Abstract
Bacterial strains of the phylum Planctomycetes occur ubiquitously, but are often found on surfaces of aquatic phototrophs, e.g. alga. Despite slower growth, planctomycetes are not outcompeted by faster-growing bacteria in biofilms on such surfaces; however, strategies allowing them to compensate for slower growth have not yet been investigated. Here, we identified stieleriacines, a class of N-acylated tyrosines produced by the novel planctomycete Stieleria maiorica Mal15T, and analysed their effects on growth of the producing strain and bacterial species likely co-occurring with strain Mal15T. Stieleriacines reduced the lag phase of Mal15T and either stimulated or inhibited biofilm formation of two bacterial competitors, indicating that Mal15T employs stieleriacines to specifically alter microbial biofilm composition. The genetic organisation of the putative stieleriacine biosynthetic cluster in strain Mal15T points towards a functional link of stieleriacine biosynthesis to exopolysaccharide-associated protein sorting and biofilm formation.
Similar content being viewed by others
Introduction
From a heterotrophic bacterial perspective, the vastness of the sea represents a hostile oligotrophic ‘desert’. In contrast, surfaces of marine macroscopic phototrophs are nutrient-rich ‘oases’, densely packed with all sorts of alluring organic compounds, which can serve as nutrient sources. In aquatic environments, such biotic surfaces represent desirable ecological niches and are thus rapidly occupied by bacterial biofilms1,2. Members of the ‘Roseobacter group’ are particularly successful in such habitats3. For example, Phaeobacter inhibens employs N-acyl homoserine lactones (AHLs) as quorum sensing signals to initiate biofilm formation4. In P. inhibens, AHLs trigger the expression of the gene encoding AHL synthase along with other genes that promote biofilm formation. This positive feedback loop is often associated with the production of bioactive small molecules, e.g., the antibiotic tropodithietic acid (TDA) by P. inhibens5,6.
While P. inhibens and other proteobacteria are well known to dominate biotic surfaces, it was found that members of the phylum Planctomycetes can sometimes also be the dominating taxon7. Species belonging to the family Pirellulaceae, including the marine model planctomycete Rhodopirellula baltica, switch between different lifestyles, in which they either live as motile free-swimming cells or attach to surfaces8. Such a complex lifecycle requires control at additional stages beyond canonical transcription factor-based regulation of gene expression. Members of the family Pirellulaceae might thus rely on similar regulatory mechanisms of secreting signalling molecules for enabling cell-to-cell communication as the Roseobacter clade and many other microorganisms9. Large planctomycetal genomes of up to 12.4 Mb and high numbers of predicted clusters involved in small molecule production are in line with the assumed portfolio of bioactive compounds with potential regulatory activities, in addition to two component systems and extracytoplasmic function sigma factors10. Although planctomycetes grow rather slowly compared to competing microorganisms occupying the same ecological niche, they are not outcompeted by their natural competitors. On the contrary, planctomycetes can even account for up to 70% of the bacterial community in certain habitats7. The deficit in growth rate is suggested to be compensated by the production of small molecules with antimicrobial properties, while the chemical nature of such molecules remains elusive11. The current knowledge gap mainly results from the insufficient number of planctomycetes available as axenic cultures7. Recently, we developed an isolation pipeline to obtain novel planctomycetal strains in axenic culture. Basis for the isolation of 79 novel strains was an optimised formulation of cultivation media, supplemented with a blend of carefully titrated antibiotics and fungicides10. In Mal15T, one of the strains isolated by this strategy, we detected production of novel compounds beloning to the class of N-acylated tyrosine derivatives, which might have regulatory activities in this strain. In order to validate our working hypothesis, we performed assays with purified stieleriacine for studying its role on intraspecies and interspecies communication. Additionally, we inferred a putative biosynthetic pathway based on the genome sequence of strain Mal15T obtained during a detailed characterisation of the novel strain.
Results
Description of Stieleria gen. nov
Stieleria (Stie.le’ri.a’ N.L. fem. n. Stieleria named in honor of Anja Heuer, née Stieler, an extraordinary skilled German technician at the Leibniz Institute DSMZ, who played a key role in the cultivation of literally hundreds of novel planctomycetal strains). The round to pear-shaped cells with a smooth cell surface form rosettes or short chains. Cells reproduce by polar budding. In liquid culture, they produce an extracellular matrix compound which interconnects cells in aggregates. Daughter cells are motile, while mother cells are non‑motile. The lifestyle is heterotrophic, obligatory aerobic and mesophilic. The genus belongs to the phylum Planctomycetes, class Planctomycetia, order Pirellulales, family Pirellulaceae. The type species is Stieleria maiorica.
Description of Stieleria maiorica sp. nov
Stieleria maiorica (ma’i.ori.ca’ N.L. fem. adj. maiorica, pertaining to the island Mallorca, Spain, on which the type strain was isolated). In addition to the features described above the species exhibits the following properties. Colonies are pink-coloured on solid medium. Cells are 1.9 ± 0.2 x 1.4 ± 0.2 µm in size. Motile daughter cells originate through budding from sessile mother cells. Gram staining delivers no clear results. The oxidase assay was negative while the catalase assay was positive. The organism can degrade a wide range of carbon sources. In particular, strong signals were observed for N-acetyl-d‑galactosamine, N-acetyl-d-glucosamine, l-arabinose, d-cellobiose, l‑fucose, d-fructose, d-galactose, gentiobiose, α-d-glucose, d-gluconic acid, glucuronamide, d-glucuronic acid, α-d-lactose, lactulose, d‑mannose, d-melibiose, β-methyl-d‑glucoside, d-raffinose, l-rhamnose, sucrose, d-trehalose, turanose and d-psicose, while for the carbon sources acetic acid, dextrin, d‑galactonic acid lactone, d-glucose-6‑phosphate, α-ketoglutaric acid, maltose, d-mannitol only weak signals were detected. The enzyme repertoire includes alkaline phosphatase, esterase (C4), esterase lipase (C8), leucine arylamidase, valine arylamidase, acid phosphatase, naphthol-AS-BI‑phosphohydrolase. Growth of the type strain occurs between pH 5.5 and 9.0 with an optimum at pH 7.5. The optimal growth temperature of the type strain was determined to be 35 °C, but cells could grow in a range from 11 °C up to at least 37 °C. The complete chromosome of the type strain comprises 9,894,293 bp, with a G + C content of 59.3%. The type strain is Mal15T (DSM 100215T = LMG 29790T, synonym Malle15), which was isolated from seawater sediment on Mallorca island, Spain.
