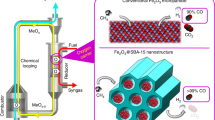
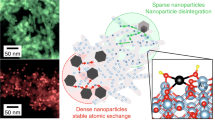
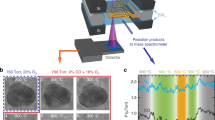
Abstract
Oxide nanoparticles with elemental and structural diversity are widely studied for catalysis and energy applications. While compositional control holds great promise for materials discovery, current oxide nanoparticles are typically limited to a few cations due to the intrinsic complexity in nanoscale multi-element mixing. Here we report the rational design and synthesis of single-phase multi-element oxide nanoparticles with tunable composition, size and structure. We have identified temperature-, oxidation- and entropy-driven synthesis strategies to mix a range of elements with largely dissimilar oxidation potentials (including palladium), thus greatly expanding the compositional space. Through rapid synthesis and screening, we obtained a denary multi-element oxide catalyst showing high performance and superior stability for catalytic methane combustion over 100 hours due to the high-entropy design and stabilization. Our work therefore provides a viable synthesis route with clear guidelines for multi-element oxide nanoparticles and enables materials design in the multi-element space towards highly stable catalysts.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Experimental procedures and material characterization of the oxide nanoparticles are available within this paper and the Methods. Powder diffraction file (PDF) cards of compounds used in this work are available at the Joint Committee on Powder Diffraction Standards. The atomic coordinates of the optimized oxide structure and initial and final configurations of the oxides are provided in the Supplementary Data. Other data that support the plots within this paper and the findings of this study are available from the corresponding authors on reasonable request.
Code availability
Density functional theory calculations on the formation of multi-element oxides were performed using VASP. The structural and thermal stabilities of multi-element oxides were computed by applying the Large-scale Atomic/Molecular Massively Parallel Simulator (LAMMPS). The open-source LAMMPS computer code is available at https://lammps.sandia.gov. The parameters used for the calculations are listed in the Methods, Supplementary Methods or available from the corresponding author upon reasonable request.
Change history
12 April 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41929-021-00613-1
References
Liao, L. et al. Efficient solar water-splitting using a nanocrystalline CoO photocatalyst. Nat. Nanotechnol. 9, 69–73 (2013).
Bergmann, A. et al. Unified structural motifs of the catalytically active state of Co(oxyhydr)oxides during the electrochemical oxygen evolution reaction. Nat. Catal. 1, 711–719 (2018).
Li, M., Schnablegger, H. & Mann, S. Coupled synthesis and self-assembly of nanoparticles to give structures with controlled organization. Nature 402, 393–395 (1999).
Feng, X. et al. Converting ceria polyhedral nanoparticles into single-crystal nanospheres. Science 312, 1504–1508 (2006).
Feng, K. et al. CuxCo1−xO nanoparticles on graphene oxide as a synergistic catalyst for high-efficiency hydrolysis of ammonia-borane. Angew. Chem. Int. Ed. 55, 11950–11954 (2016).
Rost, C. M. et al. Entropy-stabilized oxides. Nat. Commun. 6, 8485 (2015).
Gild, J., Samiee, M., Braun, L., Harrington, T. & Vega, H. High-entropy fluorite oxides. J. Eur. Ceram. Soc. 38, 3578–3584 (2018).
Oses, C., Toher, C. & Curtarolo, S. High-entropy ceramics. Nat. Rev. Mater. 5, 295–309 (2020).
Dai, S. Across the board: Sheng Dai on catalyst design by entropic factors. ChemSusChem 13, 1915–1917 (2020).
Sarkar, A. et al. High entropy oxides for reversible energy storage. Nat. Commun. 9, 3400 (2018).
Chen, H. et al. Entropy-stabilized metal oxide solid solutions as CO oxidation catalysts with high-temperature stability. J. Mater. Chem. A 6, 11129–11133 (2018).
Chen, H. et al. Mechanochemical synthesis of high entropy oxide materials under ambient conditions: dispersion of catalysts via entropy maximization. ACS Mater. Lett. 1, 83–88 (2019).
Sun, S. & Zeng, H. Size-controlled synthesis of magnetite nanoparticles. J. Am. Chem. Soc. 124, 8204–8205 (2002).
Pang, X., Zhao, L., Han, W., Xin, X. & Lin, Z. A general and robust strategy for the synthesis of nearly monodisperse colloidal nanocrystals. Nat. Nanotechnol. 8, 426–431 (2013).
