Abstract
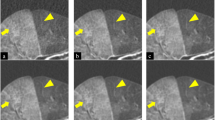
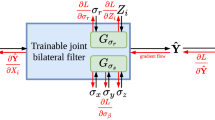
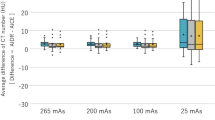
Commercial iterative reconstruction techniques help to reduce the radiation dose of computed tomography (CT), but altered image appearance and artefacts can limit their adoptability and potential use. Deep learning has been investigated for low-dose CT (LDCT). Here, we design a modularized neural network for LDCT and compare it with commercial iterative reconstruction methods from three leading CT vendors. Although popular networks are trained for an end-to-end mapping, our network performs an end-to-process mapping so that intermediate denoised images are obtained with associated noise reduction directions towards a final denoised image. The learned workflow allows radiologists in the loop to optimize the denoising depth in a task-specific fashion. Our network was trained with the Mayo LDCT Dataset and tested on separate chest and abdominal CT exams from Massachusetts General Hospital. The best deep learning reconstructions were systematically compared to the best iterative reconstructions in a double-blinded reader study. This study confirms that our deep learning approach performs either favourably or comparably in terms of noise suppression and structural fidelity, and is much faster than commercial iterative reconstruction algorithms.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The training and validation sets were obtained from the 2016 NIH-AAPM-Mayo Clinic Low-Dose CT Grand Challenge, and are publicly available (http://www.aapm.org/GrandChallenge/LowDoseCT/). The testing data from MGH for this study are protected for patient privacy and will be available for data sharing upon request and after going through an external Institutional Review Board procedure at MGH.
Code availability
The source code and the trained models for this study are publicly available on Github (https://github.com/hmshan/MAP-NN).
References
Wang, G. A perspective on deep imaging. IEEE Access 4, 8914–8924 (2016).
Wang, G., Ye, J. C., Mueller, K. & Fessler, J. A. Image reconstruction is a new frontier of machine learning. IEEE Trans. Med. Imaging 37, 1289–1296 (2018).
Zhu, B., Liu, J. Z., Cauley, S. F., Rosen, B. R. & Rosen, M. S. Image reconstruction by domain-transform manifold learning. Nature 555, 487–492 (2018).
Brenner, D. J. & Hall, E. J. Computed tomography—an increasing source of radiation exposure. New Engl. J. Med. 357, 2277–2284 (2007).
de González, A. B. et al. Projected cancer risks from computed tomographic scans performed in the United States in 2007. Arch. Intern. Med. 169, 2071–2077 (2009).
Smith-Bindman, R. et al. Radiation dose associated with common computed tomography examinations and the associated lifetime attributable risk of cancer. Arch. Intern. Med. 169, 2078–2086 (2009).
National Lung Screening Trial Research Team Reduced lung-cancer mortality with low-dose computed tomographic screening. New Engl. J. Med. 2011, 395–409 (2011).
Wang, J., Lu, H., Li, T. & Liang, Z. Sinogram noise reduction for low-dose CT by statistics-based nonlinear filters. Proc. SPIE 5747, 2059 (2005).
Manduca, A. et al. Projection space denoising with bilateral filtering and CT noise modeling for dose reduction in CT. Med. Phys. 36, 4911–4919 (2009).
Wang, J., Li, T., Lu, H. & Liang, Z. Penalized weighted least-squares approach to sinogram noise reduction and image reconstruction for low-dose X-ray computed tomography. IEEE Trans. Med. Imaging 25, 1272–1283 (2006).
Geyer, L. L. et al. State of the art: iterative CT reconstruction techniques. Radiology 276, 339–357 (2015).
Willemink, M. J. et al. Iterative reconstruction techniques for computed tomography. Part 2: Initial results in dose reduction and image quality. Eur. Radiol. 23, 1632–1642 (2013).
Zheng, X., Ravishankar, S., Long, Y. & Fessler, J. A. PWLS-ULTRA: an efficient clustering and learning-based approach for low-dose 3D CT image reconstruction. IEEE Trans. Med. Imaging 37, 1498–1510 (2018).
Chen, H. et al. Low-dose CT via convolutional neural network. Biomed. Opt. Express 8, 679–694 (2017).
Chen, H. et al. Low-dose CT with a residual encoder–decoder convolutional neural network. IEEE Trans. Med. Imaging 36, 2524–2535 (2017).
Wolterink, J. M., Leiner, T., Viergever, M. A. & Išgum, I. Generative adversarial networks for noise reduction in low-dose CT. IEEE Trans. Med. Imaging 36, 2536–2545 (2017).
Yang, Q. et al. Low-dose CT image denoising using a generative adversarial network with Wasserstein distance and perceptual loss. IEEE Trans. Med. Imaging 37, 1348–1357 (2018).
Kang, E., Chang, W., Yoo, J. & Ye, J. C. Deep convolutional framelet denosing for low-dose CT via wavelet residual network. IEEE Trans. Med. Imaging 37, 1358–1369 (2018).
Shan, H. et al. 3D convolutional encoder–decoder network for low-dose CT via transfer learning from a 2D trained network. IEEE Trans. Med. Imaging 37, 1522–1534 (2018).
