Abstract
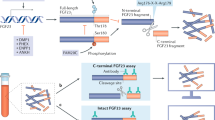
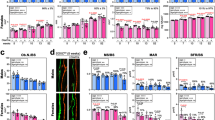
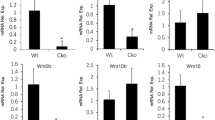
Transferrin receptor 2 (Tfr2) is mainly expressed in the liver and controls iron homeostasis. Here, we identify Tfr2 as a regulator of bone homeostasis that inhibits bone formation. Mice lacking Tfr2 display increased bone mass and mineralization independent of iron homeostasis and hepatic Tfr2. Bone marrow transplantation experiments and studies of cell-specific Tfr2-knockout mice demonstrate that Tfr2 impairs BMP-p38MAPK signalling and decreases expression of the Wnt inhibitor sclerostin, specifically in osteoblasts. Reactivation of MAPK or overexpression of sclerostin rescues skeletal abnormalities in Tfr2-knockout mice. We further show that the extracellular domain of Tfr2 binds bone morphogenic proteins (BMPs) and inhibits BMP-2-induced heterotopic ossification by acting as a decoy receptor. These data indicate that Tfr2 limits bone formation by modulating BMP signalling, possibly through direct interaction with BMP either as a receptor or as a co-receptor in complex with other BMP receptors. Finally, the Tfr2 extracellular domain may be effective in the treatment of conditions associated with pathological bone formation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data sets generated during the current study are available from the corresponding author upon reasonable request.
Change history
12 April 2019
In the version of this article initially published, affiliation 14 was incorrect, and Deutsche Forschungsgemeinschaft grants SFB1036 and SFB1118 were missing from the Acknowledgements. The errors have been corrected in the HTML and PDF versions of the article.
References
Muckenthaler, M. U., Rivella, S., Hentze, M. W. & Galy, B. A red carpet for iron metabolism. Cell 168, 344–361 (2017).
Imel, E. A. et al. Serum fibroblast growth factor 23, serum iron and bone mineral density in premenopausal women. Bone 86, 98–105 (2016).
Guggenbuhl, P. et al. Bone mineral density in men with genetic hemochromatosis and HFE gene mutation. Osteoporos. Int. 16, 1809–1814 (2005).
Nemeth, E. et al. Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science 306, 2090–2093 (2004).
Powell, L. W., Seckington, R. C. & Deugnier, Y. Haemochromatosis. Lancet 388, 706–716 (2016).
Camaschella, C. et al. The gene TFR2 is mutated in a new type of haemochromatosis mapping to 7q22. Nat. Genet. 25, 14–15 (2000).
Fleming, R. E. et al. Targeted mutagenesis of the murine transferrin receptor-2 gene produces hemochromatosis. Proc. Natl. Acad. Sci. USA 99, 10653–10658 (2002).
Forejtnikova, H. et al. Transferrin receptor 2 is a component of the erythropoietin receptor complex and is required for efficient erythropoiesis. Blood 116, 5357–5367 (2010).
Wallace, D. F., Summerville, L., Lusby, P. E. & Subramaniam, V. N. First phenotypic description of transferrin receptor 2 knockout mouse, and the role of hepcidin. Gut 54, 980–986 (2005).
Johnson, M. B. & Enns, C. A. Diferric transferrin regulates transferrin receptor 2 protein stability. Blood 104, 4287–4293 (2004).
Poli, M. et al. Transferrin receptor 2 and HFE regulate furin expression via mitogen-activated protein kinase/extracellular signal-regulated kinase (MAPK/Erk) signaling. Implications for transferrin-dependent hepcidin regulation. Haematologica 95, 1832–1840 (2010).
D’Alessio, F., Hentze, M. W. & Muckenthaler, M. U. The hemochromatosis proteins HFE, TfR2, and HJV form a membrane-associated protein complex for hepcidin regulation. J. Hepatol. 57, 1052–1060 (2012).
Wallace, D. F. et al. Combined deletion of Hfe and transferrin receptor 2 in mice leads to marked dysregulation of hepcidin and iron overload. Hepatology 50, 1992–2000 (2009).
Hogan, B. L. Bone morphogenetic proteins: multifunctional regulators of vertebrate development. Genes Dev. 10, 1580–1594 (1996).
Andriopoulos, B. et al. BMP6 is a key endogenous regulator of hepcidin expression and iron metabolism. Nat. Genet. 41, 482–487 (2009).
Babitt, J. L. et al. Bone morphogenetic protein signaling by hemojuvelin regulates hepcidin expression. Nat. Genet. 38, 531–539 (2006).
Mayeur, C., Leyton, P. A., Kolodziej, S. A., Yu, B. & Bloch, K. D. BMP type II receptors have redundant roles in the regulation of hepatic hepcidin gene expression and iron metabolism. Blood 124, 2116–2123 (2014).