Du, J. S. et al. The structural fate of individual multicomponent metal-oxide nanoparticles in polymer nanoreactors. Angew. Chem. Int. Ed. 56, 7625–7629 (2017).
Anandkumar, M., Bhattacharya, S. & Deshpande, A. S. Low temperature synthesis and characterization of single phase multi-component fluorite oxide nanoparticle sols. RSC Adv. 9, 26825–26830 (2019).
Rajeeva, B. B. et al. Accumulation-driven unified spatiotemporal synthesis and structuring of immiscible metallic nanoalloys. Matter 1, 1606–1617 (2019).
Patzke, G. R., Zhou, Y., Kontic, R. & Conrad, F. Oxide nanomaterials: synthetic developments, mechanistic studies, and technological innovations. Angew. Chem. Int. Ed. 50, 826–859 (2011).
Haber, J. A., Anzenburg, E., Yano, J., Kisielowski, C. & Gregoire, J. M. Multiphase nanostructure of a quinary metal oxide electrocatalyst reveals a new direction for OER electrocatalyst design. Adv. Energy Mater. 5, 1–11 (2015).
Cheng, F. et al. Rapid room-temperature synthesis of nanocrystalline spinels as oxygen reduction and evolution electrocatalysts. Nat. Chem. 3, 79–84 (2011).
Li, S., Ren, Y., Biswas, P. & Tse, S. D. Flame aerosol synthesis of nanostructured materials and functional devices: processing, modeling, and diagnostics. Prog. Energy Combust. Sci. 55, 1–59 (2016).
Mialon, G., Gohin, M., Gacoin, T. & Boilot, J. High temperature strategy for oxide nanoparticle synthesis. ACS Nano 2, 2505–2512 (2008).
Lu, Z. et al. Identifying the active surfaces of electrochemically tuned LiCoO2 for oxygen evolution reaction. J. Am. Chem. Soc. 139, 6270–6276 (2017).
Reed, T. B. Free Energy of Formation of Binary Compounds (MIT Press, 1971).
Shannon, R. D. Revised effective ionic radii and systematic studies of interatomie distances in halides and chaleogenides. Acta Cryst. 32, 751–767 (1976).
Yao, Y. et al. Carbothermal shock synthesis of high-entropy-alloy nanoparticles. Science 359, 1489–1494 (2018).
Neagu, D., Tsekouras, G., Miller, D. N., Ménard, H. & Irvine, J. T. S. In situ growth of nanoparticles through control of non-stoichiometry. Nat. Chem. 5, 916–923 (2013).
J Farrauto, R. Low-temperature oxidation of methane. Science 337, 659–661 (2012).
Al, F. et al. Exceptional activity for methane combustion over modular Pd@CeO2. Science 337, 713–718 (2012).
Zarur, A. J. & Ying, J. Y. Reverse microemulsion synthesis of nanostructured complex oxides for catalytic combustion. Nature 403, 65–67 (2000).
Li, W. B., Wang, J. X. & Gong, H. Catalytic combustion of VOCs on non-noble metal catalysts. Catal. Today 148, 81–87 (2009).
Peng, H. et al. Confined ultrathin Pd-Ce nanowires with outstanding moisture and SO2 tolerance in methane combustion. Angew. Chem. 130, 9091–9095 (2018).
Nguyen, T. S. et al. Correlation of the ratio of metallic to oxide species with activity of PdPt catalysts for methane oxidation. Catal. Sci. Technol. 10, 1408–1421 (2020).
Toso, A., Colussi, S., Padigapaty, S., de Leitenburg, C. & Trovarelli, A. High stability and activity of solution combustion synthesized Pd-based catalysts for methane combustion in presence of water. Appl. Catal. B 230, 237–245 (2018).
Willis, J. J. et al. Systematic identification of promoters for methane oxidation catalysts using size- and composition-controlled Pd-based bimetallic nanocrystals. J. Am. Chem. Soc. 139, 11989–11997 (2017).
Jacob, R. J. et al. High speed 2-dimensional temperature measurements of nanothermite composites: probing thermal vs. gas generation effects. J. Appl. Phys. 123, 115902 (2018).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 3–4 (1999).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Birch, F. Finite elastic strain of cubic crystals. Phys. Rev. 71, 809–824 (1947).
