Abstract
Increased aerobic exercise capacity, as a result of exercise training, has important health benefits. However, some individuals are resistant to improvements in exercise capacity, probably due to undetermined genetic and environmental factors. Here, we show that exercise-induced improvements in aerobic capacity are blunted and aerobic remodelling of skeletal muscle is impaired in several animal models associated with chronic hyperglycaemia. Our data point to chronic hyperglycaemia as a potential negative regulator of aerobic adaptation, in part, via glucose-mediated modifications of the extracellular matrix, impaired vascularization and aberrant mechanical signalling in muscle. We also observe low exercise capacity and enhanced c-Jun N-terminal kinase activation in response to exercise in humans with impaired glucose tolerance. Our work indicates that current shifts in dietary and metabolic health, associated with increasing incidence of hyperglycaemia, might impair muscular and organismal adaptations to exercise training, including aerobic capacity as one of its key health outcomes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
Source data for western blots are available online. All other data that support the findings of this study are available from the corresponding author upon reasonable request. Source data are provided with this paper.
References
Brown, L. A. et al. Late life maintenance and enhancement of functional exercise capacity in low and high responding rats after low intensity treadmill training. Exp. Gerontol. 125, 110657 (2019).
Grundy, S. M., Barlow, C. E., Farrell, S. W., Vega, G. L. & Haskell, W. L. Cardiorespiratory fitness and metabolic risk. Am. J. Cardiol. 109, 988–993 (2012).
Koch, L. G. et al. Intrinsic aerobic capacity sets a divide for aging and longevity. Circ. Res. 109, 1162–1172 (2011).
Kokkinos, P. et al. Cardiorespiratory fitness and the paradoxical BMI-mortality risk association in male veterans. Mayo Clin. Proc. 89, 754–762 (2014).
Blair, S. N. et al. Physical fitness and all-cause mortality. a prospective study of healthy men and women. JAMA 262, 2395–2401 (1989).
Ekelund, L. G. et al. Physical fitness as a predictor of cardiovascular mortality in asymptomatic North American men. The Lipid Research Clinics mortality follow-up study. N. Engl. J. Med. 319, 1379–1384 (1988).
Barry, V. W. et al. Fitness vs. fatness on all-cause mortality: a meta-analysis. Prog. Cardiovasc. Dis. 56, 382–390 (2014).
Church, T. S. et al. Exercise capacity and body composition as predictors of mortality among men with diabetes. Diabetes Care 27, 83–88 (2004).
Bouchard, C. et al. Familial aggregation of VO(2max) response to exercise training: results from the HERITAGE Family Study. J. Appl. Physiol. 87, 1003–1008 (1999).
Bouchard, C. & Rankinen, T. Individual differences in response to regular physical activity. Med. Sci. Sports Exerc. 33, S446–S451 (2001).
Koch, L. G., Pollott, G. E. & Britton, S. L. Selectively bred rat model system for low and high response to exercise training. Physiol. Genomics 45, 606–614 (2013).
Timmons, J. A. et al. Using molecular classification to predict gains in maximal aerobic capacity following endurance exercise training in humans. J. Appl. Physiol. 108, 1487–1496 (2010).
Bonafiglia, J. T. et al. Inter-individual variability in the adaptive responses to endurance and sprint interval training: a randomized crossover study. PLoS ONE 11, e0167790 (2016).
Scharhag-Rosenberger, F., Walitzek, S., Kindermann, W. & Meyer, T. Differences in adaptations to 1 year of aerobic endurance training: individual patterns of nonresponse. Scand. J. Med. Sci. Sports 22, 113–118 (2012).
Montero, D. & Lundby, C. Refuting the myth of non-response to exercise training: ‘non-responders’ do respond to higher dose of training. J. Physiol. 595, 3377–3387 (2017).
Shephard, R. J., Rankinen, T. & Bouchard, C. Test-retest errors and the apparent heterogeneity of training response. Eur. J. Appl. Physiol. 91, 199–203 (2004).
