. 210, 1137–1151 (2013).
Santos, A. M., Jung, J., Aziz, N., Kissil, J. L. & Pure, E. Targeting fibroblast activation protein inhibits tumor stromagenesis and growth in mice. J. Clin. Invest. 119, 3613–3625 (2009).
Alspach, E. et al. p38MAPK plays a crucial role in stromal-mediated tumorigenesis. Cancer Discov. 4, 716–729 (2014).
Brichkina, A. et al. p38MAPK builds a hyaluronan cancer niche to drive lung tumorigenesis. Genes Dev. 30, 2623–2636 (2016).
Matsuo, Y. et al. Involvement of p38ɑ mitogen-activated protein kinase in lung metastasis of tumor cells. J. Biol. Chem. 281, 36767–36775 (2006).
Chen, X. W. et al. CYP4A in tumor-associated macrophages promotes pre-metastatic niche formation and metastasis. Oncogene 36, 5045–5057 (2017).
Correa, D., Somoza, R. A., Lin, P., Schiemann, W. P. & Caplan, A. I. Mesenchymal stem cells regulate melanoma cancer cells extravasation to bone and liver at their perivascular niche. Int. J. Cancer 138, 417–427 (2016).
Murgai, M. et al. KLF4-dependent perivascular cell plasticity mediates pre-metastatic niche formation and metastasis. Nat. Med. 23, 1176–1190 (2017).
van Deventer, H. W., Palmieri, D. A., Wu, Q. P., McCook, E. C. & Serody, J. S. Circulating fibrocytes prepare the lung for cancer metastasis by recruiting Ly-6C+ monocytes via CCL2. J. Immunol. 190, 4861–4867 (2013).
Zheng, H. et al. Vascular endothelial growth factor-induced elimination of the type 1 interferon receptor is required for efficient angiogenesis. Blood 118, 4003–4006 (2011).
Qian, J. et al. Pathogen recognition receptor signaling accelerates phosphorylation-dependent degradation of IFNAR1. PLoS Pathog. 7, e1002065 (2011).
Kalluri, R. The biology and function of exosomes in cancer. J. Clin. Invest. 126, 1208–1215 (2016).
Lobb, R. J., Lima, L. G. & Moller, A. Exosomes: key mediators of metastasis and pre-metastatic niche formation. Semin. Cell Dev. Biol. 67, 3–10 (2017).
Sceneay, J., Smyth, M. J. & Moller, A. The pre-metastatic niche: finding common ground. Cancer Metast. Rev. 32, 449–464 (2013).
Rautela, J. et al. Loss of host type-I IFN signaling accelerates metastasis and impairs NK-cell antitumor function in multiple models of breast cancer. Cancer Immunol. Res. 3, 1207–1217 (2015).
Feig, C. et al. Targeting CXCL12 from FAP-expressing carcinoma-associated fibroblasts synergizes with anti-PD-L1 immunotherapy in pancreatic cancer. Proc. Natl Acad. Sci. USA 110, 20212–20217 (2013).
Ohlund, D., Elyada, E. & Tuveson, D. Fibroblast heterogeneity in the cancer wound. J. Exp. Med. 211, 1503–1523 (2014).
Ohlund, D. et al. Distinct populations of inflammatory fibroblasts and myofibroblasts in pancreatic cancer. J. Exp. Med. 214, 579–596 (2017).
Choudhury, S. R. et al. Dipeptidase-1 is an adhesion receptor for neutrophil recruitment in lungs and liver. Cell 178, 1205–1221 e1217 (2019).
Lo, A. et al. Fibroblast activation protein augments progression and metastasis of pancreatic ductal adenocarcinoma. JCI Insight 2, pii: 92232 (2017).
Albrengues, J. et al. Neutrophil extracellular traps produced during inflammation awaken dormant cancer cells in mice. Science 361, eaao4227 (2018).
Steeg, P. S. Targeting metastasis. Nat. Rev. Cancer 16, 201–218 (2016).
Bragado, P. et al. TGF-β2 dictates disseminated tumour cell fate in target organs through TGF-β-RIII and p38ɑ/β signalling. Nat. Cell Biol. 15, 1351–1361 (2013).
Wu, X. et al. Ubiquitin-conjugating enzyme Ubc13 controls breast cancer metastasis through a TAK1-p38 MAP kinase cascade. Proc. Natl Acad. Sci. USA 111, 13870–13875 (2014).
