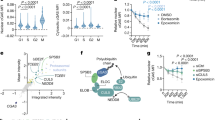
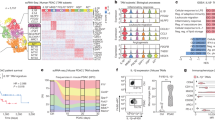
Abstract
Cancer cells express high levels of programmed death ligand 1 (PD-L1), a ligand of the programmed cell death protein 1 (PD-1) receptor on T cells, allowing tumors to suppress T cell activity. Clinical trials utilizing antibodies that disrupt the PD-1/PD-L1 checkpoint have yielded remarkable results, with anti-PD-1 immunotherapy approved as a first-line therapy for patients with lung cancer. We used CRISPR-based screening to identify regulators of PD-L1 in human lung cancer cells, revealing potent induction of PD-L1 upon disruption of heme biosynthesis. Impairment of heme production activates the integrated stress response, allowing bypass of inhibitory upstream open reading frames in the PD-L1 5′ untranslated region, resulting in enhanced PD-L1 translation and suppression of anti-tumor immunity. We demonstrate that integrated stress-response-dependent PD-L1 translation requires the translation initiation factor eIF5B. eIF5B overexpression, which is frequent in lung adenocarcinomas and associated with poor prognosis, is sufficient to induce PD-L1. These findings illuminate mechanisms of immune checkpoint activation and identify targets for therapeutic intervention.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
CRISPR screening and RNA-Seq data that support the findings of this study have been deposited in the Gene Expression Omnibus under accession codes GSE129968 and GSE139120, respectively. Kaplan–Meier survival analysis data for patients with or without EIF5B alterations are from The Cancer Genome Atlas Firehose Legacy LUAD study using cBioPortal. The EIF5B meta-analyses were performed using the Lung Cancer Explorer web portal57 (http://lce.biohpc.swmed.edu/lungcancer/). Source data for Figs. 2–6 and Extended Data Figs. 1, 2, 4–8 and 10 are provided. All other data supporting the findings of this study are available from the corresponding author on reasonable request.
References
Bray, F. et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 68, 394–424 (2018).
Akbay, E. A. et al. Activation of the PD-1 pathway contributes to immune escape in EGFR-driven lung tumors. Cancer Discov. 3, 1355–1363 (2013).
Dong, H. et al. Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat. Med. 8, 793–800 (2002).
Ribas, A. Adaptive immune resistance: how cancer protects from immune attack. Cancer Discov. 5, 915–919 (2015).
Tumeh, P. C. et al. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 515, 568–571 (2014).
Barber, D. L. et al. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature 439, 682–687 (2006).
Herbst, R. S. et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet 387, 1540–1550 (2016).
Reck, M. et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N. Engl. J. Med. 375, 1823–1833 (2016).
Atefi, M. et al. Effects of MAPK and PI3K pathways on PD-L1 expression in melanoma. Clin. Cancer Res. 20, 3446–3457 (2014).
Hu-Lieskovan, S. et al. Improved antitumor activity of immunotherapy with BRAF and MEK inhibitors in BRAFV600E melanoma. Sci. Transl. Med. 7, 279ra241 (2015).
Casey, S. C. et al. MYC regulates the antitumor immune response through CD47 and PD-L1. Science 352, 227–231 (2016).
Coelho, M. A. et al. Oncogenic RAS signaling promotes tumor immunoresistance by stabilizing PD-L1 mRNA. Immunity 47, 1083–1099.e6 (2017).
Zhang, J. et al. Cyclin D–CDK4 kinase destabilizes PD-L1 via cullin 3–SPOP to control cancer immune surveillance. Nature 553, 91–95 (2018).
Lan, B. et al. Association between PD-L1 expression and driver gene status in non-small-cell lung cancer: a meta-analysis. Oncotarget 9, 7684–7699 (2018).
Dever, T. E. et al. Phosphorylation of initiation factor 2α by protein kinase GCN2 mediates gene-specific translational control of GCN4 in yeast. Cell 68, 585–596 (1992).
Pakos-Zebrucka, K. et al. The integrated stress response. EMBO Rep. 17, 1374–1395 (2016).
Ron, D. Translational control in the endoplasmic reticulum stress response. J. Clin. Invest. 110, 1383–1388 (2002).
Donnelly, N., Gorman, A. M., Gupta, S. & Samali, A. The eIF2α kinases: their structures and functions. Cell. Mol. Life Sci. 70, 3493–3511 (2013).