Sparks, L. M. Exercise training response heterogeneity: physiological and molecular insights. Diabetologia 60, 2329–2336 (2017).
Lessard, S. J. et al. Resistance to aerobic exercise training causes metabolic dysfunction and reveals novel exercise-regulated signaling networks. Diabetes 62, 2717–2727 (2013).
Solomon, T. P., Malin, S. K., Karstoft, K., Haus, J. M. & Kirwan, J. P. The influence of hyperglycemia on the therapeutic effect of exercise on glycemic control in patients with type 2 diabetes mellitus. JAMA Intern. Med. 173, 1834–1836 (2013).
Nadeau, K. J. et al. Insulin resistance in adolescents with type 1 diabetes and its relationship to cardiovascular function. J. Clin. Endocrinol. Metab. 95, 513–521 (2010).
Nadeau, K. J. et al. Insulin resistance in adolescents with type 2 diabetes is associated with impaired exercise capacity. J. Clin. Endocrinol. Metab. 94, 3687–3695 (2009).
Solomon, T. P. et al. Association between cardiorespiratory fitness and the determinants of glycemic control across the entire glucose tolerance continuum. Diabetes Care 38, 921–929 (2015).
Hostalek, U. Global epidemiology of prediabetes—present and future perspectives. Clin. Diabetes Endocrinol. 5, 5 (2019).
Karve, A. & Hayward, R. A. Prevalence, diagnosis, and treatment of impaired fasting glucose and impaired glucose tolerance in nondiabetic U.S. adults. Diabetes Care 33, 2355–2359 (2010).
Hu, F. B. Globalization of diabetes: the role of diet, lifestyle, and genes. Diabetes Care 34, 1249–1257 (2011).
Hu, F. B. et al. Diet, lifestyle, and the risk of type 2 diabetes mellitus in women. N. Engl. J. Med. 345, 790–797 (2001).
Martinez Steele, E. et al. Ultra-processed foods and added sugars in the US diet: evidence from a nationally representative cross-sectional study. BMJ Open 6, e009892 (2016).
Roberts, C. K. & Liu, S. Effects of glycemic load on metabolic health and type 2 diabetes mellitus. J. Diabetes Sci. Technol. 3, 697–704 (2009).
Summers, L. K. et al. Substituting dietary saturated fat with polyunsaturated fat changes abdominal fat distribution and improves insulin sensitivity. Diabetologia 45, 369–377 (2002).
Uusitupa, M. et al. Effects of two high-fat diets with different fatty acid compositions on glucose and lipid metabolism in healthy young women. Am. J. Clin. Nutr. 59, 1310–1316 (1994).
Burchfield, J. G. et al. High dietary fat and sucrose results in an extensive and time-dependent deterioration in health of multiple physiological systems in mice. J. Biol. Chem. 293, 5731–5745 (2018).
Hirata, Y. et al. Hyperglycemia induces skeletal muscle atrophy via a WWP1/KLF15 axis. JCI Insight 4, e124952 (2019).
Kohsaka, A. et al. High-fat diet disrupts behavioral and molecular circadian rhythms in mice. Cell Metab. 6, 414–421 (2007).
Pendergast, J. S., Branecky, K. L., Huang, R., Niswender, K. D. & Yamazaki, S. Wheel-running activity modulates circadian organization and the daily rhythm of eating behavior. Front Psychol. 5, 177 (2014).
Jansson, E., Sjödin, B. & Tesch, P. Changes in muscle fibre type distribution in man after physical training: a sign of fibre type transformation? Acta Physiol. Scand. 104, 235–237 (1978).
Bergh, U. et al. Maximal oxygen uptake and muscle fiber types in trained and untrained humans. Med. Sci. Sport. 10, 151–154 (1978).