Katlinski, K. V. et al. Inactivation of interferon receptor promotes the establishment of immune privileged tumor microenvironment. Cancer Cell 31, 194–207 (2017).
Patnaik, A. et al. A first-in-human phase I study of the oral p38 MAPK inhibitor, ralimetinib (LY2228820 dimesylate), in patients with advanced cancer. Clin. Cancer Res. 22, 1095–1102 (2016).
Kumar, V. et al. CD45 phosphatase inhibits STAT3 transcription factor activity in myeloid cells and promotes tumor-associated macrophage differentiation. Immunity 44, 303–315 (2016).
Acknowledgements
This work was supported by the PA Department of Health 2017 Health Research Formula Fund (to S.Y.F.); by the NIH/NCI R01 grant no. CA216936 (to S.Y.F. and D.I.G.), grant no. CA092900 (to S.Y.F.), grant no. CA229803 (to B.Z.S) and grant no. P01 CA165997 (to C.K., J.A.D. and S.Y.F.); as well as by additional support from grant no. F32 CA206431 (to A.O.), grant no. T32 CA009140 (to K.V.K.) and grant no. CA121973 (to J.M.K.). We thank Y. Wang, S. Weiss and E. Brown for the reagents, and A. Gamero (Temple University) and the members of the Gabrilovich, Fuchs, Diehl and Koumenis laboratories for critical suggestions.
Author information
Authors and Affiliations
Contributions
S.Y.F. and J.G. conceived the study and designed the research. J.G. performed most of the experiments with the help of F.Z., C.C., A.O. and H.Z. and interpreted the data. K.V.K. and D.P.B performed RNA-sequencing data analysis. K.A.-T. performed the immunosuppression assay. J.L. performed ChIP experiments. C.S. and J.M.K. provided human samples and helped with analysis. B.E.S., A.C.W., T.W.M., L.T. and E.P. developed key reagents and provided technical assistance. S.Y.F. and J.G. wrote the manuscript. B.Z.S., D.I.G., E.P., C.K., S.W.R. and J.A.D. helped to design the research, discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Characterization of the metastasis in the lung tissues of B16F1 and B16F10 tumor bearing mice.
a, The primary tumor growth in WT mice s.c injected with 1 × 105 B16F1 or B16F10 tumor cells prior to surgery at equivalent tumor size (~200 mm2). Data shown as mean ± SEM (n = 7 mice per group). Repeated-measure two-way ANOVA and Sidak’s multiple comparisons test were performed. b, A representative lung images and the corresponding H&E stained lung sections in the B16F1 and B16F10 tumor bearing mice after surgery. Scale bar: 1 mm. This experiment was repeated three times independently with similar results. c, Quantification of the number of metastatic lesions and total area in the lung tissues from B16F1 and B16F10 tumor bearing mice after surgery as shown in b. Data shown as mean ± SEM (n = 7 mice per group). Two-tailed Unpaired t test was performed for the comparison. d, The primary tumor growth in the B16F1 tumor bearing mice treated with SFM or B16F10 TCM prior to surgery (100 μl every other day until primary tumor removal upon reaching the size ~200 mm2). n = 5 mice per group.
Extended Data Fig. 2 Tumor derived factors induce p38 activation and IFNAR1 downregulation.
a, A representative western blot analysis of p-p38 and total p38 in the leukocytes isolated from the peripheral blood of melanoma patients with metastasis (Met) and without metastasis (Non-Met). The ratio of p-38 to p38 was shown at bottom for each patient. This experiment was repeated three times independently with similar results. b, A representative flow cytometry histogram (left) and the quantification of surface IFNAR1 level (right) in WT lung fibroblasts 2 hr after SFM, conditioned media from normal lung fibroblasts (FCM), or TCM from different tumor cells including B16F1, B16F10, MH6499c4, and E0771. Quantitative data shown as mean ± SEM (n = 3 biologically independent samples). Two- tailed Unpaired t test was performed for the comparisons between two groups. c, A representative immunofluorescence staining of IFNAR1 (left) and the quantification of IFNAR1 level (right) in the lung tissues from WT mice treated with SFM, B16F1 TCM or B16F10 TCM (100 µl i.v., 3x per week for 3 weeks). Scale bar: 100 µm. Quantitative data shown as mean ± SEM (n = 10 mice per group). Two-tailed Unpaired t test was performed for the comparisons between two groups. d, Representative lung images and the corresponding H&E-stained lung sections in WT and Ifnar1−/− B16F1 tumor bearing mice after surgery. Lung metastases were analyzed around 30–60 days after primary tumor removal upon reaching the size ~200mm2. Scale bar: 1 mm. This experiment was repeated three times independently with similar results. e, Quantification of the number of metastatic lesions and total area in the lung tissues from WT (n = 13 mice) and Ifnar1−/− (n = 14 mice) B16F1 tumor bearing mice after surgery. Data shown as mean ± SEM. Two-tailed Unpaired t test was performed for the comparison. f, Kaplan-Meier analysis of survival of WT (n = 9 mice) and Ifnar1−/− (n = 10 mice) B16F1 tumor bearing mice after surgery by Log-rank test.
