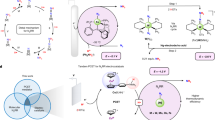
Abstract
Hydride transfers are key to a number of economically and environmentally important reactions, including H2 evolution and NADH regeneration. The electrochemical generation of hydrides can therefore drive the electrification of chemical reactions to improve their sustainability for a green economy. Catalysts containing molybdenum have recently been recognized as among the most promising non-precious catalysts for H2 evolution, but the mechanism by which molybdenum confers this activity remains debated. Here we show the presence of trapped Mo3+ hydride in amorphous molybdenum sulfide (a-MoSx) during the hydrogen evolution reaction and extend its catalytic role to the selective hydrogenation of the biologically important energy carrier NAD to its active 1,4-NADH form. Furthermore, this reactivity applies to other HER-active molybdenum sulfides. Our results demonstrate a direct role for molybdenum in heterogeneous H2 evolution. This mechanistic finding also reveals that molybdenum sulfides have potential as economic electrocatalysts for NADH regeneration in biocatalysis.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data supporting the findings in this study are available either within the paper and its Supplementary Information or from the corresponding authors on reasonable request.
References
Organization of Petroleum Exporting Countries (World Oil Outlook 2040, 2017).
Roger, I., Shipman, M. A. & Symes, M. D. Earth-abundant catalysts for electrochemical and photoelectrochemical water splitting. Nat. Rev. Chem. 1, 0003 (2017).
Montoya, J. H. et al. Materials for solar fuels and chemicals. Nat. Mater. 16, 70–81 (2017).
Seh, Z. W. et al. Combining theory and experiment in electrocatalysis: insights into materials design. Science 355, eaad4998 (2017).
Sheldon, R. A. & Woodley, J. M. Role of biocatalysis in sustainable chemistry. Chem. Rev. 118, 801–838 (2018).
Kibria, M. G. et al. Electrochemical CO2 reduction into chemical feedstocks: from mechanistic electrocatalysis models to system design. Adv. Mater. 31, 1807166 (2019).
Zhang, J. et al. Efficient hydrogen production on MoNi4 electrocatalysts with fast water dissociation kinetics. Nat. Commun. 8, 15437 (2017).
Csernica, P. M. et al. Electrochemical hydrogen evolution at ordered Mo7Ni7. ACS Catal. 7, 3375–3383 (2017).
Bau, J. A. et al. On the reconstruction of NiMo electrocatalysts by operando spectroscopy. J. Mater. Chem. A 7, 15031–15035 (2019).
Jaramillo, T. F. et al. Identification of active edge sites for electrochemical H2 evolution from MoS2 nanocatalysts. Science 317, 100–102 (2007).
Li, H. et al. Activating and optimizing MoS2 basal planes for hydrogen evolution through the formation of strained sulphur vacancies. Nat. Mater. 15, 48–53 (2016).
Ding, Q., Song, B., Xu, P. & Jin, S. Efficient electrocatalytic and photoelectrochemical hydrogen generation using MoS2 and related compounds. Chem 1, 699–726 (2016).
Chia, X. & Pumera, M. Characteristics and performance of two-dimensional materials for electrocatalysis. Nat. Catal. 1, 909 (2018).
Merki, D., Fierro, S., Vrubel, H. & Hu, X. Amorphous molybdenum sulfide films as catalysts for electrochemical hydrogen production in water. Chem. Sci. 2, 1262–1267 (2011).
Tran, P. D. et al. Coordination polymer structure and revisited hydrogen evolution catalytic mechanism for amorphous molybdenum sulfide. Nat. Mater. 15, 640–646 (2016).
Benck, J. D., Chen, Z., Kuritzky, L. Y., Forman, A. J. & Jaramillo, T. F. Amorphous molybdenum sulfide catalysts for electrochemical hydrogen production: insights into the origin of their catalytic activity. ACS Catal. 2, 1916–1923 (2012).
Escalera‐López, D., Lou, Z. & Rees, N. V. Benchmarking the activity, stability, and inherent electrochemistry of amorphous molybdenum sulfide for hydrogen production. Adv. Energy Mater. 9, 1802614 (2019).
Morales-Guio, C. G. & Hu, X. Amorphous molybdenum sulfides as hydrogen evolution catalysts. Acc. Chem. Res. 47, 2671–2681 (2014).
Zhuang, Z., Huang, J., Li, Y., Zhou, L. & Mai, L. The holy grail in platinum-free electrocatalytic hydrogen evolution: molybdenum-based catalysts and recent advances. ChemElectroChem 6, 3570–3589 (2019).
Hinnemann, B. et al. Biomimetic hydrogen evolution: MoS2 nanoparticles as catalyst for hydrogen evolution. J. Am. Chem. Soc. 127, 5308–5309 (2005).
