Abstract
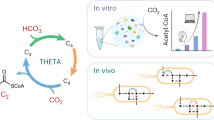
Advanced bioproduct synthesis via reductive metabolism requires coordinating carbons, ATP and reducing agents, which are generated with varying efficiencies depending on metabolic pathways. Substrate mixtures with direct access to multiple pathways may optimally satisfy these biosynthetic requirements. However, native regulation favouring preferential use precludes cells from co-metabolizing multiple substrates. Here we explore mixed substrate metabolism and tailor pathway usage to synergistically stimulate carbon reduction. By controlled cofeeding of superior ATP and NADPH generators as ‘dopant’ substrates to cells primarily using inferior substrates, we circumvent catabolite repression and drive synergy in two divergent organisms. Glucose doping in Moorella thermoacetica stimulates CO2 reduction (2.3 g gCDW−1 h−1) into acetate by augmenting ATP synthesis via pyruvate kinase. Gluconate doping in Yarrowia lipolytica accelerates acetate-driven lipogenesis (0.046 g gCDW−1 h−1) by obligatory NADPH synthesis through the pentose cycle. Together, synergistic cofeeding produces CO2-derived lipids with 38% energy yield and demonstrates the potential to convert CO2 into advanced bioproducts. This work advances the systems-level control of metabolic networks and CO2 use, the most pressing and difficult reduction challenge.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available in the Supplementary Files and from the corresponding author upon request.
Code availability
The code for the metabolic flux and free energy analysis is available on the GitHub public repository at https://github.com/jopark/moorella_yarrowia. The data that support the findings of this study are available in the Supplementary Files and from the corresponding author upon request.
References
Ledesma-Amaro, R. & Nicaud, J. M. Metabolic engineering for expanding the substrate range of Yarrowia lipolytica. Trends Biotechnol. 34, 798–809 (2016).
Atsumi, S., Hanai, T. & Liao, J. C. Non-fermentative pathways for synthesis of branched-chain higher alcohols as biofuels. Nature 451, 86–89 (2008).
Xue, Z. et al. Production of omega-3 eicosapentaenoic acid by metabolic engineering of Yarrowia lipolytica. Nat. Biotechnol. 31, 734–740 (2013).
Qiao, K. J., Wasylenko, T. M., Zhou, K., Xu, P. & Stephanopoulos, G. Lipid production in Yarrowia lipolytica is maximized by engineering cytosolic redox metabolism. Nat. Biotechnol. 35, 173–177 (2017).
Kita, A. et al. Development of genetic transformation and heterologous expression system in carboxydotrophic thermophilic acetogen Moorella thermoacetica. J. Biosci. Bioeng. 115, 347–352 (2013).
MonodJ. Recherches sur la Croissance des Cultures Bactériennes. (Hermann: 1942).
Aristilde, L., Lewis, I. A., Park, J. O. & Rabinowitz, J. D. Hierarchy in pentose sugar metabolism in Clostridium acetobutylicum. Appl. Environ. Microbiol. 81, 1452–1462 (2015).
Görke, B. & Stülke, J. Carbon catabolite repression in bacteria: many ways to make the most out of nutrients. Nat. Rev. Microbiol. 6, 613–624 (2008).
Bren, A. et al. Glucose becomes one of the worst carbon sources for E. coli on poor nitrogen sources due to suboptimal levels of cAMP. Sci. Rep. 6, 24834 (2016).
Joshua, C. J., Dahl, R., Benke, P. I. & Keasling, J. D. Absence of diauxie during simultaneous utilization of glucose and xylose by Sulfolobus acidocaldarius. J. Bacteriol. 193, 1293–1301 (2011).
Hermsen, R., Okano, H., You, C., Werner, N. & Hwa, T. A growth-rate composition formula for the growth of E. coli on co-utilized carbon substrates. Mol. Syst. Biol. 11, 801 (2015).
Kanno, M., Carroll, A. L. & Atsumi, S. Global metabolic rewiring for improved CO2 fixation and chemical production in cyanobacteria. Nat. Commun. 8, 14724 (2017).
Martinez, K. et al. Coutilization of glucose and glycerol enhances the production of aromatic compounds in an Escherichia coli strain lacking the phosphoenolpyruvate: carbohydrate phosphotransferase system. Microb. Cell Fact. 7, 1 (2008).