Identification of stieleriacines in cultures of strain Mal15T
Given our working hypothesis that planctomycetes are a promising source for novel bioactive small molecules, we continuously extend the current collection of axenic cultures from this phylum10. Here, we analysed the novel biofilm-forming strain Mal15T (DSM 100215T = LMG 29790T) isolated from sediments on Mallorca island in the Mediterranean Sea. Our phylogenetic analysis suggests that strain Mal15T belongs to a novel genus and species in the family Pirellulaceae. Hence, we propose the name Stieleria maiorica gen. nov., sp. nov. for the novel isolate. A detailed characterisation of the novel strain is presented in the Supplementary Information.
During metabolite analysis in culture supernatants of strain Mal15T, we found that it produces a distinct class of small molecules belonging to the group of long-chain N-acyl tyrosine derivatives, which we named stieleriacines (in accordance with the proposed genus name Stieleria). Stieleriacines are composed of a lauric acid or trans-2-dodecenoic acid moiety as fatty acid residue, which is ligated to a dehydrotyrosine derivative additionally C-methylated in meta-position of its aromatic ring (Fig. 1). Stieleriacines were found to be produced during laboratory-scale shaking flask cultivations of axenic Mal15T cultures, indicating that their production does not require presence of other microorganisms as an external stimulus.
Analysis of in vitro and in vivo effects of stieleriacine A1
In previous studies, it has been shown that N-acyl amino acids can display antimicrobial activities against various bacteria12,13,14,15,16,17, while N-acylated tyrosine derivatives were shown to inhibit the enzyme tyrosinase18. Employing in vivo and in vitro assays with purified stieleriacine A1, the major stieleriacine produced by strain Mal15T, we could show that these compounds exhibit only moderate antimicrobial activity against Gram-positive bacteria (Supplementary Table 1) and negligible inhibition of tyrosinase, indicating that neither of the two effects reflects the major function of stieleriacines. To get first hints on the natural function of stieleriacine A1, we thus applied time-lapse microscopy coupled to microfluidics as experimental strategy to allow observation of individual cells over time spans of several hours to days. Such unusual approaches were necessary since most of the canonical biofilm formation assays did not work for planctomycetes. Mal15T cells were treated with 1.34 µM stieleriacine A1, a ‘physiological’ concentration found to be produced in the mid-exponential phase during laboratory-scale cultivation of the strain (Fig. 2). The natural production of stieleriacines by strain Mal15T can be neglected in this experimental setup as a constant medium flow ensures that compounds secreted by the strain are immediately flushed away. We found that presence of stieleriacine A1 significantly reduced the duration of the lag phase (5.2 h) compared to untreated Mal15T cells (6.3 h; p = 0.0058) (Fig. 3, Supplementary Table 2, Supplementary Movie 1 and 2). This observation points towards a bioactivity to the advantage of its producer, however, we did not exclude any additional inhibitory effect on competing microbes at this stage.
Triplicates of Mal15T cultures were harvested every 12 h for five days, followed by harvesting every 16 h for two days. The optical density at 600 nm (OD600nm), the biomass dry weight and the stieleriacine A1 titre in the cell supernatant were quantified. Error bars indicate the standard deviation. Where error bars are not visible, they are shorter than the size of the symbols.
a Time-lapse series of untreated Mal15T cells during the 6.25 h lag-phase (1–5) and after initialisation of cell division and entrance into the exponential phase (6–8). The division as such from budding to daughter cell release required 1.9 h. b The lag phase of Mal15T cells was significantly shorter (1–5: 5.16 h) once treated with 1.34 µM stieleriacine A1, while the division as such lasted 1.9 h as well (6–8); (untreated: 6.25 h (a), treated: 5.16 h (b), p = 0.0058). The scale bar is 1 µm.
