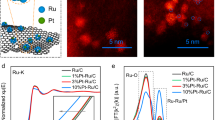
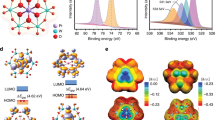
Abstract
Modifying Pt surfaces by foreign metals with a higher oxophilicity is a promising approach to improve the kinetics of hydrogen evolution and oxidation reactions. The role of foreign metals, however, is not fully understood. Here we study Ru-modified Pt as a model system to understand the underlying mechanisms in activity improvement through combining in situ infrared spectroscopy and theoretical calculations. In an alkaline solution, the hydrogen evolution and oxidation reaction activity of Ru-modified Pt is proportional to the Ru coverage due to the strain and electronic effects of the Pt substrate, which lower the energy barrier of the rate-determining Volmer step; these are the governing reasons for the improved catalytic activities. This work not only deepens our understanding of hydrogen electrocatalysis mechanisms, but also provides guidelines for the rational design of advanced electrocatalysts.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the article and its Supplementary Information files or from the corresponding author upon reasonable request. The atomic coordinates of the optimized models are provided in Supplementary Data 1. Source data are provided with this paper.
References
Bockris, J. O. M. The origin of ideas on a hydrogen economy and its solution to the decay of the environment. Int. J. Hydrog. Energy 27, 731–740 (2002).
Shao, M., Chang, Q., Dodelet, J.-P. & Chenitz, R. Recent advances in electrocatalysts for oxygen reduction reaction. Chem. Rev. 116, 3594–3657 (2016).
Diaz-Morales, O., Ledezma-Yanez, I., Koper, M. T. & Calle-Vallejo, F. Guidelines for the rational design of Ni-based double hydroxide electrocatalysts for the oxygen evolution reaction. ACS Catal. 5, 5380–5387 (2015).
Friebel, D. et al. Identification of highly active Fe sites in (Ni,Fe)OOH for electrocatalytic water splitting. J. Am. Chem. Soc. 137, 1305–1313 (2015).
Wu, G. & Zelenay, P. Nanostructured nonprecious metal catalysts for oxygen reduction reaction. Acc. Chem. Res. 46, 1878–1889 (2013).
Durst, J. et al. New insights into the electrochemical hydrogen oxidation and evolution reaction mechanism. Energy Environ. Sci. 7, 2255–2260 (2014).
Zheng, J., Sheng, W., Zhuang, Z., Xu, B. & Yan, Y. Universal dependence of hydrogen oxidation and evolution reaction activity of platinum-group metals on pH and hydrogen binding energy. Sci. Adv. 2, e1501602 (2016).
Subbaraman, R. et al. Enhancing hydrogen evolution activity in water splitting by tailoring Li+–Ni(OH)2–Pt interfaces. Science 334, 1256–1260 (2011).
Subbaraman, R. et al. Trends in activity for the water electrolyser reactions on 3d M(Ni,Co,Fe,Mn) hydr(oxy)oxide catalysts. Nat. Mater. 11, 550–557 (2012).
Strmcnik, D. et al. Improving the hydrogen oxidation reaction rate by promotion of hydroxyl adsorption. Nat. Chem. 5, 300–306 (2013).
Liu, W., Lyu, K., Xiao, L., Lu, J. & Zhuang, L. Hydrogen oxidation reaction on modified platinum model electrodes in alkaline media. Electrochim. Acta 327, 135016 (2019).
Intikhab, S., Snyder, J. D. & Tang, M. H. Adsorbed hydroxide does not participate in the Volmer step of alkaline hydrogen electrocatalysis. ACS Catal. 7, 8314–8319 (2017).
Liu, E. et al. Unifying the hydrogen evolution and oxidation reactions kinetics in base by identifying the catalytic roles of hydroxyl–water–cation adducts. J. Am. Chem. Soc. 141, 3232–3239 (2019).
Wang, Y. et al. Pt–Ru catalyzed hydrogen oxidation in alkaline media: oxophilic effect or electronic effect? Energy Environ. Sci. 8, 177–181 (2015).
Nash, J., Zheng, J., Wang, Y., Xu, B. & Yan, Y. Mechanistic study of the hydrogen oxidation/evolution reaction over bimetallic PtRu catalysts. J. Electrochem. Soc. 165, J3378–J3383 (2018).
Elbert, K. et al. Elucidating hydrogen oxidation/evolution kinetics in base and acid by enhanced activities at the optimized Pt shell thickness on the Ru core. ACS Catal. 5, 6764–6772 (2015).
Wang, X. et al. Strain effect in bimetallic electrocatalysts in the hydrogen evolution reaction. ACS Energy Lett. 3, 1198–1204 (2018).
Schwämmlein, J. N. et al. Origin of superior HOR/HER activity of bimetallic Pt–Ru catalysts in alkaline media identified via Ru@Pt core–shell nanoparticles. J. Electrochem. Soc. 165, H229–H239 (2018).
