Abstract
Non-alcoholic steatohepatitis (NASH) is characterized by lipotoxicity, inflammation and fibrosis, ultimately leading to end-stage liver disease. The molecular mechanisms promoting NASH are poorly understood, and treatment options are limited. Here, we demonstrate that hepatic expression of bone morphogenetic protein 8B (BMP8B), a member of the transforming growth factor beta (TGFβ)–BMP superfamily, increases proportionally to disease stage in people and animal models with NASH. BMP8B signals via both SMAD2/3 and SMAD1/5/9 branches of the TGFβ–BMP pathway in hepatic stellate cells (HSCs), promoting their proinflammatory phenotype. In vivo, the absence of BMP8B prevents HSC activation, reduces inflammation and affects the wound-healing responses, thereby limiting NASH progression. Evidence is featured in primary human 3D microtissues modelling NASH, when challenged with recombinant BMP8. Our data show that BMP8B is a major contributor to NASH progression. Owing to the near absence of BMP8B in healthy livers, inhibition of BMP8B may represent a promising new therapeutic avenue for NASH treatment.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding authors upon request. In vivo NGS data in this manuscript have been deposited in GEO, accession number GSE110404.
References
Younossi, Z. et al. Global burden of NAFLD and NASH: trends, predictions, risk factors and prevention. Nat. Rev. Gastroenterol. Hepatol. 15, 11–20 (2018).
Azzu, V., Vacca, M., Virtue, S., Allison, M. & Vidal-Puig, A. Adipose tissue-liver cross talk in the control of whole-body metabolism: implications in non-alcoholic fatty liver disease. Gastroenterology 158, 1899–1912 (2020).
Vacca, M., Allison, M., Griffin, J. L. & Vidal-Puig, A. Fatty acid and glucose sensors in hepatic lipid metabolism: implications in NAFLD. Semin. Liver Dis. 35, 250–261 (2015).
Bedossa, P. et al. Histopathological algorithm and scoring system for evaluation of liver lesions in morbidly obese patients. Hepatology 56, 1751–1759 (2012).
Kleiner, D. E. et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 41, 1313–1321 (2005).
Andriopoulos, B. Jr. et al. BMP6 is a key endogenous regulator of hepcidin expression and iron metabolism. Nat. Genet. 41, 482–487 (2009).
Wang, W., Yang, Y., Meng, Y. & Shi, Y. GDF-3 is an adipogenic cytokine under high fat dietary condition. Biochem. Biophys. Res. Commun. 321, 1024–1031 (2004).
Cunha, S. I., Magnusson, P. U., Dejana, E. & Lampugnani, M. G. Deregulated TGF-beta/BMP signaling in vascular malformations. Circ. Res. 121, 981–999 (2017).
David, L., Feige, J. J. & Bailly, S. Emerging role of bone morphogenetic proteins in angiogenesis. Cytokine Growth Factor Rev. 20, 203–212 (2009).
Bonnardel, J., et al. Stellate cells, hepatocytes, and endothelial cells imprint the Kupffer cell identity on monocytes colonizing the liver macrophage niche. Immunity 51, 638–654 (2019).
Herrera, B., Addante, A. & Sanchez, A. BMP signalling at the crossroad of liver fibrosis and regeneration. Int. J. Mol. Sci. 19, E39 (2017).
Hernanda, P. Y. et al. SMAD4 exerts a tumor-promoting role in hepatocellular carcinoma. Oncogene 34, 5055–5068 (2015).
Zheng, Y. et al. Bone morphogenetic protein 2 inhibits hepatocellular carcinoma growth and migration through downregulation of the PI3K/AKT pathway. Tumour Biol. 35, 5189–5198 (2014).
Zhang, Y., Alexander, P. B. & Wang, X. F. TGF-beta family signaling in the control of cell proliferation and survival. Cold Spring Harb. Perspect. Biol. 9, a022145 (2017).
Kreidl, E., Ozturk, D., Metzner, T., Berger, W. & Grusch, M. Activins and follistatins: emerging roles in liver physiology and cancer. World J. Hepatol. 1, 17–27 (2009).
