Abstract
Light is not the only stimulus that can induce linear-to-cyclic isomerization of donor-acceptor Stenhouse adducts (DASAs). Here we demonstrate the water-induced linear-to-cyclic isomerization of DASAs. The mechanism of the water-induced linear-to-cyclic isomerization of DASAs is investigated by density functional theory (DFT) calculations. Water molecules coordinate with DASAs and stabilize the intermediates and cyclic isomers, which favors cyclization thermodynamically. Moreover, the linear-to-cyclic isomerization is reversible. Heating removes the coordinated H2O molecules, which further triggers cyclic-to-linear isomerization. DASAs have been applied in information hiding/displaying and color switching under water vapor and heating control.
Similar content being viewed by others
Introduction
Donor-acceptor Stenhouse adducts (DASAs) as novel photoresponsive molecules have been attracting the attention of researchers since 20141,2,3,4,5,6,7,8. DASAs show linear-to-cyclic isomerization under visible or near-infrared (NIR) light irradiation (~480–750 nm), while cyclic-to-linear isomerization occurs upon heating in the dark9. Light irradiation switches the polarity of DASA molecules. In particular, the DASAs with alkyl-substituted electron-donating moieties show zwitterionic cyclic isomers that exhibit higher dipole moments than the linear forms6. Moreover, the DASAs with aryl-substituted electron-donating moieties show nonzwitterionic cyclic isomers6.
Compared with traditional UV-light-responsive molecules, including azobenzene10, spiropyran11, coumarin12, and o-nitrobenzyl molecules13, DASAs that are responsive to visible or NIR light are attractive, especially for potential applications in biomedicine. In fact, several groups have been working on drug-delivery systems and intelligent surfaces controlled by visible light14,15,16,17.
However, until now, most researches have only focused on the light-induced isomerization. In the current report, we demonstrate the water-induced linear-to-cyclic isomerization of DASAs, which is reversible under heating. Since the earliest report cyclic DASAs have been found to be more stable when water exists1. The linear-to-cyclic isomerization occurs under dark in aqueous environment was also noticed by Feringa’s group earlier in 2016 and 201818,19,20. However, to our knowledge no research is focusing on the mechanism of the water-induced isomerization and the relationships between DASAs and H2O molecules, which are important for the understanding, development and application of DASAs.
Herein, the mechanistic investigation is guided by density functional theory (DFT) calculations. Water molecules coordinate with DASAs and lower the energy levels of the intermediates and cyclic isomer. More importantly, the water-induced process iss demonstrated to be reversible. The cyclic-to-linear isomerization is induced by removing the coordinated H2O molecules (Fig. 1). DASAs are further applied in information hiding/displaying and color switching under controlling of water vapor and heat exposure.
Results
Water-induced linear-to-cyclic isomerization of DASAs
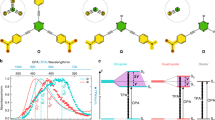
DASA-N, DASA-O with zwitterionic cyclic isomers and DASA-M, DASA-I with nonzwitterionic cyclic isomers were synthesized (Fig. 1 and Supplementary Fig. 1)1,4,6. Linear-to-cyclic isomerizations triggered by green light (530 nm) were observed for all DASAs (Fig. 2a and Supplementary Figs. 2–5). Colorless cyclic DASA-N was obtained after irradiation, and this species is unstable in toluene (Fig. 2a). Interestingly, after adding water into a toluene solution of linear DASA-N (toluene:water = 6:1, v/v), we found that the linear DASA-N spontaneously converts to cyclic DASA-N, even without light, this is in good accordance to the previous reports (Fig. 2b)1,2,18,19,20. The process is extremely slow compared with the light-induced isomerization, and only ~30% of cyclic DASA-N can be obtained after 10 h, and the process is finished in 40 h (Supplementary Fig. 7). Furthermore, the cyclic DASA-N is stabilized by H2O molecules, and therefore, it does not switch back to the linear isomer during 1H NMR studies (Fig. 2c). The results were supported by 2D 1H-1H chemical shift correlation spectroscopy (2D-COSY) (Supplementary Figs. 8–10).