Li, J. et al. Experimental proof of the bifunctional mechanism for the hydrogen oxidation in alkaline media. Angew. Chem. Int. Ed. 56, 15594–15598 (2017).
Liu, E. et al. Interfacial water shuffling the intermediates of hydrogen oxidation and evolution reactions in aqueous media. Energy Environ. Sci. 13, 3064–3074 (2020).
McCrum, I. T. & Koper, M. T. The role of adsorbed hydroxide in hydrogen evolution reaction kinetics on modified platinum. Nat. Energy 5, 891–899 (2020).
Giles, S. A. et al. Recent advances in understanding the pH dependence of the hydrogen oxidation and evolution reactions. J. Catal. 367, 328–331 (2018).
Ledezma-Yanez, I. et al. Interfacial water reorganization as a pH-dependent descriptor of the hydrogen evolution rate on platinum electrodes. Nat. Energy 2, 17031 (2017).
Chrzanowski, W. & Wieckowski, A. Ultrathin films of ruthenium on low index platinum single crystal surfaces: an electrochemical study. Langmuir 13, 5974–5978 (1997).
Ohyama, J., Sato, T., Yamamoto, Y., Arai, S. & Satsuma, A. Size specifically high activity of Ru nanoparticles for hydrogen oxidation reaction in alkaline electrolyte. J. Am. Chem. Soc. 135, 8016–8021 (2013).
Green, C. L. & Kucernak, A. Determination of the platinum and ruthenium surface areas in platinum–ruthenium alloy electrocatalysts by underpotential deposition of copper. I. Unsupported catalysts. J. Phys. Chem. B 106, 1036–1047 (2002).
Sheng, W., Myint, M., Chen, J. G. & Yan, Y. Correlating the hydrogen evolution reaction activity in alkaline electrolytes with the hydrogen binding energy on monometallic surfaces. Energy Environ. Sci. 6, 1509–1512 (2013).
Alia, S. M. & Pivovar, B. S. Evaluating hydrogen evolution and oxidation in alkaline media to establish baselines. J. Electrochem. Soc. 165, F441 (2018).
Zhu, S., Qin, X., Yao, Y. & Shao, M. pH-dependent hydrogen and water binding energies on platinum surfaces as directly probed through surface-enhanced infrared absorption spectroscopy. J. Am. Chem. Soc. 142, 8748–8754 (2020).
Kubota, J. & Aika, K.-i. FTIR spectra of terminal and bridged hydrogen atoms adsorbed on ruthenium metal particles dispersed on magnesium oxide. J. Chem. Soc. Chem. Commun. 1992, 661–662 (1992).
Adzic, R. R. et al. Platinum monolayer fuel cell electrocatalysts. Top. Catal. 46, 249–262 (2007).
ter Veen, H. R. J., Kim, T., Wachs, I. E. & Brongersma, H. H. Applications of high sensitivity–low energy ion scattering (HS-LEIS) in heterogeneous catalysis. Catal. Today 140, 197–201 (2009).
Osawa, M., Tsushima, M., Mogami, H., Samjeske, G. & Yamakata, A. Structure of water at the electrified platinum–water interface: a study by surface-enhanced infrared absorption spectroscopy. J. Phys. Chem. C 112, 4248–4256 (2008).
Danilovic, N. et al. The effect of noncovalent interactions on the HOR, ORR, and HER on Ru, Ir, and Ru0.50Ir0.50 metal surfaces in alkaline environments. Electrocatalysis 3, 221–229 (2012).
Marković, N. M. & Ross, P. N. Surface science studies of model fuel cell electrocatalysts. Surf. Sci. Rep. 45, 117–229 (2002).
Maier, S., Lechner, B. A., Somorjai, G. A. & Salmeron, M. Growth and structure of the first layers of ice on Ru(0001) and Pt(111). J. Am. Chem. Soc. 138, 3145–3151 (2016).
Ogasawara, H. et al. Structure and bonding of water on Pt(111). Phys. Rev. Lett. 89, 276102 (2002).
Lindgren, P., Kastlunger, G. & Peterson, A. A. A challenge to the G ≈ 0 interpretation of hydrogen evolution. ACS Catal. 10, 121–128 (2020).
Skúlason, E. et al. Modeling the electrochemical hydrogen oxidation and evolution reactions on the basis of density functional theory calculations. J. Phys. Chem. C 114, 18182–18197 (2010).
Brankovic, S. R. et al. Electrosorption and catalytic properties of bare and Pt modified single crystal and nanostructured Ru surfaces. J. Electroanal. Chem. 524–525, 231–241 (2002).
Herrero, E., Feliu, J. & Wieckowski, A. Scanning tunneling microscopy images of ruthenium submonolayers spontaneously deposited on a Pt(111) electrode. Langmuir 15, 4944–4948 (1999).
Jung, C., Kim, J. & Rhee, C. K. CO preoxidation on Ru-modified Pt(111). Electrochem. Commun. 12, 1363–1366 (2010).
