Abstract
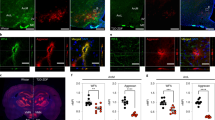
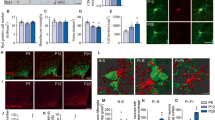
In leptin-deficient ob/ob mice, obesity and diabetes are associated with abnormal development of neurocircuits in the hypothalamic arcuate nucleus (ARC)1, a critical brain area for energy and glucose homoeostasis2,3. Because this developmental defect can be remedied by systemic leptin administration, but only if given before postnatal day 28, a critical period for leptin-dependent development of ARC neurocircuits has been proposed4. In other brain areas, critical-period closure coincides with the appearance of perineuronal nets (PNNs), extracellular matrix specializations that restrict the plasticity of neurons that they enmesh5. Here we report that in humans and rodents, subsets of neurons in the mediobasal aspect of the ARC are enmeshed in PNN-like structures. In mice, these neurons are densely packed into a continuous ring that encircles the junction of the ARC and median eminence, which facilitates exposure of ARC neurons to the circulation. Most of the enmeshed neurons are both γ-aminobutyric acid-ergic and leptin-receptor positive, including a majority of Agouti-related-peptide neurons. Postnatal formation of the PNN-like structures coincides precisely with closure of the critical period for maturation of Agouti-related-peptide neurons and is dependent on input from circulating leptin, because postnatal ob/ob mice have reduced ARC PNN-like material that is restored by leptin administration during the critical period. We conclude that neurons crucial to metabolic homoeostasis are enmeshed in PNN-like structures and organized into a densely packed cluster situated circumferentially at the ARC–median eminence junction, where metabolically relevant humoral signals are sensed.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding authors upon request. Additional detailed information on experimental design and reagents is available in the Reporting Summary.
References
Bouret, S. G., Draper, S. J. & Simerly, R. B. Trophic action of leptin on hypothalamic neurons that regulate feeding. Science 304, 108–110 (2004).
Schwartz, M. W. et al. Obesity pathogenesis: an Endocrine Society scientific statement. Endocr. Rev. 38, 267–296 (2017).
Deem, J. D., Muta, K., Scarlett, J. M., Morton, G. J. & Schwartz, M. W. How should we think about the role of the brain in glucose homeostasis and diabetes? Diabetes 66, 1758–1765 (2017).
Kamitakahara, A., Bouyer, K., Wang, C. H. & Simerly, R. A critical period for the trophic actions of leptin on AgRP neurons in the arcuate nucleus of the hypothalamus. J. Comp. Neurol. 526, 133–145 (2018).
Pizzorusso, T. et al. Reactivation of ocular dominance plasticity in the adult visual cortex. Science 298, 1248–1251 (2002).
Hensch, T. K. Critical period plasticity in local cortical circuits. Nat. Rev. Neurosci. 6, 877–888 (2005).
Wiesel, T. N. & Hubel, D. H. Single-cell responses in striate cortex of kittens deprived of vision in one eye. J. Neurophysiol. 26, 1003–1017 (1963).
Carulli, D. et al. Animals lacking link protein have attenuated perineuronal nets and persistent plasticity. Brain 133, 2331–2347 (2010).
Kwok, J. C., Dick, G., Wang, D. & Fawcett, J. W. Extracellular matrix and perineuronal nets in CNS repair. Dev. Neurobiol. 71, 1073–1089 (2011).
Balmer, T. S., Carels, V. M., Frisch, J. L. & Nick, T. A. Modulation of perineuronal nets and parvalbumin with developmental song learning. J. Neurosci. 29, 12878–12885 (2009).
Gogolla, N., Caroni, P., Lüthi, A. & Herry, C. Perineuronal nets protect fear memories from erasure. Science 325, 1258–1261 (2009).
Nowicka, D., Soulsby, S., Skangiel-Kramska, J. & Glazewski, S. Parvalbumin-containing neurons, perineuronal nets and experience-dependent plasticity in murine barrel cortex. Eur. J. Neurosci. 30, 2053–2063 (2009).
