Abstract
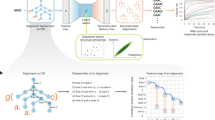
Generative adversarial networks (GANs) represent an attractive and novel approach to generate realistic data, such as genes, proteins or drugs, in synthetic biology. Here, we apply GANs to generate synthetic DNA sequences encoding for proteins of variable length. We propose a novel feedback-loop architecture, feedback GAN (FBGAN), to optimize the synthetic gene sequences for desired properties using an external function analyser. The proposed architecture also has the advantage that the analyser does not need to be differentiable. We apply the feedback-loop mechanism to two examples: generating synthetic genes coding for antimicrobial peptides, and optimizing synthetic genes for the secondary structure of their resulting peptides. A suite of metrics, calculated in silico, demonstrates that the GAN-generated proteins have desirable biophysical properties. The FBGAN architecture can also be used to optimize GAN-generated data points for useful properties in domains beyond genomics.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Demo, instructions and code for FBGAN are available at https://github.com/av1659/fbgan. All of the data used in this paper are publicly available and can be accessed at the references cited22.
References
Benner, S. & Sismour, M. Synthetic biology. Nat. Rev. Genet. 6, 533–543 (2005).
Izadpanah, A. & Gallo, R. Antimicrobial peptides. J. Am. Acad. Dermatol. 52, 381–390 (2005).
Papagianni, M. Ribosomally synthesized peptides with antimicrobial properties: biosynthesis, structure, function, and applications. Biotechnol. Adv. 21, 465–499 (2003).
Rocklin, G. J. et al. Global analysis of protein folding using massively parallel design, synthesis, and testing. Science 357, 168–175 (2017).
Abrusán, G. & Marsh, J. Alpha helices are more robust to mutations than beta strands. PLoS Comput. Biol. 12, e1005242 (2016).
Segler, M., Kogej, T., Tyrchan, C. & Waller, M. Generating focused molecule libraries for drug discovery with recurrent neural networks. ACS Cent. Sci. 4, 120–131 (2018).
Gupta, A. et al. Generative recurrent networks for de novo drug design. Mol. Inform. 37, 1700111 (2018).
Muller, A. T., Hiss, J. A. & Schneider, G. Recurrent neural network model for constructive peptide design. J. Chem. Inf. Model. 58, 472–479 (2018).
Olivecrona, M., Blaschke, T., Engkvist, O. & Chen, H. Molecular de novo design through deep reinforcement learning. J. Chemoinformatics 9, 48 (2017).
Salimans, T. et al. Improved techniques for training GANs. Preprint at abs/1606.03498 (2016).
Goldsborough, P., Pawlowski, N., Caicedo, J., Singh, S. & Carpenter, A. Cytogan: generative modeling of cell images. Preprint at https://www.biorxiv.org/content/10.1101/227645v1 (2017).
Esteban, C., Hyland, S. & Rätsch, G. Real-valued (medical) time series generation with recurrent conditional GANs. Preprint at https://arxiv.org/abs/1706.02633 (2017).
Ghahramani, A., Watt, F. & Luscombe, N. Generative adversarial networks uncover epidermal regulators and predict single cell perturbations. Preprint at https://www.biorxiv.org/content/10.1101/262501v2 (2018).
Osokin, A., Chessel, A., Carazo-Salas, R. & Vaggi, F. GANs for biological image synthesis. Preprint at http://arxiv.org/abs/1708.04692 (2017).
Zhu, J. & Bento, J. Generative adversarial active learning. Preprint at https://arxiv.org/abs/1702.07956 (2017).
Killoran, N., Lee, L., Delong, A., Duvenaud, D. & Frey, B. Generating and designing DNA with deep generative models. Preprint at https://arxiv.org/abs/1712.06148 (2017).
Arjovsky, M., Chintala, S. & Bottou, L. Wasserstein generative adversarial networks. In Proc. 34th International Conference on Machine Learning (eds Precup, D. & Teh, Y. W.) 214–223 (PMLR, 2017).
Goodfellow, I. et al. Generative adversarial nets. In Advances in Neural Information Processing Systems 27 (eds Ghahramani, Z. et al.) 2672–2680 (Curran Associates, 2014).
Gulrajani, I., Ahmed, F., Arjovsky, M., Dumoulin, V. & Courville, A. Improved training of Wasserstein GANs. Preprint at https://arxiv.org/abs/1704.00028 (2017).
Apweiler, R. et al. Uniprot: the universal protein knowledgebase. Nucleic Acids Res. 46, 2699 (2018).
Wang, G., Li, X. & Wang, Z. Apd3: the antimicrobial peptide database as a tool for research and education. Nucleic Acids Res. 44, D1087–D1093 (2016).
Muller, A., Gabernet, G., Hiss, J. & Schneider, G. modlamp: python for antimicrobial peptides. Bioinformatics 33, 2753–2755 (2017).
Buchan, D., Minneci, F., Nugent, T., Bryson, K. & Jones, D. Scalable web services for the PSIPRED protein analysis workbench. Nucleic Acids Res. 41, W349–W357 (2013).
Waghu, F., Barai, R., Gurung, P. & Idicula-Thomas, S. Campr3: a database on sequences, structures and signatures of antimicrobial peptides. Nucleic Acids Res. 44, D1094–D1097 (2016).
Xu, D. & Zhang, Y. Ab initio protein structure assembly using continuous structure fragments and optimized knowledge based force field. Proteins 80, 1715–1735 (2012).
Gupta, A. & Rush, A. Dilated convolutions for modeling long-distance genomic dependencies. Preprint at https://arxiv.org/abs/1710.01278 (2017).
Acknowledgements
The authors thank A. Kundaje for guidance when initiating the research on GANs and DNA. J.Z. is supported by a Chan-Zuckerberg Biohub Investigator grant and National Science Foundation (NSF) grant CRII 1657155.
Author information
Authors and Affiliations
Contributions
J.Z. conceived the objective of using GANs to generate genes and optimize protein functions; A.G. conceived of and implemented the feedback-loop architecture and conducted the experiments and analysis. Both authors wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures
Rights and permissions
About this article
Cite this article
Gupta, A., Zou, J. Feedback GAN for DNA optimizes protein functions. Nat Mach Intell 1, 105–111 (2019). https://doi.org/10.1038/s42256-019-0017-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42256-019-0017-4
This article is cited by
-
Discovering highly potent antimicrobial peptides with deep generative model HydrAMP
Nature Communications (2023)
-
Biological research and self-driving labs in deep space supported by artificial intelligence
Nature Machine Intelligence (2023)
-
DeepPricing: pricing convertible bonds based on financial time-series generative adversarial networks
Financial Innovation (2022)
-
The genetic and biochemical determinants of mRNA degradation rates in mammals
Genome Biology (2022)
-
Designing optimized drug candidates with Generative Adversarial Network
Journal of Cheminformatics (2022)