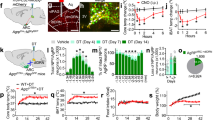
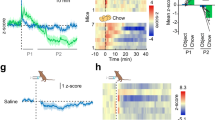
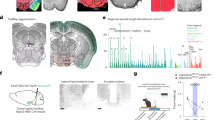
Abstract
Dopamine signalling is a crucial part of the brain reward system and can affect feeding behaviour. Dopamine receptors are also expressed in the hypothalamus, which is known to control energy metabolism in peripheral tissues. Here we show that pharmacological or chemogenetic stimulation of dopamine receptor 2 (D2R) expressing cells in the lateral hypothalamic area (LHA) and the zona incerta (ZI) decreases body weight and stimulates brown fat activity in rodents in a feeding-independent manner. LHA/ZI D2R stimulation requires an intact sympathetic nervous system and orexin system to exert its action and involves inhibition of PI3K in the LHA/ZI. We further demonstrate that, as early as 3 months after the onset of treatment, patients treated with the D2R agonist cabergoline experience an increase in energy expenditure that persists for one year, leading to total body weight and fat loss through a prolactin-independent mechanism. Our results may provide a mechanistic explanation for how clinically used D2R agonists act in the central nervous system to regulate energy balance.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon request.
References
Palmiter, R. D. Is dopamine a physiologically relevant mediator of feeding behavior? Trends Neurosci. 30, 375–381 (2007).
Mirmohammadsadeghi, Z., Shareghi Brojeni, M., Haghparast, A. & Eliassi, A. Role of paraventricular hypothalamic dopaminergic D1 receptors in food intake regulation of food-deprived rats. Eur. J. Pharmacol. 818, 43–49 (2018).
Zhu, X., Ottenheimer, D. & DiLeone, R. J. Activity of D1/2 receptor expressing neurons in the nucleus accumbens regulates running, locomotion, and food intake. Front. Behav. Neurosci. 10, 66 (2016).
Land, B. B. et al. Medial prefrontal D1 dopamine neurons control food intake. Nat. Neurosci. 17, 248–253 (2014).
Fetissov, S. O., Meguid, M. M., Sato, T. & Zhang, L. H. Expression of dopaminergic receptors in the hypothalamus of lean and obese zucker rats and food intake. Am. J. Physiol. Regulatory, Integr. Comp. Physiol. 283, R905–R910 (2002).
Johnson, P. M. & Kenny, P. J. Dopamine D2 receptors in addiction-like reward dysfunction and compulsive eating in obese rats. Nat. Neurosci. 13, 635–641 (2010).
Volkow, N. D., Wang, G. J. & Baler, R. D. Reward, dopamine and the control of food intake: implications for obesity. Trends Cogn. Sci. 15, 37–46 (2011).
Wang, G. J., Volkow, N. D. & Fowler, J. S. The role of dopamine in motivation for food in humans: implications for obesity. Expert Opin. Ther. Targets 6, 601–609 (2002).
Holt, R. I., Barnett, A. H. & Bailey, C. J. Bromocriptine: old drug, new formulation and new indication. Diabetes, Obes. Metab. 12, 1048–1057 (2010).
Cincotta, A. H. & Meier, A. H. Bromocriptine (Ergoset) reduces body weight and improves glucose tolerance in obese subjects. Diabetes Care 19, 667–670 (1996).
Pijl, H. et al. Bromocriptine: a novel approach to the treatment of type 2 diabetes. Diabetes Care 23, 1154–1161 (2000).
Gaziano, J. M. et al. Randomized clinical trial of quick-release bromocriptine among patients with type 2 diabetes on overall safety and cardiovascular outcomes. Diabetes Care 33, 1503–1508 (2010).
Wang, G. J. et al. Brain dopamine and obesity. Lancet 357, 354–357 (2001).