Beurdeley, M. et al. Otx2 binding to perineuronal nets persistently regulates plasticity in the mature visual cortex. J. Neurosci. 32, 9429–9437 (2012).
Miyata, S., Komatsu, Y., Yoshimura, Y., Taya, C. & Kitagawa, H. Persistent cortical plasticity by upregulation of chondroitin 6-sulfation. Nat. Neurosci. 15, 414–422 (2012).
Maeda, N. Structural variation of chondroitin sulfate and its roles in the central nervous system. Cent. Nerv. Syst. Agents Med. Chem. 10, 22–31 (2010).
Ahima, R. S., Prabakaran, D. & Flier, J. S. Postnatal leptin surge and regulation of circadian rhythm of leptin by feeding: implications for energy homeostasis and neuroendocrine function. J. Clin. Invest. 101, 1020–1027 (1998).
Baquero, A. F. et al. Developmental switch of leptin signaling in arcuate nucleus neurons. J. Neurosci. 34, 9982–9994 (2014).
Bouret, S. G. et al. Hypothalamic neural projections are permanently disrupted in diet-induced obese rats. Cell Metab. 7, 179–185 (2008).
Glavas, M. M. et al. Early overnutrition results in early-onset arcuate leptin resistance and increased sensitivity to high-fat diet. Endocrinology 151, 1598–1610 (2010).
Vogt, M. C. et al. Neonatal insulin action impairs hypothalamic neurocircuit formation in response to maternal high-fat feeding. Cell 156, 495–509 (2014).
Bayol, S. A., Simbi, B. H. & Stickland, N. C. A maternal cafeteria diet during gestation and lactation promotes adiposity and impairs skeletal muscle development and metabolism in rat offspring at weaning. J. Physiol. 567, 951–961 (2005).
Gorski, J. N., Dunn-Meynell, A. A., Hartman, T. G. & Levin, B. E. Postnatal environment overrides genetic and prenatal factors influencing offspring obesity and insulin resistance. Am. J. Physiol. Regul. Integr. Comp. Physiol. 291, R768–R778 (2006).
Carstens, K. E., Phillips, M. L., Pozzo-Miller, L., Weinberg, R. J. & Dudek, S. M. Perineuronal nets suppress plasticity of excitatory synapses on CA2 pyramidal neurons. J. Neurosci. 36, 6312–6320 (2016).
Köppe, G., Brückner, G., Härtig, W., Delpech, B. & Bigl, V. Characterization of proteoglycan-containing perineuronal nets by enzymatic treatments of rat brain sections. Histochem. J. 29, 11–20 (1997).
Bäckberg, M., Collin, M., Ovesjö, M. L. & Meister, B. Chemical coding of GABA(B) receptor-immunoreactive neurones in hypothalamic regions regulating body weight. J. Neuroendocrinol. 15, 1–14 (2003).
Morrison, C. D., Morton, G. J., Niswender, K. D., Gelling, R. W. & Schwartz, M. W. Leptin inhibits hypothalamic Npy and Agrp gene expression via a mechanism that requires phosphatidylinositol 3-OH-kinase signaling. Am. J. Physiol. Endocrinol. Metab. 289, E1051–E1057 (2005).
McRae, P. A., Rocco, M. M., Kelly, G., Brumberg, J. C. & Matthews, R. T. Sensory deprivation alters aggrecan and perineuronal net expression in the mouse barrel cortex. J. Neurosci. 27, 5405–5413 (2007).
Nakamura, M. et al. Expression of chondroitin sulfate proteoglycans in barrel field of mouse and rat somatosensory cortex. Brain Res. 1252, 117–129 (2009).
Dityatev, A. et al. Activity-dependent formation and functions of chondroitin sulfate-rich extracellular matrix of perineuronal nets. Dev. Neurobiol. 67, 570–588 (2007).
