Abstract
Genome editing technologies such as CRISPR–Cas9 are widely used to establish causal associations between mutations and phenotypes. However, CRISPR–Cas9 is rarely used to analyze promoter regions. The insulin promoter region (approximately 1,000 bp) directs β cell-specific expression of insulin, which in vitro studies show is regulated by ubiquitous, as well as pancreatic, β cell-specific transcription factors. However, we are unaware of any confirmatory in vivo studies. Here, we used CRISPR–Cas9 technology to generate mice with mutations in the promoter regions of the insulin I (Ins1) and II (Ins2) genes. We generated 4 homozygous diabetic mice with 2 distinct mutations in the highly conserved C1 elements in each of the Ins1 and Ins2 promoters (3 deletions and 1 replacement in total). Remarkably, all mice with homozygous or heterozygous mutations in other loci were not diabetic. Thus, the C1 element in mice is required for Ins transcription in vivo.
Similar content being viewed by others
Introduction
The promoter of the gene encoding insulin comprises sequences that are immediately upstream of the transcription initiation site1,2,3,4,5. The insulin gene is specifically expressed in pancreatic β cells4,5,6, and its promoter regulates the rate of transcription in response to physiological regulators7. In the insulin promoter, a region of ~350 bp can direct β cell-specific expression of insulin, which is regulated by ubiquitous as well as pancreatic β cell-specific transcription factors8. In β cells, insulin gene (Ins) expression is controlled by multiple regulatory elements in the basal insulin promoter, which include the A, C and E elements, and three highly conserved enhancer elements in the human insulin promoter: A3 (bases –225 to –220)9,10,11,12, A2–C1 (bases –149 to –116)13 and E1 (bases –111 to–102)14,15. Certain transcription factors that govern β cell differentiation (pancreatic and duodenal homeobox1; Pdx1, neuronal differentiation; NeuroD and v-maf musculoaponeurotic fibrosarcoma oncogene family, protein A; MafA) interact directly with insulin promoter regulatory elements9,10,11,12,13,14,15. Most of these proteins bind to specific sites and form multiprotein transcriptional complexes situated on the insulin promoter16,17. The MafA and A2-binding factors cooperatively activate insulin gene expression18. However, these events were identified through in vitro studies, and we are unaware of comparable in vivo studies.
The use of clustered regularly interspaced short palindromic repeat (CRISPR)–Cas9 systems to manipulate mammalian genomes provides enormous opportunities for curing human diseases19,20,21,22. The nonhomologous end joining (NHEJ) mechanism leads to site-specific repair of the DNA break site, which causes different unpredictable insertions and deletions of various sizes. We took advantage of these mechanisms to develop mice in which the promoter regions of Ins1 and Ins2 were partially deleted. Although these features of the CRISPR–Cas9 system are disadvantageous for the generation of knockout mice with deletion of a single protein-encoding gene, the system is advantageous for evaluating the functions of promoter regions in vivo because multiple mice with different alterations of promoter sequences can be generated concurrently.
Here, we used CRISPR–Cas9 technology to generate mice with partial deletions of the Ins1 and Ins2 promoters, which showed that only homozygous mice with mutations in the highly conserved C1 elements of the Ins1 and Ins2 promoters developed diabetes, indicating the value of genome editing in studying the regulation of insulin synthesis in vivo.
Results
Construction of CRISPR–Cas9 expression vectors
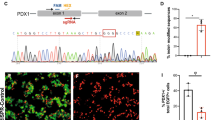
Despite the evolutionarily conserved function of insulin, the promoter and transcribed sequences of Ins are not well conserved between species23,24 (Fig. 1a, Supplementary Fig. 1). However, our detailed analysis of published mammalian promoter sequences revealed that most of the critical promoter sequence elements are well conserved25, particularly bases –151 to –103 of the mouse Ins1 and Ins2 promoters and bases –149 to –102 of the human insulin promoter (Fig. 1a, Supplementary Fig. 1). These sequences comprise the GG2–A2, C1 and E1 elements (Fig. 1a, Supplementary Fig. 1), which was previously known as the rat insulin promoter element 3 (RIPE3), to which cell-specific and ubiquitous factors bind13. For example, NeuroD binds the E1 element to form a heterodimer with the ubiquitously expressed bHLH factor E47. The C1 binding factor, which regulates pancreatic β cell-specific and glucose-regulated transcription of the insulin gene, is a member of the large family of Maf transcription factors16,17,18,23,24,25,26,27,28,29. The C1 element shares an overlapping DNA-binding region with the insulin enhancer element A2, and A2.2, a β cell-specific activator, binds to the overlapping A2 element30. Pdx1 binds to the GG2 element (bases –145 to –141 of the human insulin promoter31) (Supplementary Fig. 1).