Bray, G. A., Fruhbeck, G., Ryan, D. H. & Wilding, J. P. Management of obesity. Lancet 387, 1947–1956 (2016).
Henderson, D. C., Vincenzi, B., Andrea, N. V., Ulloa, M. & Copeland, P. M. Pathophysiological mechanisms of increased cardiometabolic risk in people with schizophrenia and other severe mental illnesses. Lancet Psychiatry 2, 452–464 (2015).
Noble, E. P. et al. D2 dopamine receptor gene and obesity. Int. J. Eat. Disord. 15, 205–217 (1994).
Sun, X., Luquet, S. & Small, D. M. DRD2: Bridging the genome and ingestive behavior. Trends Cogn. Sci. 21, 372–384 (2017).
Meier, A. H., Cincotta, A. H. & Lovell, W. C. Timed bromocriptine administration reduces body fat stores in obese subjects and hyperglycemia in type II diabetics. Experientia 48, 248–253 (1992).
Gibson, C. D., Karmally, W., McMahon, D. J., Wardlaw, S. L. & Korner, J. Randomized pilot study of cabergoline, a dopamine receptor agonist: effects on body weight and glucose tolerance in obese adults. Diabetes, Obes. Metab. 14, 335–340 (2012).
Liu, X. et al. The mechanism and pathways of dopamine and dopamine agonists in prolactinomas. Front. Endocrinol. 9, 768 (2018).
Al-Massadi, O. et al. Pharmacological and genetic manipulation of p53 in brown fat at adult but not embryonic stages regulates thermogenesis and body weight in male mice. Endocrinology 157, 2735–2749 (2016).
Fruhbeck, G., Mendez-Gimenez, L., Fernandez-Formoso, J. A., Fernandez, S. & Rodriguez, A. Regulation of adipocyte lipolysis. Nutr. Res. Rev. 27, 63–93 (2014).
Hankir, M. K., Cowley, M. A. & Fenske, W. K. A BAT-centric approach to the treatment of diabetes: Turn on the brain. Cell Metab. 24, 31–40 (2016).
Folgueira, C. et al. Uroguanylin action in the brain reduces weight gain in obese mice via different efferent autonomic pathways. Diabetes 65, 421–432 (2016).
Martinez-Sanchez, N. et al. Hypothalamic AMPK-ER stress-JNK1 axis mediates the central actions of thyroid hormones on energy balance. Cell Metab. 26, 212–229 e212 (2017).
Weiner, D. M. et al. D1 and D2 dopamine receptor mRNA in rat brain. Proc. Natl Acad. Sci. USA 88, 1859–1863 (1991).
Gomez, J. L. et al. Chemogenetics revealed: DREADD occupancy and activation via converted clozapine. Science 357, 503–507 (2017).
Stagkourakis, S., Kim, H., Lyons, D. J. & Broberger, C. Dopamine autoreceptor regulation of a hypothalamic dopaminergic network. Cell Rep. 15, 735–747 (2016).
Puighermanal, E. et al. drd2-cre:ribotag mouse line unravels the possible diversity of dopamine d2 receptor-expressing cells of the dorsal mouse hippocampus. Hippocampus 25, 858–875 (2015).
Segal-Lieberman, G. et al. Melanin-concentrating hormone is a critical mediator of the leptin-deficient phenotype. Proc. Natl Acad. Sci. USA 100, 10085–10090 (2003).
Tupone, D., Madden, C. J., Cano, G. & Morrison, S. F. An orexinergic projection from perifornical hypothalamus to raphe pallidus increases rat brown adipose tissue thermogenesis. J. Neurosci. 31, 15944–15955 (2011).
Martins, L. et al. A functional link between AMPK and orexin mediates the effect of BMP8B on energy balance. Cell Rep. 16, 2231–2242 (2016).
Yamaguchi, T. et al. Role of PKA signaling in D2 receptor-expressing neurons in the core of the nucleus accumbens in aversive learning. Proc. Natl Acad. Sci. USA 112, 11383–11388 (2015).
