Abstract
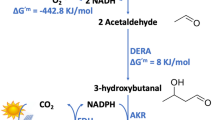
Upcycling of carbon dioxide (CO2) into value-added products represents a substantially untapped opportunity to tackle environmental issues and achieve a circular economy. Compared with easily available C1/C2 products, nevertheless, efficient and sustainable synthesis of energy-rich long-chain compounds from CO2 still remains a grand challenge. Here we describe a hybrid electro-biosystem, coupling spatially separate CO2 electrolysis with yeast fermentation, that efficiently converts CO2 to glucose with a high yield. We employ a nanostructured copper catalyst that can stably catalyse pure acetic acid production with a solid-electrolyte reactor. We then genetically engineer Saccharomyces cerevisiae to produce glucose in vitro from electro-generated acetic acid by deleting all defined hexokinase genes and overexpression of heterologous glucose-1-phosphatase. In addition, we showcase that the proposed platform can be easily extended to produce other products like fatty acids using CO2 as the carbon source. These results illuminate the tantalizing possibility of a renewable-electricity-driven manufacturing industry.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request. Source data are provided with this paper.
References
Rode, A. et al. Estimating a social cost of carbon for global energy consumption. Nature 598, 308–314 (2021).
Bushuyev, O. S. et al. What should we make with CO2 and how can we make it? Joule 2, 825–832 (2018).
Grim, R. G. et al. Transforming the carbon economy: challenges and opportunities in the convergence of low-cost electricity and reductive CO2 utilization. Energy Environ. Sci. 13, 472–494 (2020).
Zhang, B. & Sun, L. Artificial photosynthesis: opportunities and challenges of molecular catalysts. Chem. Soc. Rev. 48, 2216–2264 (2019).
Peng, C. et al. Double sulfur vacancies by lithium tuning enhance CO2 electroreduction to n-propanol. Nat. Commun. 12, 1580 (2021).
Zhu, Y. et al. Continuous artificial synthesis of glucose precursor using enzyme-immobilized microfluidic reactors. Nat. Commun. 10, 4049 (2019).
De Carvalho, C. C. C. R. & José Caramujo, M. The various roles of fatty acids. Molecules 23, 2583 (2018).
Berardi, S. et al. Molecular artificial photosynthesis. Chem. Soc. Rev. 43, 7501–7519 (2014).
Bonke, S. A., Wiechen, M., MacFarlane, D. R. & Spiccia, L. Renewable fuels from concentrated solar power: towards practical artificial photosynthesis. Energy Environ. Sci. 8, 2791–2796 (2015).
Chen, H., Dong, F. & Minteer, S. D. The progress and outlook of bioelectrocatalysis for the production of chemicals, fuels and materials. Nat. Catal. 3, 225–244 (2020).
Liu, Y. et al. Biofuels for a sustainable future. Cell 184, 1636–1647 (2021).
Cherry, J. M. et al. Genetic and physical maps of Saccharomyces cerevisiae. Nature 387, 67–73 (1997).
Liu, C. et al. Nanowire-bacteria hybrids for unassisted solar carbon dioxide fixation to value-added chemicals. Nano Lett. 15, 3634–3639 (2015).
Su, Y. et al. Close-packed nanowire-bacteria hybrids for efficient solar-driven CO2 fixation. Joule 4, 800–811 (2020).
Guo, J. et al. Light-driven fine chemical production in yeast biohybrids. Science 362, 813–816 (2018).
Tremblay, P.-L. & Zhang, T. Electrifying microbes for the production of chemicals. Front. Microbiol. 6, 201 (2015).
Claassens, N. J., Cotton, C. A. R., Kopljar, D. & Bar-Even, A. Making quantitative sense of electromicrobial production. Nat. Catal. 2, 437–447 (2019).
Haas, T., Krause, R., Weber, R., Demler, M. & Schmid, G. Technical photosynthesis involving CO2 electrolysis and fermentation. Nat. Catal. 1, 32–39 (2018).
Cai, T. et al. Cell-free chemoenzymatic starch synthesis from carbon dioxide. Science 373, 1523–1527 (2021).
Li, K., Peng, B. & Peng, T. Recent advances in heterogeneous photocatalytic CO2 conversion to solar fuels. ACS Catal. 6, 7485–7527 (2016).
Novak, K., Kutscha, R. & Pflügl, S. Microbial upgrading of acetate into 2, 3-butanediol and acetoin by E. coli W. Biotechnol. Biofuels 13, 177 (2020).
Zhang, R. et al. An in vitro synthetic biosystem based on acetate for production of phloroglucinol. BMC Biotechnol. 17, 66 (2017).
Liu, Y. et al. Steering CO2 electroreduction toward ethanol production by a surface-bound Ru polypyridyl carbene catalyst on N-doped porous carbon. Proc. Natl Acad. Sci. USA 116, 26353–26358 (2019).