a Structures and sequences of the human and mouse insulin promoters and construction of the pX330-1st/2nd gRNA vectors. Bases –170 to –147 and –145 to –104 of the promoters of mouse Ins1 and Ins2 are identical, and bases –149 to –147 (AGG) and –114 to –112 (TGG) were used as the PAM sequences of the guide RNAs (gRNAs) for the CRISPR–Cas9 system. The 20-bp double-stranded DNAs (dsDNAs) derived from positions –169 to –150 and –134 to –115 of the Ins1/2 promoters were inserted into pX330, and the resultant plasmids were designated pX330-1st gRNA and pX330-2nd gRNA, respectively. b Generation of mice with mutated insulin promoters. The mice with the four types of deletions and 1 replacement in the Ins1 promoter and the six types of deletions in the Ins2 promoter were generated. Sequences shaded in gray were deleted, and the yellow sequence indicates the replacement. c Immunohistochemical analysis of pancreatic islets (insulin, glucagon, and DAPI staining) from wild-type mice. Scale bars = 100 µm. d Blood glucose levels of fed wild-type (n = 6) and homozygous mice that had deletions or a replacement in only the Ins1 promoter or a deletion in only the Ins2 promoter (n = 6 each) at 12 weeks of age. e Blood glucose levels of fed wild-type (n = 6) homozygous mice that had any mutation in the Ins1 promoter with ≤3-base deletions in the Ins2 promoter (1C623, 1C62C1, 1C622, 1C621, 1Cm23, 1Cm2C1, 1Cm22, 1Cm21, 1C123, 1C12C1, 1C122, 1C121, 1223, 122C1, 1222, 1221, 1123, 112C1, 1122, and 1121; n = 6 each) and ≤3-base deletions in the Ins1 promoter with any deletion in the Ins2 promoter (1C12C3, 1C12C6, 122C3, 122C6,112C3, and 112C6; n = 6 each) at 12 weeks of age. f Blood glucose levels of fed wild-type (n = 6) homozygous mice with >3-base deletions in the Ins1 and Ins2 promoters (1C62C3, 1C62C6, 1Cm2C3, and 1Cm2C6; n = 6 each) at 12 weeks of age. *p < 0.01 control vs. other groups. The error bars represent the standard error.
We observed that bases –170 to –147 and –145 to –104 of the promoters of mouse Ins1 and Ins2 are identical and that bases –149 to –147 (AGG) and bases –114 to –112 (TGG) serve as protospacer adjacent motif (PAM) sequences in the guide RNA (gRNA) of the CRISPR–Cas9 system19,20,21. Therefore, we designed two gRNAs to target the promoter regions of mouse Ins1 and Ins2 (Fig. 1a). We used the pX330 vectors described in the “Methods” to express these gRNAs and Cas9 in mouse zygotes32.
Generation of mice with insulin promoter mutations
To generate mice with insulin promoter mutations, pX330-1st/2nd gRNA vectors were comicroinjected into the pronuclei of 310 one-cell-stage embryos obtained from C57BL/6J mice. Morphological abnormalities were undetectable in 252 (81.3%) of the embryos immediately after microinjection, which were consecutively transferred into the oviducts of pseudopregnant recipient the Institute for Cancer Research (ICR) mice to produce 86 neonates. Sequence analysis revealed that 23 mice had partial deletions and one replacement in either the Ins1 or Ins2 promoter or in both promoters and that 63 mice had normal or mosaic patterns. Mosaicism may be explained by mutations that occurred after the first or later embryonic divisions. Furthermore, Cas9 PCR products were not detected in 23 mice with deletions in either the Ins1 or Ins2 promoter or in both.
Among the 23 mice, we detected 5 and 6 types of mutations in the C1 elements or in the sequences bordering the C1 elements of the Ins1 and Ins2 promoters, respectively (Fig. 1b). To evaluate the importance of these promoter regions in vivo, the 11 mice with these mutations were crossed with wild-type C57BL/6J mice. Forty-one heterozygous mice with deletions in the Ins1 promoter or Ins2 promoter or in both were not diabetic (Supplementary Figs. 2, 3, Supplementary Table 1). The F1 mice were intercrossed to obtain homozygous F2 mice that had deletions in only the Ins1 promoter or the Ins2 promoter. These mice were not diabetic (Fig. 1d).