Steinbicker, A. U. et al. Perturbation of hepcidin expression by BMP type I receptor deletion induces iron overload in mice. Blood 118, 4224–4230 (2011).
Wang, R. H. et al. A role of SMAD4 in iron metabolism through the positive regulation of hepcidin expression. Cell. Metab. 2, 399–409 (2005).
Yu, P. B. et al. Dorsomorphin inhibits BMP signals required for embryogenesis and iron metabolism. Nat. Chem. Biol. 4, 33–41 (2008).
Shore, E. M. et al. A recurrent mutation in the BMP type I receptor ACVR1 causes inherited and sporadic fibrodysplasia ossificans progressiva. Nat. Genet. 38, 525–527 (2006).
Wallace, D. F. et al. A critical role for murine transferrin receptor 2 in erythropoiesis during iron restriction. Br. J. Haematol. 168, 891–901 (2015).
Nai, A. et al. The erythroid function of transferrin receptor 2 revealed by Tmprss6 inactivation in different models of transferrin receptor 2 knockout mice. Haematologica 99, 1016–1021 (2014).
Urist, M. R. Bone: formation by autoinduction. Science 150, 893–899 (1965).
Roetto, A. et al. Comparison of 3 Tfr2-deficient murine models suggests distinct functions for Tfr2-alpha and Tfr2-beta isoforms in different tissues. Blood 115, 3382–3389 (2010).
Herrmann, T. et al. Iron overload in adult Hfe-deficient mice independent of changes in the steady-state expression of the duodenal iron transporters DMT1 and Ireg1/ferroportin. J. Mol. Med. 82, 39–48 (2004).
Altamura, S. et al. Resistance of ferroportin to hepcidin binding causes exocrine pancreatic failure and fatal iron overload. Cell. Metab. 20, 359–367 (2014).
Tsay, J. et al. Bone loss caused by iron overload in a murine model: importance of oxidative stress. Blood 116, 2582–2589 (2010).
Rishi, G., Secondes, E. S., Wallace, D. F. & Subramaniam, V. N. Normal systemic iron homeostasis in mice with macrophage-specific deletion of transferrin receptor 2. Am. J. Physiol. Gastrointest. Liver Physiol. 310, G171–G180 (2016).
Kamiya, N. et al. Wnt inhibitors Dkk1 and Sost are downstream targets of BMP signaling through the type IA receptor (BMPRIA) in osteoblasts. J. Bone Miner. Res. 25, 200–210 (2010).
Simsek Kiper, P. O. et al. Cortical-bone fragility–insights from sFRP4 deficiency in Pyle’s disease. N. Engl. J. Med. 374, 2553–2562 (2016).
Wosczyna, M. N., Biswas, A. A., Cogswell, C. A. & Goldhamer, D. J. Multipotent progenitors resident in the skeletal muscle interstitium exhibit robust BMP-dependent osteogenic activity and mediate heterotopic ossification. J. Bone Miner. Res. 27, 1004–1017 (2012).
Shimono, K. et al. Potent inhibition of heterotopic ossification by nuclear retinoic acid receptor-gamma agonists. Nat. Med. 17, 454–460 (2011).
Fransen, M. et al. Safety and efficacy of routine postoperative ibuprofen for pain and disability related to ectopic bone formation after hip replacement surgery (HIPAID): randomised controlled trial. BMJ 333, 519 (2006).
Shen, G. S. et al. Hepcidin1 knockout mice display defects in bone microarchitecture and changes of bone formation markers. Calcif. Tissue Int. 94, 632–639 (2014).
Guggenbuhl, P. et al. Bone status in a mouse model of genetic hemochromatosis. Osteoporos. Int. 22, 2313–2319 (2011).
Doyard, M. et al. Decreased bone formation explains osteoporosis in a genetic mouse model of hemochromatosiss. PLoS ONE 11, e0148292 (2016).
Calzolari, A. et al. TfR2 localizes in lipid raft domains and is released in exosomes to activate signal transduction along the MAPK pathway. J. Cell. Sci. 119, 4486–4498 (2006).
Keller, S., Nickel, J., Zhang, J. L., Sebald, W. & Mueller, T. D. Molecular recognition of BMP-2 and BMP receptor IA. Nat. Struct. Mol. Biol. 11, 481–488 (2004).
Yin, H., Yeh, L. C., Hinck, A. P. & Lee, J. C. Characterization of ligand-binding properties of the human BMP type II receptor extracellular domain. J. Mol. Biol. 378, 191–203 (2008).
Canali, S., Wang, C. Y., Zumbrennen-Bullough, K. B., Bayer, A. & Babitt, J. L. Bone morphogenetic protein 2 controls iron homeostasis in mice independent of Bmp6. Am. J. Hematol. 92, 1204–1213 (2017).