Meyer, F. et al. Methanol-essential growth of Escherichia coli. Nat. Commun. 9, 1508 (2018).
Garcia Sanchez, R. et al. Improved xylose and arabinose utilization by an industrial recombinant Saccharomyces cerevisiae strain using evolutionary engineering. Biotechnol. Biofuels 3, 13 (2010).
Kim, S. M. et al. Simultaneous utilization of glucose and xylose via novel mechanisms in engineered Escherichia coli. Metab. Eng. 30, 141–148 (2015).
Jones, S. W. et al. CO2 fixation by anaerobic non-photosynthetic mixotrophy for improved carbon conversion. Nat. Commun. 7, 12800 (2016).
Bowes, G., Ogren, W. L. & Hageman, R. H. Light saturation, photosynthesis rate, RuDP carboxylase activity, and specific leaf weight in soybeans grown under different light intensities. Crop Sci. 12, 77 (1972).
Xu, J., Liu, N., Qiao, K., Vogg, S. & Stephanopoulos, G. Application of metabolic controls for the maximization of lipid production in semicontinuous fermentation. Proc. Natl Acad. Sci. USA 114, E5308–E5316 (2017).
Ratledge, C. & Wynn, J. P. The biochemistry and molecular biology of lipid accumulation in oleaginous microorganisms. Adv. Appl. Microbiol. 51, 1–51 (2002).
Qiao, K. et al. Engineering lipid overproduction in the oleaginous yeast Yarrowia lipolytica. Metab. Eng. 29, 56–65 (2015).
Fontanille, P., Kumar, V., Christophe, G., Nouaille, R. & Larroche, C. Bioconversion of volatile fatty acids into lipids by the oleaginous yeast Yarrowia lipolytica. Bioresour. Technol. 114, 443–449 (2012).
Liu, N., Qiao, K. & Stephanopoulos, G. 13C metabolic flux analysis of acetate conversion to lipids by Yarrowia lipolytica. Metab. Eng. 38, 86–97 (2016).
Gancedo, J. M. Carbon catabolite repression in yeast. Eur. J. Biochem. 206, 297–313 (1992).
Casazza, J. P. & Veech, R. L. The interdependence of glycolytic and pentose cycle intermediates in ad libitum fed rats. J. Biol. Chem. 261, 690–698 (1986).
Ragsdale, S. W. & Pierce, E. Acetogenesis and the Wood–Ljungdahl pathway of CO2 fixation. Biochim. Biophys. Acta 1784, 1873–1898 (2008).
Mall, A. et al. Reversibility of citrate synthase allows autotrophic growth of a thermophilic bacterium. Science 359, 563–567 (2018).
Nunoura, T. et al. A primordial and reversible TCA cycle in a facultatively chemolithoautotrophic thermophile. Science 359, 559–563 (2018).
Schuchmann, K. & Müller, V. Autotrophy at the thermodynamic limit of life: a model for energy conservation in acetogenic bacteria. Nat. Rev. Microbiol. 12, 809–821 (2014).
Daniell, J., Köpke, M. & Simpson, S. Commercial biomass syngas fermentation. Energies 5, 5372–5417 (2012).
Hu, P., Rismani-Yazdi, H. & Stephanopoulos, G. Anaerobic CO2 fixation by the acetogenic bacterium Moorella thermoacetica. AIChE J. 59, 3176–3183 (2013).
Blazeck, J. et al. Harnessing Yarrowia lipolytica lipogenesis to create a platform for lipid and biofuel production. Nat. Commun. 5, 3131 (2014).
Babel, W., Brinkmann, U. & Muller, R. H. The auxiliary substrate concept: an approach for overcoming limits of microbial performances. Acta Biotechnol. 13, 211–242 (1993).
Babel, W. The auxiliary substrate concept: from simple considerations to heuristically valuable knowledge. Eng. Life Sci. 9, 285–290 (2009).
Daniel, S. L., Hsu, T., Dean, S. I. & Drake, H. L. Characterization of the H2- and CO-dependent chemolithotrophic potentials of the acetogens Clostridium thermoaceticum and Acetogenium kivui. J. Bacteriol. 172, 4464–4471 (1990).
Ledesma-Amaro, R., Dulermo, R., Niehus, X. & Nicaud, J. M. Combining metabolic engineering and process optimization to improve production and secretion of fatty acids. Metab. Eng. 38, 38–46 (2016).