Extended Data Fig. 3 p38α inhibition or gene deletion impede tumor derived factors-induced downregulation of IFNAR1.
a, A representative flow cytometry analysis (left) and the quantification of surface IFNAR1 level (right) in WT lung fibroblasts pretreated with vehicle (DMSO) or p38 inhibitor Ralimetinib (LY2228820, 4 µM for 2 hr) followed by SFM, or B16F10 TCM treatment for additional 2 hr. Quantitative data shown as mean ± SEM (n = 5,and n = 6 biologically independent samples in Vehicle and LY treated group). Two-way ANOVA and Sidak’s multiple comparisons test were performed. b, A representative western blot analysis of total p38α protein level in the lung tissues of Mapk14fl/fl Ubc9-CreER− and Mapk14fl/flUbc9-CreER+ mice after tamoxifen treatment. Similar results were obtained from three independent experiments. c, A representative immunofluorescence staining of IFNAR1 (left) and the quantification of IFNAR1 level (right) in the lung tissues from Mapk14 competent mice (Mapk14fl/fl) and Mapk14 deleted mice (Mapk14Δ/Δ) treated with SFM or B16F10 TCM (100 µl i.v., 3x per week for 3 weeks). Scale bar: 100 µm. Quantitative data shown as mean ± SEM (n = 5 mice per group). Two-way ANOVA and Sidak’s multiple comparisons test were performed.
Extended Data Fig. 4 p38α-mediated IFNAR1 downregulation drives the formation of pre-metastatic niche in the lungs.
a, A representative immunofluorescence staining of IFNAR1 (left) and the quantification of IFNAR1 level (right) in the lung tissues from WT and SA mice treated with SFM or B16F10 TCM. Scale bar: 100 µm. Quantitative data shown as mean ± SEM (n = 5 mice per group). Two-way ANOVA and Sidak’s multiple comparisons test were performed. b, qPCR analysis of mRNA levels of indicated interferon stimulated genes in the lung tissues of WT and SA mice treated with SFM or B16F10 TCM. Data shown as mean ± SEM (n = 3, n = 4 mice in SFM and TCM treated group respectively). Two-way ANOVA and Tukey’s multiple comparisons test were performed. c, Schematic illustration for analysis of the lung metastasis in the B16F1 tumor bearing SA mice treated with SFM or B16F10 TCM (100 µl i.v. every other day until primary tumor removal upon reaching the size ~200 mm2). d, The primary tumor growth of B16F1 in SA mice treated with SFM or B16F10 TCM prior to surgery. Data shown as mean ± SEM (n = 4 mice per group). Repeated-measure two-way ANOVA and Sidak’s multiple comparisons test were performed. e, Representative lung images and the corresponding H&E-stained lung sections of indicated mice as described in c. Scale bar: 1 mm. Similar results were obtained from three independent experiments. f, Quantification of the number of metastatic lesions and total area in the lung tissues from B16F1 tumor bearing SA mice treated with SFM (n = 10 mice) or B16F10 TCM (n = 9 mice) after surgery. Data shown as mean ± SEM. Two-tailed Unpaired t test was performed for the comparison. g, Kaplan-Meier analysis of survival of B16F1 tumor bearing SA mice treated with SFM (n = 9 mice) or B16F10 TCM (n = 9 mice) after surgery by Log-rank test. h, Representative flow cytometry analysis of tumor cells (TdTomato+) in the lung tissues of WT, SA, and tamoxifen treated-Mapk14fl/flUbc9-CreER− (Mapk14fl/fl) and Mapk14fl/flUbc9-CreER+ (Mapk14Δ/Δ) mice pretreated with SFM or B16F10 TCM followed by intravenous injection of 5*105 B16F10-TdTomato cells. Similar results were obtained from three independent experiments.