Molecular isomerization of DASA-N. a UV/vis spectra of DASA-N in the dark (black) and after irradiation with green light (red) ([DASA-N] = 0.04 mM in toluene); b UV/vis spectra of DASA-N after water treatment at 20 °C for 0 min (black) and 480 min (red) ([DASA-N] = 0.04 mM in toluene, toluene:water = 6:1, v/v) (Due to toluene and water are not miscible, the water concentration in toluene is ~27 mM); c Time-dependent change in absorbance (570 nm) of DASA-N under water treatment at 40 °C (black, square), 20 °C (red, circle), and 0 °C (blue, triangle) ([DASA-N] = 0.04 mM in toluene, toluene:water = 6:1, v/v); d Plot of kinetic absorption data of DASA-N under water treatment at 40 °C (black, square), 20 °C (red, circle), and 0 °C (blue, triangle) ([DASA-N] = 0.04 mM in toluene, toluene:water = 6:1, v/v). The absorbance at 568 nm is normalized
These results indicated that in addition to light, water is also able to trigger the linear-to-cyclic isomerization of DASA-N. Importantly, the rate of the water-induced linear-to-cyclic isomerization is positively correlated with the temperature. The isomerization of DASA-N was investigated at 4, 20, and 40 °C (Supplementary Figs. 11–13). We learned that higher temperatures resulted in faster linear-to-cyclic isomerization rates (Fig. 2c). The water-induced isomerization of DASA-N was demonstrated to be a thermal process, which is similar to the thermal-induced cis-to-trans isomerization of azobenzene and water-induced spirocyclic-to-merocyanine isomerization of spiropyran (Fig. 2d, see the SI for details)21,22. As expected, the rate constant for the linear-to-cyclic isomerization of DASA-N at 40 °C is two orders of magnitude higher than that at 4 °C (Fig. 2d). In other words, cyclic DASA-N may be stabilized in the presence of water.
In addition to DASA-N, water-induced linear-to-cyclic isomerizations were observed for DASA-O, DASA-M and DASA-I (Supplementary Figs. 14–16)7. For both of the DASA molecules with zwitterionic cyclic isomers (DASA-N and DASA-O) and nonzwitterionic cyclic isomers (DASA-M and DASA-I), more than 60% of the linear isomers converted to the cyclic isomers after 90 min of heating at 40 °C (Supplementary Figs. 17–20). All the DASA molecules show similar isomerization rates, indicating the universality of the water-induced linear-to-cyclic isomerization in both of the DASA molecules with zwitterionic and nonzwitterionic cyclic isomers (Supplementary Fig. 21).
DASAs are not the only photoresponsive molecules that show water-induced isomerization. Solvents have been reported to favor the isomerization of photoresponsive molecules (e.g., azobenzene and spiropyran) and affect the isomer distribution23,24, and this is especially true for spiropyrans with zwitterionic merocyanine isomers. H2O molecules are believed to form hydrogen bonds with the merocyanine spiropyran, lowering its ground state energy, which allows the spirocyclic-to-merocyanine isomerization in the absence of light21.
Mechanism of the water-induced isomerization
DFT calculations were applied to investigate the mechanism of the water-induced linear-to-cyclic isomerization of the DASAs. The kinetic investigation indicated that the cyclic DASA may be stabilized by H2O molecules and show a lower ground state energy. Therefore, H2O molecules are assumed to coordinate with DASAs in toluene.
Up to 5 H2O molecules can form complexes with DASA-N (Fig. 3a and Supplementary Fig. 27). The H2O molecules are arranged between the hydroxyl and carbonyl groups of linear DASA-N due to hydrogen bonding (Supplementary Fig. 28). All the possible positions for coordinating of water molecules are investigated (Supplementary Figs. 29–39, Supplementary Tables 1, 2). For cyclic DASA-N, the H2O molecules stably surround the carbonyl in the electron-withdrawing domain (Fig. 3a). Molecular dynamics (MD) simulations indicated that the binding between H2O molecules and cyclic DASA-N is stronger than that with linear DASA-N. All the linear DASA-N·xH2O (x = 1–5) were dissociated after 9 ps MD simulations, while the molecular geometries were stable under the same conditions for cyclic DASA-N·xH2O (x = 3 and 5) (Supplementary Figs. 40–45). These results indicated cyclic DASA-N could be stabilized by H2O molecules.