Lu, P. D., Harding, H. P. & Ron, D. Translation reinitiation at alternative open reading frames regulates gene expression in an integrated stress response. J. Cell Biol. 167, 27–33 (2004).
Sendoel, A. et al. Translation from unconventional 5’ start sites drives tumour initiation. Nature 541, 494–499 (2017).
Xu, Y. et al. Translation control of the immune checkpoint in cancer and its therapeutic targeting. Nat. Med. 25, 301–311 (2019).
Shalem, O. et al. Genome-scale CRISPR–Cas9 knockout screening in human cells. Science 343, 84–87 (2014).
Burr, M. L. et al. CMTM6 maintains the expression of PD-L1 and regulates anti-tumour immunity. Nature 549, 101–105 (2017).
Mezzadra, R. et al. Identification of CMTM6 and CMTM4 as PD-L1 protein regulators. Nature 549, 106–110 (2017).
Whitby, F. G., Phillips, J. D., Kushner, J. P. & Hill, C. P. Crystal structure of human uroporphyrinogen decarboxylase. EMBO J. 17, 2463–2471 (1998).
Chen, L. et al. Metastasis is regulated via microRNA-200/ZEB1 axis control of tumour cell PD-L1 expression and intratumoral immunosuppression. Nat. Commun. 5, 5241 (2014).
Lu, L., Han, A. P. & Chen, J. J. Translation initiation control by heme-regulated eukaryotic initiation factor 2α kinase in erythroid cells under cytoplasmic stresses. Mol. Cell. Biol. 21, 7971–7980 (2001).
Rafie-Kolpin, M. et al. Two heme-binding domains of heme-regulated eukaryotic initiation factor-2α kinase. N terminus and kinase insertion. J. Biol. Chem. 275, 5171–5178 (2000).
Han, A. P. et al. Heme-regulated eIF2α kinase (HRI) is required for translational regulation and survival of erythroid precursors in iron deficiency. EMBO J. 20, 6909–6918 (2001).
Harding, H. P. et al. Regulated translation initiation controls stress-induced gene expression in mammalian cells. Mol. Cell 6, 1099–1108 (2000).
Sonenberg, N. & Hinnebusch, A. G. Regulation of translation initiation in eukaryotes: mechanisms and biological targets. Cell 136, 731–745 (2009).
Vattem, K. M. & Wek, R. C. Reinitiation involving upstream ORFs regulates ATF4 mRNA translation in mammalian cells. Proc. Natl Acad. Sci. USA 101, 11269–11274 (2004).
Boyce, M. et al. A selective inhibitor of eIF2α dephosphorylation protects cells from ER stress. Science 307, 935–939 (2005).
Sidrauski, C., McGeachy, A. M., Ingolia, N. T. & Walter, P. The small molecule ISRIB reverses the effects of eIF2α phosphorylation on translation and stress granule assembly. eLife 4, e05033 (2015).
Sidrauski, C. et al. Pharmacological dimerization and activation of the exchange factor eIF2B antagonizes the integrated stress response. eLife 4, e07314 (2015).
Blais, J. D. et al. Activating transcription factor 4 is translationally regulated by hypoxic stress. Mol. Cell. Biol. 24, 7469–7482 (2004).
Paolini, N. A. et al. Ribosome profiling uncovers selective mRNA translation associated with eIF2 phosphorylation in erythroid progenitors. PLoS ONE 13, e0193790 (2018).
Rutkowski, D. T. & Kaufman, R. J. All roads lead to ATF4. Dev. Cell 4, 442–444 (2003).
Andreev, D. E. et al. Translation of 5’ leaders is pervasive in genes resistant to eIF2 repression. eLife 4, e03971 (2015).
Hinnebusch, A. G., Ivanov, I. P. & Sonenberg, N. Translational control by 5’-untranslated regions of eukaryotic mRNAs. Science 352, 1413–1416 (2016).
Kearse, M. G. & Wilusz, J. E. Non-AUG translation: a new start for protein synthesis in eukaryotes. Genes Dev. 31, 1717–1731 (2017).
White, J. P., Reineke, L. C. & Lloyd, R. E. Poliovirus switches to an eIF2-independent mode of translation during infection. J. Virol. 85, 8884–8893 (2011).
Lignitto, L. et al. Nrf2 activation promotes lung cancer metastasis by inhibiting the degradation of Bach1. Cell 178, 316–329 e318 (2019).
Nguyen, H. G. et al. Development of a stress response therapy targeting aggressive prostate cancer. Sci. Trans. Med. 10, eaar2036 (2018).
