Abstract
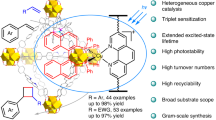
Triplet excited-state organic chromophores present countless opportunities for applications in photocatalysis. Here we describe an approach to the engineering of the triplet excited states of aromatic chromophores, which involves incorporating pyrene into pyridinium-containing mechanically interlocked molecules (MIMs). The π-extended nature of the pyrenes enforces [π···π] stacking, affording an efficient synthesis of tetrachromophoric octacationic homo[2]catenanes. These MIMs generate triplet populations and efficient intersystem crossing on account of the formation of a mixed charge-transfer/exciplex electronic state and a nanoconfinement effect, which leads to a high level of protection of the triplet state and extends the triplet lifetimes and yields. These compounds display excellent catalytic activity in photo-oxidation, as demonstrated by the aerobic oxidation of a sulfur-mustard simulant. This research highlights the benefits of using the mechanical bond to fine-tune the triplet photophysics of existing aromatic chromophores, providing an avenue for the development of unexplored MIM-based photosensitizers and photocatalysts.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
Source data related to this paper may be requested from the authors. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2098309 (2,7-2,7PyBox·4PF6), 2098310 (1,6-1,6PyBox·4PF6) and 2098670 (2,7-2,7PyHC·8PF6). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures.
References
Figueira-Duarte, T. M. & Müllen, K. Pyrene-based materials for organic electronics. Chem. Rev. 111, 7260–7314 (2011).
Mateo-Alonso, A. Pyrene-fused pyrazaacenes: from small molecules to nanoribbons. Chem. Soc. Rev. 43, 6311–6324 (2014).
Islam, M. M., Hu, Z., Wang, Q., Redshaw, C. & Feng, X. Pyrene-based aggregation-induced emission luminogens and their applications. Mater. Chem. Front. 3, 762–781 (2019).
Kinik, F. P., Ortega-Guerrero, A., Ongari, D., Ireland, C. P. & Smit, B. Pyrene-based metal-organic frameworks: from synthesis to applications. Chem. Soc. Rev. 50, 3143–3177 (2021).
Maeda, H. et al. Alkynylpyrenes as improved pyrene-based biomolecular probes with the advantages of high fluorescence quantum yields and long absorption/emission wavelengths. Chem. Eur. J. 12, 824–831 (2006).
Zych, D. Non-K region disubstituted pyrenes (1,3-, 1,6- and 1,8-) by (hetero)aryl groups. Molecules 24, 2551 (2019).
Zych, D. & Slodek, A. Pyrene derivatives with two types of substituents at positions 1, 3, 6 and 8—fad or necessity? RSC Adv. 9, 24015–24024 (2019).
Chang, X. et al. Coordination-driven self-assembled metallacycles incorporating pyrene: fluorescence mutability, tunability and aromatic amine sensing. J. Am. Chem. Soc. 141, 1757–1765 (2019).
Yang, Z. et al. Hierarchical self-assembly of a pyrene-based discrete organoplatinum(II) double-metallacycle with triflate anions via hydrogen bonding and its tunable fluorescence emission. J. Am. Chem. Soc. 142, 13689–13694 (2020).
Gan, K. P., Yoshio, M. & Kato, T. Columnar liquid-crystalline assemblies of X-shaped pyrene-oligothiophene conjugates: photoconductivities and mechanochromic functions. J. Mater. Chem. C. 4, 5073–5080 (2016).
Feng, X., Hu, J.-Y., Redshaw, C. & Yamato, T. Functionalization of pyrene to prepare luminescent materials—typical examples of synthetic methodology. Chem. Eur. J. 22, 11898–11916 (2016).
Kapf, A. et al. Alkyloxy modified pyrene fluorophores with tunable photophysical and crystalline properties. N. J. Chem. 43, 6361–6371 (2019).