Martins, L. et al. A functional link between AMPK and orexin mediates the effect of BMP8B on energy balance. Cell Rep. 16, 2231–2242 (2016).
Whittle, A. J. et al. BMP8B increases brown adipose tissue thermogenesis through both central and peripheral actions. Cell 149, 871–885 (2012).
Wu, F. J. et al. BMP8A sustains spermatogenesis by activating both SMAD1/5/8 and SMAD2/3 in spermatogonia. Sci. Signal 10, eaal1910 (2017).
Cheng, Z. et al. BMP8B mediates the survival of pancreatic cancer cells and regulates the progression of pancreatic cancer. Oncol. Rep. 32, 1861–1866 (2014).
Mima, K. et al. Gene expression of bone morphogenic protein 8B in the primary site, peripheral blood and bone marrow of patients with gastric cancer. Oncol. Lett. 6, 387–392 (2013).
Saito, T. et al. Genetic variations in humans associated with differences in the course of hepatitis C. Biochem. Biophys. Res. Commun. 317, 335–341 (2004).
Tryndyak, V. et al. Interstrain differences in the severity of liver injury induced by a choline- and folate-deficient diet in mice are associated with dysregulation of genes involved in lipid metabolism. FASEB J, 26, 4592–4602 (2012).
Stoyanov, E. et al. Chronic liver inflammation modifies DNA methylation at the precancerous stage of murine hepatocarcinogenesis. Oncotarget 6, 11047–11060 (2015).
Cubero, F. J. et al. TNFR1 determines progression of chronic liver injury in the IKKγ/Nemo genetic model. Cell Death Differ. 20, 1580–1592 (2013).
Kostrzewski, T. et al. Three-dimensional perfused human in vitro model of non-alcoholic fatty liver disease. World J. Gastroenterol. 23, 204–215 (2017).
Grefhorst, A. et al. Estrogens increase expression of bone morphogenetic protein 8b in brown adipose tissue of mice. Biol. Sex Differ. 6, 7 (2015).
Nakamura, T., Yoshimoto, K., Nakayama, Y., Tomita, Y. & Ichihara, A. Reciprocal modulation of growth and differentiated functions of mature rat hepatocytes in primary culture by cell–cell contact and cell membranes. Proc. Natl Acad. Sci. USA 80, 7229–7233 (1983).
Breher-Esch, S., Sahini, N., Trincone, A., Wallstab, C. & Borlak, J. Genomics of lipid-laden human hepatocyte cultures enables drug target screening for the treatment of non-alcoholic fatty liver disease. BMC Med. Genomics 11, 111 (2018).
Pellegrinelli, V. et al. Adipocyte-secreted BMP8b mediates adrenergic-induced remodeling of the neuro-vascular network in adipose tissue. Nat Commun 9, 4974 (2018).
Pellicoro, A., Ramachandran, P., Iredale, J. P. & Fallowfield, J. A. Liver fibrosis and repair: immune regulation of wound healing in a solid organ. Nat. Rev. Immunol. 14, 181–194 (2014).
Fausto, N., Campbell, J. S. & Riehle, K. J. Liver regeneration. Hepatology 43, S45–S53 (2006).
Kan, N. G., Junghans, D. & Izpisua Belmonte, J. C. Compensatory growth mechanisms regulated by BMP and FGF signaling mediate liver regeneration in zebrafish after partial hepatectomy. FASEB J. 23, 3516–3525 (2009).
Kostrzewski, T. et al. A microphysiological system for studying nonalcoholic steatohepatitis. Hepatol. Commun. 4, 77–91 (2020).
Schuster, S., Cabrera, D., Arrese, M. & Feldstein, A. E. Triggering and resolution of inflammation in NASH. Nat. Rev. Gastroenterol. Hepatol. 15, 349–364 (2018).
Hall, Z. et al. Lipid zonation and phospholipid remodeling in nonalcoholic fatty liver disease. Hepatology 65, 1165–1180 (2017).
Boyle, M. et al. Performance of the PRO-C3 collagen neo-epitope biomarker in non-alcoholic fatty liver disease. JHEP Rep. 1, 188–198 (2019).