Saltin, B., Henriksson, J., Nygaard, E., Andersen, P. & Jansson, E. Fiber types and metabolic potentials of skeletal muscles in sedentary man and endurance runners. Ann. N. Y. Acad. Sci. 301, 3–29 (1977).
Kolset, S. O., Reinholt, F. P. & Jenssen, T. Diabetic nephropathy and extracellular matrix. J. Histochem. Cytochem. 60, 976–986 (2012).
Russo, I. & Frangogiannis, N. G. Diabetes-associated cardiac fibrosis: cellular effectors, molecular mechanisms and therapeutic opportunities. J. Mol. Cell Cardiol. 90, 84–93 (2016).
Kang, L. et al. Diet-induced muscle insulin resistance is associated with extracellular matrix remodeling and interaction with integrin α2β1 in mice. Diabetes 60, 416–426 (2011).
Edgar, L. T., Underwood, C. J., Guilkey, J. E., Hoying, J. B. & Weiss, J. A. Extracellular matrix density regulates the rate of neovessel growth and branching in sprouting angiogenesis. PLoS ONE 9, e85178 (2014).
Mongiat, M., Andreuzzi, E., Tarticchio, G. & Paulitti, A. Extracellular matrix, a hard player in angiogenesis. Int. J. Mol. Sci. 17, 1822 (2016).
Ahmed, N. & Thornalley, P. J. Advanced glycation endproducts: what is their relevance to diabetic complications? Diabetes Obes. Metab. 9, 233–245 (2007).
Snedeker, J. G. & Gautieri, A. The role of collagen crosslinks in ageing and diabetes—the good, the bad, and the ugly. Muscles Ligaments Tendons J. 4, 303–308 (2014).
Kent, M. C., Light, N. D. & Bailey, A. J. Evidence for glucose-mediated covalent cross-linking of collagen after glycosylation in vitro. Biochem. J. 225, 745–752 (1985).
Thornalley, P. J., Langborg, A. & Minhas, H. S. Formation of glyoxal, methylglyoxal and 3-deoxyglucosone in the glycation of proteins by glucose. Biochem. J. 344, 109 (1999).
Chen, Y. H. et al. High glucose impairs early and late endothelial progenitor cells by modifying nitric oxide-related but not oxidative stress-mediated mechanisms. Diabetes 56, 1559–1568 (2007).
Dubois, S. et al. Glucose inhibits angiogenesis of isolated human pancreatic islets. J. Mol. Endocrinol. 45, 99–105 (2010).
Laufs, U. et al. Physical training increases endothelial progenitor cells, inhibits neointima formation, and enhances angiogenesis. Circulation 109, 220–226 (2004).
Schlager, O. et al. Exercise training increases endothelial progenitor cells and decreases asymmetric dimethylarginine in peripheral arterial disease: a randomized controlled trial. Atherosclerosis 217, 240–248 (2011).
Pilegaard, H., Ordway, G. A., Saltin, B. & Neufer, P. D. Transcriptional regulation of gene expression in human skeletal muscle during recovery from exercise. Am. J. Physiol. Endocrinol. Metab. 279, E806–E814 (2000).
Egan, B. & Zierath, J. R. Exercise metabolism and the molecular regulation of skeletal muscle adaptation. Cell Metab. 17, 162–184 (2013).
Lessard, S. J. et al. JNK regulates muscle remodeling via myostatin/SMAD inhibition. Nat. Commun. 9, 3030 (2018).
Martin, T. D., Dennis, M. D., Gordon, B. S., Kimball, S. R. & Jefferson, L. S. mTORC1 and JNK coordinate phosphorylation of the p70S6K1 autoinhibitory domain in skeletal muscle following functional overloading. Am. J. Physiol. Endocrin. Metab. 306, E1397–E1405 (2014).
Wojtaszewski, J. F., Nielsen, P., Hansen, B. F., Richter, E. A. & Kiens, B. Isoform-specific and exercise intensity-dependent activation of 5′-AMP-activated protein kinase in human skeletal muscle. J. Physiol. 528, 221–226 (2000).
