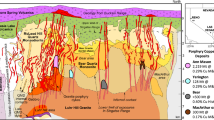
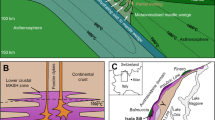
Abstract
Porphyry deposits are large, low-grade metal ore bodies that are formed from hydrothermal fluids derived from an underlying magma reservoir. They are important as major sources of critical metals for industry and society, such as copper and gold. However, the magmatic and redox processes required to form economic-grade porphyry deposits remain poorly understood. In this Review, we discuss advances in understanding crustal magmatic conditions that favour the formation of porphyry Cu deposits at subduction zones. Chalcophile metal fertility of mantle-derived arc magmas is primarily modulated by the amount and nature of residual sulfide phases in the mantle wedge during partial melting. Crustal thickness influences the longevity of lower crustal magma reservoirs and the sulfide saturation history. For example, in thick crust, prolonged magma activity with hydrous and oxidized evolving magmas increases ore potential, whereas thin crust favours high chalcophile element fertility, owing to late sulfide saturation. A shallow depth (<7 km) of fluid exsolution might play a role in increasing Au precipitation efficiency, as immiscible sulfide melts act as a transient storage of chalcophile metals and liberate them to ore fluids. Future studies should aim to identify the predominant sulfide phases in felsic systems to determine their influence on the behaviour of chalcophile elements during magma differentiation.
Key points
-
Prolonged injection of hydrous basaltic magmas and accumulation of andesitic magmas in the mid to lower crust are prerequisites to forming large porphyry deposits because these processes are required to maintain a long-lived magmatic system and associated hydrothermal activity in the shallow crust.
-
Crustal thickness influences the duration and volume of magma activity, timing of sulfide saturation, chalcophile element fertility and emplacement depth of porphyry intrusions.
-
Thick crusts (>40 km) increase porphyry Cu ore potential by producing voluminous and hydrous magmas in long-lived (≥2–3 Ma) mid to lower crustal magma reservoirs at ∼30–70 km depth, which can result in the formation of supergiant to giant porphyry Cu deposits if a combination of other ore-forming conditions is fulfilled.
-
In thin crust (<40 km), late sulfide saturation and high chalcophile element fertility in shallow magma reservoirs (∼5–15 km depth) increase Au-rich porphyry Cu ore potential.
-
Immiscible sulfide melts can act as temporary metal storage locations when the sulfide melts and exsolved fluids interact in shallow magma reservoirs.
-
Depth of porphyry emplacement (∼1–7 km), magma alkalinity and Au fertility control Au endowments in porphyry Cu deposits
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Arndt, N. T. et al. Future global mineral resources. Geochem. Perspect. Lett. 6, 1–2 (2017).
Schipper, B. W. et al. Estimating global copper demand until 2100 with regression and stock dynamics. Resour. Conserv. Recycl. 132, 28–36 (2018).
Sillitoe, R. H. Porphyry copper systems. Econ. Geol. 105, 3–41 (2010).
Chiaradia, M. Gold endowments of porphyry deposits controlled by precipitation efficiency. Nat. Commun. 11, 248 (2020). Revealed different Au precipitation efficiencies between Au-rich and Au-poor porphyry Cu deposits and suggested a link between emplacement depth and Au endowment.
Lee, C. T. A. & Tang, M. How to make porphyry copper deposits. Earth Planet. Sci. Lett. 529, 115868 (2020). Proposed that auto-oxidation by garnet fractionation in thick arcs can have important effects on the formation of porphyry Cu deposits.
Richards, J. P. The oxidation state, and sulfur and Cu contents of arc magmas: implications for metallogeny. Lithos 233, 27–45 (2015).
Chiaradia, M. Copper enrichment in arc magmas controlled by overriding plate thickness. Nat. Geosci. 7, 43–46 (2014).
Loucks, R. R. Distinctive composition of copper-ore-forming arc magmas. Aust. J. Earth Sci. 61, 5–16 (2014).
Annen, C., Blundy, J. D. & Sparks, R. S. J. The genesis of intermediate and silicic magmas in deep crustal hot zones. J. Petrol. 47, 505–539 (2005). Pioneering study that provided a model, based on experimental data and numerical modelling, for the formation of hydrous intermediate and silicic magmas in the lower crustal magma reservoir in subduction zones.
Chelle-Michou, C., Rottier, B., Caricchi, L. & Simpson, G. Tempo of magma degassing and the genesis of porphyry copper deposits. Sci. Rep. 7, 40566 (2017).
Chiaradia, M. & Caricchi, L. Stochastic modelling of deep magmatic controls on porphyry copper deposit endowment. Sci. Rep. 7, 44523 (2017). Revealed the importance of prolonged magma accumulation and evolution in the lower crustal reservoir, generating large amounts of hydrous andesitic magma that contains enough water to deliver large amounts of Cu and Au in porphyry deposits.
Blundy, J., Mavrogenes, J., Tattitch, B., Sparks, S. & Gilmer, A. Generation of porphyry copper deposits by gas–brine reaction in volcanic arcs. Nat. Geosci. 8, 235–240 (2015).
Henley, R. W. et al. Porphyry copper deposit formation by sub-volcanic sulphur dioxide flux and chemisorption. Nat. Geosci. 8, 210–215 (2015).
Li, Y. et al. An essential role for sulfur in sulfide-silicate melt partitioning of gold and magmatic gold transport at subduction settings. Earth Planet. Sci. Lett. 528, 115850 (2019).
Matjuschkin, V., Blundy, J. D. & Brooker, R. A. The effect of pressure on sulphur speciation in mid- to deep-crustal arc magmas and implications for the formation of porphyry copper deposits. Contrib. Mineral. Petrol. 171, 66 (2016).
Mungall, J. E., Brenan, J. M., Godel, B., Barnes, S. J. & Gaillard, F. Transport of metals and sulphur in magmas by flotation of sulphide melt on vapour bubbles. Nat. Geosci. 8, 216–219 (2015). First experimental study to demonstrate a mechanism for sulfur and metal transfer from sulfide melt to magmatic vapour.