Koch, P. S. et al. Angiocrine Bmp2 signaling in murine liver controls normal iron homeostasis. Blood 129, 415–419 (2017).
Ramos, E. et al. Evidence for distinct pathways of hepcidin regulation by acute and chronic iron loading in mice. Hepatology 53, 1333–1341 (2011).
Li, X. et al. Targeted deletion of the sclerostin gene in mice results in increased bone formation and bone strength. J. Bone Miner. Res. 23, 860–869 (2008).
MacDonald, B. T. et al. Bone mass is inversely proportional to Dkk1 levels in mice. Bone 41, 331–339 (2007).
van Bezooijen, R. L. et al. Sclerostin is an osteocyte-expressed negative regulator of bone formation, but not a classical BMP antagonist. J. Exp. Med. 199, 805–814 (2004).
Kamiya, N. et al. BMP signaling negatively regulates bone mass through sclerostin by inhibiting the canonical Wnt pathway. Development 135, 3801–3811 (2008).
Yu, C. et al. Advanced oxidation protein products induce apoptosis, and upregulate sclerostin and RANKL expression, in osteocytic MLO-Y4 cells via JNK/p38 MAPK activation. Mol. Med. Rep. 15, 543–550 (2017).
Croons, V., Martinet, W., Herman, A. G., Timmermans, J. P. & De Meyer, G. R. The protein synthesis inhibitor anisomycin induces macrophage apoptosis in rabbit atherosclerotic plaques through p38 mitogen-activated protein kinase. J. Pharmacol. Exp. Ther. 329, 856–864 (2009).
Kamiya, N., Kaartinen, V. M. & Mishina, Y. Loss-of-function of ACVR1 in osteoblasts increases bone mass and activates canonical Wnt signaling through suppression of Wnt inhibitors SOST and DKK1. Biochem. Biophys. Res. Commun. 414, 326–330 (2011).
Biswas, S. et al. BMPRIA is required for osteogenic differentiation and RANKL expression in adult bone marrow mesenchymal stromal cells. Sci. Rep. 8, 8475 (2018).
Witcher, P. C. et al. Sclerostin neutralization unleashes the osteoanabolic effects of Dkk1 inhibition. JCI Insight 3, e98673 (2018).
Lowery, J. W. et al. Loss of BMPR2 leads to high bone mass due to increased osteoblast activity. J. Cell Sci. 128, 1308–1315 (2015).
Bao, Q. et al. Disruption of bone morphogenetic protein type IA receptor in osteoblasts impairs bone quality and bone strength in mice. Cell Tissue Res. 374, 263–273 (2018).
Zhang, Y. et al. Loss of BMP signaling through BMPR1A in osteoblasts leads to greater collagen cross-link maturation and material-level mechanical properties in mouse femoral trabecular compartments. Bone 88, 74–84 (2016).
Forsberg, J. A. et al. Heterotopic ossification in high-energy wartime extremity injuries: prevalence and risk factors. J. Bone Jt. Surg. Am. 91, 1084–1091 (2009).
Regis, D., Sandri, A. & Sambugaro, E. Incidence of heterotopic ossification after surface and conventional total hip arthroplasty: a comparative study using anterolateral approach and indomethacin prophylaxis. Biomed. Res. Int. 2013, 293528 (2013).
Chakkalakal, S. A. et al. Palovarotene inhibits heterotopic ossification and maintains limb mobility and growth in mice with the human ACVR1(R206H) fibrodysplasia ossificans progressiva (FOP) mutation. J. Bone Miner. Res. 31, 1666–1675 (2016).
Agarwal, S. et al. mTOR inhibition and BMP signaling act synergistically to reduce muscle fibrosis and improve myofiber regeneration. JCI Insight 1, e89805 (2016).
Hino, K. et al. Activin-A enhances mTOR signaling to promote aberrant chondrogenesis in fibrodysplasia ossificans progressiva. J. Clin. Invest. 127, 3339–3352 (2017).
Rodda, S. J. & McMahon, A. P. Distinct roles for Hedgehog and canonical Wnt signaling in specification, differentiation and maintenance of osteoblast progenitors. Development 133, 3231–3244 (2006).
Nakamura, T. et al. Estrogen prevents bone loss via estrogen receptor alpha and induction of Fas ligand in osteoclasts. Cell 130, 811–823 (2007).
Clausen, B. E., Burkhardt, C., Reith, W., Renkawitz, R. & Forster, I. Conditional gene targeting in macrophages and granulocytes using LysMcre mice. Transgenic Res. 8, 265–277 (1999).