Extended Data Fig. 5 p38α-mediated IFNAR1 downregulation induces CXCL1/CXCL3/CXCL5-CXCR2 axis which is critical for the neutrophil recruitment.
a, Representative flow cytometry analysis of myeloid cell subpopulations (above) and the quantification of these cell subpopulations (below) in the lung tissues of WT and SA mice treated with SFM or B16F10 TCM. Quantitative data shown as mean ± SEM (n = 4 mice per group). Two-way ANOVA and Sidak’s multiple comparisons test were performed. b, Representative flow cytometry analysis of the purity of the isolated granulocytes from lung tissues of WT and SA mice treated with B16F10 TCM. Similar results were obtained from three independent experiments. c, Antigen-specific proliferation of CD8+ T cells in the presence of isolated granulocytes (Ly-6G+) from the lung tissues of WT and SA mice treated with B16F10 TCM at a ratio of 1:1, 1:2 or 1:4 measured as the uptake of 3H thymidine and presented relative to that in the absence of granulocytes which set as 100%. Splenic PMN-MDSC isolated from MC38 tumor-bearing mice served as a positive control for the suppression. Data shown as mean ± SEM (n = 6 mice for WT and SA group, n = 3 mice for PMN-MDSC). Two-way ANOVA and Sidak’s multiple comparisons test were performed. d, qPCR analysis of mRNA levels of the indicated chemokines in WT lung fibroblasts 6 hr after SFM, conditioned media from normal lung fibroblasts (FCM), or TCM from different tumor cells including B16F1, B16F10, MH6499c4, and E0771. Data shown as mean ± SEM (n = 3 biologically independent samples). Two-tailed Unpaired t test was performed for the comparisons between two groups. e, qPCR analysis of mRNA levels of the indicated chemokines in the lung tissues of WT mice treated with SFM (n = 3 mice), B16F1 TCM (n = 4 mice) or B16F10 TCM (n = 4 mice). Data shown as mean ± SEM. Two-way ANOVA and Tukey’s multiple comparisons test were performed. f, Representative flow cytometry analysis (left) and the quantification of percent of neutrophils (right) in the lung tissues of WT mice treated with SFM (n = 5 mice), B16F10 TCM plus vehicle (n = 4 mice) or CXCR2 inhibitor (n = 5 mice). Quantitative data shown as mean ± SEM. Two-tailed Unpaired t test was performed for the comparisons between two groups. g, qPCR analysis of mRNA level of Cxcl1 in the lung tissues of SA mice intranasally administered with control (n = 3 mice) or CXCL1 (n = 7 mice) expressing adenovirus. Data shown as mean ± SEM. Two-tailed Unpaired t test was performed for the comparison. h, The percent of total myeloid cells in the lung tissues of SA mice intranasally administered with control (n = 4 mice) or CXCL1 (n = 5 mice) expressing adenovirus. Data shown as mean ± SEM. Two-tailed Unpaired t test was performed for the comparison.
Extended Data Fig. 6 p38α-mediated IFNAR1 downregulation induces FAP expression.
a, A representative western blot analysis of p-p38 and total p38 in the lung fibroblasts of WT, SA, Ifnar1 null (Ifnar1−/−), Mapk14 deletion (Mapk14Δ/Δ), Mapk14 deletion along with Ifnar1 null (Mapk14Δ/Δ Ifnar1−/−) 1 hr after SFM or B16F10 TCM treatment in vitro. b, A representative flow cytometry analysis of surface IFNAR1 level in the lung fibroblasts of WT, SA, Ifnar1−/−, Mapk14Δ/Δ, Mapk14Δ/Δ Ifnar1−/− 2 hr after SFM or B16F10 TCM treatment in vitro. c, The decay of Cxcl1 mRNA in WT and SA lung fibroblasts treated with SFM, or B16F10 TCM for 12 hr, then exposed to Actinomycin D (5 µg/ml) to terminate transcription. Cxcl1 mRNA level was determined by qPCR. Data shown as mean ± SEM (n = 3 independent experiments). Repeated-measure two-way ANOVA and Sidak’s multiple comparisons test were performed. d, ChIP analysis of H3K4me3 binding to the Cxcl1 promoter in the isolated WT and SA lung fibroblasts treated with SFM or B16F10 TCM for 12 hr. Data shown as mean ± SEM (n = 3 independent experiments). Two-way ANOVA and Sidak’s multiple comparisons test were performed. e, The mRNA level of Setdb2 in WT and SA lung fibroblasts treated with PBS or murine IFNβ (1000 IU/ml) for 4 hr. Data shown as mean ± SEM (n = 3 