Cao, Y. Roadmap and direction toward high-performance MoS2 hydrogen evolution catalysts. ACS Nano 15, 11014–11039 (2021).
Lassalle-Kaiser, B. et al. Evidence from in situ X-ray absorption spectroscopy for the involvement of terminal disulfide in the reduction of protons by an amorphous molybdenum sulfide electrocatalyst. J. Am. Chem. Soc. 137, 314–321 (2015).
Xi, F. et al. Structural transformation identification of sputtered amorphous mosx as an efficient hydrogen-evolving catalyst during electrochemical activation. ACS Catal. 9, 2368–2380 (2019).
Huang, Y., Nielsen, R. J., Goddard, W. A. & Soriaga, M. P. The reaction mechanism with free energy barriers for electrochemical dihydrogen evolution on MoS2. J. Am. Chem. Soc. 137, 6692–6698 (2015).
Karunadasa, H. I., Chang, C. J. & Long, J. R. A molecular molybdenum-oxo catalyst for generating hydrogen from water. Nature 464, 1329–1333 (2010).
Prior, C. et al. EPR detection and characterisation of a paramagnetic Mo(III) dihydride intermediate involved in electrocatalytic hydrogen evolution. Dalton Trans. 45, 2399–2403 (2016).
Ahn, H. S. & Bard, A. J. Electrochemical surface interrogation of a MoS2 hydrogen-evolving catalyst: in situ determination of the surface hydride coverage and the hydrogen evolution kinetics. J. Phys. Chem. Lett. 7, 2748–2752 (2016).
Sellés Vidal, L., Kelly, C. L., Mordaka, P. M. & Heap, J. T. Review of NAD(P)H-dependent oxidoreductases: properties, engineering and application. Biochim. Biophys. Acta Proteins Proteom. 1866, 327–347 (2018).
Wu, H. et al. Methods for the regeneration of nicotinamide coenzymes. Green. Chem. 15, 1773–1789 (2013).
Faber, K. in Biotransformations in Organic Chemistry: A Textbook (ed. Faber, K.) 31–313 (Springer, 2011); https://doi.org/10.1007/978-3-642-17393-6_2
Wang, X. et al. Cofactor NAD(P)H regeneration inspired by heterogeneous pathways. Chem. 2, 621–654 (2017).
Ali, I., Khan, T. & Omanovic, S. Direct electrochemical regeneration of the cofactor NADH on bare Ti, Ni, Co and Cd electrodes: the influence of electrode potential and electrode material. J. Mol. Catal. A 387, 86–91 (2014).
Trasatti, S. Work function, electronegativity, and electrochemical behaviour of metals: III. Electrolytic hydrogen evolution in acid solutions. J. Electroanal. Chem. Interfacial Electrochem. 39, 163–184 (1972).
González, J. R., Alcántara, R., Tirado, J. L., Fielding, A. J. & Dryfe, R. A. W. Electrochemical interaction of few-layer molybdenum disulfide composites vs sodium: new insights on the reaction mechanism. Chem. Mater. 29, 5886–5895 (2017).
Sathiya, M. et al. Electron paramagnetic resonance imaging for real-time monitoring of Li-ion batteries. Nat. Commun. 6, 6276 (2015).
Yin, Y. et al. Contributions of phase, sulfur vacancies, and edges to the hydrogen evolution reaction catalytic activity of porous molybdenum disulfide nanosheets. J. Am. Chem. Soc. 138, 7965–7972 (2016).
Cai, L. et al. Vacancy-Induced ferromagnetism of MoS2 nanosheets. J. Am. Chem. Soc. 137, 2622–2627 (2015).
Meija, J. et al. Isotopic compositions of the elements 2013 (IUPAC technical report). Pure Appl. Chem. 88, 293–306 (2016).
Stoll, S. & Schweiger, A. EasySpin, a comprehensive software package for spectral simulation and analysis in EPR. J. Magn. Reson. 178, 42–55 (2006).
Lu, Z. et al. Ultrahigh hydrogen evolution performance of under-water ‘superaerophobic’ MoS2 nanostructured electrodes. Adv. Mater. 26, 2683–2687 (2014).
Aljarb, A. et al. Ledge-directed epitaxy of continuously self-aligned single-crystalline nanoribbons of transition metal dichalcogenides. Nat. Mater. 19, 1300–1306 (2020).
Busetto, L., Vaccari, A. & Martini, G. Electron spin resonance of paramagnetic species as a tool for studying the thermal decomposition of molybdenum trisulfide. J. Phys. Chem. 85, 1927–1930 (1981).
Bullock, R. M. in Catalysis Without Precious Metals 51–81 (Wiley, 2010); https://doi.org/10.1002/9783527631582.ch3
Micheletti Moracci, F. et al. Electrochemical reduction of 1-benzyl-3-carbamoylpyridinium chloride, a nicotinamide adenine dinucleotide model compound. J. Org. Chem. 43, 3420–3422 (1978).