Hayer, A. et al. Highly fluorescent crystalline and liquid crystalline columnar phases of pyrene-based structures. J. Phys. Chem. B 110, 7653–7659 (2006).
Winnik, F. M. Photophysics of preassociated pyrenes in aqueous polymer solutions and in other organized media. Chem. Rev. 93, 587–614 (1993).
Pokhrel, M. R. & Bossmann, S. H. Synthesis, characterization and first application of high molecular weight polyacrylic acid derivatives possessing perfluorinated side chains and chemically linked pyrene labels. J. Phys. Chem. B 104, 2215–2223 (2000).
Li, D. et al. Inorganic-organic hybrid vesicles with counterion- and pH-controlled fluorescent properties. J. Am. Chem. Soc. 133, 14010–14016 (2011).
Ni, X.-L., Wang, S., Zeng, X., Tao, Z. & Yamato, T. Pyrene-linked triazole-modified homooxacalix[3]arene: a unique C3 symmetry ratiometric fluorescent chemosensor for Pb2+. Org. Lett. 13, 552–555 (2011).
Ueno, A., Suzuki, I. & Osa, T. Host-guest sensory systems for detecting organic compounds by pyrene excimer fluorescence. Anal. Chem. 62, 2461–2466 (1990).
Ghosh, P. et al. ‘Extra stabilisation’ of a pyrene based molecular couple by γ-cyclodextrin in the excited electronic state. Phys. Chem. Chem. Phys. 14, 11500–11507 (2012).
Vullev, V. I., Jiang, H. & Jones, G. in Advanced Concepts in Fluorescence Sensing: Part B: Macromolecular Sensing (eds Geddes, C. D. & Lakowicz, J. R.) 211–239 (Springer, 2005).
McNelles, S. A., Thoma, J. L., Adronov, A. & Duhamel, J. Quantitative characterization of the molecular dimensions of flexible dendritic macromolecules in solution by pyrene excimer fluorescence. Macromolecules 51, 1586–1590 (2018).
Takaya, T. et al. Excited-state dynamics of pyrene incorporated into poly(substituted methylene)s: effects of dense packing of pyrenes on excimer formation. Macromolecules 51, 5430–5439 (2018).
Hu, J.-Y. et al. A single-molecule excimer-emitting compound for highly efficient fluorescent organic light-emitting devices. Chem. Commun. 48, 8434–8436 (2012).
Jiang, W. et al. A single-molecule conformation modulating crystalline polymorph of a physical π–π pyrene dimer: blue and green emissions of a pyrene excimer. J. Mater. Chem. C 8, 3367–3373 (2020).
Miyoshi, N. & Tomita, G. Production and reaction of singlet oxygen in aqueous micellar solutions using pyrene as photosensitizer. Z. Naturforsch. B 33, 622–627 (1978).
Liu, Y. et al. Efficient and selective oxidation of sulfur mustard using singlet oxygen generated by a pyrene-based metal-organic framework. J. Mater. Chem. A 4, 13809–13813 (2016).
Atilgan, A., Islamoglu, T., Howarth, A. J., Hupp, J. T. & Farha, O. K. Detoxification of a sulfur mustard simulant using a BODIPY-functionalized zirconium-based metal-organic framework. ACS Appl. Mater. Interfaces 9, 24555–24560 (2017).
Howarth, A. J. et al. Postsynthetic incorporation of a singlet oxygen photosensitizer in a metal-organic framework for fast and selective oxidative detoxification of sulfur mustard. Chem. Eur. J. 23, 214–218 (2017).
McClure, D. S. Triplet‐singlet transitions in organic molecules. Lifetime measurements of the triplet state. J. Chem. Phys. 17, 905–913 (1949).
Kasha, M. Collisional perturbation of spin‐orbital coupling and the mechanism of fluorescence quenching. A visual demonstration of the perturbation. J. Chem. Phys. 20, 71–74 (1952).
El‐Sayed, M. A. Effect of spin-orbit interactions on the dipolar nature of the radiative microwave zero‐field transitions in aromatic molecules. J. Chem. Phys. 60, 4502–4507 (1974).