Ho, J. J. D. et al. Oxygen-sensitive remodeling of central carbon metabolism by archaic eIF5B. Cell Rep. 22, 17–26 (2018).
Golden, R. J. et al. An Argonaute phosphorylation cycle promotes microRNA-mediated silencing. Nature 542, 197–202 (2017).
Hooda, J., Alam, M. & Zhang, L. Measurement of heme synthesis levels in mammalian cells. J. Vis. Exp. https://doi.org/10.3791/51579 (2015).
Gandin, V. et al. Polysome fractionation and analysis of mammalian translatomes on a genome-wide scale. J. Vis. Exp. https://doi.org/10.3791/51455 (2014).
Aronesty, E. Comparison of sequencing utility programs. Open Bioinformatics J. 7, 1–8 (2013).
Dobin, A. et al. STAR: ultrafast universal RNA-Seq aligner. Bioinformatics 29, 15–21 (2013).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Mootha, V. K. et al. PGC-1α-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat. Genet. 34, 267–273 (2003).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl Acad. Sci. USA 102, 15545–15550 (2005).
Kramer, A., Green, J., Pollard, J. Jr & Tugendreich, S. Causal analysis approaches in ingenuity pathway analysis. Bioinformatics 30, 523–530 (2014).
Liberzon, A. et al. Molecular signatures database (MSigDB) 3.0. Bioinformatics 27, 1739–1740 (2011).
Cai, L. et al. LCE: an open web portal to explore gene expression and clinical associations in lung cancer. Oncogene 38, 2551–2564 (2019).
DerSimonian, R. & Laird, N. Meta-analysis in clinical trials. Control. Clin. Trials 7, 177–188 (1986).
Paule, R. C. & Mandel, J. Consensus values and weighting factors. J. Res. Nat. Bur. Stand. 87, 377–385 (1982).
Acknowledgements
We thank the McDermott Center Sequencing Core for sequencing, R. Kaufman (SBP Discovery Institute) for sharing the eIF2α wild-type (S/S) and mutant (A/A) MEFs, D. Gibbons (MD Anderson Cancer Center) for sharing the LLC cells, V. Tagliabracci and A. Sreelatha for assistance with the heme synthesis experiments, J. Jewell and C. Melick for assistance with 35S labeling, M. Buszczak and members of his laboratory for assistance with the polysome profiling, and H. Sadek and members of his laboratory for assistance with the hypoxia treatments. We thank members of the O’Donnell laboratory for critical reading of the manuscript. Funding to K.A.O. was supported by the NCI (R01 CA207763 and P50CA70907), Sidney Kimmel Foundation (SKF-15-067), Cancer Prevention and Research Institute of Texas (CPRIT) (R1101 and RP150676), Welch Foundation (I-1881-20180324), LUNGevity Foundation (2015-03) and UTSW Friends of the Comprehensive Cancer Center. J.T.M. is an investigator of the Howard Hughes Medical Institute and was also supported by the NCI (R35CA197311, P30CA142543 and P50CA196516), CPRIT (RP160249) and Welch Foundation (I-1961-20180324). S.S. was supported by RP140110 and RP160157 (CPRIT), an NIH T32 grant (1T32GM10977601) and an HHMI Med into Grad Grant (56006776). N.N. was supported by RP160157 (CPRIT).
Author information
Authors and Affiliations
Contributions
K.A.O. and S.S. conceived of and designed the study. S.S., J.Z., S.J., C.M.K., B.S. and N.N. performed the investigations. R.J.G. and J.T.M. developed the methodology. K.A.O., S.S., V.S., M.P., J.D.M., Y.-X.F., C.B. and I.I.W. provided resources. B.C., B.M.E., X.Z., G.X., Y.X., C.L., L.Z., Z.W., A.A.S. and C.X. performed the formal analysis. K.A.O. supervised the work. K.A.O. and S.S. wrote the original draft of the manuscript. K.A.O., S.S., R.J.G., C.L., B.M.E., X.Z., C.M.K., M.P., J.D.M. and J.T.M. reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Validation of positive and negative regulators of PD-L1 identified in the CRISPR-Cas9 screen.
a, Flow cytometry analysis of cell surface PD-L1 in H358 cells expressing control sgRNA or sgRNAs targeting CMTM6, SMAD4, DPAGT1 or DNAJC13. b, Western blot analysis confirming loss of target protein and a decrease in PD-L1 in cells from (a). Experiments in a and b were repeated two independent times with similar results, data from a representative experiment are shown. Validation was performed with an independent sgRNA for CMTM6 and SMAD4. c, Western blot analysis confirming loss of target protein and an increase in PD-L1 in cells from Fig. 1f. Experiments were performed three independent times with similar results, data from a representative experiment are shown.