Schmakel, C. O., Santhanam, K. S. V. & Elving, P. J. Nicotinamide and N′‐methylnicotinamide: electrochemical redox pattern: behavior of free radical, dimeric, and dihydropyridine species. J. Electrochem. Soc. 121, 345 (1974).
Erb, C. et al. Formation of N-methylnicotinamide in the brain from a dihydropyridine-type prodrug: effect on brain choline. Biochem. Pharmacol. 57, 681–684 (1999).
Burnett, J. W. H., Howe, R. F. & Wang, X. Cofactor NAD(P)H regeneration: how selective are the reactions? Trends Chem. 2, 488–492 (2020).
Wilson, D. F., Erecińska, M. & Dutton, P. L. Thermodynamic relationships in mitochondrial oxidative phosphorylation. Annu. Rev. Biophysics Bioeng. 3, 203–230 (1974).
Godtfredsen, S. E., Ottesen, M. & Andersen, N. R. On the mode of formation of 1,6-dihydro-NAD in NADH preparations. Carlsberg Res. Commun. 44, 65 (1979).
Burnett, J. W. H. et al. Directing the H2-driven selective regeneration of NADH via Sn-doped Pt/SiO2. Green. Chem. 24, 1451–1455 (2022).
Steckhan, E. in Electrochemistry V (ed. Steckhan, E.) 83–111 (Springer, 1994); https://doi.org/10.1007/3-540-57729-7_3
Ali, I., Soomro, B. & Omanovic, S. Electrochemical regeneration of NADH on a glassy carbon electrode surface: the influence of electrolysis potential. Electrochem. Commun. 13, 562–565 (2011).
Forni, L. G. & Willson, R. L. Thiyl and phenoxyl free radicals and NADH Direct observation of one-electron oxidation. Biochem. J. 240, 897–903 (1986).
Vera-Hidalgo, M., Giovanelli, E., Navío, C. & Pérez, E. M. Mild covalent functionalization of transition metal dichalcogenides with maleimides: a ‘click’ reaction for 2H-MoS2 and WS2. J. Am. Chem. Soc. 141, 3767–3771 (2019).
Deng, Y. et al. Operando raman spectroscopy of amorphous molybdenum sulfide (MoSx) during the electrochemical hydrogen evolution reaction: identification of sulfur atoms as catalytically active sites for H+ reduction. ACS Catal. 6, 7790–7798 (2016).
Chen, J. et al. Ag@MoS2 core–shell heterostructure as sers platform to reveal the hydrogen evolution active sites of single-layer MoS2. J. Am. Chem. Soc. 142, 7161–7167 (2020).
Ting, L. R. L. et al. Catalytic activities of sulfur atoms in amorphous molybdenum sulfide for the electrochemical hydrogen evolution reaction. ACS Catal. 6, 861–867 (2016).
Han, J. et al. Design, synthesis, and biological activity of novel dicoumarol glucagon-like peptide 1 conjugates. J. Med. Chem. 56, 9955–9968 (2013).
Fu, X. et al. Targeted determination of tissue energy status by LC-MS/MS. Anal. Chem. 91, 5881–5887 (2019).
Acknowledgements
This work has been supported by King Abdullah University of Science and Technology (KAUST). J.A.B. acknowledges S. Sioud from KAUST Analytical Core Labs for assistance with UHPLC/MS, and D. Renn of KAUST for assistance with procuring materials, images and equipment for biocatalytic experiments. V.T. and A.A. are indebted to the support from the King Abdullah University of Science and Technology (KAUST) Office of Sponsored Research (OSR) under award no. OSR-2018-CARF/CCF-3079.
Author information
Authors and Affiliations
Contributions
J.A.B. and M.R. conceived and designed the experiments and wrote the manuscript. J.A.B. performed the experiments and analysed the data. A.-H.E. assisted with the design and carrying out of the EPR and NMR experiments. P.N. assisted with the design of the NMR experiments for NMN reduction. A.A.A. and V.T. provided defect-free MoS2. J.A.B, V.T., and M.R. discussed and revised the manuscript. All authors read the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Vasily Oganesyan, Jiafu Shi and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–24, and Tables 1 and 2.
Rights and permissions
About this article
Cite this article
Bau, J.A., Emwas, AH., Nikolaienko, P. et al. Mo3+ hydride as the common origin of H2 evolution and selective NADH regeneration in molybdenum sulfide electrocatalysts. Nat Catal 5, 397–404 (2022). https://doi.org/10.1038/s41929-022-00781-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-022-00781-8
This article is cited by
-
Catalytic hydrogenation of olefins by a multifunctional molybdenum-sulfur complex
Nature Communications (2024)