Luquet, S., Perez, F. A., Hnasko, T. S. & Palmiter, R. D. NPY/AgRP neurons are essential for feeding in adult mice but can be ablated in neonates. Science 310, 683–685 (2005).
Xu, J. et al. Genetic identification of leptin neural circuits in energy and glucose homeostases. Nature 556, 505–509 (2018).
de Winter, F. et al. The chemorepulsive protein semaphorin 3A and perineuronal net-mediated plasticity. Neural Plast. 2016, 3679545 (2016).
Barker, D. J. The fetal and infant origins of adult disease. BMJ 301, 1111 (1990).
Ravelli, G. P., Stein, Z. A. & Susser, M. W. Obesity in young men after famine exposure in utero and early infancy. N. Engl. J. Med. 295, 349–353 (1976).
Ravelli, A. C. et al. Glucose tolerance in adults after prenatal exposure to famine. Lancet 351, 173–177 (1998).
Vickers, M. H. et al. Neonatal leptin treatment reverses developmental programming. Endocrinology 146, 4211–4216 (2005).
Miyata, S. & Kitagawa, H. Formation and remodeling of the brain extracellular matrix in neural plasticity: roles of chondroitin sulfate and hyaluronan. Biochim. Biophys. Acta Gen. Subj. 1861, 2420–2434 (2017).
Saghatelyan, A. K. et al. Reduced perisomatic inhibition, increased excitatory transmission, and impaired long-term potentiation in mice deficient for the extracellular matrix glycoprotein tenascin-R. Mol. Cell. Neurosci. 17, 226–240 (2001).
Hylin, M. J., Orsi, S. A., Moore, A. N. & Dash, P. K. Disruption of the perineuronal net in the hippocampus or medial prefrontal cortex impairs fear conditioning. Learn. Mem. 20, 267–273 (2013).
Thaler, J. P. et al. Obesity is associated with hypothalamic injury in rodents and humans. J. Clin. Invest. 122, 153–162 (2012).
Douglass, J. D., Dorfman, M. D., Fasnacht, R., Shaffer, L. D. & Thaler, J. P. Astrocyte IKKβ/NF-κB signaling is required for diet-induced obesity and hypothalamic inflammation. Mol. Metab. 6, 366–373 (2017).
Valdearcos, M. et al. Microglial inflammatory signaling orchestrates the hypothalamic immune response to dietary excess and mediates obesity susceptibility. Cell Metab. 26, 185–197.e3 (2017).
Balland, E. et al. Hypothalamic tanycytes are an ERK-gated conduit for leptin into the brain. Cell Metab. 19, 293–301 (2014).
Gao, Y. et al. Hormones and diet, but not body weight, control hypothalamic microglial activity. Glia 62, 17–25 (2014).
Tamamaki, N. et al. Green fluorescent protein expression and colocalization with calretinin, parvalbumin, and somatostatin in the GAD67-GFP knock-in mouse. J. Comp. Neurol. 467, 60–79 (2003).
Leshan, R. L., Björnholm, M., Münzberg, H. & Myers, M. G.Jr. Leptin receptor signaling and action in the central nervous system. Obesity (Silver Spring) 14, 208S–212S (2006).
Madisen, L. et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat. Neurosci. 13, 133–140 (2010).
van den Pol, A. N. et al. Neuromedin B and gastrin-releasing peptide excite arcuate nucleus neuropeptide Y neurons in a novel transgenic mouse expressing strong renilla green fluorescent protein in NPY neurons. J. Neurosci. 29, 4622–4639 (2009).
Cowley, M. A. et al. Leptin activates anorexigenic POMC neurons through a neural network in the arcuate nucleus. Nature 411, 480–484 (2001).
Mirzadeh, Z., Doetsch, F., Sawamoto, K., Wichterle, H. & Alvarez-Buylla, A. The subventricular zone en-face: wholemount staining and ependymal flow. J. Vis. Exp. 39, 1938 (2010).