Havlas, Z. & Michl, J. Prediction of an inverse heavy-atom effect in H–C–CH2Br: bromine substituent as a π acceptor. J. Am. Chem. Soc. 124, 5606–5607 (2002).
Sasikumar, D., John, A. T., Sunny, J. & Hariharan, M. Access to the triplet excited states of organic chromophores. Chem. Soc. Rev. 49, 6122–6140 (2020).
Filatov, M. A. et al. BODIPY-pyrene and perylene dyads as heavy-atom-free singlet oxygen sensitizers. ChemPhotoChem 2, 606–615 (2018).
Beldjoudi, Y. et al. Supramolecular porous organic nanocomposites for heterogeneous photocatalysis of a sulfur mustard simulant. J. Adv. Mater. 32, 2001592 (2020).
Stoddart, J. F. Mechanically interlocked molecules (MIMs)—molecular shuttles, switches and machines (Nobel Lecture). Angew. Chem. Int. Ed. 56, 11094–11125 (2017).
Sauvage, J.-P. From chemical topology to molecular machines (Nobel Lecture). Angew. Chem. Int. Ed. 56, 11080–11093 (2017).
Barnes, J. C. et al. Solid-state characterization and photoinduced intramolecular electron transfer in a nanoconfined octacationic homo[2]catenane. J. Am. Chem. Soc. 136, 10569–10572 (2014).
Inouye, M. et al. A doubly alkynylpyrene-threaded [4]rotaxane that exhibits strong circularly polarized luminescence from the spatially restricted excimer. Angew. Chem. Int. Ed. 53, 14392–14396 (2014).
Ryan, S. T. J. et al. Energy and electron transfer dynamics within a series of perylene diimide/cyclophane systems. J. Am. Chem. Soc. 137, 15299–15307 (2015).
Hayashi, K. et al. Observation of circularly polarized luminescence of the excimer from two perylene cores in the form of [4]rotaxane. Chem. Eur. J. 24, 14613–14616 (2018).
Sagara, Y. et al. Rotaxanes as mechanochromic fluorescent force transducers in polymers. J. Am. Chem. Soc. 140, 1584–1587 (2018).
Gong, X. et al. Toward a charged homo[2]catenane employing diazaperopyrenium homophilic recognition. J. Am. Chem. Soc. 140, 6540–6544 (2018).
Sagara, Y. et al. Rotaxane-based mechanophores enable polymers with mechanically switchable white photoluminescence. ACS Cent. Sci. 5, 874–881 (2019).
David, A. H. G., Casares, R., Cuerva, J. M., Campaña, A. G. & Blanco, V. A. [2]rotaxane-based circularly polarized luminescence switch. J. Am. Chem. Soc. 141, 18064–18074 (2019).
Özkan, M., Keser, Y., Hadi, S. E. & Tuncel, D. A. [5]rotaxane-based photosensitizer for photodynamic therapy. Eur. J. Org. Chem. 21, 3534–3541 (2019).
Garci, A. et al. Mechanical-bond-induced exciplex fluorescence in an anthracene-based homo[2]catenane. J. Am. Chem. Soc. 142, 7956–7967 (2020).
Li, W.-J. et al. Rotaxane-branched dendrimers with enhanced photosensitization. J. Am. Chem. Soc. 142, 16748–16756 (2020).
Li, W.-J. et al. AIE-active chiral [3]rotaxanes with switchable circularly polarized luminescence. Angew. Chem. Int. Ed. 60, 9507–9515 (2021).
Rajamalli, P. et al. Using the mechanical bond to tune the performance of a thermally activated delayed fluorescence emitter. Angew. Chem. Int. Ed. 60, 12066–12073 (2021).
Stoddart, J. F. The chemistry of the mechanical bond. Chem. Soc. Rev. 38, 1802–1820 (2009).