Bonito-Oliva, A. et al. Haloperidol promotes mTORC1-dependent phosphorylation of ribosomal protein S6 via dopamine- and cAMP-regulated phosphoprotein of 32 kDa and inhibition of protein phosphatase-1. Neuropharmacology 72, 197–203 (2013).
Mighiu, P. I. et al. Hypothalamic glucagon signaling inhibits hepatic glucose production. Nat. Med. 19, 766–772 (2013).
Quinones, M. et al. Hypothalamic CaMKKbeta mediates glucagon anorectic effect and its diet-induced resistance. Mol. Metab. 4, 961–970 (2015).
Meacci, E. et al. Molecular cloning and expression of human myocardial cGMP-inhibited cAMP phosphodiesterase. Proc. Natl Acad. Sci. USA 89, 3721–3725 (1992).
Zhao, A. Z., Huan, J. N., Gupta, S., Pal, R. & Sahu, A. A phosphatidylinositol 3-kinase phosphodiesterase 3B-cyclic AMP pathway in hypothalamic action of leptin on feeding. Nat. Neurosci. 5, 727–728 (2002).
Sahu, M., Anamthathmakula, P. & Sahu, A. Hypothalamic phosphodiesterase 3B pathway mediates anorectic and body weight-reducing effects of insulin in male mice. Neuroendocrinology 104, 145–156 (2017).
Sahu, M., Anamthathmakula, P. & Sahu, A. Phosphodiesterase-3B-cAMP pathway of leptin signalling in the hypothalamus is impaired during the development of diet-induced obesity in FVB/N mice. J. Neuroendocrinol. 27, 293–302 (2015).
Moore, C. E., Xie, J., Gomez, E. & Herbert, T. P. Identification of cAMP-dependent kinase as a third in vivo ribosomal protein S6 kinase in pancreatic beta-cells. J. Mol. Biol. 389, 480–494 (2009).
Valjent, E. et al. Haloperidol regulates the state of phosphorylation of ribosomal protein S6 via activation of PKA and phosphorylation of DARPP-32. Neuropsychopharmacol. 36, 2561–2570 (2011).
Blouet, C., Ono, H. & Schwartz, G. J. Mediobasal hypothalamic p70 S6 kinase 1 modulates the control of energy homeostasis. Cell Metab. 8, 459–467 (2008).
Richard, D. Cognitive and autonomic determinants of energy homeostasis in obesity. Nat. Rev. Endocrinol. 11, 489–501 (2015).
Meguid, M. M., Yang, Z. J. & Montante, A. Lateral hypothalamic dopaminergic neural activity in response to total parenteral nutrition. Surgery 114, 400–405 (1993). discussion 405-406.
Meguid, M. M., Yang, Z. J. & Koseki, M. Eating induced rise in LHA-dopamine correlates with meal size in normal and bulbectomized rats. Brain Res. Bull. 36, 487–490 (1995).
Yang, Z. J., Koseki, M., Meguid, M. M. & Laviano, A. Eating-related increase of dopamine concentration in the LHA with oronasal stimulation. Am. J. Physiol. 270, R315–R318 (1996).
Najam, N. Involvement of dopaminergic systems in the ventromedial hypothalamic hyperphagia. Brain Res. Bull. 21, 571–574 (1988).
Baptista, T., Parada, M. & Hernandez, L. Long term administration of some antipsychotic drugs increases body weight and feeding in rats. Are D2 dopamine receptors involved? Pharmacol. Biochem. Behav. 27, 399–405 (1987).
Meguid, M. M., Yang, Z. J. & Laviano, A. Meal size and number: relationship to dopamine levels in the ventromedial hypothalamic nucleus. Am. J. Physiol. 272, R1925–R1930 (1997).
Meguid, M. M. et al. Hypothalamic dopamine and serotonin in the regulation of food intake. Nutrition 16, 843–857 (2000).