Extended Data Fig. 2 Effects of heme synthesis inhibition on PD-L1 mRNA and protein, and analysis of Pd-l1 mRNA and tumors from LLC cells.
a, Western blot analysis in Calu-6 and H2030 human lung cancer cell lines expressing the indicated sgRNAs. b, Western blot analysis in cells treated with 10 mM succinyl acetone (ALAD inhibitor) for 48 h. c, qRT-PCR analysis of PD-L1 mRNA relative to ACTIN, in cells from (b). Error bars represent SDs from the mean for n = 3 independent experiments. A student’s two-tailed t-test was used to assess statistical significance with ***p = 0.0002. d, Western blot analysis of PD-L1 in H1944 cells treated with 50 μM or 100 μM N-Methyl Protoporphyrin IX (FECH inhibitor) for 48 h. e, qRT-PCR analysis of PD-L1 mRNA in cells from (d). Data represent mean PD-L1 mRNA expression normalized to ACTIN across n = 3 technical replicates, shown as individual data points. All experiments in a-b and d-e were performed two independent times, with similar results. Data from representative experiments are shown. f, qRT-PCR analysis of Pd-l1 mRNA normalized to Actin in LLC cells expressing control or Urod shRNA. Bar graph represents mean normalized Pd-l1 mRNA from n = 3 technical replicates, shown as individual data points from a representative experiment. Experiment was performed two independent times with similar results. g, Representative images of LLC tumors expressing control or Urod shRNA. This experiment was repeated twice with similar results. h, Gating strategy for TIL staining of tumors. Cells were gated based on SSC and FSC. Singlet cells were then gated for APC-Cy7-CD45+ and eFLUOR V500+ dead cells were excluded. i, Representative gating for CD4+ and CD8+ cells in one tumor from each experimental group. CD45+ live cells from each group was obtained by gating as in (h) and then gated for FITC-CD8+ and PE-CD4+ cells to obtain % CD8+ (of CD45+ cells).
Extended Data Fig. 3 Urod depletion induces an immune suppressive and pro-tumorigenic transcriptional program in LLC cells.
a-b, Gene Set Enrichment Analysis highlighting expression of top gene sets significantly increased in LLC cells expressing Urod shRNA. c, Ingenuity Pathway Analysis demonstrating a decrease in expression of genes involved in immune cell recruitment and migration. Graph represents biological processes plotted against their bias adjusted z-scores. Transcriptional analysis was performed on n = 3 biological replicates per group.
Extended Data Fig. 4 Investigating the mechanism of PD-L1 regulation in UROD depleted cells.
a, qRT-PCR analysis of PD-L1 mRNA decay in H1944 cells expressing control or UROD sgRNA after treatment with 10 μM Actinomycin D in n = 3 technical replicates per time point, across 4 time points. b, Quantification of PD-L1 levels, determined by western blot, in H1944 cells expressing control or UROD sgRNA after treatment with 20 μM cycloheximide across 5 time points. c, Immunoprecipitation of endogenous PD-L1 in H1944 cells transfected with control or UROD siRNA treated with 10 μM Velcade for 24 hours. Protein abundance and ubiquitylation monitored by western blot. Experiment in (c) was performed two independent times, with similar results. Data from a representative experiment are shown.
Extended Data Fig. 5 The ISR pathway regulates PD-L1 expression independently of IFN-γ.
a, Western blot analysis in H1944 cells expressing control or UROD sgRNA after treatment with 200 nM ISRIB for 24 h. b, Western blot analysis of UROD in H358 and H1944 cells treated with 10 ng/mL IFN-γ for 24 h. c, Western blot analysis of PD-L1 in H1944 cells treated with DMSO or 100uM Salubrinal for 24 h +/− 10 ng/mL IFN-γ (left) or 10 uM Ruxolitinib (right). d, Western blot analysis of PD-L1 in H1944 cells expressing a control UROD sgRNA +/− 10 ng/mL IFN-γ (left) or 10 uM Ruxolitinib (right). All experiments were performed three independent times with similar results. Representative data are shown.