biologically independent samples). Two-way ANOVA and Sidak’s multiple comparisons test were performed. f, The mRNA level of Setdb2 in WT and SA lung fibroblasts treated with SFM or B16F10 TCM for 4 hr. Data shown as mean ± SEM (n = 3 biologically independent samples). Two-way ANOVA and Sidak’s multiple comparisons test were performed. g, A representative flow cytometry histogram (left) and the quantification of FAP expression (right) in the lung fibroblasts of WT, SA, and Fap−/− 24 hr after SFM or B16F10 TCM treatment. Quantitative data shown as mean ± SEM (n = 3 biologically independent samples). Two-way ANOVA and Sidak’s multiple comparisons test were performed. h, A representative immunofluorescence staining of FAP (left) and the quantification of FAP+ cells (right) in the lung tissues from WT mice treated with B16F10 TCM plus vehicle or p38 inhibitor LY2228820. Scale bar: 100 µm. Quantitative data shown as mean ± SEM (n = 5 mice per group). Two-tailed Unpaired t test was performed for the comparison. i, A representative western blot analysis of p-p38 and total p38 in the lung fibroblasts of WT and Fap−/− 1 hr after SFM or B16F10 TCM treatment in vitro. j, A representative flow cytometry histogram (above) and the quantification of surface IFNAR1 level (bottom) in the lung fibroblasts of WT and Fap−/− 2 hr after SFM or B16F10 TCM treatment in vitro. Quantitative data shown as mean ± SEM (n = 3 biologically independent samples). Two-way ANOVA and Sidak’s multiple comparisons test were performed. k, Genotyping of FapS624A mutant mice. A 677 bp amplicon was amplified by PCR both in the wild-type and FapS624A knock-in mouse. The 1 bp change generated a Stu1 restriction site (AGG | CCT) only in the knock-in mice. The PCR products were cleaned up and then digested with Stu1 producing the cut size of 210 bp and 467 bp. l, A representative flow cytometry analysis of FAP levels in the adult dermal fibroblasts isolated from Fap+/+, Fap−/−, FapS624A/S624A and FapS624A/+ mice. The experiments in a, b, i, k, l were repeated three times independently with similar results, and the results of one representative experiment are shown.
Extended Data Fig. 7 P38α expression in fibroblasts is critical for the generation of pre-metastatic niche.
a, Representative western blot analysis of p38α protein level in the isolated lung fibroblasts from tamoxifen-treated Mapk14fl/flCol1a2-CreER− (Mapk14fl/fl) and Mapk14fl/flCol1a2-CreER+ (Mapk14ΔFib, p38α knock out specific in fibroblasts) mice. b, A representative flow cytometry gating and histogram of IFNAR1 level in the lung fibroblasts of Mapk14fl/fl and Mapk14ΔFib mice injected with SFM, B16F10 TCM, or MH6499c4 TCM. c, Representative flow cytometry analysis of neutrophils in the lung tissues of Mapk14fl/fl and Mapk14ΔFib mice injected with SFM, B16F10 TCM, MH6499c4 TCM. d, qPCR analysis of the indicated chemokines in the lung tissues of Mapk14fl/fl and Mapk14ΔFib mice pretreated with SFM, B16F10 TCM, or MH6499c4 TCM followed by intravenous injection of 5*105 B16F10-TdTomato cells. Data shown as mean ± SEM (n = 4 mice for SFM treated Mapk14ΔFib group, n = 5 mice for all the other groups). Two-way ANOVA and Tukey’s multiple comparisons test were performed. e, Representative flow cytometry analysis (left) and the quantification of percent and absolute number (right) of neutrophils in the lung tissues of Mapk14fl/fl and Mapk14ΔFib mice pretreated with SFM, B16F10 TCM, MH6499c4 TCM followed by intravenous injection of 5*105 B16F10-TdTomato cells. Quantitative data shown as mean ± SEM (n = 4 mice for SFM treated group, n = 5 mice for TCM treated group). Two-way ANOVA and Tukey’s multiple comparisons test were performed. f, Representative flow cytometry analysis of tumor cells (TdTomato+) in the lung tissues of Mapk14fl/fl and Mapk14ΔFib mice pretreated with SFM, B16F10 TCM, or MH6499c4 TCM followed by intravenous injection of 5*105 B16F10-TdTomato cells. The experiments in a, b, c, f were repeated three times independently with similar results, and the results of one representative experiment are shown.