DFT calculations. a Molecular structures of DASA-N coordinated with 0, 1, and 3 H2O molecules by DFT. Full geometry optimizations were carried out at the M06-2X/6–311++g(d, p) level of theory, and toluene was selected as the solvent. b ΔE between the linear and cyclic DASA-N coordinated to various numbers of H2O molecules. c Schematic illustration of the putative mechanism of the water-induced linear-to-cyclic isomerization of DASA-N
Similar results were concluded by calculating the energy difference (ΔE) between the linear and cyclic isomers (Ecyclic − Elinear), which shows a decreased ΔE as the number of surrounding H2O molecules increases (Fig. 3b and Supplementary Table 3). The ΔE goes to the lowest when 3 H2O molecules coordinate with DASA-N, indicating that the cyclic DASA-N is stabilized by H2O molecules (Fig. 3b, c). However, by further increasing the number of coordinated H2O molecules (DASA-N·4H2O and DASA-N·5H2O), the ΔE does not further decrease (Fig. 3b and Supplementary Table 3). These results are in good accordance with MD simulations.
Similar results were noticed by applying water as the solvent. Actually, the ΔE is negative when applying water as the solvent, which is thermodynamically favorable for the linear-to-cyclic isomerization of DASA-N (Supplementary Figs. 46, 51 and Supplementary Table 4). Moreover, for the cyclic DASA-N, the coordinated H2O molecules may transfer to quaternary ammonium salt in the electron-donating domain, which further lowers the ΔE (Supplementary Fig. 47, 52 and Supplementary Table 5). Therefore, for the thermodynamic concern, water molecules play two roles during the linear-to-cyclic isomerization of DASA-N: (1) water molecules coordinated around the hydroxyl and carbonyl decrease ΔE between linear and cyclic DASA-N, which favors the linear-to-cyclic isomerization; (2) water molecules coordinated on quaternary ammonium salt further decrease the molecular energy and stabilize cyclic DASA-N. In this work, all the coordinated H2O molecules were set around the carbonyl in the electron-withdrawing domain for a direct comparison.
Similar results were observed for DASA-O, DASA-M and DASA-I through DFT calculations (Supplementary Figs. 48–50). H2O molecules coordinate with DASAs through hydrogen bonding to stabilize the cyclic isomers (Supplementary Figs. 53–55 and Supplementary Table 6–8). However, for DASA-M, even without H2O molecules, the linear isomers are not more stable than the cyclic isomers (Supplementary Fig. 54). Considering that isomerization only occurs in the presence of water, the process of the water-induced linear-to-cyclic isomerization was investigated.
Feringa’s group demonstrated a intermediates-included linear-to-cyclic isomerization process of DASAs18,20,25. Linear DASAs (A1) switches to A2 via a light-induced C–C isomerization (Fig. 4a). Afterwards, A2 switches to A3 via a thermal-induced C–C rotation, and through a A2-A3 transition state. A3 then switches to cyclic DASAs (A4) through the cyclization (Supplementary Fig. 56). The Gibbs free energy of the intermediates of DASA-N were calculated by DFT methods (see the Supplementary Methods for details). With the process of linear-to-cyclic isomerization, the free energy of DASA-N increases step by step, and reaches the highest for A4 (Fig. 4c). These results are in good accordance with the previous reports25. After coordinated with 3 H2O molecules, the linear-to-cyclic isomerization is induced by thermal (Fig. 4b). The Gibbs free energy decreases step by step with the process, which thermodynamically favors the linear-to-cyclic isomerization of DASA-N·3H2O (Fig. 4d). These results also explain why the cyclic-to-linear isomerization of DASA-N·3H2O does not occur spontaneously. The process of linear-to-cyclic isomerization of DASA-M and DASA-M·3H2O is similar to DASA-N and DASA-N·3H2O (Fig. 4a, b). H2O molecules coordination causes a decreased free energy of the intermediates, which favors the isomerization (Fig. 4c, d).