Extended Data Fig. 6 ISR pathway activation enhances PD-L1 translation, and regulation of the PD-L1 5′ UTR reporter by UROD depletion.
a, qRT-PCR analysis of mean Pd-l1 mRNA, normalized to Actin, in S/S or A/A MEFs expressing either control or Urod shRNA with 3 technical replicates per sample. Experiment was performed two independent times with similar results. Data from a representative experiment are shown. b,c, qRT-PCR analysis of PD-L1 mRNA in 6 individual ribosomal fractions, with 3 technical replicates per sample for two independent primer pairs. Quantification of combined fractions (<3 and >3) from (b) are shown in main Fig. 5c. d,e, qRT-PCR analysis of PD-L1 mRNA in 6 individual ribosomal fractions, with 3 technical replicates per sample for two independent primer pairs. Quantification of combined fractions (<3 and >3) from (d) are shown in main Fig. 5d. f,g, qRT-PCR analysis of ATF4 mRNA in ribosomal fractions from main Fig. 5a (f) or main Fig. 5b (g). ATF4 mRNA abundance was calculated as described in main Fig. 5c and d. Error bars in f and g represent SDs from the mean from three independent fractions (<3 or >3 ribosomes). A student’s two-tailed t-test was performed to determine statistical significance. ***p = 0.00062 (f); ***p = 0.0008 (g). h,i, qRT-PCR analysis of ATF4 mRNA in 6 individual ribosomal fractions. j, qRT-PCR analysis of mean Luciferase mRNA, normalized to Actin, in MEF cells transfected with reporter constructs shown in main Fig. 5e, with 3 technical replicates, shown as individual data points. Experiment was performed two independent times, with similar results. Data from a representative experiment are shown. k, qRT-PCR analysis of mean Luciferase mRNA normalized to ACTIN in H358 cells from main Fig. 5g across n = 3 technical replicates, shown as individual data points. l, qRT-PCR analysis of mean Luciferase mRNA normalized to Actin in MEF cells from main Fig. 5h across n = 3 technical replicates, shown as individual data points. Experiment in (k) was performed in an independent cell line, shown in (l).
Extended Data Fig. 7 eIF5B is necessary for PD-L1 upregulation in UROD depleted human lung cancer cells and the KLN205 syngeneic mouse model.
a, Flow cytometry analysis of cell-surface PD-L1 in independent human lung cancer cells. Bar graph represents Mean Fluorescence Intensity of cell-surface PD-L1 in PD-L1 Low (Calu-6, H2030) and PD-L1 High (H358, H441, Hop62) cell lines. b, Western blot analysis of PD-L1 and eIF5B in Calu-6 and H2030 cells expressing a control or UROD sgRNA transfected with control or EIF5B siRNA. c, Western blot analysis of PD-L1 and eIF5B in H441, H358 and Hop62 cells expressing a control or UROD sgRNA transfected with control or EIF5B siRNA. Experiments in b-c were performed two independent times with similar results. Data from a representative experiment are shown. d, Western blot analysis of PD-L1 in KLN205 cells expressing a Scrambled shRNA or two independent Urod shRNAs. e, Western blot analysis of PD-L1, UROD, and eIF5B in KLN205 cells expressing a control shRNA or Urod shRNA with and without Eif5b shRNA. Experiments in d and e were performed two independent times with similar results. Data from a representative experiment are shown. f, Quantification of tumor volumes of KLN205 cells shown in (e) transplanted to syngeneic DBA/2 mice (n = 8 mice for Scrambled shRNA, n = 5 for Eif5b shRNA, n = 9 for Urod shRNA and n = 8 for Urod + Eif5b shRNA). Linear mixed model was used to assess statistical significance, as described in Methods. p value (Scrambled vs Urod shRNA) = 0.0002, p value (Urod shRNA vs Urod + Eif5b shRNA) = 4.9351e-05.
Extended Data Fig. 8 EIF5B depletion reduces cell proliferation without affecting global translation of human lung cancer cells.
a, MTS cell proliferation assay measuring proliferation of H358, H1944, H2030, Calu-6 and H441 cells transfected with control or EIF5B siRNA with n = 3 biological replicates. Student’s t-test with Holm-Sidak adjustment was performed per time point to assess statistical significance. ** = p < 0.01; *** = p < 0.001; **** = p < 0.0001. Adjusted p values were as follows for Day 1, 2, 3, 4 and 5 respectively. H358: 0.74, 0.096, 0.064, 0.013 and 0.0001, H1944: 0.495, 0.044, 0.042, 0.0449, 0.018, H2030: 0.801, 0.068, 0.06, 0.039, 0.008, Calu6: 0.57, 0.42, 0.002, 0.016 and 0.008, H441: 0.258, 0.079, 0.01, 0.0002 and 0.012. b, Autoradiography analysis of newly synthesized S-35 labeled proteins in cells from (a). Experiments were performed two independent times for H358 and H1944 cells with similar results. Data from a representative experiment are shown.