The artificial laboratory cultivation conditions and the tested concentration of stieleriacine A1 probably only insufficiently reflect natural conditions in seawater, in which densities of planktonic Mal15T cells are much lower. From an ecological perspective, stieleriacine production by individual swarming cells or smaller aggregates appears irrational as the secreted molecules are immediately diluted to inactive concentrations. We hypothesised that sufficient stieleriacine concentrations can only be reached by larger numbers of cells inhabiting a local environment, such as a biofilm with a rather gel-like texture and that stieleriacines might thus play a more important role in such micro-environments. Following this argumentation, we were curious to test for a potential effect of stieleriacine A1 on biofilm formation capabilities of natural competitors of strain Mal15T. We chose two members of the ‘Roseobacter group’, P. inhibens DSM 17395 and Sulfitobacter dubius DSM 16472T, which were shown to co-occur with species of the familiy Pirellulaceae19,20. The biofilm assays were performed with isolated stieleriacine A1 in absence of Mal15T cells to exclude any additional effects, which might e.g. be caused by other natural compounds produced by strain Mal15T. Unexpectedly, application of stieleriacine A1 led to an increased biofilm formation of P. inhibens (+35%, p < 0.001), but reduced biofilm formation of S. dubius (−15%, p < 0.001) (Fig. 4a). This effect was observed when 134 µM stieleriacine A1 was used, a 100-fold higher concentration than in the microfluidics experiment performed with strain Mal15T. The biofilm assay required empirical optimisation of the stieleriacine A1 concentration; since we were aware that concentrations in liquid cultures might not properly reflect local concentrations in biofilms, in which cells are in very close proximity and local metabolite concentrations are thus considerably higher. We decided to follow the empirical optimisation approach of the stieleriacine A1 concentration to reconstruct the natural conditions in the biofilm as good as possible. Natural local concentrations of stieleriacines in biofilms are difficult to assess and largely dependent on size and microbial community composition of the biofilm. The observed stimulating effect of stieleriacine A1 on P. inhibens was surprising and was thus analysed further. To this end, we tested for a potential interaction of stieleriacine A1 with the AHL-dependent quorum sensing system in P. inhibens, in particular the AHL-responsive transcriptional regulator protein LuxR21. Mutant analysis with a luxR-deficient P. inhibens strain (Supplementary Fig. 1) demonstrated that the positive effect on biofilm formation is largely independent from LuxR, indicating that stieleriacine A1 does not directly interfere with the quorum sensing system of P. inhibens (Fig. 4b). But, why should strain Mal15T produce a molecule that stimulates biofilm formation of its natural competitor P. inhibens, while reducing the fitness of its other competitor S. dubius? The reason possibly relates to production of TDA22. Promoting the biofilm formation of P. inhibens would in return lead to increased production of AHLs and TDA23,24. This benefits strain Mal15T in two ways: (i) Mal15T turned out to be resistant against TDA while other competitors are not. (ii) TDA production reduces the growth speed of P. inhibens approximately by 41% due to the increased metabolic burden25 and may ensure that strain Mal15T is not outcompeted. Thus, we suggest that strain Mal15T ‘invites’ P. inhibens via stieleriacine production to join the biofilm and to produce TDA in order to challenge other faster growing competitors. In contrast, biofilm formation of S. dubius is reduced by stieleriacine A1 as this bacterium does not produce TDA and is in that regard useless for strain Mal15T. The hypothesis suggests that planctomycetes actively shape the biofilm community to gain advantages employing a novel type of natural products: stieleriacines. Our results thus point towards the capability of certain slow-growing bacteria to mediate colonisation of their surrounding by production of small molecules. The hypothesis fits to the observation that slow-growing planctomycetes can dominate biofilms on competitive marine surfaces7.
a Box plot of biofilm formation capacity of P. inhibens (P) and S. dubius (S). b Box plot of biofilm formation capacity of P. inhibens (P) compared to P. inhibens ΔluxR transposon mutant (M). Experimental setup: P.C+: P. inhibens in MB medium; P.C++: P. inhibens in MB with acetone; P.A11: P. inhibens in MB with stieleriacine A1 1.34 µM in acetone; P.A12: P. inhibens in MB with stieleriacine A1 134 µM in acetone; S.C+: S. dubius in MB medium; S.C++: S. dubius in MB medium with acetone; SA11: S. dubius in MB with stieleriacine A1 1.34 µM in acetone; S.A12: S. dubius in MB with stieleriacine A1 134 µM in acetone; M.C+: ΔluxR transposon mutant in MB medium; P.C++: ΔluxR transposon mutant in MB with acetone; MA11: ΔluxR transposon mutant in MB with stieleriacine A1 1.34 µM in acetone; MA12: ΔluxR transposon mutant in MB with stieleriacine A1 134 µM in acetone; C−: MB medium. For each experiment, four biological replicates and two technical replicates were performed. The minimum and maximum value, sample median, and the first and third quartiles are shown (box-and-whisker plot).
Genome analysis identifies a putative stieleriacine biosynthesis pathway
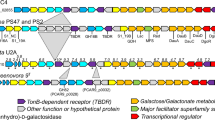
Although microbial synthesis of N-acyl tyrosine molecules has been reported in several recent studies18,26,27, the genetic basis for biosynthesis has not been elucidated in greater detail. The key enzyme for stieleriacine biosynthesis in strain Mal15T is likely an N-acyl amino acid synthase (NAS) (Fig. 5). We sequenced the 9.9 Mb genome of strain Mal15T and identified a cluster (locus tags Mal15_37240 to Mal15_37430, Fig. 6), which contains not only four putative NAS-encoding genes, but all genes required for a postulated stieleriacine biosynthesis pathway. The NAS-encoding genes (Mal15_37340, Mal15_37370, Mal15_37380 and Mal15_37430) were originally annotated as hypothetical proteins, but were identified as candidates by BLASTp and InterPro scan. The four enzymes might have different substrate spectra, which can explain the production of five slightly different stieleriacines in strain Mal15T (Fig. 1). However, it should not be excluded that the strain is capable of producing other N-acyl amino acids. The C-methylation in meta-position of the aromatic ring and an uncommon double bond in the α,β-position of the tyrosine moiety were identified as characteristic molecular features separating stieleriacines from other N-acyl tyrosines identified so far18,26. In the biosynthetic cluster, Mal15_37250 encodes a putative methyltransferase similar to the l-tyrosine C3-methyltransferase SfmM2 of Streptomyces lavendulae. This enzyme catalyses an early step in saframycin biosynthesis28 identical to the reaction required during synthesis of stieleriacines (Fig. 5). The gene encoded immediately downstream, Mal15_37560, codes for a putative tRNA threonylcarbamoyladenosine dehydratase. When only taking the putative protein annotation into consideration, involvement in stieleriacine biosynthesis appears rather unlikely. However, the substrate of the dehydratase (tRNA threonylcarbamoyladenosine) strongly resembles the amide bond obtained after the N-acylation reaction in the postulated stieleriacine pathway. The catalysed lactonization and dehydration reaction would give rise to a cyclic pathway intermediate, in which the remaining double bond can be introduced by a coupled oxidation and hydrolysis reaction induced by a keto-enol tautomeric rearrangement (Fig. 5). This reaction may be catalysed by the putative oxidoreductase encoded by Mal15_37310. Other proteins in the cluster code for a putative transcriptional regulator or putative transporters (Supplementary Table 3), which could have additional function for regulation of the pathway and product export, however, we had no clear indication for this based on published information. Mal15_37240, encoding a putative fatty acid desaturase, is likely involved in the introduction of the double bond in the fatty acid part of the molecule (Fig. 5), however it remains to be elucidated whether this reaction takes place before or after the N-acylation.