Extended Data Fig. 8 The effect of p38α deficiency specifically in fibroblasts and p38 inhibitors on the primary tumor.
a, Representative flow cytometry analysis of IFNAR1 level in the isolated lung fibroblasts from tamoxifen treated Ifnar1fl/flCol1a2-CreER− (Ifnar1fl/fl) and Ifnar1fl/flCol1a2-CreER+ (Ifnar1ΔFib, Ifnar1 knock out specific in fibroblasts) mice. Similar results were obtained from three independent experiments. b, qPCR analysis of the indicated chemokines in the lung tissues of Ifnar1fl/fl (n = 7 mice) and Ifnar1ΔFib mice (n = 6 mice) after intravenous injection with 5*105 B16F10-TdTomato cells. Data shown as mean ± SEM. Two-way ANOVA and Sidak’s multiple comparisons test were performed. c, Representative flow cytometry analysis of neutrophils in the lung tissues of Ifnar1fl/fl and Ifnar1ΔFib mice after intravenous injection with 5*105 B16F10-TdTomato cells. Similar results were obtained from three independent experiments. d, Quantification of the percent (left) and the absolute number of neutrophils (right) in the lung tissues as described in c. Data shown as mean ± SEM (n = 6 mice for Ifnar1fl/fl group, n = 7 for Ifnar1ΔFib group). Two-tailed Unpaired t test was performed for the comparison. e, The primary tumor growth of B16F10 in the Mapk14fl/fl (n = 5 mice) and Mapk14ΔFib (n = 8 mice) mice s.c injected with 1 × 105 B16F10 tumor cells prior to surgery at equivalent tumor size (~200 mm2). f, A representative western blot analysis of p-p38 and total p38 in the WT lung fibroblasts 1 hr after treatment with SFM, or TCM from B16F10 tumor cells pretreated with vehicle (DMSO) or p38 inhibitor LY2228820 (4 µM) for 24 hr. Similar results were obtained from three independent experiments. g, A representative flow cytometry histogram (left) and the quantification of surface IFNAR1 level (right) in WT lung fibroblasts 2 hr after treatment with SFM, or TCM from B16F10 tumor cells pretreated with vehicle (DMSO) or p38 inhibitor LY2228820 (4 µM) for 24 hr. Quantitative data shown as mean ± SEM (n = 3 biologically independent samples). Two- tailed Unpaired t test was performed for the comparisons between two groups. h, Primary tumor growth of B16F10 in mice that received vehicle, LY2228820 (LY), or SB203580 (SB) treatment prior to surgery at equivalent tumor size (~200 mm2). Data shown as mean ± SEM (n = 6 mice per group).
Supplementary information
Supplementary Table 1
Genotyping primers and qPCR primers.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 2
Unprocessed western blots.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 6
Statistical source data.
Source Data Fig. 7
Statistical source data.
Source Data Fig. 8
Statistical source data.
Source Data Extended Data Fig. 1
Statistical source data.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 2
Unprocessed western blots.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 3
Unprocessed western blots.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 6
Unprocessed western blots.
Source Data Extended Data Fig. 7
Statistical source data.
Source Data Extended Data Fig. 7
Unprocessed western blots.
Source Data Extended Data Fig. 8
Statistical source data.
Source Data Extended Data Fig. 8
Unprocessed western blots.
Rights and permissions
About this article
Cite this article
Gui, J., Zahedi, F., Ortiz, A. et al. Activation of p38α stress-activated protein kinase drives the formation of the pre-metastatic niche in the lungs. Nat Cancer 1, 603–619 (2020). https://doi.org/10.1038/s43018-020-0064-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s43018-020-0064-0
This article is cited by
-
Targeted nanostrategies eliminate pre-metastatic niche of cancer
Nano Research (2024)
-
RGCC-mediated PLK1 activity drives breast cancer lung metastasis by phosphorylating AMPKα2 to activate oxidative phosphorylation and fatty acid oxidation
Journal of Experimental & Clinical Cancer Research (2023)
-
Coadaptation fostered by the SLIT2-ROBO1 axis facilitates liver metastasis of pancreatic ductal adenocarcinoma
Nature Communications (2023)
-
Induction of the activating transcription factor-4 in the intratumoral CD8+ T cells sustains their viability and anti-tumor activities
Cancer Immunology, Immunotherapy (2023)
-
A palmitate-rich metastatic niche enables metastasis growth via p65 acetylation resulting in pro-metastatic NF-κB signaling
Nature Cancer (2023)