Sheng, W., Gasteiger, H. A. & Shao-Horn, Y. Hydrogen oxidation and evolution reaction kinetics on platinum: acid vs alkaline electrolytes. J. Electrochem. Soc. 157, B1529 (2010).
Yan, Y.-G. et al. Ubiquitous strategy for probing ATR surface-enhanced infrared absorption at platinum group metal–electrolyte interfaces. J. Phys. Chem. B 109, 7900–7906 (2005).
Zhu, S., Jiang, B., Cai, W.-B. & Shao, M. Direct observation on reaction intermediates and the role of bicarbonate anions in CO2 electrochemical reduction reaction on Cu surfaces. J. Am. Chem. Soc. 139, 15664–15667 (2017).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558–561 (1993).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Hammer, B., Hansen, L. B. & Nørskov, J. K. Improved adsorption energetics within density-functional theory using revised Perdew–Burke–Ernzerhof functionals. Phys. Rev. B 59, 7413–7421 (1999).
Methfessel, M. & Paxton, A. High-precision sampling for Brillouin-zone integration in metals. Phys. Rev. B 40, 3616–3621 (1989).
Klimeš, J., Bowler, D. R. & Michaelides, A. Chemical accuracy for the van der Waals density functional. J. Phys. Condens. Matter 22, 022201 (2009).
Klimeš, J., Bowler, D. R. & Michaelides, A. Van der Waals density functionals applied to solids. Phys. Rev. B 83, 195131 (2011).
Sakong, S., Forster-Tonigold, K. & Groß, A. The structure of water at a Pt(111) electrode and the potential of zero charge studied from first principles. J. Chem. Phys. 144, 194701 (2016).
Henkelman, G. & Jónsson, H. Improved tangent estimate in the nudged elastic band method for finding minimum energy paths and saddle points. J. Chem. Phys. 113, 9978–9985 (2000).
Henkelman, G., Uberuaga, B. P. & Jónsson, H. A climbing image nudged elastic band method for finding saddle points and minimum energy paths. J. Chem. Phys. 113, 9901–9904 (2000).
Sheppard, D., Terrell, R. & Henkelman, G. Optimization methods for finding minimum energy paths. J. Chem. Phys. 128, 134106 (2008).
Acknowledgements
This work was supported by the Research Grant Council (16308420, 16310419 and C6011-20G, received by M.S.) of the Hong Kong Special Administrative Region, Hong Kong Branch of Southern Marine Science and Engineering Guangdong Laboratory (Guangzhou) (SMSEGL20SC01, received by M.S.), Hong Kong Innovation and Technology Commission (ITC-CNERC14EG03, received by M.S.), Opening Project of the State Key Laboratory of Physical Chemistry of Solid Surfaces at Xiamen University (201903, received by M.S.) collaborated with B. Ren, the Tianhe-2 National Supercomputer Center in Guangzhou and the Research Grants Council Postdoctoral Fellowship Scheme (PDFS2021-6S08, received by S.Z.). The authors thank L. Weng from HKUST for fruitful discussions, and N. Ho from MCPF of HKUST for XPS measurements.
Author information
Authors and Affiliations
Contributions
M.S. supervised the project. S.Z. and M.S. conceived the idea and designed the experiments. S.Z. performed the electrochemical and spectroscopic measurements. X.Q. carried out the simulations. S.Z., F.X., Z.T., J.L. and J.Y. performed the material preparation and general characterizations. Y.X. and Q.C. conducted the atomic force microscopy characterizations. S.Y. and M.C. performed the high-sensitivity low-energy ion scattering measurements. S.Z., X.Q. and M.S. analysed the data and wrote the manuscript. All the authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Catalysis thanks Eduardo Gracia-Espino and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–14, Discussion and Tables 1–4.
Supplementary Data 1
Atomic coordinates of the optimized models.
Source data
Source Data Fig. 1
Statistical Source Data.
Source Data Fig. 2
Statistical Source Data.
Source Data Fig. 3
Statistical Source Data.
Source Data Fig. 4
Statistical Source Data.
Rights and permissions
About this article
Cite this article
Zhu, S., Qin, X., Xiao, F. et al. The role of ruthenium in improving the kinetics of hydrogen oxidation and evolution reactions of platinum. Nat Catal 4, 711–718 (2021). https://doi.org/10.1038/s41929-021-00663-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-021-00663-5
This article is cited by
-
A strongly coupled Ru–CrOx cluster–cluster heterostructure for efficient alkaline hydrogen electrocatalysis
Nature Catalysis (2024)
-
Implanting oxophilic metal in PtRu nanowires for hydrogen oxidation catalysis
Nature Communications (2024)
-
Facilitating alkaline hydrogen evolution reaction on the hetero-interfaced Ru/RuO2 through Pt single atoms doping
Nature Communications (2024)
-
Unveiling the nature of Pt-induced anti-deactivation of Ru for alkaline hydrogen oxidation reaction
Nature Communications (2024)
-
Atomic interface regulation of rare-marth metal single atom catalysts for energy conversion
Nano Research (2024)