Aronson, D. et al. Exercise stimulates the mitogen-activated protein kinase pathway in human skeletal muscle. J. Clin. Invest. 99, 1251–1257 (1997).
Thomson, D. M. & Gordon, S. E. Impaired overload‐induced muscle growth is associated with diminished translational signalling in aged rat fast‐twitch skeletal muscle. J. Physiol. 574, 291–305 (2006).
Baar, K. & Esser, K. Phosphorylation of p70S6kcorrelates with increased skeletal muscle mass following resistance exercise. Am. J. Physiol. Cell Physiol. 276, C120–C127 (1999).
Park, S. W. et al. Decreased muscle strength and quality in older adults with type 2 diabetes: the health, aging, and body composition study. Diabetes 55, 1813–1818 (2006).
Ghachem, A., Brochu, M. & Dionne, I. J. Differential clusters of modifiable risk factors for impaired fasting glucose versus impaired glucose tolerance in adults 50 years of age and older. Ther. Adv. Chronic Dis. 10, 2040622319854239 (2019).
Messa, G. A. M. et al. The impact of a high-fat diet in mice is dependent on duration and age, and differs between muscles. J. Exp. Biol. 223, jeb217117 (2020).
Perry, B. D. et al. Muscle atrophy in patients with Type 2 diabetes mellitus: roles of inflammatory pathways, physical activity and exercise. Exercise Immunol. Rev. 22, 94 (2016).
National Diabetes Statistics Report, 2020 (Centers for Disease Control and Prevention, US Department of Health and Human Services, 2020).
Lillioja, S. et al. Skeletal muscle capillary density and fiber type are possible determinants of in vivo insulin resistance in man. J. Clin. Invest. 80, 415–424 (1987).
Walton, R. G. et al. Insulin‐resistant subjects have normal angiogenic response to aerobic exercise training in skeletal muscle, but not in adipose tissue. Physiol. Rep. 3, e12415 (2015).
Hickey, M. S. et al. Skeletal muscle fiber composition is related to adiposity and in vitro glucose transport rate in humans. Am. J. Physiol. 268, E453–E457 (1995).
Stuart, C. A. et al. Slow-twitch fiber proportion in skeletal muscle correlates with insulin responsiveness. J. Clin. Endocrinol. Metab. 98, 2027–2036 (2013).
Timmons, J. A. et al. Modulation of extracellular matrix genes reflects the magnitude of physiological adaptation to aerobic exercise training in humans. BMC Biol. 3, 19 (2005).
Pillon, N. J. et al. Transcriptomic profiling of skeletal muscle adaptations to exercise and inactivity. Nat. Commun. 11, 470 (2020).
Martineau, L. C. & Gardiner, P. F. Insight into skeletal muscle mechanotransduction: MAPK activation is quantitatively related to tension. J. Appl. Physiol. 91, 693–702 (2001).
Wang, N., Tytell, J. D. & Ingber, D. E. Mechanotransduction at a distance: mechanically coupling the extracellular matrix with the nucleus. Nat. Rev. Mol. Cell Biol. 10, 75 (2009).
Pandey, A. et al. Metabolic effects of exercise training among fitness-nonresponsive patients with type 2 diabetes: the HART-D study. Diabetes Care 38, 1494–1501 (2015).
Berria, R. et al. Increased collagen content in insulin-resistant skeletal muscle. Am J. Physiol. Endocrinol. Metab. 290, E560–E565 (2006).
Williams, A. S., Kang, L. & Wasserman, D. H. The extracellular matrix and insulin resistance. Trends Endocrinol. Metab. 26, 357–366 (2015).
Bonafiglia, J. T., Brennan, A. M., Ross, R. & Gurd, B. J. An appraisal of the SDIR as an estimate of true individual differences in training responsiveness in parallel-arm exercise randomized controlled trials. Physiol. Rep. 7, e14163 (2019).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(ΔΔC(T)) method. Methods 25, 402–408 (2001).