Cocker, H. A., Valente, D. L., Park, J. W. & Campbell, I. H. Using platinum group elements to identify sulfide saturation in a porphyry Cu system: the El Abra porphyry Cu deposit, Northern Chile. J. Petrol. 56, 2491–2514 (2015).
Park, J. W. et al. Chalcophile element fertility and the formation of porphyry Cu ± Au deposits. Miner. Deposita 54, 657–670 (2019). Found the correlation between chalcophile element fertility and porphyry ore type (Cu-Au versus Cu versus barren) using a platinum group element as a chalcophile element fertility indicator.
Richards, J. P. Tectono-magmatic precursors for porphyry Cu-(Mo-Au) deposit formation. Econ. Geol. Bull. Soc. 98, 1515–1533 (2003).
Wilkinson, J. J. Triggers for the formation of porphyry ore deposits in magmatic arcs. Nat. Geosci. 6, 917–925 (2013).
Cooke, D. R., Hollings, P. & Walsh, J. L. Giant porphyry deposits: Characteristics, distribution, and tectonic controls. Econ. Geol. 100, 801–818 (2005).
Burnham, C. W. in Geochemistry of Hydrothermal Ore Deposits (ed. Barnes, H. L.) 71–136 (Wiley, 1979).
Sillitoe, R. Some metallogenic features of gold and copper deposits related to alkaline rocks and consequences for exploration. Miner. Deposita 37, 4–13 (2002).
Audétat, A., Simon, A. C., Hedenquist, J. W., Harris, M. & Camus, F. in Geology and Genesis of Major Copper Deposits and Districts of the World: A Tribute to Richard H. Sillitoe Vol. 16 (Society of Economic Geologists, 2012).
Candela, P. A. et al. in One Hundredth Anniversary Volume (Society of Economic Geologists, 2005).
Hedenquist, J. W. & Lowenstern, J. B. The role of magmas in the formation of hydrothermal ore-deposits. Nature 370, 519–527 (1994).
Richards, J. P. Magmatic to hydrothermal metal fluxes in convergent and collided margins. Ore Geol. Rev. 40, 1–26 (2011).
Richards, J. P. Giant ore deposits formed by optimal alignments and combinations of geological processes. Nat. Geosci. 6, 911–916 (2013).
Seedorff, E. et al. in Economic Geology and the Bulletin of the Society. One Hundredth Anniversary Volume (eds Hedenquist, J. W., Thompson, J. F. H., Goldfarb, R. J. & Richards, J. P.) 251–298 (Society of Economic Geologists, 2005).
Botcharnikov, R. E. et al. Behavior of gold in a magma at sulfide-sulfate transition: Revisited. Am. Mineral. 98, 1459–1464 (2013).
Kiseeva, E. S., Fonseca, R. O. C. & Smythe, D. J. Chalcophile elements and sulfides in the upper mantle. Elements 13, 111–116 (2017).
McInnes, B. I. A., McBride, J. S., Evans, N. J., Lambert, D. D. & Andrew, A. S. Osmium isotope constraints on ore metal recycling in subduction zones. Science 286, 512–516 (1999).
Mungall, J. E. Roasting the mantle: Slab melting and the genesis of major Au and Au-rich Cu deposits. Geology 30, 915–918 (2002). Proposed that highly oxidizing slab-derived melts or supercritical fluids play an essential role in producing Cu-rich and Au-rich primary magmas, increasing porphyry Cu-Au ore potential.
Rehkämper, M. et al. Ir, Ru, Pt, and Pd in basalts and komatiites: New constraints for the geochemical behavior of the platinum-group elements in the mantle. Geochim. Cosmochim. Acta 63, 3915–3934 (1999).
Yao, Z., Qin, K. & Mungall, J. E. Tectonic controls on Ni and Cu contents of primary mantle-derived magmas for the formation of magmatic sulfide deposits. Am. Mineral. 103, 1545–1567 (2018).
Candela, P. A., Brown, P. E. & Chappell, B. W. in The Second Hutton Symposium on the Origin of Granites and Related Rocks Vol. 272 (Geological Society of America, 1992).
Cline, J. S. & Bodnar, R. J. Can economic porphyry copper mineralization be generated by a typical calc-alkaline melt. J. Geophys. Res. Solid 96, 8113–8126 (1991).
Richards, J. P. A shake-up in the porphyry world? Econ. Geol. 113, 1225–1233 (2018).
Spooner, E. T. C. Magmatic sulphide/volatile interaction as a mechanism for producing chalcophile element enriched, Archean Au-quartz, epithermal Au-Ag and Au skarn hydrothermal ore fluids. Ore Geol. Rev. 7, 359–379 (1993).
Richards, J. P., Spell, T., Rameh, E., Razique, A. & Fletcher, T. High Sr/Y magmas reflect arc maturity, high magmatic water content, and porphyry Cu ± Mo ± Au potential: examples from the tethyan arcs of central and eastern Iran and western Pakistan. Econ. Geol. 107, 295–332 (2012).
Francis, R. D. Sulfide globules in mid-ocean ridge basalts (MORB), and the effect of oxygen abundance in Fe-S-O liquids on the ability of those liquids to partition metals from MORB and komatiite magmas. Chem. Geol. 85, 199–213 (1990).
Mungall, J. E. & Brenan, J. M. Partitioning of platinum-group elements and Au between sulfide liquid and basalt and the origins of mantle-crust fractionation of the chalcophile elements. Geochim. Cosmochim. Acta 125, 265–289 (2014).
Alard, O., Griffin, W. L., Lorand, J. P., Jackson, S. E. & O’Reilly, S. Y. Non-chondritic distribution of the highly siderophile elements in mantle sulphides. Nature 407, 891–894 (2000).