Investigation of linear-to-cyclic isomerization process. a Schematic illustration of the reported mechanism of the light-induced linear-to-cyclic isomerization of DASA-N and DASA-M. b Schematic illustration of the hypothesized mechanism of the water-induced linear-to-cyclic isomerization of DASA-N·3H2O and DASA-M·3H2O. c Relative Gibbs energy levels of DASA-N and DASA-M in toluene. d Relative Gibbs energy levels of DASA-N·3H2O and DASA-M·3H2O in toluene
Moreover, previous research demonstrated that an intramolecular proton transfer mechanism was involved in the light-induced linear-to-cyclic isomerization of DASAs19. Protons transfer from the hydroxyl group to the carbonyl group, which favors cyclization (Supplementary Fig. 58). Moreover, Feringa’s group very recently reported that protic solvents mainly affect the thermal process in the light-induced linear-to-cyclic isomerization11. In this work, we hypothesized the coordinated H2O molecules could assist in the intramolecular proton transfer from the hydroxyl group to the carbonyl group (Supplementary Fig. 58), further promoting the cyclization (Fig. 58, B3–B4).
Heat-induced cyclic-to-linear isomerization of DASAs
The water-induced linear-to-cyclic isomerization of DASA is reversible. Removing the coordinated H2O molecules induces cyclic-to-linear isomerization. The cyclic DASA-N·xH2O was prepared in two steps, namely, heating the mixture of linear DASA-N and water at 70 °C, and then gently evaporating the solvent (see the SI for details). Anhydrous MgSO4 and a microwave oven were tested for removing the coordinated H2O molecules. However, no cyclic-to-linear isomerization was observed by UV-vis, indicating that the complexes are stable. Actually, cyclic DASA-N·xH2O does not switch back to linear DASA-N in DCM (Supplementary Fig. 59). The thermal gravimetric analysis (TGA) curves of the cyclic DASA-N·xH2O showed that weight loss began at 145 °C, which can be attributed to the loss of coordinated H2O molecules (Fig. 5a). In contrast, the linear DASA-N begins to lose weight at 224 °C and almost reaches equilibrium at 500 °C. The number of H2O molecules (x) coordinated to the cyclic DASA-N·xH2O was calculated to be >2.6, which is in good agreement with the DFT results (Fig. 5a).
Cyclic-to-linear isomerization. a Thermal gravimetric analysis curves for linear DASA-N (black) and cyclic DASA-N·xH2O (red). b UV/vis spectra of cyclic DASA-N·xH2O before (black) and after (red) heating ([DASA-N] = 0.04 mM in dichloromethane (DCM)). c Putative mechanism of the cyclic-to-linear isomerization of DASA-N induced by removing the coordinated H2O molecules
We tried to heat the cyclic DASA-N·xH2O to remove the coordinated H2O molecules. A droplet of a colorless solution of cyclic DASA-N·xH2O in methanol (0.1 mM) was dropped on filter paper. After heating with a heat gun at 180 °C for ~10 s, the colorless spot on the filter paper became bright purple (Supplementary Fig. 61). UV/vis spectra of cyclic DASA-N·xH2O showed a sharp increase of the absorbance in visible region after heating, indicating the formation of linear DASA-N (Fig. 5b). These were further demonstrated by 1H NMR and Fourier transform infrared (FTIR) spectra (Supplementary Figs. 65, 66). Therefore, heating can remove the coordinated H2O molecules of cyclic DASA-N·xH2O, and further triggers the cyclic-to-linear isomerization (Fig. 5c). DASA-N is reversible in 5 linear-to-cyclic and cyclic-to-linear cycles, and thermally stable under 180 °C (Supplementary Figs. 67, 68). The reversible isomerization of DASA-N by adding and removing coordinated H2O molecules is important for DASAs, which are potential to be applied as hydrochromic and thermochromic molecules.
The cyclic-to-linear isomerization triggered by removing the coordinated H2O molecules occurs for all the DASA-N, DASA-O, DASA-M and DASA-I, indicating the universality of the phenomena (Supplementary Figs. 62–64, 72, 73).
Application in information hiding/displaying and color-switched inks
Due to the reversibility of the water-induced linear-to-cyclic isomerization, DASA-N was applied in information hiding/displaying and color switching under controlling of water vapor and heat exposure (Fig. 6). A “9” was written on filter paper using normal purple pigment, and a vertical line was added with the DASA-N ink ([DASA-N] = 0.3 mM in dichloromethane) to form an “8” (Fig. 6a). Water vapor treatment switched the linear DASA-N to colorless cyclic DASA-N, changing the “8” to a “9” (Fig. 6a and Supplementary Movie 1). The cyclic DASA-N is stable on filter paper under ambient conditions for months and does not switch back to the linear form. Heating at 180 °C for ~30 s removed the coordinated H2O molecules and changed the “9” back to an “8” (Fig. 6a and Supplementary Movie 2).