Acknowledgements
Research reported in this publication was supported by a Pilot and Feasibility award granted to S.J.L., and Diabetes Research Center core facilities funded by the NIH (NIDDK) award number P30DK036836. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. This work was also supported by an American Heart Association grant to S.J.L. (award number 15SDG25560057) and the Boston Nutrition and Obesity Research Center (BNORC) Pilot Program (P30DK046200, subaward no. 7513). T.L.M. was supported by a postdoctoral fellowship from the American Heart Association (no. 19POST34381036). P.Pattamaprapanont was supported by a Mary K. Iacocca Senior Visiting Fellowship. E.C.F. was supported by the São Paulo Research Foundation (grant no. FAPESP 2017/21676-3). Core facilities used for histological analysis were supported in part by NCI Cancer Center Support grant no. NIH 5 P30 CA06516 and NINDS P30 Core Center grant no. NS072030. The LRT-HRT rat model is funded by the Office of Infrastructure Programs grant no. P40ODO21331 (to L.G.K. and S.L.B.) from the NIH. Rat models for low and high response to exercise training are maintained as an international resource with support from the Department of Physiology and Pharmacology, The University of Toledo College of Medicine, Toledo, OH. Contact L.G.K. (lauren.koch2@utoledo.edu) or S.L.B. (brittons@umich.edu) for information on the rat models. For human studies, we acknowledge support by the Joslin Clinical Research Center and thank its philanthropic donors.
Author information
Authors and Affiliations
Contributions
Conceptualization of the idea came from T.L.M. and S.J.L. The methodology was devised by T.L.M., P. Pattamaprapanont, P. Pathak, S.H., J.M., S.L.B., L.G.K. and S.J.L. Validation was conducted by T.L.M., P. Pattamaprapanont, P. Pathak, N.F., E.C.F. and S.J.L. Formal analysis was conducted by T.L.M., P. Pattamaprapanont, P. Pathak, N.F., E.C.F. and S.J.L. Investigation was done by T.L.M., P. Pattamaprapanont, P. Pathak, N.F., E.C.F., S.H., J.M., S.L.B., L.G.K. and S.J.L. Resources were provided by S.L.B., L.G.K. and S.J.L. The original draft was written by T.L.M. and S.J.L. Reviews and editing were done by T.L.M., P. Pattamaprapanont, P. Pathak, N.F., E.C.F., S.H., J.M., S.L.B., L.G.K. and S.J.L. The visualization was done by T.L.M. and S.J.L. Supervision was done by T.L.M. and S.J.L. Funding acquisition was done by T.L.M., P. Pattamaprapanont, E.C.F. and S.J.L.
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflicts of interest.
Additional information
Peer review information Primary Handling Editor: Christoph Schmitt.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Extended Data Figure 1: 24 h wheel running behaviour.
A cohort of mice in control (CON, n=4), Western Diet-fed (WD, n=4) and Streptozotocin (STZ, n=4) groups undergoing exercise training were placed in cages to assess wheel running patterns. Wheel revolutions were counted per 1 h interval and recorded. a, An average trace over 24 h is shown; and mean for time spent running in the dark (%) is shown in Table b. Data are shown as mean ± SEM. Group differences in time spent dark running were compared by one-way ANOVA.
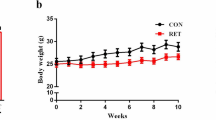
Extended Data Fig. 2 Extended Data Figure 2: Baseline exercise capacity.