Genes with a predicted role in the stieleriacine biosynthesis cluster are shown in different colours. For these genes the automated gene annotation and the postulated function are shown in the table. The complete list of automated gene annotations of the cluster can be found in Supplementary Table 3.
Discussion
In this study, we isolated and characterised Stieleria maiorica Mal15T, which represents a novel species and genus within the family Pirellulaceae, order Pirellulales, class Planctomycetia, phylum Planctomycetes. The strain was shown to produce N-acylated tyrosine deriatives, which we designated stieleriacines and which belong to the class of N-acylated amino acids. In previous studies, compounds of this class showed antimicrobial activities against various bacteria12,13,14,15,16,17 and probably act as signalling molecules mediating intraspecies and interspecies communication4,15,29. N-acylated tyrosine derivatives isolated from the marine γ-proteobacterium Thalassotalea sp. PP2-459 (Alteromonadales) were also shown to inhibit the enzyme tyrosinase18. Recently, an N-acyl tyrosine congener harbouring an uncommon α-methyl group in the tyrosine moiety was identified in Alteromonas sp. RKMC-009 isolated from the surface of a marine sponge26, while its natural function remains to be elucidated. Stieleriacine A1 showed only moderate antimicrobial activity against Gram-positive bacteria and negligible tyrosinase inhibitory activity. At this early stage of research with the novel isolate, we can only speculate on potential quorum sensing activity of stieleriacines. Analysis of potential target genes of a respective regulator part of a putative quorum sensing system is challenging given that 42% (2897 out of 6920) proteins encoded in the genome of strain Mal15T are of unknown function. Experiments with the strain itself instead of isolated stieleriacine are particularly interesting, however, these require a Mal15T mutant incapable of stieleriacine biosynthesis as negative control. Such a mutant can also provide important information on the stieleriacine biosynthesis pathway, however, due to lacking experience in terms of the genetic accessibility of strain Mal15T, this question needs to be addressed in follow-up studies.
Although some N-acyl amino acid compounds have been identified, the current knowledge on the underlying biosynthetic pathways is limited. Evidence for the presence of N-acylated derivatives of aromatic or basic amino acids was mainly based on a multitude of environmental DNA libraries. Characterisation of biosynthetic pathways for N-acyl amino acids is an important task in the future given that such compounds find potential biotechnological applications as antimicrobial surfactants and precursors for biodegradable polyesters in pharmaceutical and biomedical applications30,31. In case of the surfactants, the C12 chain as acyl residue (as present in stieleriacine A1) proved to have the optimum antibacterial activity among the single chain surfactants tested31. In that regard, genes of strain Mal15T are interesting target for expression in heterologous microbial hosts.
A report on screening of soil metagenomic libraries revealed that genes coding for NAS proteins are frequently found adjacent to genes involved in the PEP-CTERM/exosortase protein sorting system (an exopolysaccharide-associated protein targeting system), indicating a functional link between both pathways29. Indeed, the same situation is also found in the genome of strain Mal15T. A PEP-CTERM motif protein-encoding gene (Mal15_37300) was found within the proposed stieleriacine biosynthetic gene cluster (Fig. 6), and many exopolysaccharide biosynthesis-related and secretion-related genes are encoded in the region upstream (Mal15_36520 to Mal17_37230, Supplementary Table 3). The conserved genetic organisation supports the notion of a functional link between stieleriacine biosynthesis, exopolysaccharide assembly and biofilm formation, which requires additional attention in future studies.
In conclusion, the identified stieleriacines stimulated growth initiation of Mal15T cells and either promoted or inhibited biofilm formation of two different microorganisms, likely naturally competing with strain Mal15T for biotic surfaces in marine environments. Although the two tested competitors might only be a small fraction of microorganisms dwelling in such environments, it allows first insights into strategies how slow-growing microorganisms compensate for lower growth rates and avoid being outcompeted. Stieleriacines are likely only one of several strategies how the survival of planctomycetes in aquatic environments is ensured and their high abundance on several marine biotic surfaces can be explained.
Methods
Sample collection and preparation
Seawater sediment samples from 30 cm depth were collected from Spain, Mallorca island, El Arenal, between Balneario 4 and 5 (39° 30′ 45.2″ N, 2° 44′ 49.1″ E) on September 23th, 2014. Samples were collected in sterile 50 mL polypropylene tubes, transferred to the laboratory, homogenised and processed.
Culture and isolation conditions
For maintenance of strain Mal15T M1 medium with HEPES (H) as buffering agent and additionally supplemented with N-acetyl glucosamine (NAG) and artificial seawater (ASW) (M1H NAG ASW medium) was used and prepared as described before32. Solid medium was prepared with 12 g/L (w/v) agar, washed three times with double distilled H2O and cooled to 55 °C prior to the addition of heat-sensitive solutions. For the initial strain isolation, solid M1H NAG ASW medium was supplemented with 100 µL of an ampicillin (50 mg/mL) and cycloheximide stock solution (20 mg/mL), dried for 30 min, inoculated with 100 µL homogenised sample material per plate and incubated at 20 °C in the dark until colony formation became visible. Single colonies were inoculated on fresh solid medium with the respective antibiotics. Axenic cultures were cryo-preserved in M1H NAG ASW medium supplemented with 50% glycerol or 5% DMSO and stored at −80 °C. Exponentially growing Mal15T cells were used for analysis of carbon source utilisation (Supplementary Table 4) and enzymatic activities (Supplementary Table 5).