Applications of water-induced isomerization of DASAs. a Schematic illustration and photographs of switching between “8” and “9” using DASA-N under controlling of water vapor and heat exposure. b Schematic illustration and photographs of switching the color between purple and cyan by DASA-N under controlling of water vapor and heat exposure. c UV/vis spectra of color-switched inks on paper after treating with vapor (black) and heat (red). Insert shows the color change. d Absorbance (500 nm) of color-switched inks on paper after sequential vapor and heat treating
By mixing the DASA-N with stimuli-nonresponsive dyes, color switching can be achieved by controlling exposure to vapor and heating (Fig. 6b). DASA-N and azure II were dissolved in dichloromethane ([DASA-N] = 0.3 mM, [azure II] = 0.2 mM) to prepare color-switching ink. The smiley face on the filter paper switched from purple to cyan after water vapor treatment for ~5 min due to the water-induced linear-to-cyclic isomerization of DASA-N (Fig. 6b and Supplementary Movie 3). Heating at 180 °C triggered the cyclic-to-linear isomerization and switched the smiley face back to purple in ~30 s (Fig. 6b and Supplementary Movie 4). Peak shifting at visible light region was observed for the DASAs/azure II color-switched inks, which is reversible for several cycles after sequential treatment of vapor and heat (Fig. 6c, d). Moreover, a series of color-switching inks were prepared by mixing DASA-N with various stimuli-nonresponsive dyes, including dimethyl yellow, methylene blue, Fast Green FCF, and methyl violet (Supplementary Figs. 74, 75, see the SI for details).
Discussion
In summary, we report the reversible, water-induced linear-to-cyclic isomerization of DASAs. Both the DASAs with zwitterionic (i.e., DASA-N and DASA-O) and nonzwitterionic (i.e., DASA-M and DASA-I) cyclic isomers show water-induced linear-to-cyclic isomerizations. The isomerization process is positively correlated with temperature. H2O molecules coordinate with the DASAs and stabilize their cyclic isomers. Approximately three H2O molecules coordinate with the DASA-N and switch the linear isomer to the colorless and stable cyclic DASA-N·xH2O. Removing the coordinated H2O molecules by heating induces the cyclic-to-linear isomerization of DASAs, indicating that the water-induced process is reversible. The DASA-N was applied in information hiding/displaying and color-switched inks under controlling of water vapor and heating exposure.
As DASAs are reported as novel photoresponsive molecules, the investigations of their applications are still in their infancy. In addition to the light-induced process, the reported reversible water-induced linear-to-cyclic isomerization deepens our understanding and broadens the applications of the DASAs. Therefore, the role of H2O molecules to induce linear-to-cyclic isomerization of DASAs is encouraged to be understood from different perspective and needs further researches. We envision that DASAs can be widely applied in supramolecular materials, energy and printing applications based on the water-induced process. Light and water can be used to switch the DASAs in an orthogonal manner3,26. Moreover, DASAs that are responsive to visible or even NIR light have the potential to be applied in biomedicine27. Therefore, in this case, limiting the water-induced linear-to-cyclic isomerization is important. Understanding the mechanism of the water-induced process is thus helpful for synthesizing a DASA that is stable in water and is only responsive to light.
Methods
General synthesis of DASAs
Step I: electron-withdrawing part (Meldrum’s acid or 1,3-dimethylbarbituric acid) (1 euqiv.) and furfural (1 equiv.) were dissolved in water. The solution was stirred under room temperature for 2 h. The yellow crude product was filtrated out and washed with cold water two times. Then the precipitate was collected and dissolved into DCM, and washed with saturated NaHSO3 solution, saturated NaCl solution and water sequentially. After drying with MgSO4, the yellow solid was further purified by chromatography to obtain product 1 or 2 (Supplementary Fig. 1).
Step II: product 1 or 2 (1 equiv.) and electron-donating part (diethylamine, N-methylaniline or indoline) (1 equiv.) were dissolved in DCM. The mixed solution was stirred under 30 °C for 2 h. After removing the solvent by a rotavapor, the rough product was purified by chromatography to obtain DASAs (Supplementary Fig. 1).