Acknowledgements
This project is not directly funded. We thank D. J. Kline and M. R. Zachariah for their assistance with temperature measurements. R.S.-Y. and Z.H., and electron microscopy studies were supported by NSF-DMR award no.1809439. Z.L. and G.W. acknowledge computational resources provided by the University of Pittsburgh Center for Research Computing as well as the Extreme Science and Engineering Discovery Environment (XSEDE), which is supported by National Science Foundation grant no. ACI-1053575. M.C. acknowledges the Center for Nanophase Materials Sciences, which is a US Department of Energy Office of Science User Facility.
Author information
Authors and Affiliations
Contributions
L.H., T.L. and Y.Y. contributed to the idea and experimental design. T.L., Y.Y., J.G., A.H.B., G.P., M.J., Q.D., J.D. and S.L. conducted materials preparation and characterization. Z.H., M.C. and R.S.-Y. carried out the high-resolution microscopy. P.X., K.Z., H.Z. and C.W. performed the catalysis measurements. J.L. contributed to experimental analysis. Z.L. and G.W. conducted the DFT simulations. M.Y. and Y.M. conducted the MD simulations. L.H., T.L. and Y.Y. wrote the paper, and all authors commented on the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Catalysis thanks Wangcheng Zhan and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 10-MEO-MgMn sample before and after the high-temperature synthesis and size control.
a, SEM images of the sample prior to synthesis. Mixed salt precursors were uniformly loaded onto the CNF substrate. b, SEM images of the 10-MEO-MgMn nanoparticles uniformly dispersed on the CNFs by the high-temperature conditions (1500 K, 50 ms). c, d, SEM images of the 10-MEO-MgMn nanoparticles synthesized at different durations, including 10 ms (c) and 500 ms (d) at 1500 K. e, Particle size distribution of the 10-MEO-MgMn nanoparticles made at 1500 K and at different durations. f, Temperature profiles used in our synthesis. High temperature (~1500 K), short time (within 1 second), and high cooling rate (~104 K/s) are beneficial for the MEO nanoparticle formation.
Extended Data Fig. 2 XRD and SEM patterns of unary metal and oxide nanoparticles dispersed on the CNF substrate.
a, XRD patterns. The formation of unary metal (for example, Co, Ni) and oxide (for example, MnO, CaO) nanoparticles is closely related to the Ellingham diagram (Fig. 2a in the main text). Elements above the carbothermic reduction curve (2C+O2=2CO↑) are easily reduced to metallic states, while those elements beneath the curve can maintain their oxidized states after the high-temperature synthesis (~1500 K). b, c, SEM images of metal In (b) and oxide ZnO (c) nanoparticles on CNFs. The SEM images and their XRD patterns demonstrate the formation of unary metal or oxide nanoparticles using our high-temperature synthesis.
Extended Data Fig. 3 MEO nanoparticles synthesized by temperature- and oxidation-driven strategies.
a, b, Elemental mapping of single-phase (Zr,Ce,Hf,Ti,La,Y)O2-x (a) and denary (Zr,Ce,Hf,Ti,La,Y,V,Nb,Ca,Mn)O2-x (b) nanoparticles by temperature-driven mixing. c, d, XRD (c) and elemental mapping (d) of (Mg,Zn,Co,Ni,Cu)O nanoparticles synthesized by oxidation-driven mixing, showing a single-phase rocksalt (indexed to MgO (PDF#65-0476)) structure.
Extended Data Fig. 4 The stabilization of Pd in the denary oxide nanoparticles.
a, XRD patterns of samples synthesized by our rapid high-temperature synthesis, including denary MEOs with noble Pd content of 10at% (single phase), 15at% (phase separation), and 20at% (phase separation). In these samples with higher Pd contents, although the strategy of entropy-driven mixing and rapid synthesis was used, the metallic Pd phase starts to emerge due to increasing concentration of Pd in MEOs, which lowers the diffusion distance and eventually causes phase separation and reduction of Pd. b, Elemental mapping of the denary MEO with 10at% Pd, showing uniform elemental mixing. c, XRD and d, SEM of the denary MEO with 10at% Pd synthesized by the conventional heating method (annealed under Ar at 1,273 K for 2 h), showing phase separation and particle aggregation. This result suggests the capability of our high-temperature non-equilibrium method to synthesize and stabilize the noble element into single-phase MEO nanoparticles.