Barnes, S.-J., van Achterbergh, E., Makovicky, E. & Li, C. Proton microprobe results for the partitioning of platinum-group elements between monosulphide solid solution and sulphide liquid. S. Afr. J. Geol. 104, 275–286 (2001).
Li, C., Barnes, S. J., Makovicky, E., Rose-Hansen, J. & Makovicky, M. Partitioning of nickel, copper, iridium, rhenium, platinum, and palladium between monosulfide solid solution and sulfide liquid: Effects of composition and temperature. Geochim. Cosmochim. Acta 60, 1231–1238 (1996).
Mungall, J. E., Andrews, D. R. A., Cabri, L. J., Sylvester, P. J. & Tubrett, M. Partitioning of Cu, Ni, Au, and platinum-group elements between monosulfide solid solution and sulfide melt under controlled oxygen and sulfur fugacities. Geochim. Cosmochim. Acta 69, 4349–4360 (2005).
Du, J. & Audétat, A. Early sulfide saturation is not detrimental to porphyry Cu-Au formation. Geology 48, 519–524 (2020).
Zhang, D. & Audétat, A. What caused the formation of the giant Bingham Canyon porphyry Cu-Mo-Au deposit? Insights from melt inclusions and magmatic sulfides. Econ. Geol. Bull. Soc. 112, 221–244 (2017).
Sun, W.-d et al. The link between reduced porphyry copper deposits and oxidized magmas. Geochim. Cosmochim. Acta 103, 263–275 (2013).
Kiseeva, E. S. & Wood, B. J. A simple model for chalcophile element partitioning between sulphide and silicate liquids with geochemical applications. Earth Planet. Sci. Lett. 383, 68–81 (2013).
Ripley, E. M., Brophy, J. G. & Li, C. Copper solubility in a basaltic melt and sulfide liquid/silicate melt partition coefficients of Cu and Fe. Geochim. Cosmochim. Acta 66, 2791–2800 (2002).
Zhang, Z. & Hirschmann, M. M. Experimental constraints on mantle sulfide melting up to 8 GPa. Am. Mineral. 101, 181–192 (2016).
Li, Y. & Audétat, A. Partitioning of V, Mn, Co, Ni, Cu, Zn, As, Mo, Ag, Sn, Sb, W, Au, Pb, and Bi between sulfide phases and hydrous basanite melt at upper mantle conditions. Earth Planet. Sci. Lett. 355-356, 327–340 (2012).
Aulbach, S., Mungall, J. E. & Pearson, D. G. Distribution and processing of highly siderophile elements in cratonic mantle lithosphere. Rev. Mineral. Geochem. 81, 239–304 (2016).
Hamlyn, P. R., Keays, R. R., Cameron, W. E., Crawford, A. J. & Waldron, H. M. Precious metals in magnesian low-Ti lavas: Implications for metallogenesis and sulfur saturation in primary magmas. Geochim. Cosmochim. Acta 49, 1797–1811 (1985).
McDonough, W. F. & Sun, S. S. The composition of the Earth. Chem. Geol. 120, 223–253 (1995).
Mavrogenes, J. A. & O’Neill, H. S. C. The relative effects of pressure, temperature and oxygen fugacity on the solubility of sulfide in mafic magmas. Geochim. Cosmochim. Acta 63, 1173–1180 (1999).
Jugo, P. J. Sulfur content at sulfide saturation in oxidized magmas. Geology 37, 415–418 (2009).
Jugo, P. J., Luth, R. W. & Richards, J. P. Experimental data on the speciation of sulfur as a function of oxygen fugacity in basaltic melts. Geochim. Cosmochim. Acta 69, 497–503 (2005).
Lee, C.-T. A. et al. The redox state of arc mantle using Zn/Fe systematics. Nature 468, 681–685 (2010).
Lee, C. T. A., Leeman, W. P., Canil, D. & Li, Z. X. A. Similar V/Sc systematics in MORB and arc basalts: Implications for the oxygen fugacities of their mantle source regions. J. Petrol. 46, 2313–2336 (2005).
Lee, C. T. A. et al. Copper systematics in arc magmas and implications for crust-mantle differentiation. Science 336, 64–68 (2012).
Mallmann, G. & O’Neill, H. S. C. The crystal/melt partitioning of V during mantle melting as a function of oxygen fugacity compared with some other elements (Al, P, Ca, Sc, Ti, Cr, Fe, Ga, Y, Zr and Nb). J. Petrol. 50, 1765–1794 (2009).
Salters, V. J. M. & Stracke, A. Composition of the depleted mantle. Geochem. Geophys. Geosyst. 5, Q05B07 (2004).
Hildreth, W. & Moorbath, S. Crustal contributions to arc magmatism in the Andes of central Chile. Contrib. Mineral. Petrol. 98, 455–489 (1988).
Lee, C.-T. A., Lee, T. C. & Wu, C.-T. Modeling the compositional evolution of recharging, evacuating, and fractionating (REFC) magma chambers: Implications for differentiation of arc magmas. Geochim. Cosmochim. Acta 143, 8–22 (2014). Proposed a model to produce hydrous and oxidized arc magma in the long-lived lower crustal magma chamber in subduction zones.
Lee, C.-T. A. & Anderson, D. L. Continental crust formation at arcs, the arclogite “delamination” cycle, and one origin for fertile melting anomalies in the mantle. Sci. Bull. 60, 1141–1156 (2015).
Alonso-Perez, R., Müntener, O. & Ulmer, P. Igneous garnet and amphibole fractionation in the roots of island arcs: experimental constraints on andesitic liquids. Contrib. Mineral. Petrol. 157, 541–558 (2008).
Tang, M., Erdman, M., Eldridge, G. & Lee, C.-T. A. The redox “filter” beneath magmatic orogens and the formation of continental crust. Sci. Adv. 4, eaar4444 (2018).
Tang, M., Lee, C.-T. A., Costin, G. & Höfer, H. E. Recycling reduced iron at the base of magmatic orogens. Earth Planet. Sci. Lett. 528, 115827 (2019).