Strain characterisation
Electron microscopy and basic light microscopy were performed as previously described33,34. For time-lapse microscopy, cells were observed using the CellASIC ONIX Microfluidic Platform (Merck Millipore). Physiological tests as well as phylogenetic and genome analysis were performed as previously described35. The results are shown in Supplementary Figs. 2–6.
Statistics and reproducibility
All experiments were performed at least in biological triplicates. If not stated otherwise, data represent average and standard deviation. In all cases, detailed information is given in the respective figure captions. Significance of results is indicated by p-values given in the text.
Phylogenetic analysis
16S rRNA gene sequence-based phylogeny was computed for strain Mal15T (GenBank acc. no. MK554562), the type strains of all described planctomycetal species (assessed in May 2019) and all isolates recently published10 and described1,36,37. Phylogenetic inference based on 16S rRNA gene sequences and multi-locus sequence analysis (MLSA) was performed as described36. The 16S rRNA gene sequences were aligned with SINA38 and the phylogenetic inference was calculated with RAxML39 with a maximum likelihood approach with 1000 bootstraps, nucleotide substitution model GTR, gamma distributed rate variation and estimation of proportion of invariable sites (GTRGAMMAI option). Three 16S rRNA genes of bacterial strains from the PVC superphylum outside of the phylum Planctomycetes were used as outgroup. For MLSA the unique single-copy core genome of the analysed genomes (GenBank acc. no. for strain Mal15T: CP036264.1) was determined with proteinortho540 with the ‘selfblast’ option enabled. The protein sequences of the resulting orthologous groups were aligned using MUSCLE v.3.8.3141. After clipping, partially aligned C-terminal and N-terminal regions and poorly aligned internal regions were filtered using Gblocks42. The final alignment was concatenated and clustered using the maximum likelihood method implemented by RAxML39 with the ‘rapid bootstrap’ method and 500 bootstrap replicates. Four planctomycetal genomes from different families in the class Planctomycetia were used as outgroup. The average nucleotide identity (ANI) was calculated using OrthoANI43. The average amino acid identity (AAI) was calculated using the aai.rb script of the enveomics collection44 and the percentage of conserved proteins (POCP) was calculated as described45. The rpoB nucleotide sequences were taken from publicly available planctomycetal genome annotations and the sequence identities were determined as described46. Alignment and matrix calculation were done with Clustal Omega47.
Isolation and purification of stieleriacines from culture broth
For getting sufficient amounts of Mal15T biomass, nutrient content of the culture broth was increased (1.0 g/L peptone, 1.0 g/L yeast extract, 40 mL/L 2.5% (w/v) glucose solution). Six hundred microlitre Mal15T cultures were incubated for three days at 28 °C and 80 rpm in 2 L baffled flasks until 2% (w/v) of purified adsorbent resin XAD-16N (Rohm and Haas) were added. Cultures were further incubated for three days. In total, XAD-16N from 32 L culture was harvested and processed as previously described32. The combined crude extract was filtered with water/methanol (1:1) using a Strata-X 33 mm, Polymeric Reverse Phase Solid-Phase cartridge (Phenomenex). The solid phase was eluted with acetone and hexane yielding 308.25 mg crude product, which was fractionated using preparative RP-HPLC (PLC 2020, Gilson with Kromasil C18 column 250 × 20 mm, 7 µm; MZ-Analysetechnik) with deionised water and 0.1% formic acid (solvent A), and acetonitrile with 0.1% formic acid (solvent B). The following elution gradient was used: 40% B for 10 min, increased to 50% B in 3 min, and a gradient from 50–70% B in 60 min, thereafter 100% B for 10 min. Ultraviolet (UV) detection was carried out at 215 and 310 nm. Fractions were collected yielding five pure compounds. Stieleriacine A1 (1) (6.3 mg) was obtained at a retention time (tR) of 32.5 min, B1 (2) (2.0 mg) at tR = 34.0 min, C (3) (0.8 mg) at tR = 36.0 min, A2 (4) (4.9 mg) at tR = 39.5 min, B2 (5) (3.5 mg) at tR = 41.1 min (Fig. 1, Supplementary Fig. 7).
Structure elucidation
HRESIMS mass spectra were measured with an Agilent 1200 series HPLC-UV system in combination with an ESI-TOF-MS (Maxis, Bruker) [column 2.1 × 50 mm, 1.7 µm, C18 Acquity UPLC BEH (Waters), solvent A: water with 0.1% formic acid, solvent B: acetonitrile with 0.1% formic acid, gradient: 5% B for 0.5 min increasing to 100% B in 19.5 min, maintaining 100% B for another 5 min, RF = 0.6 mL/min, UV detection 200–600 nm. Nuclear magnetic resonance spectra were recorded on a Bruker Avance III 500 MHz spectrometer with a BBFO(plus) SmartProbe (1H 500 MHz, 13C 126 MHz), and a Bruker Avance III 700 MHz spectrometer with a 5 mm TCI cryoprobe (1H 700 MHz, 13C 175 MHz, 15N 71 MHz). UV spectra were recorded using a Shimadzu UV-VIS spectrophotometer UV-2450. Optical rotation was determined using a PerkinElmer 241 polarimeter. Correlations are shown in Supplementary Fig. 8.
Antimicrobial activity assay
Determination of minimum inhibitory concentrations (MIC) was performed using serial dilution assays in 96-well microtiter plates. YMG medium (4 g/L yeast extract, 10 g/L malt extract, 4 g/L glucose, pH 7.2) was used for yeasts and filamentous fungi, while EBS medium (5 g/L casein peptone, 1 g/L meat extract, 1 g/L yeast extract, 5 g/L glucose, 50 mM HEPES, pH 7.0) was used for bacteria. Tested species were inoculated to initial cell densities of 105–106 colony-forming units per mL in the respective growth medium. MIC were assessed after 16 and 48 h of cultivation at 37 °C (bacteria) and 28 °C (yeasts and moulds), respectively. The MIC values represent the lowest concentration of stieleriacine at which no visible growth was observed.