Zhang, X. & van den Pol, A. N. Hypothalamic arcuate nucleus tyrosine hydroxylase neurons play orexigenic role in energy homeostasis. Nat. Neurosci. 19, 1341–1347 (2016).
Labouesse, M. A. et al. Striatal dopamine 2 receptor upregulation during development predisposes to diet-induced obesity by reducing energy output in mice. Proc. Natl Acad. Sci. USA 115, 10493–10498 (2018).
Carlin, J., Hill-Smith, T. E., Lucki, I. & Reyes, T. M. Reversal of dopamine system dysfunction in response to high-fat diet. Obesity 21, 2513–2521 (2013).
Friend, D. M. et al. Basal ganglia dysfunction contributes to physical inactivity in obesity. Cell Metab. 25, 312–321 (2017).
Lopez, M., Nogueiras, R., Tena-Sempere, M. & Dieguez, C. Hypothalamic AMPK: a canonical regulator of whole-body energy balance. Nat. Rev. Endocrinol. 12, 421–432 (2016).
Kohlie, R. et al. Dopamine directly increases mitochondrial mass and thermogenesis in brown adipocytes. J. Mol. Endocrinol. 58, 57–66 (2017).
Burt, J., Alberto, C. O., Parsons, M. P. & Hirasawa, M. Local network regulation of orexin neurons in the lateral hypothalamus. Am. J. Physiol. Regulatory, Integr. Comp. Physiol. 301, R572–R580 (2011).
Bubser, M. et al. Dopaminergic regulation of orexin neurons. Eur. J. Neurosci. 21, 2993–3001 (2005).
Yasuda, T. et al. Dual regulatory effects of orexins on sympathetic nerve activity innervating brown adipose tissue in rats. Endocrinology 146, 2744–2748 (2005).
Sellayah, D., Bharaj, P. & Sikder, D. Orexin is required for brown adipose tissue development, differentiation, and function. Cell Metab. 14, 478–490 (2011).
Stoof, J. C. & Kebabian, J. W. Opposing roles for D-1 and D-2 dopamine receptors in efflux of cyclic AMP from rat neostriatum. Nature 294, 366–368 (1981).
Yang, L. & McKnight, G. S. Hypothalamic PKA regulates leptin sensitivity and adiposity. Nat. Commun. 6, 8237 (2015).
Cummings, D. E. et al. Genetically lean mice result from targeted disruption of the RII beta subunit of protein kinase A. Nature 382, 622–626 (1996).
Zheng, R. et al. Deficiency of the RIIbeta subunit of PKA affects locomotor activity and energy homeostasis in distinct neuronal populations. Proc. Natl Acad. Sci. USA 110, E1631–E1640 (2013).
Inancli, S. S. et al. Effect of cabergoline on insulin sensitivity, inflammation, and carotid intima media thickness in patients with prolactinoma. Endocrine 44, 193–199 (2013).
Pala, N. A., Laway, B. A., Misgar, R. A. & Dar, R. A. Metabolic abnormalities in patients with prolactinoma: response to treatment with cabergoline. Diabetol. Metab. Syndr. 7, 99 (2015).
Lamos, E. M., Levitt, D. L. & Munir, K. M. A review of dopamine agonist therapy in type 2 diabetes and effects on cardio-metabolic parameters. Prim. Care Diabetes 10, 60–65 (2016).
Nogueiras, R. et al. Direct control of peripheral lipid deposition by CNS GLP-1 receptor signaling is mediated by the sympathetic nervous system and blunted in diet-induced obesity. J. Neurosci. 29, 5916–5925 (2009).
Nogueiras, R. et al. The central melanocortin system directly controls peripheral lipid metabolism. J. Clin. Investig. 117, 3475–3488 (2007).
Gangarossa, G. et al. Characterization of dopamine D1 and D2 receptor-expressing neurons in the mouse hippocampus. Hippocampus 22, 2199–2207 (2012).