Extended Data Fig. 9 Meta-analysis of EIF5B expression correlated with survival across lung cancer datasets.
a, Oncoprint plot depicting mRNA upregulation, amplifications, and mutations in EIF5B in human lung adenocarcinoma samples from the TCGA, Firehose Legacy. b, Kaplan-Meier survival analysis of patients with or without EIF5B alterations shown in (a) from the TCGA Firehose Legacy lung adenocarcinoma study using cBioPortal. n = 230 patients, Log rank test p-value = 0.0238. c, Summary of meta analyses results. d, Forest plot of meta-analysis showing association between EIF5B mRNA expression and patient survival outcome in all lung cancer patients across different studies (26 studies with 4,528 patients in total). e, Forest plot showing association between EIF5B expression and patient outcome in lung adenocarcinoma patients across different studies (20 studies with 2,882 patients in total). f, forest plot showing no association between EIF5B and patient survival in lung squamous cell carcinoma patients (26 studies with 1,277 patients in total). The confidence interval was calculated based on two-sided tests.
Extended Data Fig. 10 Luciferase mRNA levels from the PD-L1 5′ UTR reporter with EIF5B overexpression.
a, qRT-PCR analysis of mean Luciferase mRNA normalized to ACTIN in cells from Fig. 6i across three technical replicates, shown as individual data points. b, qRT-PCR analysis of Luciferase mRNA in cells from Fig. 6j, normalized to Actin across three technical replicates, shown as individual data points.
Supplementary information
Supplementary Information
Supplementary Tables 3 and 4.
Supplementary Tables
Supplementary Table 1: Upregulated gene sets in LLC Urod shRNA cells. Supplementary Table 2: Ingenuity pathway analysis of downregulated genes in LLC Urod shRNA cells.
Source data
Source Data Fig. 2
Statistical Source Data
Source Data Fig. 3
Statistical Source Data
Source Data Fig. 4
Statistical Source Data
Source Data Fig. 5
Statistical Source Data
Source Data Fig. 6
Statistical Source Data
Source Data Extended Data Fig. 2
Statistical Source Data
Source Data Extended Data Fig. 4
Statistical Source Data
Source Data Extended Data Fig. 6
Statistical Source Data
Source Data Extended Data Fig. 7
Statistical Source Data
Source Data Extended Data Fig. 8
Statistical Source Data
Source Data Extended Data Fig. 10
Statistical Source Data
Source Data Fig. 2
Unprocessed western blots or gels
Source Data Fig. 3
Unprocessed western blots or gels
Source Data Fig. 4
Unprocessed western blots or gels
Source Data Fig. 6
Unprocessed western blots or gels
Source Data Extended Data Fig. 1
Unprocessed western blots or gels
Source Data Extended Data Fig. 2
Unprocessed western blots or gels
Source Data Extended Data Fig. 4
Unprocessed western blots or gels
Source Data Extended Data Fig. 5
Unprocessed western blots or gels
Source Data Extended Data Fig. 7
Unprocessed western blots or gels
Source Data Extended Data Fig. 8
Unprocessed western blots or gels
Rights and permissions
About this article
Cite this article
Suresh, S., Chen, B., Zhu, J. et al. eIF5B drives integrated stress response-dependent translation of PD-L1 in lung cancer. Nat Cancer 1, 533–545 (2020). https://doi.org/10.1038/s43018-020-0056-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s43018-020-0056-0
This article is cited by
-
Decoding dynamic miRNA:ceRNA interactions unveils therapeutic insights and targets across predominant cancer landscapes
BioData Mining (2024)
-
Non-invasive plasma testing for CD274 UTR structural variations by next-generation sequencing in cancer
Cell Death Discovery (2023)
-
In vivo CRISPR screens reveal Serpinb9 and Adam2 as regulators of immune therapy response in lung cancer
Nature Communications (2023)
-
Improvement of the anticancer efficacy of PD-1/PD-L1 blockade via combination therapy and PD-L1 regulation
Journal of Hematology & Oncology (2022)
-
Cysteine dioxygenase 1 attenuates the proliferation via inducing oxidative stress and integrated stress response in gastric cancer cells
Cell Death Discovery (2022)