Cytotoxicity assay
In vitro cytotoxicity (IC50) was investigated using mouse fibroblast cell line L929 and HeLa KB3.1 cells as previously described48. Briefly, cells were cultivated in EBM-2 supplemented with 10% foetal bovine serum under 10% CO2 at 37 °C. Sixty microlitre of serial dilutions from an initial stock of 1 mg/mL stieleriacine in acetone was added to 120 μL aliquots of a cell suspension (50,000 cells per mL) in 96-well microtiter plates. After 5 days of incubation, a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) (MTT) assay was performed. The absorbance was measured at 590 nm using an ELISA plate reader (Victor). The concentration, at which the growth of cells was inhibited to 50% of the control (IC50), was obtained from the dose−response curves. Acetone served as negative control.
Tyrosinase inhibition assay
The tyrosinase inhibition assay was performed as previously described18 with the following modifications: 80 µL of compound, dissolved in PBS (0.1 M, pH 6.8) with 10% methanol at various concentrations, were added to 80 µL of PBS. PBS with 10% methanol served as a negative control. Eighty microlitre of mushroom-tyrosinase (100 units per mL) and 40 µL of l-tyrosine (2.5 mM) were added to each well. Phosphate buffer and tyrosinase served as a negative control. Reactions were incubated at 37 °C for 45 min and the absorbance was measured at 490 nm using the Infinite 200 Pro multimode reader (Tecan). The percentage of tyrosinase inhibition was calculated as follows: [(ΔAcontrol – ΔAsample)/ΔAcontrol] × 100.
Time-lapse microscopy
To analyse the physiological effect of stieleriacine A1 with time-lapse microscopy, a preculture of Mal15T cells was inoculated 1:10 in fresh M1H NAG ASW medium and incubated for 72 h (28 °C, 80 rpm). Before loading, the CellASIC® microfluidic chamber was washed with a constant flow of medium (2 × 5 psi, 5 min). Stieleriacine A1 was provided dissolved in 132 µL acetone at 1.34 µM, while medium supplemented with the same volume of acetone was used as negative control. Cells were monitored for up to 36 h at 28 °C at 2 × 1 psi flow rate. Phase contrast images were acquired and processed as previously described49. In total, 1264 cells were analysed and all experiments were performed in three biological replicates.
Biofilm formation assay
Biofilm formation of P. inhibens and S. dubius was analysed by a crystal violet assay3,50. P. inhibens and S. dubius were grown in 10 mL MB medium (MB, Carl Roth) at 28 °C with vigorous shaking to the early exponential phase (OD600 of 0.4). Cultures were subsequently diluted to an OD600 of 0.065. 100 μL of the diluted culture was transferred to a respective well of a sterile polystyrene 96-well assay plate (Corning, New York, NY, USA; Costar 3370). 1.34 µM or 134 µM of stieleriacine A1 solved in acetone or pure acetone were added to the wells prior to statically culturing of the plates for 24 h. After incubation, medium along with planktonic cells was removed and wells were washed twice with H2O. Two hundred microlitre of 0.5% crystal violet (CV) was added to the wells, which were incubated at room temperature for 10 min. After staining, the CV solution was removed and each well was washed twice with H2O to remove the residual dye and dried overnight. For scoring of cell attachment, the CV was extracted from the biofilm with 200 μL 95% (v/v) ethanol, of which 100 μL were transferred to a new 96-well plate before the absorbance was determined at 595 nm using the Tecan microplate reader (Infinite® 200 PRO). For each experiment, four biological replicates and two technical replicates were performed.
Analysis of the stieleriacine production rate
To analyse the production rate of stieleriacine A1, precultures of strain Mal15T were inoculated 1:10 in fresh M1H NAG ASW medium in triplicates and incubated for 7 days (152 h, 28 °C, 80 rpm). For the next five days 30 mL of each culture were harvested every 12 h, followed by 2 days harvesting after every 16 h. The harvested material was analysed for optical density (OD600), biomass and stieleriacine A1 concentration.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
The genome and 16S rRNA gene sequence of strain Mal15T are available from GenBank under accession numbers CP036264.1 and MK554562.1, respectively. Other relevant data are available from the corresponding authors upon request. The putative stieleriacine biosynthetic gene cluster was deposited at the MiBiG (Minimum Information about a Biosynthetic Gene cluster) database and can be found under accession number BGC0002080.
Change history
17 February 2021
A Correction to this paper has been published: https://doi.org/10.1038/s42003-020-01271-y
17 February 2021
A Correction to this paper has been published: https://doi.org/10.1038/s42003-020-01228-1
References
Kohn, T. et al. Planctopirus ephydatiae, a novel Planctomycete isolated from a freshwater sponge. Syst. Appl. Microbiol. 43, 126022 (2019).
Lage, O. M. & Bondoso, J. Planctomycetes and macroalgae, a striking association. Front. Microbiol. 5, 267 (2014).
Frank, O. et al. Plasmid curing and the loss of grip—the 65-kb replicon of Phaeobacter inhibens DSM 17395 is required for biofilm formation, motility and the colonization of marine algae. Syst. Appl. Microbiol. 38, 120–127 (2014).
Van Wagoner, R. M. & Clardy, J. FeeM, an N-acyl amino acid synthase from an uncultured soil microbe: structure, mechanism, and acyl carrier protein binding. Structure 14, 1425–1435 (2006).
Whiteley, M., Diggle, S. P. & Greenberg, E. P. Progress in and promise of bacterial quorum sensing research. Nature 551, 313–320 (2017).
Papenfort, K. & Bassler, B. L. Quorum sensing signal-response systems in Gram-negative bacteria. Nat. Rev. Microbiol. 14, 576–588 (2016).