Cox, D., Watt, S. F. L., Jenner, F. E., Hastie, A. R. & Hammond, S. J. Chalcophile element processing beneath a continental arc stratovolcano. Earth Planet. Sci. Lett. 522, 1–11 (2019).
Cox, D. et al. Elevated magma fluxes deliver high-Cu magmas to the upper crust. Geology 48, 957–960 (2020).
Etschmann, B. E. et al. An in situ XAS study of copper(I) transport as hydrosulfide complexes in hydrothermal solutions (25–592 °C, 180–600 bar): Speciation and solubility in vapor and liquid phases. Geochim. Cosmochim. Acta 74, 4723–4739 (2010).
Pokrovski, G. S., Borisova, A. Y. & Harrichoury, J.-C. The effect of sulfur on vapor–liquid fractionation of metals in hydrothermal systems. Earth Planet. Sci. Lett. 266, 345–362 (2008).
Seo, J. H., Guillong, M. & Heinrich, C. A. The role of sulfur in the formation of magmatic–hydrothermal copper–gold deposits. Earth Planet. Sci. Lett. 282, 323–328 (2009).
Seo, J. H., Guillong, M. & Heinrich, C. A. Separation of molybdenum and copper in porphyry deposits: the roles of sulfur, redox, and ph in ore mineral deposition at bingham canyon. Econ. Geol. 107, 333–356 (2012).
Ballard, J. R., Palin, M. J. & Campbell, I. H. Relative oxidation states of magmas inferred from Ce(IV)/Ce(III) in zircon: application to porphyry copper deposits of northern Chile. Contrib. Mineral. Petrol. 144, 347–364 (2002).
Hao, H. D., Campbell, I. H., Richards, J. P., Nakamura, E. & Sakaguchi, C. Platinum-group element geochemistry of the Escondida igneous suites, Northern chile: implications for ore formation. J. Petrol. 60, 487–514 (2019).
Stern, C. R., Skewes, M. A. & Arévalo, A. Magmatic evolution of the giant El Teniente Cu–Mo deposit, central Chile. J. Petrol. 52, 1591–1617 (2010).
Zimmer, M. M. et al. The role of water in generating the calc-alkaline trend: new volatile data for Aleutian magmas and a new tholeiitic index. J. Petrol. 51, 2411–2444 (2010).
Chapman, J. B., Ducea, M. N., DeCelles, P. G. & Profeta, L. Tracking changes in crustal thickness during orogenic evolution with Sr/Y: An example from the North American Cordillera. Geology 43, 919–922 (2015).
Chiaradia, M. Crustal thickness control on Sr/Y signatures of recent arc magmas: an Earth scale perspective. Sci. Rep. 5, 8115 (2015).
Profeta, L. et al. Quantifying crustal thickness over time in magmatic arcs. Sci. Rep. 5, 17786 (2015).
Defant, M. J. & Drummond, M. S. Derivation of some modern arc magmas by melting of young subducted lithosphere. Nature 347, 662–665 (1990).
Sun, W. et al. The genetic association of adakites and Cu–Au ore deposits. Int. Geol. Rev. 53, 691–703 (2011).
Oyarzun, R., Márquez, A., Lillo, J., López, I. & Rivera, S. Giant versus small porphyry copper deposits of Cenozoic age in northern Chile: adakitic versus normal calc-alkaline magmatism. Miner. Deposita 36, 794–798 (2001).
Sajona, F. G. & Maury, R. C. Association of adakites with gold and copper mineralization in the Philippines. C. R. Acad. Sci. 326, 27–34 (1998).
Macpherson, C. G., Dreher, S. T. & Thirlwall, M. F. Adakites without slab melting: High pressure differentiation of island arc magma, Mindanao, the Philippines. Earth Planet. Sci. Lett. 243, 581–593 (2006).
Richards, J. P. & Kerrich, R. Special paper: adakite-like rocks: their diverse origins and questionable role in metallogenesis. Econ. Geol. 102, 537–576 (2007).
Chiaradia, M., Ulianov, A., Kouzmanov, K. & Beate, B. Why large porphyry Cu deposits like high Sr/Y magmas? Sci. Rep. 2, 685 (2012).
Richards, J. P. High Sr/Y arc magmas and porphyry Cu ± Mo ± Au deposits: Just add water. Econ. Geol. 106, 1075–1081 (2011).
Singer, D. A. World class base and precious metal deposits; a quantitative analysis. Econ. Geol. 90, 88–104 (1995).
Ariskin, A. A. et al. Modeling solubility of Fe-Ni sulfides in basaltic magmas: the effect of nickel. Econ. Geol. 108, 1983–2003 (2013).
Li, C. & Ripley, E. M. Empirical equations to predict the sulfur content of mafic magmas at sulfide saturation and applications to magmatic sulfide deposits. Miner. Deposita 40, 218–230 (2005).
O’Neill, H. S. C. & Mavrogenes, J. A. The sulfide capacity and the sulfur content at sulfide saturation of silicate melts at 1400 °C and 1 bar. J. Petrol. 43, 1049–1087 (2002).
Park, J.-W., Campbell, I. H. & Arculus, R. J. Platinum-alloy and sulfur saturation in an arc-related basalt to rhyolite suite: Evidence from the Pual Ridge lavas, the Eastern Manus Basin. Geochim. Cosmochim. Acta 101, 76–95 (2013).
Park, J. W., Campbell, I. H., Kim, J. & Moon, J. W. The role of late sulfide saturation in the formation of a Cu- and Au-rich magma: insights from the platinum group element geochemistry of Niuatahi–Motutahi lavas, Tonga rear arc. J. Petrol. 56, 59–81 (2015).
Lowczak, J. N., Campbell, I. H., Cocker, H., Park, J. W. & Cooke, D. R. Platinum-group element geochemistry of the Forest Reef Volcanics, southeastern Australia: Implications for porphyry Au-Cu mineralisation. Geochim. Cosmochim. Acta 220, 385–406 (2018).