Thirty homozygous mice with mutations in both the Ins1 and Ins2 promoters were obtained by crossing the F3 and F4 mice (Supplementary Table 2). Homozygous mice with ≤3-base deletions in the Ins1 promoter and any deletion in the Ins2 promoter were not diabetic (Fig. 1e). Homozygous mice with any deletion in the Ins1 promoter and ≤3-base deletions in the Ins2 promoter were not diabetic (Fig. 1e). The most significant outcome of these studies was that the four types of mice with homozygous genotypes (1C62C6, 1Cm2C6, 1C62C3, and 1Cm2C3 mice) with >3-base deletions in both the Ins1 and Ins2 promoters were diabetic (Fig. 1f).
Quantitative RT-PCR analysis of mRNAs extracted from the pancreatic islets of the 4 types of mice (fasted) with diabetes showed that the mRNA levels of Ins1 and Ins2 were decreased compared with those of normal mice (fasted) (Fig. 2a, b). There were no significant differences in the levels of the Pdx1, NeuroD, and MafA mRNAs (Supplementary Fig. 4). Moreover, insulin promoter activity was significantly decreased for the two types of Ins1 and Ins2 promoters (Fig. 2c–f). MafA, Pdx1, and NeuroD synergistically activated the wild-type promoters. Mutations in C1 result in a sharp decrease in synergistic activation by MafA, Pdx1, and NeuroD. In particular, MafA did not induce the activation of promoters with C1 mutations (Fig. 2d, f).
a, b qRT-PCR analysis of Ins1 (a) and Ins2 (b) in the pancreatic islets of mice with mutations of the insulin promoter. Pancreatic islets (purity > 95%) of wild-type mice served as a control. The data are expressed as the target gene-to-Gapdh ratio, and that of the control cells was arbitrarily defined as 1 (n = 8). **p < 0.05 control vs. other groups c Mouse Ins1 promoter-luciferase plasmid containing ~1000 bp of the 5′-flanking sequences of the wild-type or mutated Ins1 promoter region was transfected into βTC6 cells (β cell line). Forty-eight hours after transfection, cells were harvested and assayed (n = 3). *p < 0.01 control vs. other groups d Mouse Ins1 promoter-luciferase plasmid containing approximately 1000 bp of the 5′-flanking sequences of the wild-type or mutated Ins1 promoter region was cotransfected with combinations of the expression plasmids for MafA, Pdx1, and NeuroD into βTC6 cells (β cell line). Forty-eight hours after transfection, cells were harvested and assayed (n = 3). N.S. no significant differences. e Mouse Ins2 promoter-luciferase plasmid containing approximately 1000-bp of the 5′-flanking sequences of the wild-type or Ins2 deletion-containing promoter region was transfected into βTC6 cells (β cell line). Forty-eight hours after transfection, cells were harvested and assayed (n = 3). *p < 0.01 control vs. other groups f Mouse Ins2 promoter-luciferase plasmid containing ~1000 bp of the 5′-flanking sequences of the wild-type or mutated Ins2 promoter region was cotransfected with different combinations of the expression plasmids for MafA, Pdx1, and NeuroD into βTC6 cells (β cell line). Forty-eight hours after transfection, cells were harvested and assayed (n = 3). N.S. no significant differences. The error bars represent the standard error.
Phenotypes of 1C62C6 mice
1C62C6 mice (Fig. 3a) have deletions in the C1 elements of the Ins1 and Ins2 promoters. Immunohistochemical analysis of the pancreatic tissue of 1C62C6 mice showed reduced insulin expression in islets (Fig. 3b) compared with that in islets of wild-type C57BL/6 mice (Fig. 1c). Fasting C-peptide levels were significantly reduced in 1C62C6 mice at 12 weeks of age compared with those in wild-type mice (Fig. 3c). Blood glucose levels of fasted and fed 1C62C6 mice at 6 and 12 weeks of age were elevated compared with those of wild-type C57BL/6 mice (Fig. 3d–g). Ins1 and Ins2 mRNA levels in pancreatic islets of 1C62C6 mice (fasted) were significantly decreased compared with those of wild-type C57BL/6 mice (fasted) (Fig. 3h, i). Glucose tolerance tests showed significantly elevated glucose levels in male and female 1C62C6 mice (Fig. 3j). The 1C62C6 mice lost body weight at 6 and 12 weeks (Fig. 3k). The fertility of 1C62C6 female mice was significantly reduced (40.0%).