Rong, W. et al. Size-dependent activity and selectivity of atomic-level copper nanoclusters during CO/CO2 electroreduction. Angew. Chem. Int. Ed. 60, 466–472 (2021).
Saxena, A., Liyanage, W., Masud, J., Kapila, S. & Nath, M. Selective electroreduction of CO2 to carbon-rich products with a simple binary copper selenide electrocatalyst. J. Mater. Chem. A 9, 7150–7161 (2021).
Wei, X. et al. Highly selective reduction of CO2 to C2+ hydrocarbons at copper/polyaniline interfaces. ACS Catal. 10, 4103–4111 (2020).
Jouny, M., Luc, W. & Jiao, F. High-rate electroreduction of carbon monoxide to multi-carbon products. Nat. Catal. 1, 748–755 (2018).
Ripatti, D. S., Veltman, T. R. & Kanan, M. W. Carbon monoxide gas diffusion electrolysis that produces concentrated C2 products with high single-pass conversion. Joule 3, 240–256 (2019).
Bushuyev, O. S. et al. What should we make with CO2 and how can we make it? Joule 2, 825–832 (2018).
Orella, M. J., Brown, S. M., Leonard, M. E., Román-Leshkov, Y. & Brushett, F. R. A general technoeconomic model for evaluating emerging electrolytic processes. Energy Technol. 8, 1900994 (2019).
Wang, X. et al. Efficient upgrading of CO to C3 fuel using asymmetric CC coupling active sites. Nat. Commun. 10, 5186 (2019).
Zheng, T. et al. Large-scale and highly selective CO2 electrocatalytic reduction on nickel single-atom catalyst. Joule 3, 265–278 (2019).
Kortlever, R., Shen, J., Schouten, K. J. P., Calle-Vallejo, F. & Koper, M. T. Catalysts and reaction pathways for the electrochemical reduction of carbon dioxide. J. Phys. Chem. Lett. 6, 4073–4082 (2015).
Schouten, K., Kwon, Y., Van der Ham, C., Qin, Z. & Koper, M. A new mechanism for the selectivity to C1 and C2 species in the electrochemical reduction of carbon dioxide on copper electrodes. Chem. Sci. 2, 1902–1909 (2011).
Bao, H. et al. Isolated copper single sites for high-performance electroreduction of carbon monoxide to multicarbon products. Nat. Commun. 12, 238 (2021).
Li, C. W., Ciston, J. & Kanan, M. W. Electroreduction of carbon monoxide to liquid fuel on oxide-derived nanocrystalline copper. Nature 508, 504–507 (2014).
Luc, W. et al. Two-dimensional copper nanosheets for electrochemical reduction of carbon monoxide to acetate. Nat. Catal. 2, 423–430 (2019).
Zhu, P. et al. Direct and continuous generation of pure acetic acid solutions via electrocatalytic carbon monoxide reduction. Proc. Natl Acad. Sci. USA 118, e2010868118 (2021).
Zhu, Q. et al. Carbon dioxide electroreduction to C2 products over copper-cuprous oxide derived from electrosynthesized copper complex. Nat. Commun. 10, 3851 (2019).
Lum, Y., Cheng, T., Goddard, W. A. III & Ager, J. W. Electrochemical CO reduction builds solvent water into oxygenate products. J. Am. Chem. Soc. 140, 9337–9340 (2018).
Choi, C. et al. Highly active and stable stepped Cu surface for enhanced electrochemical CO2 reduction to C2H4. Nat. Catal. 3, 804–812 (2020).
Goodpaster, J. D., Bell, A. T. & Head-Gordon, M. Identification of possible pathways for C–C bond formation during electrochemical reduction of CO2: new theoretical insights from an improved electrochemical model. J. Phys. Chem. Lett. 7, 1471–1477 (2016).
Montoya, J. H., Peterson, A. A. & Nørskov, J. K. Insights into C—C coupling in CO2 electroreduction on copper electrodes. ChemCatChem 5, 737–742 (2013).
Ferreira, I., Pinho, O., Vieira, E. & Tavarela, J. Brewer’s Saccharomyces yeast biomass: characteristics and potential applications. Trends Food Sci. Tech. 21, 77–84 (2010).
Sharif, M. et al. Single cell protein: sources, mechanism of production, nutritional value and its uses in aquaculture nutrition. Aquaculture 531, 735885 (2021).
Johnson, E. A. & Echavarri-Erasun, C. The Yeasts 21–44 (Elsevier, 2011).
Eibl, R. et al. Cellular agriculture: opportunities and challenges. Annu. Rev. Food Sci. Technol. 12, 51–73 (2021).
Yu, T. et al. Reprogramming yeast metabolism from alcoholic fermentation to lipogenesis. Cell 174, 1549–1558 (2018).
Westfall, P. J. et al. Production of amorphadiene in yeast, and its conversion to dihydroartemisinic acid, precursor to the antimalarial agent artemisinin. Proc. Natl Acad. Sci. USA 109, 111–118 (2012).