Wiegand, S., Jogler, M. & Jogler, C. On the maverick planctomycetes. FEMS Microbiol. Rev. 42, 739–760 (2018).
Frank, C. S., Langhammer, P., Fuchs, B. M. & Harder, J. Ammonium and attachment of Rhodopirellula baltica. Arch. Microbiol. 193, 365–372 (2011).
Waters, C. M. & Bassler, B. L. Quorum sensing: cell-to-cell communication in bacteria. Annu. Rev. Cell Dev. Biol. 21, 319–346 (2005).
Wiegand, S. et al. Cultivation and functional characterization of 79 planctomycetes uncovers their unique biology. Nat. Microbiol. 5, 126–140 (2020).
Jeske, O., Jogler, M., Petersen, J., Sikorski, J. & Jogler, C. From genome mining to phenotypic microarrays: planctomycetes as source for novel bioactive molecules. Antonie Van. Leeuwenhoek 104, 551–567 (2013).
Clardy, J. & Brady, S. F. Cyclic AMP directly activates NasP, an N-acyl amino acid antibiotic biosynthetic enzyme cloned from an uncultured beta-proteobacterium. J. Bacteriol. 189, 6487–6489 (2007).
Thies, S. et al. Metagenomic discovery of novel enzymes and biosurfactants in a slaughterhouse biofilm microbial community. Sci. Rep. 6, 27035 (2016).
Brady, S. F., Clardy, J. & Long-Chain N-Acyl amino acid antibiotics isolated from heterologously expressed environmental DNA. J. Am. Chem. Soc. 122, 12903–12904 (2000).
Brady, S. F., Chao, C. J., Clardy, J. & Long-chain N-acyltyrosine synthases from environmental DNA. Appl. Environ. Microbiol. 70, 6865–6870 (2004).
Brady, S. F. & Clardy, J. N-acyl derivatives of arginine and tryptophan isolated from environmental DNA expressed in Escherichia coli. Org. Lett. 7, 3613–3616 (2005).
Brady, S. F., Chao, C. J. & Clardy, J. New natural product families from an environmental DNA (eDNA) gene cluster. J. Am. Chem. Soc. 124, 9968–9969 (2002).
Deering, R. W. et al. N-acyl dehydrotyrosines, tyrosinase inhibitors from the marine bacterium Thalassotalea sp. PP2-459. J. Nat. Prod. 79, 447–450 (2016).
Morris, R., Longnecker, K. & Giovannoni, S. Pirellula and OM43 are among the dominant lineages identified in an Oregon coast diatom bloom. Environ. Microbiol. 8, 1361–1370 (2006).
Hengst, M. B., Andrade, S., González, B. & Correa, J. A. Changes in epiphytic bacterial communities of intertidal seaweeds modulated by host, temporality, and copper enrichment. Microb. Ecol. 60, 282–290 (2010).
Zan, J., Liu, Y., Fuqua, C. & Hill, R. T. Acyl-homoserine lactone quorum sensing in the Roseobacter clade. Int. J. Mol. Sci. 15, 654–669 (2014).
Wilson, M. Z., Wang, R., Gitai, Z. & Seyedsayamdost, M. R. Mode of action and resistance studies unveil new roles for tropodithietic acid as an anticancer agent and the γ-glutamyl cycle as a proton sink. Proc. Natl Acad. Sci. USA 113, 1630–1635 (2016).
Berger, M., Neumann, A., Schulz, S., Simon, M. & Brinkhoff, T. Tropodithietic acid production in Phaeobacter gallaeciensis is regulated by N-acyl homoserine lactone-mediated quorum sensing. J. Bacteriol. 193, 6576–6585 (2011).
Beyersmann, P. G. et al. Dual function of tropodithietic acid as antibiotic and signaling molecule in global gene regulation of the probiotic bacterium Phaeobacter inhibens. Sci. Rep. 7, 730 (2017).
Will, S. E. et al. The limits to growth—energetic burden of the endogenous antibiotic tropodithietic acid in Phaeobacter inhibens DSM 17395. PLoS ONE 12, e0177295 (2017).
MacIntyre, L. W., Charles, M. J., Haltli, B. A., Marchbank, D. H. & Kerr, R. G. An Ichip-domesticated sponge bacterium produces an N-acyltyrosine bearing an α-methyl substituent. Org. Lett. 21, 7768–7771 (2019).
Lee, C.-M. et al. Characterization of a novel antibacterial N-acyl amino acid synthase from soil metagenome. J. Biotechnol. 294, 19–25 (2019).
Li, L. et al. Characterization of the saframycin A gene cluster from Streptomyces lavendulae NRRL 11002 revealing a nonribosomal peptide synthetase system for assembling the unusual tetrapeptidyl skeleton in an iterative manner. J. Bacteriol. 190, 251–263 (2008).
Craig, J. W., Cherry, M. A. & Brady, S. F. Long-chain N-acyl amino acid synthases are linked to the putative PEP-CTERM/exosortase protein-sorting system in Gram-negative bacteria. J. Bacteriol. 193, 5707–5715 (2011).
Taresco, V. et al. New N-acyl amino acid-functionalized biodegradable polyesters for pharmaceutical and biomedical applications. RSC Adv. 6, 109401–109405 (2016).
Joondan, N., Jhaumeer‐Laulloo, S., Caumul, P. & Akerman, M. Synthesis, physicochemical, and biological activities of novel N‐acyl tyrosine monomeric and gemini surfactants in single and SDS/CTAB–mixed micellar system. J. Phys. Org. Chem. 30, e3675 (2017).
Jeske, O. et al. Developing techniques for the utilization of planctomycetes as producers of bioactive molecules. Front. Microbiol. 7, 1242 (2016).
Kohn, T. et al. Fuerstia marisgermanicae gen. nov., sp. nov., an unusual member of the phylum planctomycetes from the German Wadden Sea. Front. Microbiol 7, 2079 (2016).