Three separate cohorts of CD-1 mice were fed with Western Diet (WD), injected with streptozotocin (STZ) or maintained on a control diet (CON). After 8 weeks of treatment, all mice underwent aerobic exercise capacity testing in a and were found to have similar exercise capacities prior to being allocated to treatment groups for the training intervention (CON n=34, WD n=32, STZ n=42). Mice from each treatment group (CON, WD, STZ) were then allocated to remain sedentary or undergo voluntary wheel running (exercise-training) for a further 8 weeks. Baseline (pretraining) exercise capacity, shown in b, was similar among all six treatment groups (CON SED n=17, WD SED n=16, STZ SED n=22; CON EXT n=17, WD EXT n=16, STZ EXT n=20). Main effects were determined by one-way ANOVA relative to CON group in a and by two-way ANOVA in b. Data is represented as a point for the result of each individual animal, or mean ± SEM.
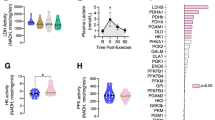
Extended Data Fig. 3 Extended Data Figure 3: JNK activation with treadmill running during acute and chronic hyperglycaemia.
a, To determine whether JNK activation with exercise is due to hyperglycemia, NOD mice completed an acute exercise bout (AEX n=18; 30 min treadmill running) or remained sedentary (SED n=6) and JNK signaling was measured in gastrocnemius muscle to determine correlation with random blood glucose. Representative blots (n= 1 SED; n= 2 AEX) shown here correspond with data shown in Figure 6g. To determine whether JNK activation with exercise is affected by acute increases in blood glucose, CD-1 mice were maintained on a control chow diet and fasted for 2 h prior to being split into four groups: a) control sedentary (n=5), b) acute glucose sedentary (n=5), c) control 30 min exercise (n=4) and d) acute glucose 30 min exercise (n=5). Mice allocated to glucose treatment were injected with 3 g/kg glucose to rapidly increase circulating glucose levels. b, JNK is activated with acute exercise (AEX) in gastrocnemius muscle, but acute glucose elevation does not alter JNK signaling with moderate treadmill running. c, Relative increases in blood glucose in control animals versus mice injected with 3 g/kg glucose. d, Mice were maintained on control diet (CON) or Western Diet (WD) for 8 (CON n=4; WD n=5), 16 (CON n=10; WD n=7), or 32 weeks (CON n=9, WD n=10) and an acute running experiment was performed at each time point. JNK activation with moderate treadmill running (30 min) was significantly higher in WD mice vs. CON mice by 16 weeks and worsened by 32 weeks; (right) representative blots showing n= 2/group per time point. Data is represented as mean ± SEM in all panels. Main effects were determined by two-way ANOVA in a and c. In d, differences in JNK activation between groups and over time were determined by two-way repeated measured ANOVA and indicated with “P”; differences between CON and WD groups at each 16 and 32 wk timepoint are indicated with “p.”.
Supplementary information
Source data
Source Data Fig. 2
Unprocessed western blots
Source Data Fig. 6
Unprocessed western blots
Source Data Fig. 7
Unprocessed western blots
Source Data Extended Data Fig. 3
Unprocessed western blots
Rights and permissions
About this article
Cite this article
MacDonald, T.L., Pattamaprapanont, P., Pathak, P. et al. Hyperglycaemia is associated with impaired muscle signalling and aerobic adaptation to exercise. Nat Metab 2, 902–917 (2020). https://doi.org/10.1038/s42255-020-0240-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-020-0240-7
This article is cited by
-
Continuous Glucose Monitoring in Endurance Athletes: Interpretation and Relevance of Measurements for Improving Performance and Health
Sports Medicine (2024)
-
Geroprotector drugs and exercise: friends or foes on healthy longevity?
BMC Biology (2023)
-
Effects of short-term endurance and strength exercise in the molecular regulation of skeletal muscle in hyperinsulinemic and hyperglycemic Slc2a4+/− mice
Cellular and Molecular Life Sciences (2023)
-
Sugar not so sweet for training-enhanced fitness
Nature Metabolism (2020)
-
Aerobics — decades of data for future hypothesis-testing research
Nature Reviews Endocrinology (2020)