Matsumoto, M. et al. An improved mouse model that rapidly develops fibrosis in non-alcoholic steatohepatitis. Int. J. Exp. Pathol. 94, 93–103 (2013).
Baeck, C. et al. Pharmacological inhibition of the chemokine CCL2 (MCP-1) diminishes liver macrophage infiltration and steatohepatitis in chronic hepatic injury. Gut 61, 416–426 (2012).
Krenkel, O. et al. Therapeutic inhibition of inflammatory monocyte recruitment reduces steatohepatitis and liver fibrosis. Hepatology 67, 1270–1283 (2018).
Bosselut, N. et al. Including osteoprotegerin and collagen IV in a score-based blood test for liver fibrosis increases diagnostic accuracy. Clin. Chim. Acta 415, 63–68 (2013).
Yilmaz, Y. et al. Serum levels of osteoprotegerin in the spectrum of nonalcoholic fatty liver disease. Scand. J. Clin. Lab. Invest. 70, 541–546 (2010).
Mann, J. & Mann, D. A. Transcriptional regulation of hepatic stellate cells. Adv. Drug Deliv. Rev. 61, 497–512 (2009).
Faillaci, F. et al. Liver angiopoietin-2 is a key predictor of de novo or recurrent hepatocellular cancer after hepatitis C virus direct-acting antivirals. Hepatology 68, 1010–1024 (2018).
Scheving, L. A., Zhang, X., Stevenson, M. C., Threadgill, D. W. & Russell, W. E. Loss of hepatocyte EGFR has no effect alone but exacerbates carbon tetrachloride-induced liver injury and impairs regeneration in hepatocyte Met-deficient mice. Am. J. Physiol. Gastrointest. Liver Physiol. 308, G364–G377 (2015).
Franklin, M. P., Sathyanarayan, A. & Mashek, D. G. Acyl-CoA thioesterase 1 (ACOT1) regulates PPARα to couple fatty acid flux with oxidative capacity during fasting. Diabetes 66, 2112–2123 (2017).
Thomas, M. et al. Peroxisome proliferator-activated receptor alpha, PPARα, directly regulates transcription of cytochrome P450 CYP2C8. Front Pharmacol. 6, 261 (2015).
Feng, L. et al. Identification and characterization of a novel PPARalpha-regulated and 7alpha-hydroxyl bile acid-preferring cytosolic sulfotransferase mL-STL (Sult2a8). J. Lipid Res. 58, 1114–1131 (2017).
Hardy, T., Anstee, Q. M. & Day, C. P. Nonalcoholic fatty liver disease: new treatments. Curr. Opin. Gastroenterol. 31, 175–183 (2015).
Brunt, E. M. et al. Nonalcoholic fatty liver disease. Nat. Rev. Dis. Primers 1, 15080 (2015).
Anstee, Q. M., Reeves, H. L., Kotsiliti, E., Govaere, O. & Heikenwalder, M. From NASH to HCC: current concepts and future challenges. Nat. Rev. Gastroenterol. Hepatol. 16, 411–428 (2019).
Zhao, G. Q., Deng, K., Labosky, P. A., Liaw, L. & Hogan, B. L. The gene encoding bone morphogenetic protein 8B is required for the initiation and maintenance of spermatogenesis in the mouse. Genes Dev. 10, 1657–1669 (1996).
FELASA working group on revision of guidelines for health monitoring of rodents and rabbits et al. FELASA recommendations for the health monitoring of mouse, rat, hamster, guinea pig and rabbit colonies in breeding and experimental units. Lab Anim. 48, 178–192 (2014)..
Virtue, S. et al. Peroxisome proliferator-activated receptor γ2 controls the rate of adipose tissue lipid storage and determines metabolic flexibility. Cell Rep. 21, 2005–2012.e7 (2018).
Elia, M. & Livesey, G. Energy expenditure and fuel selection in biological systems: the theory and practice of calculations based on indirect calorimetry and tracer methods. World Rev. Nutr. Diet 70, 68–131 (1992).