a Sequences of the Ins1 and Ins2 promoters in 1C62C6 mice. b Immunohistochemical analysis of pancreatic islets (insulin, glucagon, and DAPI staining) in 1C62C6 mice. Scale bars = 100 µm. c Fasting C-peptide levels of wild-type (n = 8) and 1C62C6 mice (n = 8) at 12 weeks of age. d, e Fasting blood glucose concentrations of wild-type (n = 6) and 1C62C6 mice (n = 6) at 6 and 12 weeks of age ((d): male, (e): female). f, g Blood glucose levels of fed wild-type (n = 6) and 1C62C6 mice (n = 6) at 6 and 12 weeks of age ((f): male, (g): female). h, i qRT-PCR analysis of Ins1 and Ins2 in pancreatic islets of 1C62C6 mice. Pancreatic islets (purity > 95%) of wild-type mice served as a control ((h): male, (i): female). The data are expressed as the gene-to-Gapdh ratio; that of the control cells was arbitrarily defined as 1 (n = 8). j Intraperitoneal glucose tolerance testing of wild-type (blue: n = 6) and 1C62C6 mice (white: n = 6) at 12 weeks of age. k Body weights of wild-type (n = 6) and 1C62C6 mice (n = 6) at 6 and 12 weeks of age. The error bars represent the standard error. *p < 0.01, **p < 0.05.
Phenotypes of 1Cm2C6, 1C62C3, and 1Cm2C3 mice
Analyses of the pancreatic tissues of 1Cm2C6, 1C62C3, and 1Cm2C3 mice revealed similar alterations as those found in 1C62C6 mice (Figs. 4a, b, 5a, b, 6a, b). The fasting C-peptide levels, fasting and fed blood glucose levels at 6 and 12 weeks of age, fasting Ins1 mRNA levels in pancreatic islets, glucose tolerance test results, and body weights at 6 and 12 weeks of 1Cm2C6, 1C62C3, and 1Cm2C3 mice were similar to those of 1C62C6 mice and significantly different from those of wild-type C57BL/6 mice (Figs. 4c–g, j–k, 5c–g, j–k, 6c–g, j–k). In contrast, Ins2 mRNA levels in the pancreatic islets of 1C62C3 and 1Cm2C3 mice were relatively but not significantly decreased compared with those of wild-type C57BL/6 mice (Figs. 5h, i, 6h, i), while Ins2 mRNA levels in the pancreatic islets of 1C62C6 mice were significantly decreased compared with those of wild-type C57BL/6 mice (Fig. 4h, i). The fertility of 1Cm2C6, 1C62C3, and 1Cm2C3 female mice was significantly reduced and was similar to that of 1C62C6 female mice.
a Sequences of the Ins1 and Ins2 promoters in 1Cm2C6 mice. b Immunohistochemical analysis of pancreatic islets (insulin, glucagon, and DAPI staining) in 1Cm2C6 mice. Scale bars = 100 µm. c Fasting C-peptide levels of wild-type (n = 8) and 1Cm2C6 mice (n = 8) at 12 weeks of age. d, e Fasting blood glucose concentrations of wild-type (n = 6) and 1Cm2C6 mice (n = 6) at 6 and 12 weeks of age ((d): male, (e): female). f, g Blood glucose levels of fed wild-type (n = 6) and 1Cm2C6 mice (n = 6) at 6 and 12 weeks of age ((f): male, (g): female). h, i qRT-PCR analysis of Ins1 and Ins2 in pancreatic islets of 1Cm2C6 mice. Pancreatic islets (purity > 95%) of wild-type mice served as a control ((h): male, (i): female). The data are expressed as the gene-to-Gapdh ratio; that of the control cells was arbitrarily defined as 1 (n = 8). j Intraperitoneal glucose tolerance testing of wild-type (blue: n = 6) and 1Cm2C6 mice (white: n = 6) at 12 weeks of age. k Body weights of wild-type (n = 6) and 1Cm2C6 mice (n = 6) at 6 and 12 weeks of age. The error bars represent the standard error. *p < 0.01, **p < 0.05.
a Sequences of the Ins1 and Ins2 promoters in 1C62C3 mice. b Immunohistochemical analysis of pancreatic islets (insulin, glucagon, and DAPI staining) in 1C62C3 mice. Scale bars = 100 µm. c Fasting C-peptide levels of wild-type (n = 8) and 1C62C3 mice (n = 8) at 12 weeks of age. d, e Blood glucose concentrations of fasting wild-type (n = 6) and 1C62C3 mice (n = 6) at 6 and 12 weeks of age ((d): male, (e): female). f, g Blood glucose levels of fed of wild-type (n = 6) and 1C62C3 mice (n = 6) at 6 and 12 weeks of age ((f): male, (g): female). h, i qRT-PCR analysis of Ins1 and Ins2 in pancreatic islets of 1C62C3 mice. Pancreatic islets (purity > 95%) of wild-type mice served as a control ((h): male, (i): female). The data are expressed as the gene-to-Gapdh ratio; that of the control cells was arbitrarily defined as 1 (n = 8). j Intraperitoneal glucose tolerance testing of wild-type (blue: n = 6) and 1C62C3 mice (white: n = 6) at 12 weeks of age. k Body weights of wild-type (n = 6) and 1C62C3 mice (n = 6) at 6 and 12 weeks of age. The error bars represent the standard error. *p < 0.01, **p < 0.05.
