Abstract
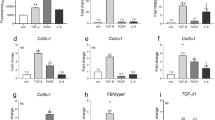
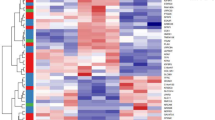
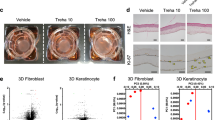
Extracellular matrix (ECM) homeostasis is essential for normal tissue function, and its disruption by iatrogenic injury, trauma, or disease results in fibrosis. Skin ECM homeostasis is maintained by a complex process that involves an integration of cytokine and environmental mediators. However, it is unclear, in both normal and disease states, how these multifactorial processes converge to shift ECM homeostasis towards accumulation or degradation. Here we show a consistent downregulation in fatty acid oxidation (FAO) and upregulation of glycolysis in fibrotic skin and in normal skin with abundant ECM. Perturbation of glycolysis and FAO pathway enzymes reveals their reciprocal effects in ECM upregulation and downregulation, respectively. Increasing peroxisome proliferator-activated receptor (PPAR) signalling, an inducer of the FAO pathway, generates a catabolic fibroblast phenotype characterised by inhibition of ECM transcription and enhanced ECM internalization and lysosomal degradation. In contrast, suppression of glycolysis inhibits ECM gene transcription and protein levels, independently of an intact FAO pathway or PPAR signalling. Moreover, we show that CD36, a multifunctional fatty acid transporter, connects the metabolic state of fibroblasts with their capacity for ECM regulation, as internalization and degradation of collagen-1 is abrogated in fibroblasts lacking CD36. Finally, restoring FAO and upregulating CD36 reduces ECM accumulation in murine skin fibrosis. These findings indicate that metabolic perturbation of ECM homeostasis may have broad implications for therapies aimed at ECM regulation, such as fibrosis, regenerative medicine, and ageing.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Transcriptomic data can be found on the National Center for Biotechnology Information (NCBI) gene expression omnibus (GEO) using ID GSE98157 for murine RNA-seq data and GSE98159 for human HT-12 array data. Software parameters have been uploaded onto Github: https://github.com/bhklab/SkinFibrosis. The data that support the plots within this paper and other findings of this study are available from the corresponding authors upon reasonable request.
References
Watt, F. M. Mammalian skin cell biology: at the interface between laboratory and clinic. Science 346, 937–940 (2014).
Vander Heiden, M. G., Cantley, L. C. & Thompson, C. B. Understanding the Warburg Effect: the metabolic requirements of cell proliferation. Science 324, 1029–1033 (2009).
Kelly, B. & O’Neill, L. A. Metabolic reprogramming in macrophages and dendritic cells in innate immunity. Cell Res. 25, 771–784 (2015).
Stone, H. B., Coleman, C. N., Anscher, M. S. & McBride, W. H. Effects of radiation on normal tissue: consequences and mechanisms. Lancet Oncol. 4, 529–536 (2003).
Mehta, A. S. & Blodgett, T. M. Retroperitoneal fibrosis as a cause of positive FDG PET/CT. J. Radiol. Case Rep. 5, 35–41 (2011).
Justet, A. et al. [18F]FDG PET/CT predicts progression-free survival in patients with idiopathic pulmonary fibrosis. Respir. Res. 18, 74 (2017).
Kok, H. M., Falke, L. L., Goldschmeding, R. & Nguyen, T. Q. Targeting CTGF, EGF and PDGF pathways to prevent progression of kidney disease. Nat. Rev. Nephrol. 10, 700–711 (2014).
Higgins, D. F. et al. Hypoxic induction of Ctgf is directly mediated by Hif-1. Am. J. Physiol. Ren. Physiol. 287, F1223–F1232 (2004).
Lamb, J. et al. The Connectivity Map: using gene-expression signatures to connect small molecules, genes, and disease. Science 313, 1929–1935 (2006).
Vanella, L. et al. Caffeic acid phenethyl ester regulates PPAR’s levels in stem cells-derived Adipocytes. PPAR Res. 2016, 7359521 (2016).
Isono, M., Chen, S., Hong, S. W., Iglesias-de la Cruz, M. C. & Ziyadeh, F. N. Smad pathway is activated in the diabetic mouse kidney and Smad3 mediates TGF-beta-induced fibronectin in mesangial cells. Biochem. Biophys. Res. Commun. 296, 1356–1365 (2002).
McCulloch, C. A. G. & Knowles, G. C. Deficiencies in collagen phagocytosis by human fibroblasts in vitro: a mechanism for fibrosis? J. Cell. Physiol. 155, 461–471 (1993).
Febbraio, M., Hajjar, D. P. & Silverstein, R. L. CD36: a class B scavenger receptor involved in angiogenesis, atherosclerosis, inflammation, and lipid metabolism. J. Clin. Invest. 108, 785–791 (2001).
Zhang, P., Tang, Y., Li, N.-G., Zhu, Y. & Duan, J.-A. Bioactivity and chemical synthesis of caffeic acid phenethyl ester and its derivatives. Molecules 19, 16458–16476 (2014).
Coburn, C. T. et al. Defective uptake and utilization of long chain fatty acids in muscle and adipose tissues of CD36 knockout mice. J. Biol. Chem. 275, 32523–32529 (2000).
Vincent, A. S. et al. Human skin keloid fibroblasts display bioenergetics of cancer cells. J. Invest. Dermatol. 128, 702–709 (2008).