Doetsch, F., García-Verdugo, J. M. & Alvarez-Buylla, A. Cellular composition and three-dimensional organization of the subventricular germinal zone in the adult mammalian brain. J. Neurosci. 17, 5046–5061 (1997).
Acknowledgements
The authors are grateful to the original providers of the transgenic mouse lines used in this work: N. Tamamaki (GAD67-GFP), M. Myers (LepRb-Cre), H. Zeng (Ai14), B. Lowell (NPY-GFP) and M. Low (POMC-GFP). This work was supported by the US National Institutes of Health National Institute of Diabetes and Digestive and Kidney Diseases (grant nos. DK108596 (Z.M.), DK114474 (J.M.S.), DK083042 (M.W.S.), DK090320 (M.W.S.) and DK101997 (M.W.S.)), the National Institute of Neurological Disorders and Stroke Neurosurgeon Research Career Development Program K Award (Z.M.), the American Diabetes Association (grant no. 7–11-BS-179 (L.Z.)), the Russell Berrie Foundation (R.H.) and the Barrow Neurological Foundation (grant no. 18-0025-30-05 (Z.M.)).
Author information
Authors and Affiliations
Contributions
Z.M., K.M.A. and M.W.S. conceived and designed the study; Z.M., K.M.A., E.C., V.H.P., J.M.S., J.M.B., R.H., M.E.M., H.T.N., J.M.G.V., L.M.Z. and M.W.S. acquired, analysed and interpreted the data; Z.M. and M.W.S. drafted and revised the manuscript. All authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures. 1–9
Supplementary Video 1
Imaris three-dimensional (3D) surface rendering of an isolated PNN-enmeshed (red) GAD67-GFP+ ARC neuron (green). This rendering was performed on a 63× image taken from a coronal section, through the ARC of a GAD67-GFP mouse, stained with WFA (red) and GFP antibody (green). This 3D image is representative of renderings performed on five other GAD67-GFP+ ARC neurons
Supplementary Video 2
Imaris 3D surface rendering of an isolated PNN-enmeshed (red) GAD67-GFP+ V1 interneuron (green). This rendering was performed on a 63× image taken from a coronal section, through V1 of a GAD67-GFP mouse, stained with WFA (red) and GFP antibody (green). This 3D image is representative of renderings performed on three other GAD67-GFP+ V1 neurons.
Supplementary Video 3
Imaris 3D surface rendering of an isolated PNN-enmeshed (red) Npy-GFP+ ARC neuron (green). This rendering was performed on a 63× image taken from a coronal section, through the ARC of a Npy-GFP mouse, stained with WFA (red) and GFP antibody (green). This 3D image is representative of renderings performed on five other NPY-GFP+ neurons.
Supplementary Video 4
Imaris 3D surface rendering of an isolated PNN-enmeshed (red) NPY+ human ARC neuron (green). This rendering was performed on a 63× image taken from a coronal section, through the ARC of a human brain, stained with WFA (red) and NPY antibody (green). This 3D image is representative of renderings performed on four other NPY+ human ARC neurons.
Rights and permissions
About this article
Cite this article
Mirzadeh, Z., Alonge, K.M., Cabrales, E. et al. Perineuronal net formation during the critical period for neuronal maturation in the hypothalamic arcuate nucleus. Nat Metab 1, 212–221 (2019). https://doi.org/10.1038/s42255-018-0029-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-018-0029-0
This article is cited by
-
Microglia shape AgRP neuron postnatal development via regulating perineuronal net plasticity
Molecular Psychiatry (2023)
-
The ventromedial hypothalamic nucleus: watchdog of whole-body glucose homeostasis
Cell & Bioscience (2022)
-
Metabolic Messengers: fibroblast growth factor 1
Nature Metabolism (2022)
-
POMC neuronal heterogeneity in energy balance and beyond: an integrated view
Nature Metabolism (2021)
-
Central nervous system regulation of organismal energy and glucose homeostasis
Nature Metabolism (2021)