Kohn, T. et al. Corrigendum: Fuerstia marisgermanicae gen. nov., sp. nov., an unusual member of the phylum planctomycetes from the German Wadden Sea. Front. Microbiol. 10, 1029 (2019).
Rast, P. et al. Three novel species with peptidoglycan cell walls form the new genus Lacunisphaera gen. nov. in the family Opitutaceae of the verrucomicrobial subdivision 4. Front. Microbiol. 8, 202 (2017).
Kallscheuer, N. et al. Description of three bacterial strains belonging to the new genus Novipirellula gen. nov., reclassificiation of Rhodopirellula rosea and Rhodopirellula caenicola and readjustment of the genus threshold of the phylogenetic marker rpoB for Planctomycetaceae. Antonie van Leeuwenhoek https://doi.org/10.1007/s10482-019-01374-51-17 (2019).
Boersma, A. et al. Alienimonas californiensis gen. nov. sp. nov., a novel planctomycete isolated from the kelp forest in Monterey Bay. Antonie van Leeuwenhoek. https://doi.org/10.1007/s10482-019-01367-4 (2019).
Pruesse, E., Peplies, J. & Glöckner, F. O. SINA: accurate high-throughput multiple sequence alignment of ribosomal RNA genes. Bioinformatics 28, 1823–1829 (2012).
Stamatakis, A. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30, 1312–1313 (2014).
Lechner, M. et al. Proteinortho: detection of (co-)orthologs in large-scale analysis. BMC Bioinform. 12, 124 (2011).
Edgar, R. C. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32, 1792–1797 (2004).
Castresana, J. Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol. Biol. Evol. 17, 540–552 (2000).
Lee, I., Ouk Kim, Y., Park, S. C. & Chun, J. OrthoANI: an improved algorithm and software for calculating average nucleotide identity. Int. J. Syst. Evol. Microbiol. 66, 1100–1103 (2016).
Rodriguez-R, L. M. & Konstantinidis, K. T. The enveomics collection: a toolbox for specialized analyses of microbial genomes and metagenomes. PeerJ Prepr. 4, e1900v1 (2016).
Qin, Q.-L. et al. A proposed genus boundary for the prokaryotes based on genomic insights. J. Bacteriol. 196, 2210–2215 (2014).
Bondoso, J., Harder, J. & Lage, O. M. rpoB gene as a novel molecular marker to infer phylogeny in Planctomycetales. Antonie Van. Leeuwenhoek 104, 477–488 (2013).
Sievers, F. et al. Fast, scalable generation of high‐quality protein multiple sequence alignments using Clustal Omega. Mol. Syst. Biol. 7, 539 (2011).
Surup, F. et al. Deconins A–E: cuparenic and mevalonic or propionic acid conjugates from the basidiomycete Deconica sp. 471. J. Nat. Prod. 78, 934–938 (2015).
Boedeker, C. et al. Determining the bacterial cell biology of planctomycetes. Nat. Commun. 8, 14853 (2017).
O’Toole, G. A. & Kolter, R. Initiation of biofilm formation in Pseudomonas fluorescens WCS365 proceeds via multiple, convergent signalling pathways: a genetic analysis. Mol. Microbiol. 28, 449–461 (1998).
Acknowledgements
We thank especially Anja Heuer and Cäcilia Bergmann, Wera Collisi, Christel Kakoschke, Simone Severitt and Nicole Heyer for excellent technical assistance. We further thank Cathrin Spröer for general support during sequencing, Daniela Borchert for help with time-lapse microscopy and Marc Stadler for generous support. This work was funded by the Deutsche Forschungsgemeinschaft DFG JO 893/4-1, SU 936/1-1, TRR 51 and KA 4967/1-1.
Author information
Authors and Affiliations
Contributions
N.K. wrote the final version of the manuscript with help of C.J. based on an initial version written by O.J., B.S. and F.S. O.J. performed most biological experiments. N.K. analysed the genome and proposed the biosynthesis pathway with help from M.H.M. B.S. performed most of the chemical experiments with the help of F.S. C.B. performed time-lapse and light microscopy experiments. S.W. sequenced and characterised the genome and performed the phylogenetic analysis. P.B. performed Roseobacter-related experiments and constructed the mutants with help from J.P. M.J. developed the novel isolation strategy with help from C.J., isolated the strain and performed general microbiological experiments. M.R. took electron micrographs. J.P. designed the Roseobacter-related experiments and supervised P.B. M.H.M. supported N.K. and both analysed genes related to small molecule production. F.S. supervised B.S. and guided structure elucidation. C.J. and F.S. designed the concept of this study. C.J. supervised O.J. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kallscheuer, N., Jeske, O., Sandargo, B. et al. The planctomycete Stieleria maiorica Mal15T employs stieleriacines to alter the species composition in marine biofilms. Commun Biol 3, 303 (2020). https://doi.org/10.1038/s42003-020-0993-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-020-0993-2
This article is cited by
-
Planctoellipticum variicoloris gen. nov., sp. nov., a novel member of the family Planctomycetaceae isolated from wastewater of the aeration lagoon of a sugar processing plant in Northern Germany
Scientific Reports (2024)
-
A type III polyketide synthase cluster in the phylum Planctomycetota is involved in alkylresorcinol biosynthesis
Applied Microbiology and Biotechnology (2024)
-
Uncovering the biotechnological capacity of marine and brackish water Planctomycetota
Antonie van Leeuwenhoek (2024)
-
Stieleria tagensis sp. nov., a novel member of the phylum Planctomycetota isolated from Tagus River in Portugal
Antonie van Leeuwenhoek (2023)
-
Stratiformator vulcanicus gen. nov., sp. nov., a marine member of the family Planctomycetaceae isolated from a red biofilm in the Tyrrhenian Sea close to the volcanic island Panarea
Antonie van Leeuwenhoek (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.