a Sequences of the Ins1 and Ins2 promoters in 1Cm2C3 mice. b Immunohistochemical analysis of pancreatic islets (insulin, glucagon, and DAPI staining) in 1Cm2C3 mice. Scale bars = 100 µm. c Fasting C-peptide levels of wild-type (n = 8) and 1Cm2C3 mice (n = 8) at 12 weeks of age. d, e Blood glucose concentrations of fasting wild-type (n = 6) and 1Cm2C3 mice (n = 6) at 6 and 12 weeks of age ((d): male, (e): female). f and g Blood glucose levels of fed wild-type (n = 6) and 1Cm2C3 mice (n = 6) at 6 and 12 weeks of age ((f): male, (g): female). h, i qRT-PCR analysis of Ins1 and Ins2 in pancreatic islets of 1Cm2C3 mice. Pancreatic islets (purity > 95%) of wild-type mice served as a control ((h): male, (i): female). The data are expressed as the gene-to-Gapdh ratio; that of the control cells was arbitrarily defined as 1 (n = 8). j Intraperitoneal glucose tolerance testing of wild-type (blue: n = 6) and 1Cm2C3 mice (white: n = 6) at 12 weeks of age. k Body weights of wild-type (n = 6) and 1Cm2C3 mice (n = 6) at 6 and 12 weeks of age. The error bars represent the standard error. *p < 0.01, **p < 0.05.
Discussion
Here, we used the CRISPR–Cas9 system to evaluate the functions of the promoter regions of Ins1 and Ins2 in vivo. We generated mice with 5 and 6 distinct deletions in the Ins1 and Ins2 promoters, respectively (Fig. 1b). Homozygous mice with deletions of ≤3 bases in the Ins1 promoter with any deletion in the Ins2 promoter (1C12any, 122any, and 112any) or Ins1 promoter and with deletions ≤3 bases of the Ins2 promoter (1any23, 1any2C1, 1any22, and 1any21) were not diabetic (Fig. 1e, Supplementary Table 2). The 1–3 deleted bases were located between the C1 and E1 elements or within the sequences bordering the C1 element (Fig. 1b). Therefore, deletions of these 1–3 bases in the Ins1/2 promoters did not affect insulin promoter activity. The other homozygous mice with deletions in both the Ins1 and Ins2 promoters were diabetic (Figs. 3–6). These data suggest that these sequences are important for insulin promoter activity in vivo. In vitro studies show that the C1 elements of the Ins1 and Ins2 promoters are required for Ins expression13. Furthermore, these sequences contain cell-specific and ubiquitous motifs that confer enhancer activity13, and proteins that bind to these sequences, such as MafA, show synergistic activity to activate Ins expression17,18,31. Our results provide compelling evidence that the sequences of the C1 elements in the Ins1 and Ins2 promoters are required for Ins expression in vivo.
Heterozygous mice with deletions in either the Ins1 promoter or Ins2 promoter or in both were not diabetic (Supplementary Figs. 2, 3, Supplementary Table 1). Moreover, homozygous mice with deletions in only the Ins1 promoter or the Ins2 promoter were not diabetic (Fig. 1d). It has been reported that mice with single homozygous null mutations of Ins1 or Ins2 were not diabetic because of compensatory responses, and there was a dramatic increase in Ins1 transcripts in the Ins2−/− mice33. Our data also showed that compensatory transcription of a functional insulin gene in homozygous mice with a deletion in the Ins1 or Ins2 promoter (Supplementary Fig. 3c, d) did not lead to diabetes.
In conclusion, we used the CRISPR–Cas9 system to analyze the insulin promoter by taking advantage of the site-specific repair mediated by NHEJ at the DNA break site, which leads to different unpredictable insertions or deletions of various sizes. Our findings show, to the best of our knowledge and for the first time, that the C1 elements of the insulin promoter in mice are required for its activity in vivo. More importantly, the CRISPR–Cas9 technique will provide a tool to generate knockout mice that can be used to evaluate promoter regions.