Martin, V. J., Pitera, D. J., Withers, S. T., Newman, J. D. & Keasling, J. D. Engineering a mevalonate pathway in Escherichia coli for production of terpenoids. Nat. Biotechnol. 21, 796–802 (2003).
Haynes, C. A. & Gonzalez, R. Rethinking biological activation of methane and conversion to liquid fuels. Nat. Chem. Biol. 10, 331–339 (2014).
Tai, M. & Stephanopoulos, G. Engineering the push and pull of lipid biosynthesis in oleaginous yeast Yarrowia lipolytica for biofuel production. Metab. Eng. 15, 1–9 (2013).
Michaelis, L. & Guzman Barron, E. S. Oxidation-reduction systems of biological significance. II. Reducing effect of cysteine induced by free metals. J. Biol. Chem. 81, 29–40 (1929).
Rabinowitz, J. D. & Kimball, E. Acidic acetonitrile for cellular metabolome extraction from Escherichia coli. Anal. Chem. 79, 6167–6173 (2007).
Clasquin, M. F., Melamud, E. & Rabinowitz, J. D. LC–MS data processing with MAVEN: a metabolomic analysis and visualization engine. Curr. Protoc. Bioinformatics Chapter 14, Unit14.11 (2012).
Tracy, B. P., Jones, S. W., Fast, A. G., Indurthi, D. C. & Papoutsakis, E. T. Clostridia: the importance of their exceptional substrate and metabolite diversity for biofuel and biorefinery applications. Curr. Opin. Biotechnol. 23, 364–381 (2012).
Islam, M. A., Zengler, K., Edwards, E. A., Mahadevan, R. & Stephanopoulos, G. Investigating Moorella thermoacetica metabolism with a genome-scale constraint-based metabolic model. Integr. Biol. (Camb.) 7, 869–882 (2015).
Schellenberger, J. et al. Quantitative prediction of cellular metabolism with constraint-based models: the COBRA Toolbox v2.0. Nat. Protoc. 6, 1290–1307 (2011).
Antoniewicz, M. R., Kelleher, J. K. & Stephanopoulos, G. Elementary metabolite units (EMU): a novel framework for modeling isotopic distributions. Metab. Eng. 9, 68–86 (2007).
Antoniewicz, M. R., Kelleher, J. K. & Stephanopoulos, G. Determination of confidence intervals of metabolic fluxes estimated from stable isotope measurements. Metab. Eng. 8, 324–337 (2006).
Acknowledgements
The authors would like to thank C. Lewis and E. Freinkman for their help with the LC–MS. This research was supported by the U.S. Department of Energy grant nos. DE-AR0000433, DE-SC0008744 and DE-SC0012377, as well as a Mobility Plus Fellowship no. 1284/MOB/IV/2015/0.
Author information
Authors and Affiliations
Contributions
J.O.P, N.L. and G.S. designed the study and wrote the paper. J.O.P., N.L. and K.M.H performed the experiments and flux analysis. J.O.P., N.L., B.M.W. and C.V. developed the methods for the LC–MS and gas chromatography–mass spectrometry. J.O.P., N.L., D.F.E. and J.X. designed the bioreactors. J.O.P. and M.A.I. developed the updated metabolic model. J.O.P., N.L., K.Q., Z.L., P.R.G. and G.S. analysed the data.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information: Primary Handling Editor: Ana Mateus.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–9, Supplementary Tables 1–7 and Supplementary Note
Rights and permissions
About this article
Cite this article
Park, J.O., Liu, N., Holinski, K.M. et al. Synergistic substrate cofeeding stimulates reductive metabolism. Nat Metab 1, 643–651 (2019). https://doi.org/10.1038/s42255-019-0077-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-019-0077-0
This article is cited by
-
Formate-induced CO tolerance and methanogenesis inhibition in fermentation of syngas and plant biomass for carboxylate production
Biotechnology for Biofuels and Bioproducts (2023)
-
Metabolic engineering of Pichia pastoris for myo-inositol production by dynamic regulation of central metabolism
Microbial Cell Factories (2022)
-
Metabolic reconfiguration enables synthetic reductive metabolism in yeast
Nature Metabolism (2022)
-
Synthetic metabolism without the TCA cycle
Nature Metabolism (2022)
-
Bio-oil production for biodiesel industry by Yarrowia lipolytica from volatile fatty acids in two-stage batch culture
Applied Microbiology and Biotechnology (2022)