Hao, H., Campbell, I. H., Arculus, R. J. & Perfit, M. R. Using precious metal probes to quantify mid-ocean ridge magmatic processes. Earth Planet. Sci. Lett. 553, 116603 (2021).
Chen, K. et al. Sulfide-bearing cumulates in deep continental arcs: The missing copper reservoir. Earth Planet. Sci. Lett. 531, 115971 (2020).
Chin, E. J., Shimizu, K., Bybee, G. M. & Erdman, M. E. On the development of the calc-alkaline and tholeiitic magma series: A deep crustal cumulate perspective. Earth Planet. Sci. Lett. 482, 277–287 (2018).
Jenner, F. E. Cumulate causes for the low contents of sulfide-loving elements in the continental crust. Nat. Geosci. 10, 524–529 (2017).
Straub, S. M., Gómez-Tuena, A. & Vannucchi, P. Subduction erosion and arc volcanism. Nat. Rev. Earth Environ. 1, 574–589 (2020).
Wykes, J. L., O’Neill, H. S. C. & Mavrogenes, J. A. The effect of FeO on the sulfur content at sulfide saturation (SCSS) and the selenium content at selenide saturation of silicate melts. J. Petrol. 56, 1407–1424 (2015).
Iacono-Marziano, G., Ferraina, C., Gaillard, F., Di Carlo, I. & Arndt, N. T. Assimilation of sulfate and carbonaceous rocks: Experimental study, thermodynamic modeling and application to the Noril’sk-Talnakh region (Russia). Ore Geol. Rev. 90, 399–413 (2017).
Ripley, E. M. & Li, C. Sulfide saturation in mafic magmas: Is external sulfur required for magmatic Ni-Cu-(PGE) ore genesis? Econ. Geol. 108, 45–58 (2013).
Tomkins, A. G., Rebryna, K. C., Weinberg, R. F. & Schaefer, B. F. Magmatic sulfide formation by reduction of oxidized arc basalt. J. Petrol. 53, 1537–1567 (2012).
Core, D. P., Kesler, S. E. & Essene, E. J. Unusually Cu-rich magmas associated with giant porphyry copper deposits: evidence from Bingham, Utah. Geology 34, 41–44 (2006).
Richards, J. P. Postsubduction porphyry Cu-Au and epithermal Au deposits: Products of remelting of subduction-modified lithosphere. Geology 37, 247–250 (2009).
Karlstrom, L., Lee, C.-T. A. & Manga, M. The role of magmatically driven lithospheric thickening on arc front migration. Geochem. Geophys. Geosyst. 15, 2655–2675 (2014).
Cao, M. et al. Physicochemical processes in the magma chamber under the Black Mountain porphyry Cu-Au deposit, Philippines: Insights from mineral chemistry and implications for mineralization. Econ. Geol. 113, 63–82 (2018).
Dugmore, M. A., Leaman, P. W. & Philip, R. Discovery of the Mt Bini porphyry copper-gold-molybdenum deposit in the Owen Stanley Ranges, Papua New Guinea—A geochemical case history. J. Geochem. Explor. 57, 89–100 (1996).
Olson, N. H., Dilles, J. H., Kent, A. J. R. & Lang, J. R. Geochemistry of the Cretaceous Kaskanak batholith and genesis of the Pebble porphyry Cu-Au-Mo deposit, southwest Alaska. Am. Mineral. 102, 1597–1621 (2017).
Shinohara, H. & Hedenquist, J. W. Constraints on magma degassing beneath the Far Southeast porphyry Cu–Au deposit, Philippines. J. Petrol. 38, 1741–1752 (1997).
van Dongen, M., Weinberg, R. F., Tomkins, A. G., Armstrong, R. A. & Woodhead, J. D. Recycling of Proterozoic crust in Pleistocene juvenile magma and rapid formation of the Ok Tedi porphyry Cu–Au deposit, Papua New Guinea. Lithos 114, 282–292 (2010).
Hao, H. D., Campbell, I. H., Park, J. W. & Cooke, D. R. Platinum-group element geochemistry used to determine Cu and Au fertility in the Northparkes igneous suites, New South Wales, Australia. Geochim. Cosmochim. Acta 216, 372–392 (2017).
Crocket, J. H., Fleet, M. E. & S, W. E. Implications of composition for experimental partitioning of platinum-group elements and gold between sulfide liquid and basalt melt: The significance of nickel content. Geochim. Cosmochim. Acta 61, 4139–4149 (1997).
Crocket, J. H. PGE in fresh basalt, hydrothermal alteration products, and volcanic incrustations of Kilauea volcano, Hawaii. Geochim. Cosmochim. Acta 64, 1791–1807 (2000).
Park, J. W., Campbell, I. H. & Kim, J. Abundances of platinum group elements in native sulfur condensates from the Niuatahi-Motutahi submarine volcano, Tonga rear arc: Implications for PGE mineralization in porphyry deposits. Geochim. Cosmochim. Acta 174, 236–246 (2016).
Cocker, H. Platinum Group Elements: Indicators of Sulfide Saturation in Intermediate to Felsic Magmatic Systems and Implications for Porphyry Deposit Formation. PhD thesis, Australian National University (2016).
Halter, W. E., Heinrich, C. A. & Pettke, T. Magma evolution and the formation of porphyry Cu–Au ore fluids: evidence from silicate and sulfide melt inclusions. Miner. Deposita 39, 845–863 (2005).
Halter, W. E., Pettke, T. & Heinrich, C. A. The origin of Cu/Au ratios in porphyry-type ore deposits. Science 296, 1844–1846 (2002).
Keith, J. D. et al. The role of magmatic sulfides and mafic alkaline magmas in the Bingham and Tintic mining districts, Utah. J. Petrol. 38, 1679–1690 (1997).