Beiroa, D. et al. GLP-1 agonism stimulates brown adipose tissue thermogenesis and browning through hypothalamic AMPK. Diabetes 63, 3346–3358 (2014).
Contreras, C. et al. Central ceramide-induced hypothalamic lipotoxicity and ER stress regulate energy balance. Cell Rep. 9, 366–377 (2014).
Czyzyk, T. A. et al. Mice lacking delta-opioid receptors resist the development of diet-induced obesity. FASEB J. 26, 3483–3492 (2012).
Alvarez-Crespo, M. et al. Essential role of UCP1 modulating the central effects of thyroid hormones on energy balance. Mol. Metab. 5, 271–282 (2016).
Imbernon, M. et al. Hypothalamic kappa opioid receptor mediates both diet-induced and melanin concentrating hormone-induced liver damage through inflammation and endoplasmic reticulum stress. Hepatology 64, 1086–1104 (2016).
Alvarez-Crespo, M. et al. The orexigenic effect of orexin-A revisited: dependence of an intact growth hormone axis. Endocrinology 154, 3589–3598 (2013).
de Jong, J. W. et al. Reducing ventral tegmental dopamine D2 receptor expression selectively boosts incentive motivation. Neuropsychopharmacol. 40, 2085–2095 (2015).
Messina, A. et al. A microRNA switch regulates the rise in hypothalamic GnRH production before puberty. Nat. Neurosci. 19, 835–844 (2016).
Konner, A. C. et al. Insulin action in AgRP-expressing neurons is required for suppression of hepatic glucose production. Cell Metab. 5, 438–449 (2007).
Lopez, M. et al. Hypothalamic AMPK and fatty acid metabolism mediate thyroid regulation of energy balance. Nat. Med. 16, 1001–1008 (2010).
Imbernon, M. et al. Central melanin-concentrating hormone influences liver and adipose metabolism via specific hypothalamic nuclei and efferent autonomic/JNK1 pathways. Gastroenterology 144, 636–649 e636 (2013).
Seoane, L. M. et al. Agouti-related peptide, neuropeptide Y, and somatostatin-producing neurons are targets for ghrelin actions in the rat hypothalamus. Endocrinology 144, 544–551 (2003).
Melmed, S. et al. Diagnosis and treatment of hyperprolactinemia: an Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 96, 273–288 (2011).
Webster, J. et al. A comparison of cabergoline and bromocriptine in the treatment of hyperprolactinemic amenorrhea. Cabergoline comparative study group. N. Engl. J. Med. 331, 904–909 (1994).
Gómez-Ambrosi, J. et al. Body adiposity and type 2 diabetes: increased risk with a high body fat percentage even having a normal BMI. Obesity 19, 1439–1444 (2011).
Sabater, M. et al. Circulating pigment epithelium-derived factor levels are associated with insulin resistance and decrease after weight loss. J. Clin. Endocrinol. Metab. 95, 4720–4728 (2010).
Gómez-Ambrosi, J. et al. Involvement of leptin in the association between percentage of body fat and cardiovascular risk factors. Clin. Biochem. 35, 315–320 (2002).
Miller, J. Reaction time analysis with outlier exclusion: bias varies with sample size. Q. J. Exp. Psychol. A, Hum. Exp. Psychol. 43, 907–912 (1991).
Razali, N. M. & Wah, Y. B. Power comparisons of shapiro-wilk, kolmogorov-smirnov, lilliefors and anderson-darling tests. J. Stat. Modeling Analytics 2, 21–33 (2011).
Student. The probable error of a mean. Biometrika 6, 1–25 (1908).
Fay, D. S. & Gerow, K. A biologist’s guide to statistical thinking and analysis. WormBook: the Online Review of C. elegans Biology (2013); https://doi.org/10.1895/wormbook.1891.1159.1891
Charan, J. & Biswas, T. How to calculate sample size for different study designs in medical research? Indian J. Psychological Med. 35, 121–126 (2013).