DFT calculations
All the DFT calculations were performed with Gaussian 16 package. Full geometry optimizations were carried out at M06-2X/6–31++g(d,p) level of theory, whereas the reaction pathways were obtained with 6–311++G(d,p) atomic basis set after a basis set test (Supplementary Fig. 22). The choice of this combination was used by several previous benchmarks that demonstrated their performances for weak interactions like hydrogen bonds18,20,28. Vibration frequencies were calculated to confirm if all of the structures optimized were at stationary points or true minima on the potential energy surfaces. The solvation model density (SMD) solvation model was chosen to calculate the electronic energy in solution. To describe the hydrogen bonding interaction between SA and water molecules, various number of water molecules (1–5) were modeled and optimized with DASA molecules, and also the obtained stationary points were characterized by frequency calculations. Further data can be found in Supplementary Data 1.
MD simulations
Ab initio molecular dynamics simulations (AIMD) were performed using first-principles DFT as implemented in the Vienna ab initio simulation package (VASP)29,30,31. The exchange and correlation were obtained within the generalized gradient approximation (GGA), using the functional of Perdew, Burke, and Ernzerhof (PBE)32. The projector augmented wave (PAW) method was used to describe the potential of the nuclear and the core33,34. For the electronic structure calculations, plane waves with a kinetic energy cutoff of 500 eV were adopted to expand the valance electron (2s22p2) wavefunctions. Brillouin zone (BZ) integrations were carried using Γ centered sampling grids with 1 × 1 × 1. Structural relaxation was performed using the conjugate gradients (CG) scheme until the force components were converged to within 0.02 eV/Å, and the self-consistent field calculations were stopped when the energy difference between two steps was smaller than 1 × 10−6 eV/atom. Temperature was set to 300 K and volume (NVT) were performed to exam thermal stability.
Mass-production of cyclic DASA·xH2O
Linear DASAs were mixed with water. The mixture was heated to 70 °C for 5 h to obtain clear light brown solution. After removing the solvent smoothly by a rotavapor under room temperature, the solid cyclic DASA·xH2O could be obtained.
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its supplementary information files.
References
Helmy, S. et al. Photoswitching using visible light: a new class of organic photochromic molecules. J. Am. Chem. Soc. 136, 8169–8172 (2014).
Helmy, S. et al. Design and synthesis of donor-acceptor Stenhouse adducts: a visible light photoswitch derived from furfural. J. Org. Chem. 79, 11316–11329 (2014).
Lerch, M. M. et al. Orthogonal photoswitching in a multifunctional molecular system. Nat. Commun. 7, 12054–12063 (2016).
Hemmer, J. R. et al. Tunable visible and near infrared photoswitches. J. Am. Chem. Soc. 138, 13960–13966 (2016).
Hemmer, J. R. et al. Controlling dark equilibria and enhancing donor-acceptor Stenhouse adduct photoswitching properties through carbon acid design. J. Am. Chem. Soc. 140, 10425–10429 (2018).
Mallo, N. et al. Photochromic switching behavior of donor-acceptor Stenhouse adducts in organic solvents. Chem. Commun. 52, 13576–13579 (2016).
Mallo, N. et al. Structure-function relationships of donor-acceptor Stenhouse adduct photochromic switches. Chem. Sci. 9, 8242–8252 (2018).
Lerch, M. M. et al. The (photo)chemistry of stenhouse photoswitches: guiding principles and system design. Chem. Soc. Rev. 47, 1910–1937 (2018).
Mason, B. P. et al. A temperature-mapping molecular sensor for polyurethane-based elastomers. Appl. Phys. Lett. 108, 041906 (2016).
Bandara, H. M. D. & Burdette, S. C. Photoisomerization in different classes of azobenzene. Chem. Soc. Rev. 41, 1809–1825 (2012).
Klajn, R. Spiropyran-based dynamic materials. Chem. Soc. Rev. 43, 148–184 (2014).
Tunc, D. et al. Reversible cross-linking of aliphatic polyamides bearing thermos- and photoresponsive cinnamoyl moieties. Macromolecules 47, 8247–8254 (2014).
Yan, B. et al. Near infrared light triggered release of biomacromolecules from hydrogels loaded with upconversion nanoparticles. J. Am. Chem. Soc. 134, 16558–16561 (2012).