Nielsen, M. J. et al. The neo-epitope specific PRO-C3 ELISA measures true formation of type III collagen associated with liver and muscle parameters. Am. J. Transl. Res. 5, 303–315 (2013).
Barascuk, N. et al. A novel assay for extracellular matrix remodeling associated with liver fibrosis: An enzyme-linked immunosorbent assay (ELISA) for a MMP-9 proteolytically revealed neo-epitope of type III collagen. Clin. Biochem. 43, 899–904 (2010).
Girish, V. & Vijayalakshmi, A. Affordable image analysis using NIH Image/ImageJ 1.8.0. Indian J. Cancer 41, 47 (2004).
Wilson, C. L. et al. Ubiquitin C-terminal hydrolase 1: a novel functional marker for liver myofibroblasts and a therapeutic target in chronic liver disease. J. Hepatol. 63, 1421–1428 (2015).
Folch, J., Lees, M. & Stanley, G. H. S. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 226, 497–509 (1957).
Gibson-Corley, K. N., Olivier, A. K. & Meyerholz, D. K. Principles for valid histopathologic scoring in research. Vet. Pathol. 50, 1007–1015 (2013).
Anders, S., Pyl, P. T. & Huber, W. HTSeq–a Python framework to work with high-throughput sequencing data. Bioinformatics 31, 166–169 (2015).
Benjamini, Y., Drai, D., Elmer, G., Kafkafi, N. & Golani, I. Controlling the false discovery rate in behavior genetics research. Behav. Brain Res. 125, 279–284 (2001).
Frishberg, A., Brodt, A., Steuerman, Y. & Gat-Viks, I. ImmQuant: a user-friendly tool for inferring immune cell-type composition from gene-expression data. Bioinformatics 32, 3842–3843 (2016).
Acknowledgements
The authors are indebted to the following colleagues and institutions: MRC Metabolic Diseases Unit (MC_UU_00014/5): M. Dale, M. Campbell, R. Dias, the Disease Model Core (DMC; H. Webber, D. Hart, S. Grocott, C. Beresford, D. Jessop, E. Rasijeff and A. Warner), the Biochemistry Assay Lab (K. Burling and collaborators), the Genomics and Transcriptomics Core (M. Ma), the Histology Core (J. Warner) and the Imaging Core (G. Strachan). MRC Human Nutrition Research and Department of Biochemistry, University of Cambridge: F. Sanders, Z. Hall and J. West. Histopathology/ISH core facility of Cancer Research UK—Cambridge Institute: J. Jones for assistance with in situ hybridization. The human NASH histological samples come from the Human Research Tissue Bank of the Cambridge University Hospitals, which is supported by the NIHR Cambridge Biomedical Research Centre. M.V., J.L.G. and A.V.P. are supported by MRC programs (MRC MDU Programme Grant. PO 4050281695 ‘Lipotoxicity and the Metabolic Syndrome’ and MRC DMC MC UU 12012/2 to A.V.P.; Lipid Profiling and Signalling, MC UP A90 1006 to J.L.G.) and MRC adjunct funding as part of the Cambridge Initiative in Metabolic Diseases (Lipid Dynamics and Regulation: MC_PC_13030). M.V., M.A. and A.V.P. are also supported by the Cambridge NIHR Biomedical Research Center (Gastroenterology); M.V. is a recipient of the BRC Gastroenterology Pump-Priming award 2018/2019 that funded part of this study. F.O. is supported by MRC program Grants MR/K0019494/1 and MR/R023026/1. J.L. is supported by Medical Research Council PhD studentship and a CRUK program grant (C18342/A23390). Q.M.A., M.V., A.V.P., V.R., M.A. and D.T. are contributing members of the European NAFLD Registry. Q.M.A. is supported by the Newcastle NIHR Biomedical Research Centre (BRC). M.V. has been fellow of the Fondazione Umberto Veronesi in 2014. M.A., A.V.P. and J.L.G. received funding from the Evelyn Trust. M.V., O.G., D.T., M.A., F.O., Q.M.A., M.J.N., D.J.L. and A.V.P. are members of the EPoS (Elucidating Pathways of Steatohepatitis) consortium, which is funded by the Horizon 2020 Framework Program of the European Union under Grant Agreement 634413.