Larocque, A. C. L., Stimac, J. A., Keith, J. D. & Huminicki, M. A. E. Evidence for open-system behavior in immiscible Fe–S–O liquids in silicate magmas: Implications for contributions of metals and sulfur to ore-forming fluids. Can. Mineral. 38, 1233–1249 (2000).
Nadeau, O., Williams-Jones, A. E. & Stix, J. Sulphide magma as a source of metals in arc-related magmatic hydrothermal ore fluids. Nat. Geosci. 3, 501–505 (2010).
Reekie, C. D. J. et al. Sulfide resorption during crustal ascent and degassing of oceanic plateau basalts. Nat. Commun. 10, 82 (2019).
Stavast, W. J. A. et al. The fate of magmatic sulfides during intrusion or eruption, Bingham and Tintic districts, Utah. Econ. Geol. 101, 329–345 (2006).
Yao, Z. & Mungall, J. E. Flotation mechanism of sulphide melt on vapour bubbles in partially molten magmatic systems. Earth Planet. Sci. Lett. 542, 116298 (2020).
Barnes, S. J., Le Vaillant, M., Godel, B. & Lesher, C. M. Droplets and bubbles: solidification of sulphide-rich vapour-saturated orthocumulates in the Norilsk-Talnakh Ni–Cu–PGE ore-bearing Intrusions. J. Petrol. 60, 269–300 (2018).
Le Vaillant, M., Barnes, S. J., Mungall, J. E. & Mungall, E. L. Role of degassing of the Noril’sk nickel deposits in the Permian–Triassic mass extinction event. Proc. Natl Acad. Sci. USA 114, 2485–2490 (2017).
Candela, P. A. A review of shallow, ore-related granites: textures, volatiles, and ore metals. J. Petrol. 38, 1619–1633 (1997).
Murakami, H. et al. The relation between Cu/Au ratio and formation depth of porphyry-style Cu–Au ± Mo deposits. Miner. Deposita 45, 11–21 (2010).
D’Angelo, M. et al. Petrogenesis and magmatic evolution of the Guichon Creek batholith: Highland Valley porphyry Cu ± (Mo) district, south-central British Columbia. Econ. Geol. 112, 1857–1888 (2017).
Dilles, J. H. Petrology of the Yerington Batholith, Nevada; evidence for evolution of porphyry copper ore fluids. Econ. Geol. 82, 1750–1789 (1987).
Schöpa, A., Annen, C., Dilles, J. H., Sparks, R. S. J. & Blundy, J. D. Magma emplacement rates and porphyry copper deposits: Thermal modeling of the Yerington batholith, Nevada. Econ. Geol. 112, 1653–1672 (2017).
Heinrich, C. A., Driesner, T., Stefánsson, A. & Seward, T. M. Magmatic vapor contraction and the transport of gold from the porphyry environment to epithermal ore deposits. Geology 32, 761–764 (2004).
Kay, S. M., Mpodozis, C., Tittler, A. & Cornejo, P. Tertiary magmatic evolution of the Maricunga mineral belt in Chile. Int. Geol. Rev. 36, 1079–1112 (1994).
Vila, T. & Sillitoe, R. H. Gold-rich porphyry systems in the Maricunga belt, northern Chile. Econ. Geol. 86, 1238–1260 (1991).
Leys, C. A. et al. in Geology and Genesis of Major Copper Deposits and Districts of the World: A Tribute to Richard H. Sillitoe Vol. 16 (Society of Economic Geologists, 2012).
Garwin, S. The geological characteristics, geochemical signature and geophysical expression of porphyry copper-(gold) deposits in the circum-Pacific region. ASEG Ext. Abstr. 2019, 1–4 (2019).
Grondahl, C. & Zajacz, Z. Magmatic controls on the genesis of porphyry Cu–Mo–Au deposits: The Bingham Canyon example. Earth Planet. Sci. Lett. 480, 53–65 (2017).
Holliday, J. R. et al. Porphyry gold–copper mineralisation in the Cadia district, eastern Lachlan Fold Belt, New South Wales, and its relationship to shoshonitic magmatism. Miner. Deposita 37, 100–116 (2002).
Jensen, E. P., Barton, M. D., Hagemann, S. G. & Brown, P. E. in Gold Vol. 13 (Society of Economic Geologists, 2000).
Sillitoe, R. H., Hagemann, S. G. & Brown, P. E. in Gold Vol. 13 (Society of Economic Geologists, 2000).
Wainwright, A. J., Tosdal, R. M., Wooden, J. L., Mazdab, F. K. & Friedman, R. M. U–Pb (zircon) and geochemical constraints on the age, origin, and evolution of Paleozoic arc magmas in the Oyu Tolgoi porphyry Cu–Au district, southern Mongolia. Gondwana Res. 19, 764–787 (2011).
Rock, N. M. S. & Groves, D. I. Do lamprophyres carry gold as well as diamonds? Nature 332, 253–255 (1988).
Zajacz, Z. et al. Alkali metals control the release of gold from volatile-rich magmas. Earth Planet. Sci. Lett. 297, 50–56 (2010).
Holwell, D. A. et al. A metasomatized lithospheric mantle control on the metallogenic signature of post-subduction magmatism. Nat. Commun. 10, 3511 (2019).
Heinrich, C. A. & Candela, P. A. in Treatise on Geochemistry 2nd edn (eds Holland, H. D. & Turekian, K. K.) 1–28 (Elsevier, 2014).
Landtwing, M. R. et al. The Bingham Canyon porphyry Cu-Mo-Au deposit. III. Zoned copper-gold ore deposition by magmatic vapor expansion. Econ. Geol. 105, 91–118 (2010).
Bodnar, R. J., Lecumberri-Sanchez, P., Moncada, D. & Steele-MacInnis, M. in Treatise on Geochemistry 2nd edn (eds Holland, H. D. & Turekian, K. K.) 119–142 (Elsevier, 2014).