Poelma, S. O. et al. Controlled drug release to cancer cells from modular one-photon visible light-responsive micellar system. Chem. Commun. 52, 10525–10528 (2016).
Singh, S. et al. Spatiotemporal photopatterning on polycarbonate surface through visible light responsive polymer bound DASA compounds. ACS Macro Lett. 4, 1273–1277 (2015).
Zhao, H. Q. et al. Surface with reversible green-light-switched wettability by donor-acceptor Stenhouse adducts. Langmuir 34, 15537–15543 (2018).
Sinawang, G. et al. Polystyrene based visible light responsive polymer with donor-acceptor stenhouse adduct pendants. Macromol. Chem. Phys. 217, 2409–2414 (2016).
Lerch, M. M. et al. Unraveling the photoswitching mechanism in donor-accepotor stenhouse adducts. J. Am. Chem. Soc. 138, 6344–6347 (2016).
Lerch, M. M. et al. Tailoring photoisomerization pathways in donor-acceptor Stenhouse adducts: the role of the hydroxy group. J. Phys. Chem. A 122, 955–964 (2018).
Lerch, M. M. et al. Solvent effects on the actinic step of donor-acceptor Stenhouse adduct photoswitching. Angew. Chem. Int. Ed. 57, 8063–8068 (2018).
Shiraishi, Y. et al. Thermal isomerization of spiropyran to merocyanine in aqueous media and its application to colorimetric temperature indication. Phys. Chem. Chem. Phys. 12, 13737–13745 (2010).
Rau, H. Spectroscopic properties of organic Azo compounds. Angew. Chem., Int. Ed. Engl. 12, 224–235 (1973).
Burke, K. et al. Thermosolvatochromism of nitrospiropyran and merocyanine free and bound to cyclodextrin. J. Phys. Chem. B 116, 2483–2491 (2012).
Minkin, V. I. et al. Photo-, thermo-, solvato-, and electrochromic spiroheterocyclic compounds. Chem. Rev. 104, 2751–2776 (2004).
Donato, M. D. et al. Shedding light on the photoisomerization pathway of donor-acceptor Stenhouse adducts. J. Am. Chem. Soc. 139, 15596–15599 (2017).
Wang, D. et al. Orthogonal photo-switching of supramolecular patterned surfaces. Chem. Commun. 54, 3403–3406 (2018).
Jia, S. et al. Photoswitchable molecules in long-wavelength light-responsive drug delivery: from molecular design to applications. Chem. Mater. 30, 2873–2887 (2018).
Laurent, A. D. et al. Using time-dependent density functional theory to probe the nature of donor-acceptor stenhouse adduct photochromes. ChemPhysChem 17, 1846–1851 (2016).
Kohn, W. & Jeu, S. L. Self-consistent equations including exchange and correlation effects. Phys. Rev. 140, A1133–A1138 (1965).
Kresse, G. et al. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Kresse, G. & Jürgen, F. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Perdew, J. P. et al. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Kresse, G. et al. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B. 59, 1758–1775 (1999).
Acknowledgements
We would like to thank D. Vollmer (Max-Planck-Institute for Polymer Research) and C. Zhao (College of Polymer Science and Engineering, Sichuan University) for their kind supports and discussions. We would also acknowledge C.He, W.Zhao (College of Polymer Science and Engineering, Sichuan University), and C.Xie (Max-Planck-Institute for Polymer Research) for their help in the measurements of NMR. We gratefully acknowledge the financial support from the National Key R & D Program (2018YFA0307400) and Opening Project of State Key Laboratory of Polymer Materials Engineering (Sichuan University) (Grant No. sklpme2018-4-28).
Author information
Authors and Affiliations
Contributions
D.W. and Y.Z. conceived the project and designed the experiments. H.Z. contributed to the synthesis of the DASAs. L.Z. and M-S.M.contributed to the DFT calculation of DASAs. J.W. and X.L. designed, prepared and measured the organic solar cells. M-S.W. and W.S. measured and analyzed the NMR data. D.W. and Y.Z. analyzed the data and co-wrote the paper. All authors discussed the results and commented on the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, D., Zhao, L., Zhao, H. et al. Inducing molecular isomerization assisted by water. Commun Chem 2, 118 (2019). https://doi.org/10.1038/s42004-019-0221-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42004-019-0221-5
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.