Methods
Generation of mice with insulin promoter mutations
To construct two Cas9–single-guide RNA (sgRNA) expression vectors, deoxyoligonucleotides (Fig. 1a: 1st gRNA and 2nd gRNA) were inserted into pX330 vectors34 (Addgene, Watertown, MA). Female C57BL/6J mice were injected with pregnant mare serum gonadotropin and human CG (hCG) at 48-h intervals and were then mated with male C57BL/6J mice. The fertilized one-cell embryos were collected from the oviducts. Subsequently, 5 ng/μL pX330-1st/2nd gRNA vectors were injected into the pronuclei of these one-cell embryos, which were transferred into pseudopregnant Institute of Cancer Research (ICR) mice. F0 mice were genotyped to detect the presence of mutations in the Ins1/2 promoters. F0 mice were checked for the Cas9 transgene and for off-target effects. F0 mice were mated with C57BL/6N mice to obtain the F1 offspring.
The mutant mice and littermate wild-type mice were maintained on a C57BL/6 background. Mice were housed under a 12:12-h light/dark cycle in a room with controlled temperature and humidity. Food and water were provided without restrictions. The Institutional Animal Care and Use Committee of the University of the Ryukyus and the University of Tsukuba approved the animal studies.
Construction of insulin promoter-luciferase plasmids
Wild-type or mutated insulin promoter cDNAs containing ~1000 bp of the 5′-flanking sequences of the Ins1/2 promoter regions were amplified using PCR with appropriate linker-containing primers, which were then used to replace the promoter region of the Rat Ins2 promoter-reporter (luciferase) plasmid35 using a ligation kit (TaKaRa, Tokyo, Japan).
Gene transfection and luciferase assay
Mouse Ins1/2 promoter-reporter (luciferase) plasmids (1.0 μg) containing ~1000 bp of the 5′-flanking sequences of the wild-type or mutated Ins1/2 promoter region were cotransfected with a combination of plasmids (MafA, Pdx1, and NeuroD, 100 ng of each plasmid) into βTC6 cells (β cell line) (ATCC, Manassas, VA) using Lipofectamine (Thermo Fisher Scientific, Tokyo, Japan) according to the conditions recommended by the manufacturer. Forty-eight hours after transfection, the cells were harvested and assayed (Promega, Madison, WI).
Immunohistochemistry
Cells were fixed with 4% paraformaldehyde in PBS. After blocking with 20% AquaBlock (EastCoast Bio, North Berwick, ME, USA) for 30 min at room temperature, the cells were incubated overnight at 4 °C with a guinea pig anti-insulin antibody (1:100; Abcam, Tokyo, Japan) or a mouse anti-glucagon antibody (1:200; Merck, Tokyo, Japan) and then for 1 h at room temperature with FITC-conjugated anti-guinea pig IgG (1:250; Abcam) or TRITC-conjugated anti-mouse IgG (1:250; Merck). Sections were treated with mounting medium to detect the fluorescence emitted by DAPI (Vector Laboratories, Peterborough, UK).
Isolation of mouse pancreatic islets
Islets were isolated from the pancreata of normal and genome-edited mice36,37. For islet isolation, the common bile duct was cannulated and injected with cold Hank’s Balanced Salt Solution (HBSS, Thermo Fisher Scientific) containing 1.5 mg/ml collagenase (Roche Boehringer Mannheim, Indianapolis, IN). The islets were separated with a Histopaque 1077 (Merck) density gradient, hand-picked using a dissecting microscope to ensure pure islet preparation and used immediately.
Quantitative RT-PCR
Total RNA was extracted from the isolated islets using an RNeasy Mini Kit (Qiagen, Tokyo, Japan). After quantifying the RNA using spectrophotometry, 2.5 µg of RNA was heated at 85 °C for 3 min and then reverse-transcribed in a 25-µl reaction mixture containing 200 units of Superscript II RNase H-RT (Thermo Fisher Scientific), 50 ng random hexamers (Thermo Fisher Scientific), 160 µmol/l dNTP and 10 nmol/l dithiothreitol. The reaction mixture was incubated for 10 min at 25 °C, 60 min at 42 °C and 10 min at 95 °C.
The mRNAs were quantitated using the TaqMan real-time PCR system according to the manufacturer’s instructions (Applied Biosystems, Foster City, CA, USA). PCR was performed for 40 cycles, and the reactions were incubated for 2 min at 50 °C and 10 min at 95 °C for the initial steps. In each cycle, denaturation was performed for 15 s at 95 °C, and annealing/extension was performed for 1 min at 60 °C. PCR was performed in 20 µl of solution using cDNAs synthesized from 1.11 ng of total RNA. The amount of mRNA was normalized by dividing the amount of the mRNA of interest by that of Gapdh mRNA. Primers specific for mouse Ins1, Ins2, Pdx1, NeuroD, MafA, and Gapdh were purchased as Assays-on-Demand Gene Expression Products (Applied Biosystems).