Shinohara, H. Exsolution of immiscible vapor and liquid phases from a crystallizing silicate melt: Implications for chlorine and metal transport. Geochim. Cosmochim. Acta 58, 5215–5221 (1994).
Webster, J. D. The exsolution of magmatic hydrosaline chloride liquids. Chem. Geol. 210, 33–48 (2004).
Guo, H. & Audétat, A. Transfer of volatiles and metals from mafic to felsic magmas in composite magma chambers: An experimental study. Geochim. Cosmochim. Acta 198, 360–378 (2017).
Zajacz, Z., Candela, P. A., Piccoli, P. M., Wälle, M. & Sanchez-Valle, C. Gold and copper in volatile saturated mafic to intermediate magmas: Solubilities, partitioning, and implications for ore deposit formation. Geochim. Cosmochim. Acta 91, 140–159 (2012).
Candela, P. A. & Holland, H. D. The partitioning of copper and molybdenum between silicate melts and aqueous fluids. Geochim. Cosmochim. Acta 48, 373–380 (1984).
Simon, A. C. et al. Gold partitioning in melt-vapor-brine systems. Geochim. Cosmochim. Acta 69, 3321–3335 (2005).
Simon, A. C., Pettke, T., Candela, P. A., Piccoli, P. M. & Heinrich, C. A. Copper partitioning in a melt–vapor–brine–magnetite–pyrrhotite assemblage. Geochim. Cosmochim. Acta 70, 5583–5600 (2006).
Williams, T. J., Candela, P. A. & Piccoli, P. M. The partitioning of copper between silicate melts and two-phase aqueous fluids: An experimental investigation at 1 kbar, 800 °C and 0.5 kbar, 850 °C. Contrib. Mineral. Petrol. 121, 388–399 (1995).
Rusk, B. G., Reed, M. H. & Dilles, J. H. Fluid inclusion evidence for magmatic-hydrothermal fluid evolution in the porphyry copper-molybdenum deposit at Butte, Montana. Econ. Geol. 103, 307–334 (2008).
Frank, M. R., Simon, A. C., Pettke, T., Candela, P. A. & Piccoli, P. M. Gold and copper partitioning in magmatic-hydrothermal systems at 800 °C and 100 MPa. Geochim. Cosmochim. Acta 75, 2470–2482 (2011).
Lerchbaumer, L. & Audétat, A. High Cu concentrations in vapor-type fluid inclusions: An artifact? Geochim. Cosmochim. Acta 88, 255–274 (2012). Discovered that the conventional vapour–brine partition coefficients of Cu inferred from natural fluid inclusions are an artefact, showing Cu’s higher affinity for brine than vapour.
Zajacz, Z., Candela, P. A. & Piccoli, P. M. The partitioning of Cu, Au and Mo between liquid and vapor at magmatic temperatures and its implications for the genesis of magmatic-hydrothermal ore deposits. Geochim. Cosmochim. Acta 207, 81–101 (2017).
Driesner, T. & Heinrich, C. A. The system H2O–NaCl. Part I: Correlation formulae for phase relations in temperature–pressure–composition space from 0 to 1000 °C, 0 to 5000 bar, and 0 to 1 XNaCl. Geochim. Cosmochim. Acta 71, 4880–4901 (2007).
Gregory, M. J. A fluid inclusion and stable isotope study of the Pebble porphyry copper-gold-molybdenum deposit, Alaska. Ore Geol. Rev. 80, 1279–1303 (2017).
Crerar, D. A. & Barnes, H. L. Ore solution chemistry; V, Solubilities of chalcopyrite and chalcocite assemblages in hydrothermal solution at 200 degrees to 350 degrees C. Econ. Geol. 71, 772–794 (1976).
Landtwing, M. R. et al. Copper deposition during quartz dissolution by cooling magmatic–hydrothermal fluids: the Bingham porphyry. Earth Planet. Sci. Lett. 235, 229–243 (2005).
Henley, R. W. & Berger, B. R. Nature’s refineries — Metals and metalloids in arc volcanoes. Earth Sci. Rev. 125, 146–170 (2013).
Giggenbach, W. F. Redox processes governing the chemistry of fumarolic gas discharges from White Island, New Zealand. Appl. Geochem. 2, 143–161 (1987).
Gustafson, L. B. & Hunt, J. P. The porphyry copper deposit at El Salvador, Chile. Econ. Geol. 70, 857–912 (1975).
Li, Y. & Audétat, A. Gold solubility and partitioning between sulfide liquid, monosulfide solid solution and hydrous mantle melts: Implications for the formation of Au-rich magmas and crust–mantle differentiation. Geochim. Cosmochim. Acta 118, 247–262 (2013).
Liu, Y. & Brenan, J. Partitioning of platinum-group elements (PGE) and chalcogens (Se, Te, As, Sb, Bi) between monosulfide-solid solution (MSS), intermediate solid solution (ISS) and sulfide liquid at controlled fO2–fS2 conditions. Geochim. Cosmochim. Acta 159, 139–161 (2015).
Costa, S. et al. Tracking metal evolution in arc magmas: Insights from the active volcano of La Fossa, Italy. Lithos 380–381, 105851 (2021).
Wang, Z. et al. Evolution of copper isotopes in arc systems: Insights from lavas and molten sulfur in Niuatahi volcano, Tonga rear arc. Geochim. Cosmochim. Acta 250, 18–33 (2019).
Rottier, B., Audétat, A., Koděra, P. & Lexa, J. Magmatic evolution of the mineralized Štiavnica volcano (Central Slovakia): Evidence from thermobarometry, melt inclusions, and sulfide inclusions. J. Volcanol. Geotherm. Res. 401, 106967 (2020).
Rottier, B., Audétat, A., Koděra, P. & Lexa, J. Origin and evolution of magmas in the porphyry Au-mineralized Javorie volcano (Central Slovakia): Evidence from thermobarometry, melt Inclusions and sulfide inclusions. J. Petrol. 60, 2449–2482 (2020).