Author information
Authors and Affiliations
Contributions
M.V. and A.V.P. conceived and designed the study and wrote the manuscript. M.V., S.V. and V.P. designed and performed the WD experiment. J.L., F.O., M.V. and S.V. designed and performed the PHx and CCl4 experiments. M.V., S.S., T.K., Z.T. and K.P. performed the in vitro experiments. O.G., Q.M.A., V.R., M.E.D.A. and S.D. contributed with human data and samples. D.T. and S.D. scored the liver histology. Z.T. and W.L. contributed to the design of some of the in vitro experiments. M.J.N. and D.J.L. performed the collagen-3/MMP9 assays. Z.A. and J.L.G. performed liver lipidomics. B.Y.H.L. and M.V. analysed the NGS sequencing data. M.V. performed most of the analyses in all the models. All the authors provided useful criticism during the study, and critically reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
At the time of this study, T.K. and S.S. were employees of CN Bio Innovations; M.J.N. and D.J.L. of Nordic Bioscience and are among original inventors and patent holders for the PRO-C3, C3M and MMP9 assays. F.O. is a director of Fibrofind. J.L. and F.O. are shareholders in Fibrofind. The other authors declare no competing interests.
Additional information
Peer review information Primary Handling Editor: Christoph Schmitt.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
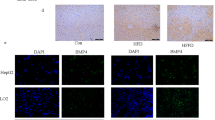
Extended Data Fig. 1 In liver disease, BMP8b is overexpressed in Albumin and αSma positive cells.
a, Relative mRNA expression levels of Bmp8b measured by quantitative real-time polymerase chain reaction (RTqPCR) in murine livers. The levels of BMP8b mRNA transcript increase in Western Diet (WD)-induced NASH [(CTRL) n: 8; (WD) n:8], but not in High Fat Diet (HFD)-induced NAFL [(CTRL) n:5; (HFD) n:6], and in acute [3 days; (CTRL) n: 6; (CCl4) n:4] (3 days; n:4-5/group) and chronic [8 weeks; (CTRL) n: 5; (CCl4) n:5] carbon tetrachloride (CCl4) -induced liver damage. All the results are shown as mean ± s.e.m.; expression data of biological replicates represented as dot plots; statistical significance (vs. control treatment) assessed by two-sided Student’s t-test. b-c, Representative IF of Bmp8b and Albumin (Alb) or αSma protein expression: in Western Diet (WD)-induced NASH and following acute (3 days) CCl4 challenge Bmp8b is expressed in the liver, and co-localizes with both αSma and Albumin thus suggesting that Bmp8b is expressed in hepatocytes and activated HSC following hepatic damage (2 replicates / condition; staining repeated twice).
Extended Data Fig. 2 Bmp8b is overexpressed by primary hepatocytes when cultured in vitro.
a, Relative mRNA gene expression levels of Bmp8b measured by quantitative real-time polymerase chain reaction (RTqPCR) in PH cultured at low (20,000 cells/cm2) or high (100,000 cells/cm2) confluence to model a highly proliferative (b; Cyclin E1, Ccne1) or differentiated (c; Albumin, Alb) behavior, respectively. Bmp8b mRNA expression levels are very low at baseline, and induced from 24h after culturing. All the results are shown as mean ± s.e.m. Statistical significance was assessed by Multivariate Analysis of Variance (MANOVA; 5 replicates/group). d, Microarray data from publicly available database GSE122660 of human primary hepatocytes (PH) and human hepatocytes cell lines (HepG2 and Huh7) cultured in 2D and challenged for 72h with DMSO or a mixture of oleic (OA) and palmitic (PA) fatty acids (with/without TNFα) suggest that also human hepatocytes express BMP8B and that the challenges with fatty acids and/or pro-inflammatory factors do not influence BMP8B expression. Expression data were retrieved using the tool Geo2R from NCBI, and statistical significance was assessed using One-Way Analysis of Variance (ANOVA; 3 replicates/group).