Nielsen, J. & Keasling, J. D. Engineering cellular metabolism. Cell 164, 1185–1197 (2016).
Wilson, J. Hexokinases. Rev. Physiol. Biochem. Pharmacol. 126, 65–198 (1995).
Suleimanova, A. D. et al. Novel glucose-1-phosphatase with high phytase activity and unusual metal ion activation from soil bacterium Pantoea sp. strain 3.5.1. Appl. Environ. Microbiol. 81, 6790–6799 (2015).
Pfeiffer, M., Wildberger, P. & Nidetzky, B. Yihx-encoded haloacid dehalogenase-like phosphatase HAD4 from Escherichia coli is a specific alpha-d-glucose 1-phosphate hydrolase useful for substrate-selective sugar phosphate transformations. J. Mol. Catal. B Enzym. 110, 39–46 (2014).
Dimian, A. C. & Kiss, A. A. Novel energy efficient process for acetic acid production by methanol carbonylation. Chem. Eng. Res. Des. 159, 1–12 (2020).
Wysocki, R. & Tamás, M. J. How Saccharomyces cerevisiae copes with toxic metals and metalloids. FEMS Microbiol. Rev. 34, 925–951 (2010).
Xia, C. et al. Continuous production of pure liquid fuel solutions via electrocatalytic CO2 reduction using solid-electrolyte devices. Nat. Energy 4, 776–785 (2019).
Xia, C., Xia, Y., Zhu, P., Fan, L. & Wang, H. Direct electrosynthesis of pure aqueous H2O2 solutions up to 20% by weight using a solid electrolyte. Science 366, 226–231 (2019).
Zarkevich, N. A. & Johnson, D. D. Nudged-elastic band method with two climbing images: finding transition states in complex energy landscapes. J. Chem. Phys. 142, 024106 (2015).
Acknowledgements
J. Zeng acknowledges the National Key Research and Development Program of China (2021YFA1500500, 2019YFA0405600), National Science Fund for Distinguished Young Scholars (21925204), National Natural Science Foundation of China (NSFC; U19A2015), the Dalian National Laboratory (DNL) Cooperation Fund, Chinese Academy of Science (CAS; DNL202003), K. C.Wong Education (GJTD-2020-15), Fundamental Research Funds for the Central Universities, Provincial Key Research and Development Program of Anhui (202004a05020074) and University of Science and Technology of China (USTC) Research Funds of the Double First-Class Initiative (YD2340002002). C.X. acknowledges the NSFC (22102018 and 52171201), the Central Government Funds of Guiding Local Scientific and Technological Development for Sichuan Province (no. 2021ZYD0043) and the University of Electronic Science and Technology of China for startup funding (A1098531023601264). T.Y. acknowledges the NSFC (32071416), the National Key Research and Development Program of China (2020YFA0907800 and 2021YFA0911000), the Shenzhen Institute of Synthetic Biology Scientific Research Program (grant no. JCHZ20200003) and Shenzhen Key Laboratory for the Intelligent Microbial Manufacturing of Medicines. T.Z. acknowledges the NSFC (22005291) and University of Electronic Science and Technology of China for startup funding (A1098531023601356). We thank beamline BL14W1 of Shanghai Synchrotron Radiation Facility for providing the beamtime. We also thank F. Jin for helpful discussions.
Author information
Authors and Affiliations
Contributions
The project was conceptualized and supervised by J. Zeng, T.Y. and C.X.; T.Z. and M.Z. prepared the catalysts and performed the catalytic tests. L.W., X. Liu and S.G. performed the yeast fermentation. T.Z., M.Z., W.X. and J.L. performed the catalyst characterizations. J. Zhao carried out density functional theory calculations. X. Li, C.L. and Q.J. performed the X-ray absorption fine structure measurements. J.B. helped in the analysis of data. T.Z., C.X., T.Y. and J. Zeng wrote the paper with input from all authors. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Jens Nielsen and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–24 and Tables 1–9.
Source data
Source Data Fig. 2
Statistical source data.
Rights and permissions
About this article
Cite this article
Zheng, T., Zhang, M., Wu, L. et al. Upcycling CO2 into energy-rich long-chain compounds via electrochemical and metabolic engineering. Nat Catal 5, 388–396 (2022). https://doi.org/10.1038/s41929-022-00775-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-022-00775-6
This article is cited by
-
Microbial conversion of carbon dioxide into premium medium-chain fatty acids: the progress, challenges, and prospects
npj Materials Sustainability (2024)
-
Synthesis of hydroxylamine from air and water via a plasma-electrochemical cascade pathway
Nature Sustainability (2024)
-
Solar-driven sugar production directly from CO2 via a customizable electrocatalytic–biocatalytic flow system
Nature Communications (2024)
-
Compositional and temporal division of labor modulates mixed sugar fermentation by an engineered yeast consortium
Nature Communications (2024)
-
Cascade electrolysis and thermocatalysis: a reliable system for upgrading C1 to C4 hydrocarbons
Rare Metals (2024)