ELISA
The mice were fasted overnight. C-peptide was measured with a C-peptide ELISA kit (FUJIFILM Wako Shibayagi, Gunma, Japan).
Intraperitoneal glucose tolerance testing
Intraperitoneal glucose tolerance testing was performed on 6- and 12-week-old mice. The mice were fasted overnight, after which glucose (2.0 g/kg body weight) was intraperitoneally injected. The blood glucose levels were measured before injection and at 5, 30, 60, 90, and 120 min after injection.
Statistics and reproducibility
Error bars indicate standard error of at least triplicate measurements. Student’s t test was used to compare two samples from independent groups using Microsoft Excel. Repeated measures ANOVA was performed to compare data among groups. The differences between the groups were considered to be significant when p < 0.05.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
Raw data used to generate the figures can be found in Supplementary Data 1. Additional data that support the findings of this study are available from the corresponding author, H.N., upon reasonable request.
References
Clark, A. R. & Docherty, K. How is the developmental timing and tissue-specificity of insulin gene expression controlled? J. Endocrinol. 136, 187–190 (1993).
Docherty, K. 1992 R.D. Lawrence Lecture. The regulation of insulin gene expression. Diabet. Med. 9, 792–798 (1992).
Docherty, K. & Clark, A. R. Nutrient regulation of insulin gene expression. FASEB J. 8, 20–27 (1994).
Edlund, T., Walker, M. D., Barr, P. J. & Rutter, W. J. Cell-specific expression of the rat insulin gene: evidence for role of two distinct 5’ flanking elements. Science 230, 912–916 (1985).
Walker, M. D., Edlund, T., Boulet, A. M. & Rutter, W. J. Cell-specific expression controlled by the 5’-flanking region of insulin and chymotrypsin genes. Nature 306, 557–561 (1983).
Hanahan, D. Heritable formation of pancreatic beta-cell tumours in transgenic mice expressing recombinant insulin/simian virus 40 oncogenes. Nature 315, 115–122 (1985).
German, M. S., Moss, L. G. & Rutter, W. J. Regulation of insulin gene expression by glucose and calcium in transfected primary islet cultures. J. Biol. Chem. 265, 22063–22066 (1990).
Melloul, D., Marshak, S. & Cerasi, E. Regulation of insulin gene transcription. Diabetologia 45, 309–326 (2002).
German, M. S., Moss, L. G., Wang, J. & Rutter, W. J. The insulin and islet amyloid polypeptide genes contain similar cell-specific promoter elements that bind identical beta-cell nuclear complexes. Mol. Cell Biol. 12, 1777–1788 (1992).
Peshavaria, M. et al. XIHbox 8, an endoderm-specific Xenopus homeodomain protein, is closely related to a mammalian insulin gene transcription factor. Mol. Endocrinol. 8, 806–816 (1994).
Ohlsson, H., Thor, S. & Edlund, T. Novel insulin promoter- and enhancer-binding proteins that discriminate between pancreatic alpha- and beta-cells. Mol. Endocrinol. 5, 897–904 (1991).
Boam, D. S. & Docherty, K. A tissue-specific nuclear factor binds to multiple sites in the human insulin-gene enhancer. Biochem J. 264, 233–239 (1989).
Shieh, S. Y. & Tsai, M. J. Cell-specific and ubiquitous factors are responsible for the enhancer activity of the rat insulin II gene. J. Biol. Chem. 266, 16708–16714 (1991).
Karlsson, O., Edlund, T., Moss, J. B., Rutter, W. J. & Walker, M. D. A mutational analysis of the insulin gene transcription control region: expression in beta cells is dependent on two related sequences within the enhancer. Proc. Natl Acad. Sci. USA 84, 8819–8823 (1987).
Crowe, D. T. & Tsai, M. J. Mutagenesis of the rat insulin II 5’-flanking region defines sequences important for expression in HIT cells. Mol. Cell Biol. 9, 1784–1789 (1989).
Ohneda, K., Ee, H. & German, M. Regulation of insulin gene transcription. Semin Cell Dev. Biol. 11, 227–233 (2000).
Qiu, Y., Guo, M., Huang, S. & Stein, R. Insulin gene transcription is mediated by interactions between the p300 coactivator and PDX-1, BETA2, and E47. Mol. Cell Biol. 22, 412–420 (2002).
Nishimura, W., Salameh, T., Kondo, T. & Sharma, A. Regulation of insulin gene expression by overlapping DNA-binding elements. Biochem J. 392, 181–189 (2005).