Extended Data Fig. 3 Bmp8b is overexpressed by Primary Hepatic Stellate Cells when cultured in vitro.
Relative mRNA gene expression levels measured by RNA sequencing (a-c) or quantitative real-time polymerase chain reaction (RTqPCR; d) in murine primary hepatic stellate cells cultured at a density of 35,000 cells/cm2 and harvested before (day 0) and after culture (days 1, 4, 8, 12). Average mRNA abundance (Log2CPM) of BMP/TGFβ receptors and effectors (a), and of TGFβ/BMP family members (b): Artn, Bmp 2/8a, and Inhb b/c/e were suppressed; Bmp3/5/7/9/10 and Gdf9/10 were suppressed after a transient upregulation at day 1 of culture; Tgfβ2/3, Gdf6 and Inhba were upregulated (Nodal and Gdf3 were not expressed by HSC). c, IPA “upstream regulator” analysis based on time-dependent GE changes (NGS) in HSC at different stages of the trans-activation program compared to Day 1 (D1) of culturing shows activation of multiple TGFβ-related effectors Detailed NGS analysis (significantly modulated genes, IPA analysis) available in Supplementary Table 5; statistical significance was assessed by GLM likelihood ratio (edgeR) and then adjusted by the Benjamini-Hochberg procedure to control the False Discovery Rate (FDR). “Upstream Regulators” shown are all significantly enriched (P<0.05 - and with a 2≤Z-Score≥2) in at least one comparison. d, Time-dependent changes of Bmp8b expression in HSC treated with drivers of HSC activation (PDGFB 10ng/mL; Oleic Acid 100 μM; Palmitic Acid 100 μM; TNFα 30ng/mL; LPS 50 ng/mL). Bmp8b mRNA expression levels are very low at baseline and induced 24h after culturing (Area Under the Curve, AUC, in the small panel); Palmitic Acid significantly induces Bmp8b, while LPS reduces its expression over the time. All the results are shown as mean ± s.e.m. or in a heatmap format (representing gene abundance expressed as Log2CPM, or the degree of “Upstream Regulator” activation -Z-score- at the IPA “Upstream Regulator” Analysis). Sample Size: 4 biological replicates/group for panels a-c (each biological replicate is a pool of 3 livers); 3 replicates/time point for panel (d). Statistical significance was assessed by one-way analysis of variance (ANOVA) plus Fisher’s least significant difference test (d). Lowercase letters indicate post hoc analysis significance: a, reference group; groups with different letters are statistically different per post hoc comparison; differences between groups with the same letter are statistically not significant per post hoc comparison.
Extended Data Fig. 4 Recombinant BMP8 rescues the defect in HSC activation observed in Bmp8b KO HSC cultured in vitro.
Freshly isolated HSC from Bmp8b KO mice and WT littermates were cultured for 4 and 6 days at a density of 35K cells/cm2 in presence/absence of recombinant BMP8 (30 mg/mL) to study the effect of BMP8b gain of function/recovery in both KO and WT cells. αSMA protein expression (a,b, IF; staining repeated twice in each biological replicate; magnification: 10X), and gene expression (RTqPCR) of multiple markers of HSC transactivation (c), inflammation (d) and TGFβ/BMP targets (e) were checked to study HSC’s activation status (4 biological replicates/group; each biological replicate is a pool of three livers). All the results are shown as mean ± s.e.m. Statistical significance was assessed by Multivariate Analysis of Variance (MANOVA).
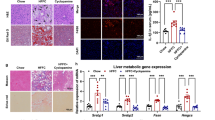
Extended Data Fig. 5 BMP8 stimulates TGFβ/BMP signaling in a human 3D in vitro NASH model promoting inflammation and proliferation (continues from Figure 6).