Choi, K., Kim, S. W. & Lim, J. S. Real-time image reconstruction for low-dose CT using deep convolutional generative adversarial networks (GANs). In Medical Imaging 2018: Physics of Medical Imaging Vol. 10573, 1057332 (International Society for Optics and Photonics, 2018).
Kim, B., Han, M., Shim, H. & Baek, J. Performance comparison of convolutional neural network based denoising in low dose CT images for various loss functions. In Medical Imaging 2019: Physics of Medical Imaging Vol. 10948, 1094849 (International Society for Optics and Photonics, 2019).
Liu, Y. & Zhang, Y. Low-dose CT restoration via stacked sparse denoising autoencoders. Neurocomputing 284, 80–89 (2018).
You, C. et al. Structurally-sensitive multi-scale deep neural network for low-dose CT denoising. IEEE Access 6, 41839–41855 (2018).
Hu, Z. et al. Artifact correction in low-dose dental CT imaging using Wasserstein generative adversarial networks. Med. Phys. 46, 1686–1696 (2019).
Gholizadeh-Ansari, M., Alirezaie, J. & Babyn, P. Deep learning for low-dose CT denoising using perceptual loss and edge detection layer. Preprint at https://arxiv.org/abs/1902.10127 (2019).
Yi, X. & Babyn, P. Sharpness-aware low-dose CT denoising using conditional generative adversarial network. J. Digit. Imaging 31, 655–669 (2018).
Ronneberger, O., Fischer, P. & Brox, T. U-Net: convolutional networks for biomedical image segmentation. In Proceedings of Medical Image Computing and Computer Assisted Intervention (MICCAI) 234–241 (Springer, 2015).
Pascanu, R., Mikolov, T. & Bengio, Y. On the difficulty of training recurrent neural networks. In Proceedings of the International Conference on Machine Learning 1310–1318 (JMLR, 2013).
Tai, Y., Yang, J. & Liu, X. Image super-resolution via deep recursive residual network. In Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition (CVPR) 2790–2798 (IEEE, 2017).
Gulrajani, I., Ahmed, F., Arjovsky, M., Dumoulin, V. & Courville, A. C. Improved training of Wasserstein GANs. In Advances in Neural Information Processing Systems (NIPS), 5769–5779 (Curran Associates, 2017).
Goodfellow, I. et al. Generative adversarial nets. In Advances in Neural Information Processing Systems (NIPS) 2672–2680 (Curran Associates, 2014).
Arjovsky, M., Chintala, S. & Bottou, L. Wasserstein generative adversarial networks. In Proceedings of the International Conference on Machine Learning (ICML) 214–223 (JMLR, 2017).
Kingma, D. & Ba, J. Adam: a method for stochastic optimization. Preprint at https://arxiv.org/abs/1412.6980 (2014).
Abadi, M. et al. Tensorflow: large-scale machine learning on heterogeneous distributed systems. Preprint at https://arxiv.org/abs/1603.04467 (2016).
Conover, W. J. & Conover, W. J. Practical Nonparametric Statistics Ch. 3 (Wiley, 1980).
Acknowledgements
The authors thank M. Vannier and B. De Man for helpful discussions, and NVIDIA Corporation for the donation of GPUs used for this research. H.S. and G.W. were partially supported by the National Institutes of Health/National Institute of Biomedical Imaging and Bioengineering (U01EB017140).
Author information
Authors and Affiliations
Contributions
G.W. initiated the project. G.W., H.S. and M.K.K. designed the experiments. H.S. and G.W. performed machine learning research. A.P. and F.H. collected CT data and conducted the reader studies. M.K.K., C.N. and R.D.K. were the readers. H.S., G.W., U.K. and M.K.K. analysed the data. H.S. and G.W. wrote the paper. M.K.K. and U.K. edited the paper.
Corresponding authors
Ethics declarations
Competing interests
G.W. and H.S. have received unrelated industrial research grants from General Electric and Hologic Inc. M.K.K. has received unrelated industrial research grants from Siemens Healthineers and Riverain Inc.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Notes 1–3, Supplementary Figs. 1–4, Supplementary Tables 1 and 2
Rights and permissions
About this article
Cite this article
Shan, H., Padole, A., Homayounieh, F. et al. Competitive performance of a modularized deep neural network compared to commercial algorithms for low-dose CT image reconstruction. Nat Mach Intell 1, 269–276 (2019). https://doi.org/10.1038/s42256-019-0057-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42256-019-0057-9
This article is cited by
-
Generative deep learning furthers the understanding of local distributions of fat and muscle on body shape and health using 3D surface scans
Communications Medicine (2024)
-
SwinCT: feature enhancement based low-dose CT images denoising with swin transformer
Multimedia Systems (2024)
-
Subjective and objective image quality of low-dose CT images processed using a self-supervised denoising algorithm
Radiological Physics and Technology (2024)
-
A multi-attention Uformer for low-dose CT image denoising
Signal, Image and Video Processing (2024)
-
Design and manufacture of an X-ray generator by support vector machines
Evolutionary Intelligence (2024)