Doudna, J. A. & Charpentier, E. Genome editing. The new frontier of genome engineering with CRISPR-Cas9. Science 346, 1258096 (2014).
Hsu, P. D., Lander, E. S. & Zhang, F. Development and applications of CRISPR-Cas9 for genome engineering. Cell 157, 1262–1278 (2014).
Cox, D. B., Platt, R. J. & Zhang, F. Therapeutic genome editing: prospects and challenges. Nat. Med. 21, 121–131 (2015).
Pickar-Oliver, A. & Gersbach, C. A. The next generation of CRISPR-Cas technologies and applications. Nat. Rev. Mol. Cell Biol. https://doi.org/10.1038/s41580-019-0131-5 (2019).
Lomedico, P. et al. The structure and evolution of the two nonallelic rat preproinsulin genes. Cell 18, 545–558 (1979).
Steiner, D. F., Chan, S. J., Welsh, J. M. & Kwok, S. C. Structure and evolution of the insulin gene. Annu. Rev. Genet. 19, 463–484 (1985).
German, M. et al. The insulin gene promoter. A simplified nomenclature. Diabetes 44, 1002–1004 (1995).
Kataoka, K. et al. MafA is a glucose-regulated and pancreatic beta-cell-specific transcriptional activator for the insulin gene. J. Biol. Chem. 277, 49903–49910 (2002).
Olbrot, M., Rud, J., Moss, L. G. & Sharma, A. Identification of beta-cell-specific insulin gene transcription factor RIPE3b1 as mammalian MafA. Proc. Natl Acad. Sci. USA 99, 6737–6742 (2002).
Matsuoka, T. A. et al. Members of the large Maf transcription family regulate insulin gene transcription in islet beta cells. Mol. Cell Biol. 23, 6049–6062 (2003).
Zhao, L. et al. The islet beta cell-enriched MafA activator is a key regulator of insulin gene transcription. J. Biol. Chem. 280, 11887–11894 (2005).
Harrington, R. H. & Sharma, A. Transcription factors recognizing overlapping C1-A2 binding sites positively regulate insulin gene expression. J. Biol. Chem. 276, 104–113 (2001).
Le Lay, J., Matsuoka, T. A., Henderson, E. & Stein, R. Identification of a novel PDX-1 binding site in the human insulin gene enhancer. J. Biol. Chem. 279, 22228–22235 (2004).
Cong, L. et al. Multiplex genome engineering using CRISPR/Cas systems. Science 339, 819–823 (2013).
Leroux, L. et al. Compensatory responses in mice carrying a null mutation for Ins1 or Ins2. Diabetes 50, S150–S153 (2001).
Mashiko, D. et al. Generation of mutant mice by pronuclear injection of circular plasmid expressing Cas9 and single guided RNA. Sci. Rep. 3, 3355 (2013).
Noguchi, H., Kaneto, H., Weir, G. C. & Bonner-Weir, S. PDX-1 protein containing its own antennapedia-like protein transduction domain can transduce pancreatic duct and islet cells. Diabetes 52, 1732–1737 (2003).
Noguchi, H. et al. A new cell-permeable peptide allows successful allogeneic islet transplantation in mice. Nat. Med. 10, 305–309 (2004).
Noguchi, H. et al. RCAN-11R peptide provides immunosuppression for fully mismatched islet allografts in mice. Sci. Rep. 7, 3043 (2017).
Acknowledgements
We thank Naomi Kakazu (University of the Ryukyus) for administrative support and Maki Higa, Yuki Kawahira, Ikue Honda, Yuka Onishi, Saori Adaniya, Youichi Toyokawa (University of the Ryukyus), Seiya Mizuno, Yoko Tanimoto, Satoru Takahashi, and Fumihiro Sugiyama (University of Tsukuba) for technical support. This work was supported in part by JSPS KAKENHI Grant Numbers JP17H00769 and JP19K09051 and Okinawa Science and Technology Innovation System Construction Project.
Author information
Authors and Affiliations
Contributions
H.N. designed the experiments and analyzed the data. H.N. carried out most of the experimental work with the help of C.M-S., Y.N., and T.K. I.S. and M.W. provided materials and participated in discussion. H.N. wrote the manuscript. All authors discussed and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Noguchi, H., Miyagi-Shiohira, C., Nakashima, Y. et al. Mutations in the C1 element of the insulin promoter lead to diabetic phenotypes in homozygous mice. Commun Biol 3, 309 (2020). https://doi.org/10.1038/s42003-020-1040-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-020-1040-z
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.