Ring, A., Le Lay, S., Pohl, J., Verkade, P. & Stremmel, W. Caveolin-1 is required for fatty acid translocase (FAT/CD36) localization and function at the plasma membrane of mouse embryonic fibroblasts. Biochim. Biophys. Acta 1761, 416–423 (2006).
Del Galdo, F. et al. Decreased expression of caveolin 1 in patients with systemic sclerosis: crucial role in the pathogenesis of tissue fibrosis. Arthritis Rheum. 58, 2854–2865 (2008).
Castello-Cros, R. et al. Scleroderma-like properties of skin from caveolin-1-deficient mice: implications for new treatment strategies in patients with fibrosis and systemic sclerosis. Cell Cycle 10, 2140–2150 (2011).
Xie, N. et al. Glycolytic reprogramming in myofibroblast differentiation and lung fibrosis. Am. J. Respir. Crit. Care. Med. 192, 1462–1474 (2015).
Kang, H. M. et al. Defective fatty acid oxidation in renal tubular epithelial cells has a key role in kidney fibrosis development. Nat. Med. 21, 37–46 (2015).
Lovisa, S., Zeisberg, M. & Kalluri, R. Partial epithelial-to-mesenchymal transition and other new mechanisms of kidney fibrosis. Trends Endocrinol. Metab. 27, 681–695 (2016).
Stone, H. B. Leg contracture in mice: an assay of normal tissue response. Int. J. Radiat. Oncol. Biol. Phys. 10, 1053–1061 (1984).
Trapnell, C. et al. Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat. Protoc. 7, 562–578 (2012).
Dutta, T. et al. Concordance of changes in metabolic pathways based on plasma metabolomics and skeletal muscle transcriptomics in type 1 diabetes. Diabetes 61, 1004–1016 (2012).
Pfaffl, M. W. Relative expression software tool (REST(C)) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Res. 30, 36e–36e (2002).
Ran, F. A. et al. Genome engineering using the CRISPR-Cas9 system. Nat. Protoc. 8, 2281–2308 (2013).
Dennler, S. et al. Direct binding of Smad3 and Smad4 to critical TGF beta-inducible elements in the promoter of human plasminogen activator inhibitor-type 1 gene. EMBO J. 17, 3091–3100 (1998).
Khan, M. et al. mTORC2 controls cancer cell survival by modulating gluconeogenesis. Cell Death Discov. 1, 15016 (2015).
Jiang, P. et al. P53 regulates biosynthesis through direct inactivation of glucose-6-phosphate dehydrogenase. Nat. Cell Biol. 13, 310–316 (2011).
Irizarry, R. A. Exploration, normalization, and summaries of high density oligonucleotide array probe level data. Biostatistics 4, 249–264 (2003).
Smirnov, P. et al. PharmacoGx: an R package for analysis of large pharmacogenomic datasets. Bioinformatics 32, 1244–1246 (2016).
Rousseeuw, P. J. Silhouettes: A graphical aid to the interpretation and validation of cluster analysis. J. Comput. Appl. Math. 20, 53–65 (1987).
Acknowledgements
This research was funded by the Canadian Institutes of Health Research, the Canadian Cancer Society Research Institute, the Physicians Services Incorporated Foundation, the Harry Barbarian Research Grant, the Mariano Elia Chair in Head and Neck Cancer Research, the Campbell Family Institute for Cancer Research, the Ministry of Health and Long-Term Care, and the Princess Margaret Cancer Centre Head and Neck Translational Program, with philanthropic funding from the Wharton family and Joe’s Team.
Author information
Authors and Affiliations
Contributions
X.Z., L.S.G., K.Y., D.G., R.G., I.W., S.V.B., L.A., B.H.-K., and F.-F.L. participated in the research design. X.Z, P.P., .H.P., A.H., J.H.L., and J.W. conducted experiments. X.Z., L.S.G., L.A., B.H.-K. contributed new reagents or analytic tools. G.Z., L.S.G., P.P., H.P., A.H., J.H.L. and J.W. performed data analysis. X.Z., L.S.G., P.P., K.Y., D.G., R.G., S.V.B., L.A., B.H.-K., and F.-F.L. wrote or contributed to the writing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–35 and Supplementary Tables 1 and 2
Supplementary Dataset 1
qRT–PCR primers
Rights and permissions
About this article
Cite this article
Zhao, X., Psarianos, P., Ghoraie, L.S. et al. Metabolic regulation of dermal fibroblasts contributes to skin extracellular matrix homeostasis and fibrosis. Nat Metab 1, 147–157 (2019). https://doi.org/10.1038/s42255-018-0008-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-018-0008-5
This article is cited by
-
The HIF-1α/PLOD2 axis integrates extracellular matrix organization and cell metabolism leading to aberrant musculoskeletal repair
Bone Research (2024)
-
PHDs-seq: a large-scale phenotypic screening method for drug discovery through parallel multi-readout quantification
Cell Regeneration (2023)
-
Tissue fibrosis induced by radiotherapy: current understanding of the molecular mechanisms, diagnosis and therapeutic advances
Journal of Translational Medicine (2023)
-
Mesenchymal stem cells in fibrotic diseases—the two sides of the same coin
Acta Pharmacologica Sinica (2023)
-
Mechanical stiffness promotes skin fibrosis through Piezo1-mediated arginine and proline metabolism
Cell Death Discovery (2023)