Kao, L. S. & Green, C. E. Analysis of variance: Is there a difference in means and what does it mean? J. Surgical Res. 144, 158–170 (2008).
Bridge, P. D. & Sawilowsky, S. S. Increasing physicians’ awareness of the impact of statistics on research outcomes. J. Clin. Epidemiol. 52, 229–235 (1999).
Dunn, O. J. Multiple comparisons using rank sums. Technometrics 6, 241–252 (1964).
Campbell, G. & Skillings, J. H. Nonparametric stepwise multiple comparison procedures. J. Am. Stat. Assoc. 80, 998–1003 (1985).
Acknowledgements
We would like to thank L. Casas for her excellent technical assistance. This work has been supported by grants from FEDER, Ministerio de Ciencia, Innovación y Universidades-Agencia Estatal de Investigación (C.D.: BFU2017-87721; M.L.: SAF2015-71026-R and BFU2015-70454-REDT/Adipoplast; R.N.: BFU2015-70664R), and Centro Singular de Investigación de Galicia accreditation 2016–2019, ED431G/05) and the European Regional Development Fund (ERDF), Xunta de Galicia (M.L.: 2015-CP079 and 2016-PG068; R.N.: 2015-CP080 and 2016-PG057), Fundación BBVA (R.N.), Fundación Atresmedia (M.L. and R.N.), Instituto de Salud Carlos III and cofunded by FEDER (L.M.S.:PI15/01272 and PI18/01890). The research leading to these results has also received funding from the European Community’s H2020 Framework Programme under the following grant: ERC Synergy Grant-2019-WATCH-810331 to V.P. and R.N. Centro de Investigación Biomédica en Red (CIBER) de Fisiopatología de la Obesidad y Nutrición (CIBERobn). CIBERobn is an initiative of the Instituto de Salud Carlos III (ISCIII) of Spain, which is supported by FEDER funds. This work was supported by Inserm, Fondation pour la Recherche Médicale, ANR-EPITRACES (E.V.).
Author information
Authors and Affiliations
Contributions
C.F., D.B., B.P., M.D., E.P., M.F.-F., S.B.-F., R.G., R.H.-B., C.C., A.S., P.S.-C., N.G.-L., P.A., D.G., M.F., A.R.-R., I.K. and Z.L. carried out the experiments. R.A., C.B., J.L.L.-B. and F.J. generated viral vectors and animal models. J.S. and G.F. performed the assays in patients. C.F., V.P., C.D., M.L., E.V., L.M.S. and R.N. designed and planned the study. All authors contributed to the preparation of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information: Primary Handling Editor: Elena Bellafante
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–11
Supplementary Table 1
Antisense oligonucleotide for rat MCH and rat prepro-OX.
Rights and permissions
About this article
Cite this article
Folgueira, C., Beiroa, D., Porteiro, B. et al. Hypothalamic dopamine signalling regulates brown fat thermogenesis. Nat Metab 1, 811–829 (2019). https://doi.org/10.1038/s42255-019-0099-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-019-0099-7
This article is cited by
-
The effects of ghrelin and LEAP-2 in energy homeostasis are modulated by thermoneutrality, high-fat diet and aging
Journal of Endocrinological Investigation (2024)
-
Astrocytic GABA in LHA is an obesity ‘thermostat’
Nature Metabolism (2023)
-
Dopamine receptor D4 (DRD4) negatively regulates UCP1- and ATP-dependent thermogenesis in 3T3-L1 adipocytes and C2C12 muscle cells
Pflügers Archiv - European Journal of Physiology (2023)
-
Prolonged lactation benefits offspring metabolism
Nature Metabolism (2022)
-
A hypothalamic dopamine locus for psychostimulant-induced hyperlocomotion in mice
Nature Communications (2022)