Rhee, Y. et al. PTH receptor signaling in osteocytes governs periosteal bone formation and intracortical remodeling. J. Bone Miner. Res. 26, 1035–1046 (2011).
Liu, X. et al. A novel mouse model of trauma induced heterotopic ossification. J. Orthop. Res. 32, 183–188 (2014).
Norden, D. M. et al. Ibuprofen ameliorates fatigue- and depressive-like behavior in tumor-bearing mice. Life Sci. 143, 65–70 (2015).
Bassett, J. H. et al. Optimal bone strength and mineralization requires the type 2 iodothyronine deiodinase in osteoblasts. Proc. Natl. Acad. Sci. USA 107, 7604–7609 (2010).
Rauner, M. et al. Increased EPO levels are associated with bone loss in mice lacking PHD2 in EPO-producing cells. J. Bone Miner. Res. 31, 1877–1887 (2016).
Theurl, I. et al. On-demand erythrocyte disposal and iron recycling requires transient macrophages in the liver. Nat. Med. 22, 945–951 (2016).
Liberzon, A. et al. The Molecular Signatures Database hallmark gene set collection. Cell Syst. 1, 417–425 (2015).
Park, K. W. et al. The small molecule phenamil induces osteoblast differentiation and mineralization. Mol. Cell. Biol. 29, 3905–3914 (2009).
Liberzon, A. et al. Molecular signatures database (MSigDB) 3.0. Bioinformatics 27, 1739–1740 (2011).
Acknowledgements
We would like to thank our technicians for their excellent work. We thank the Core Facility Cellular Imaging of TU Dresden for their support with the confocal microscope and the acquisition of immunofluorescence images and A. Drescher and J. Nickel for critical suggestions regarding SPR analyses. This work was supported by the German Research Foundation (DFG-SFB655 to L.C.H. and U.P.; TRR-67 to V.H. and L.C.H.; µBONE to M.R. and L.C.H.; RA1923/12-1 to M.R.) and MedDrive start-up grants from the Medical Faculty of the Technische Universität Dresden (M.R. and U.B.). M.R. was supported by the Support-the-Best Initiative of the TUD funded through the Excellence initiative of the German Federal and State Governments. J.H.D.B. and G.R.W. received a Wellcome Trust Joint Investigator Award (110141/Z/15/Z and 110140/Z/15/Z). M.U.M. acknowledges support by grants from the Deutsche Forschungsgemeinschaft (SFB1036 and SFB1118).
Author information
Authors and Affiliations
Contributions
M.R., U.B., A.R., J.S-H., S.R., U.P., and L.C.H. designed experiments. M.R., U.B., A.R., R.M.P., J.S.-H., H.W., S.R., G.C., A.P., R.L., I.H., S.C., and D.K.E. performed experiments and analysed data. T.B., S.A., S.C., M.U.M., and I.T. provided mouse bone samples. M.R., U.B., A.R., J.S.-H., H.W., S.R., V.H., I.H., T.B., M.U.M., J.H.D.B., G.R.W., G.S., I.T., U.P., and L.C.H. interpreted the data and provided critical comments on the manuscript. M.R., U.B., and L.C.H. wrote the manuscript. All authors provided critical review of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The Technische Universität Dresden holds a patent for the use of Tfr2-ECD to treat heterotopic ossification and other related bone excess diseases (PCT/EP2018/065846). Moreover, a patent application has been filed at the European Patent Office for the use of Tfr2 blockade for the treatment of bone and haematological diseases (#18 177 441.5, 19.06.2018). M.R., U.B., U.P., and L.C.H. are the inventors of both patents. I.T. is a consultant for Kymab Ltd. The other authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–11 and Supplementary Tables 2–4
Supplementary Table 1
Summary of the results obtained from gene ontology analysis
Rights and permissions
About this article
Cite this article
Rauner, M., Baschant, U., Roetto, A. et al. Transferrin receptor 2 controls bone mass and pathological bone formation via BMP and Wnt signalling. Nat Metab 1, 111–124 (2019). https://doi.org/10.1038/s42255-018-0005-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-018-0005-8
Keywords
This article is cited by
-
Comparison of the effects of high dietary iron levels on bone microarchitecture responses in the mouse strains 129/Sv and C57BL/6J
Scientific Reports (2024)
-
The Role of Neuromodulation and Potential Mechanism in Regulating Heterotopic Ossification
Neurochemical Research (2024)
-
Proteomics-based evaluation of the mechanism underlying vascular injury via DNA interstrand crosslinks, glutathione perturbation, mitogen-activated protein kinase, and Wnt and ErbB signaling pathways induced by crotonaldehyde
Clinical Proteomics (2022)
-
Dickkopf1 fuels inflammatory cytokine responses
Communications Biology (2022)
-
The mutual crosstalk between iron and erythropoiesis
International Journal of Hematology (2022)