Barin, G., Coskun, A., Fouda, M. M. G. & Stoddart, J. F. Mechanically interlocked molecules assembled by π–π recognition. ChemPlusChem 77, 159–185 (2012).
Lewis, J. E. M., Galli, M. & Goldup, S. M. Properties and emerging applications of mechanically interlocked ligands. Chem. Commun. 53, 298–312 (2017).
Barnes, J. C. et al. A radically configurable six-state compound. Science 339, 429–433 (2013).
Jiao, Y. et al. A donor–acceptor [2]catenane for visible light photocatalysis. J. Am. Chem. Soc. 143, 8000–8010 (2021).
Barnes, J. C. et al. ExBox: a polycyclic aromatic hydrocarbon scavenger. J. Am. Chem. Soc. 135, 183–192 (2013).
Juríček, M. et al. Ex2Box: interdependent modes of binding in a two-nanometer-long synthetic receptor. J. Am. Chem. Soc. 135, 12736–12746 (2013).
Juríček, M. et al. An Exbox[2]catenane. Chem. Sci. 5, 2724–2731 (2014).
Lu, Q. et al. Comparison study of the site-effect on regioisomeric pyridyl-pyrene conjugates: synthesis, structures and photophysical properties. J. Org. Chem. 85, 4256–4266 (2020).
Austin, A. et al. A density functional with spherical atom dispersion terms. J. Chem. Theory Comput. 8, 4989–5007 (2012).
Acknowledgements
We thank Northwestern University (NU) for their continued support of this research. This research was also supported by the National Science Foundation under grant no. DMR-2003739 (M.R.W. and R.M.Y., photophysical studies). O.K.F. acknowledges support from the Defense Threat Reduction Agency under award no. HDTRA1-19-1-0010. L.O.J and G.C.S were supported by the Department of Energy, Office of Basic Energy Science as part of the Center for Bioinspired Energy Science under grant DE-SC0000989. We thank C. Lin for his assistance with fluorescence quantum yield measurements and S. Abid and A. H. G. David for helpful discussions. The research made use of the Integrated Molecular Structure and Educational Research Center (IMSERC) at NU, which receives support from the State of Illinois and the International Institute for Nanotechnology (IIN). The research was also supported in part through the computational resources and staff contributions provided for the Quest High Performance Computing Facility at Northwestern University, which is jointly supported by the Office of the Provost, the Office for Research, and Northwestern University Information Technology.
Author information
Authors and Affiliations
Contributions
A.G. and J.F.S. conceived the project. A.G. carried out the synthesis. J.A.W. and L.O.J. conducted the computational study. A.G. and R.M.Y. performed the optical study. A.G., Y.B. and A.A. carried out the photocatalytic experiments. M.O. contributed to the graphical design in the figures. A.G. and W.L. studied the dynamic behaviour of the catenane. C.L.S. resolved the crystal structures. M.K.-R. contributed to the HPLC, electrospray ionization mass spectrometry and NMR titrations. A.G., J.A.W., R.M.Y. and J.F.S. wrote the draft manuscript. All other co-authors contributed to various stages of manuscript preparation.
Corresponding author
Ethics declarations
Competing interests
A.G. and J.F.S. have filed a patent application lodged with Northwestern University based on this work (Invention Disclosure: Disc-ID-22-04-22-002). The other authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Diego Troya and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–68, Tables 1–3, Methods and Note 1.
Supplementary Data 1
Crystal structure of 1,6-1,6PyBox
Supplementary Data 1
Crystal structure of 2,7-2,7PyBox
Supplementary Data 1
Crystal structure of 2,7-2,7PyHC
Supplementary Data 1
Atomic coordinates of DFT models
Rights and permissions
About this article
Cite this article
Garci, A., Weber, J.A., Young, R.M. et al. Mechanically interlocked pyrene-based photocatalysts. Nat Catal 5, 524–533 (2022). https://doi.org/10.1038/s41929-022-00799-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-022-00799-y