a, BMP8b mRNA expression (RTqPCR) in the cells cultured without/with medium containing a mixture of saturated and unsaturated FFAs (Lean vs. Fat; n: 6 replicates/group). b, Targeted phospho-proteomics data of cells studied 30 min after BMP8 challenge (vs. Control; 3 biological replicates/group) in cells cultured in “Fat” medium; c, TGFβ/BMP targets studied by BMP8b mRNA expression (RTqPCR) studied after 2 challenges (every 24h) of BMP8, TGFβ, or BMP7 (in “Fat” medium; cells and media were harvested 48h after commencing the challenges; n: 4 replicates/group). d, Secreted proteins quantified in the culturing media of 48h treated cells (n: 4 replicates/group); e, IPA “upstream regulator” analysis of the RNA sequencing of cells treated with recombinant BMP8 for 5h vs 48h (4 replicates/group; full list of genes differentially regulated and statistical design are provided in Supplementary Table 7). All the results are shown as mean ± s.e.m. (expression data of biological replicates are represented as dot plots), or in a heatmap format [representing the degree of “Upstream Regulator” activation (-2 ≤ Z-score ≥2) at the IPA “Upstream Regulator” Analysis]. To provide a framework of interpretation of the sequencing data, we clustered the results in “early response” regulators (modulated at 5h; not modulated at 48h), “persistent response” regulators (modulated both at 5h and 48h with the same direction), “late response” regulators (mildly/not regulated at 5h and modulated at 48h), “biphasic” regulators (showing opposite direction of regulation between 5h and 48h data). Statistical significance was assessed by two-sided Student T-Test (a,b), or by One-Way analysis of variance (ANOVA; c, d) plus Fisher’s least significant difference test (n: 4 replicates/group). Lowercase letters indicate post hoc analysis significance: a, reference group; groups with different letters are statistically different per post hoc comparison; differences between groups with the same letter are statistically not significant per post hoc comparison.
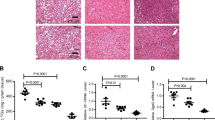
Extended Data Fig. 6 Bmp8b KO mice challenged with CDHFD model (NASH F2 fibrosis).
BMP8b KO and wild-type littermates mice were treated for 14 weeks with a choline deficient high-fat diet (CDHFD – N: 9WT & 6KO). CDHFD -treated Bmp8b KO mice show no difference in BW (a), Liver to body weight percent ratio (b: LW/BW%), glucose and lipid metabolism (c), ALT (d), NASH activity (e, H&E; f, NASH activity score) and Fibrosis (g, Picro-Sirius Red, PSR; h, “Kleiner” Fibrosis Stage; i, PSR quantification (% of stained area quantified using HALO imaging software, Indica Lab) on the whole-tissue scanned slide; j, Procollagen C3, PRO-C3). However, the relative mRNA expression levels of key genes measured by RTqPCR in the livers (k), and in freshly isolated HSC (l; Sample size: 6WT & 3 KO HSC pools) show impaired activation of TGFβ/BMP signaling, reduced inflammation, and defective HSC activation in Bmp8b KO mice compared to WT littermates. a-l, All the results are shown as mean ± s.e.m. (biological replicates are represented as dot plots). Statistical significance was assessed by two-sided Student T-Test.
Supplementary information
Supplementary Information
Supplementary Figures 1–8 and Supplementary Tables 1–4
Supplementary Data
Supplementary Tables 5–9
Rights and permissions
About this article
Cite this article
Vacca, M., Leslie, J., Virtue, S. et al. Bone morphogenetic protein 8B promotes the progression of non-alcoholic steatohepatitis. Nat Metab 2, 514–531 (2020). https://doi.org/10.1038/s42255-020-0214-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-020-0214-9
This article is cited by
-
Adipocyte YTH N(6)-methyladenosine RNA-binding protein 1 protects against obesity by promoting white adipose tissue beiging in male mice
Nature Communications (2023)
-
Circulating bone morphogenetic protein 8A is a novel biomarker to predict advanced liver fibrosis
Biomarker Research (2023)
-
BMP4 and Gremlin 1 regulate hepatic cell senescence during clinical progression of NAFLD/NASH
Nature Metabolism (2022)
-
Modelling human liver fibrosis in the context of non-alcoholic steatohepatitis using a microphysiological system
Communications Biology (2021)
-
Novel regulatory pathway in NASH identified
Nature Reviews Endocrinology (2020)