Proffett, J. M. High Cu grades in porphyry Cu deposits and their relationship to emplacement depth of magmatic sources. Geology 37, 675–678 (2009).
Hou, Z. et al. A genetic linkage between subduction- and collision-related porphyry Cu deposits in continental collision zones. Geology 43, 247–250 (2015).
Hou, Z. et al. The Miocene Gangdese porphyry copper belt generated during post-collisional extension in the Tibetan Orogen. Ore Geol. Rev. 36, 25–51 (2009).
Hou, Z. et al. Contribution of mantle components within juvenile lower-crust to collisional zone porphyry Cu systems in Tibet. Miner. Deposita 48, 173–192 (2013).
Blanks, D. E. et al. Fluxing of mantle carbon as a physical agent for metallogenic fertilization of the crust. Nat. Commun. 11, 4342 (2020).
Singer, D. A., Berger, V. I. & Moring, B. C. Porphyry copper deposits of the world: Database and grade and tonnage models, 2008. U.S. Geological Survey open-file report 2008-1155. USGS https://pubs.usgs.gov/of/2008/1155/ (2008).
Clark, A. H., Whiting, B. H., Hodgson, C. J. & Mason, R. in Giant Ore Deposits (Society of Economic Geologists, 1993).
Bai, Z.-J., Zhong, H., Hu, R.-Z. & Zhu, W.-G. Early sulfide saturation in arc volcanic rocks of southeast China: Implications for the formation of co-magmatic porphyry–epithermal Cu–Au deposits. Geochim. Cosmochim. Acta 280, 66–84 (2020).
Huang, M.-L. et al. The role of early sulfide saturation in the formation of the Yulong porphyry Cu-Mo deposit: evidence from mineralogy of sulfide melt inclusions and platinum-group element geochemistry. Ore Geol. Rev. 124, 103644 (2020).
Park, J.-W., Campbell, I. H. & Eggins, S. M. Enrichment of Rh, Ru, Ir and Os in Cr spinels from oxidized magmas: Evidence from the Ambae volcano, Vanuatu. Geochim. Cosmochim. Acta 78, 28–50 (2012).
Dale, C. W., Macpherson, C. G., Pearson, D. G., Hammond, S. J. & Arculus, R. J. Inter-element fractionation of highly siderophile elements in the Tonga Arc due to flux melting of a depleted source. Geochim. Cosmochim. Acta 89, 202–225 (2012).
Acknowledgements
J.-W.P. was supported by a fund from the Korea Government Ministry of Science and ICT (NRF-2019R1A2C1009809). I.H.C. was supported by an Australian Research Council Discovery Grant (DP17010340). H.H. acknowledges the support from Brain Pool Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (2019H1D3A1A01102977). M.C. acknowledges support from the Swiss National Science Foundation (200020_162415, 200021_169032). The authors thank J. H. Seo for their discussion and comments on the manuscript.
Author information
Authors and Affiliations
Contributions
J.-W.P., I.H.C. and M.C. substantially contributed to the discussion and writing of the manuscript. H.H. and C.-T.L. contributed to the discussion of the content and reviewed the manuscript before submission. H.H. and M.C. compiled the data sets and drafted the figures.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Earth & Environment thanks E. Melekhova, J. Mungall and J. Dilles (who co-reviewed with M. Campbell) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Glossary
- Sulfide saturation
-
Silicate melt becomes saturated with a sulfide phase, normally an immiscible sulfide melt, and segregates from the silicate melt.
- Hydrothermal system
-
A system that redistributes energy and mass by circulation of hot, water-rich fluid.
- Differentiation
-
Processes that lead to changes in magma composition, such as fractional crystallization, crustal assimilation, recharge and mixing.
- Fluid exsolution
-
A process through which water-rich fluid separates from silicate melt.
- Metasomatized
-
Metamorphic processes that change the chemical composition of a rock in a pervasive manner by interaction with aqueous fluids.
- Chalcophile elements
-
Elements that have a high affinity with sulfur and form sulfide minerals or partition strongly into immiscible sulfide melts.
- Monosulfide solid solution
-
A high-temperature (>∼600 °C) sulfide phase that is mainly composed of Fe with minor Ni and Cu.
- Fractionation
-
Removal and segregation of a mineral from a melt.
- Oxygen fugacity (fO2)
-
Partial pressure of oxygen in a given environment.
- Cumulates
-
Igneous rocks formed by accumulation of crystals from magma.
- Adakite
-
An intermediate to felsic volcanic rock that has geochemical signatures of magma thought to be produced by partial melting of altered basalt.
- Subduction erosion
-
Removal of upper plate materials in active continental margins.
- Delamination
-
Detachment of lower crust and/or mantle lithosphere from the continental crust.
Rights and permissions
About this article
Cite this article
Park, JW., Campbell, I.H., Chiaradia, M. et al. Crustal magmatic controls on the formation of porphyry copper deposits. Nat Rev Earth Environ 2, 542–557 (2021). https://doi.org/10.1038/s43017-021-00182-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s43017-021-00182-8
This article is cited by
-
The Magma Emplacement of a Composite Volcanic-Intrusive System and Its Mineralization
Surveys in Geophysics (2024)
-
Magma mixing and magmatic-to-hydrothermal fluid evolution revealed by chemical and boron isotopic signatures in tourmaline from the Zhunuo–Beimulang porphyry Cu-Mo deposits
Mineralium Deposita (2024)
-
Copper behavior in arc-back-arc systems: Insights into the porphyry Cu metallogeny of the Gangdese belt, southern Tibet
Mineralium Deposita (2024)
-
Imaging the subsurface architecture in porphyry copper deposits using local earthquake tomography
Scientific Reports (2023)
-
Sulfide minerals and chalcophile elements in The Pleiades Volcanic Field, Antarctica: implications for the distribution of chalcophile metals in continental intraplate alkaline magma systems
Contributions to Mineralogy and Petrology (2023)