Extended Data Fig. 5 MEO nanoparticles synthesized by entropy-driven strategies.
a, b, Elemental mapping of ternary oxide (Ti,Mn,Co)Ox (a) showing phase separation of Co from other elements. By adding 7 more elements, the denary oxide (Zr,Ce,Hf,Ti,La,Y,Gd,Mn,Fe,Co)O2-x (b) shows single-phase MEO formation, indicating entropy-driven mixing effect. Scale bar = 50 nm. c, d, Elemental mapping of senary oxide (Zr,Hf,Ti,Fe,Co,Ni)Ox (phase separation) (c) and denary oxide (Zr,Hf,Ti,La,V,Nb,Zn,Fe,Co,Ni)O2-x (single phase) (d) nanoparticles, showing the similar entropy-driven effect. Scale bar = 10 nm.
Extended Data Fig. 6 Optimized synthesis temperature and high-temperature reduction.
a, b, c, SEM images of (Zr,Ce,Hf,Ti,Mn,Co)O2-x nanoparticles synthesized at low (I, ~1200 K) (a), optimized (II, ~1500 K) (b), and high (III, ~1800 K) (c) temperatures for 500 ms. Scale bar = 100 nm. d, Elemental mapping of (Zr,Ce,Hf,Ti,Mn,Co)O2-x synthesized at 1800 K. Too high-temperature (1800 K) synthesis may lead to reduction of Co metals (phase separation) and particle overgrowth. Scale bar = 50 nm.
Extended Data Fig. 7 The thermal stability of denary oxide nanoparticles.
a, The microscale stress distribution for quaternary (Zr,Ce,Hf,Ti)O2 and denary 10-MEO-MgMn. The 10-MEO-MgMn shows a more uniform stress distribution than the (Zr,Ce,Hf,Ti)O2, suggesting a relatively more stable structure. b, The thermal stability (that is, after 2×106 Monte Carlo (MC) steps at 1073 K) for quaternary (Zr,Ce,Hf,Ti)O2, which shows phase separation after thermal annealing at 1073 K. c, TEM elemental mapping of in situ heat-treated (Ce,Y,Gd)O2-x, (Zr,Ce,Hf,Ti)O2, and 10-MEO-MgMn nanoparticles. The denary oxide 10-MEO-MgMn nanoparticles remain almost the same after in situ annealing at 1073 K for 1 h, with little change in the particle size, dispersity, and elemental mixing, while (Ce,Y,Gd)O2-x and (Zr,Ce,Hf,Ti)O2 show elemental segregation. Scale bar = 50 nm. d, The elemental mapping of 10-MEO-MgMn nanoparticles at 298 K and 773 K. Scale bar = 50 nm. e, The elemental concentration evolution in the 10-MEO-MgMn nanoparticles, derived from the single nanoparticle in the red box of Extended Data Fig. 7d. The composition ratio also remained similar during the in situ stability test.
Extended Data Fig. 8 Multi-element mixing to stabilize Pd in the oxidized state in 10-MEO-PdO nanoparticles.
a, The temperature-dependence of the Gibbs free energy of (Zr, Ce)0.6Mg0.3Pd0.1Ox (4-MEO-Pd) and (Zr,Ce)0.6(Hf,Ti,La,Y,Mg,Cr,Mn)0.3Pd0.1O2-x (10-MEO-PdO). The 10-MEO-PdO (a higher entropy) has a lower formation temperature (~790 K) at which the free energy of mixing becomes zero. However, the 4-MEO-Pd (with a lower entropy) cannot form single-phase fluorite structure below the temperature of ~2473 K. The calculated study demonstrates the entropy stabilization effect in the Pd-contained MEO systems. Thermodynamically, the increased mixing entropy and high-temperature synthesis provide a high entropic driving force (ΔG=ΔH‒T×ΔS) which enables single-phase MEO formation and stabilization of PdO in the resulting MEOs. Kinetically, the PdO is trapped and stabilized in the denary MEO nanoparticles due to the high entropy structure with random mixing, lattice distortion, and slow kinetics. Detailed calculation models, parameters, and simulation process can be found in the Supplementary Methods. b, c, Elemental mapping of 4-MEO-Pd (b) (elemental segregation) and 10-MEO-PdO (c) (uniform mixing) nanoparticles. Scale bar = 10 nm. d, XRD of 4-MEO-Pd (phase separation) and 10-MEO-PdO (single phase) nanoparticles. e, XPS of the Pd element in 4-MEO-Pd (Metallic and oxidized Pd) and 10-MEO-PdO (Oxidized Pd). The evolution of Pd from a mixture phase (in the 4-element system) to a stable oxide PdO in the denary MEO (10-element system), demonstrating an entropy-driven mixing and stabilization of noble metal Pd.
Extended Data Fig. 9 Stability of the 10-MEO-PdO catalyst.
a, XRD patterns before and after the methane combustion reaction for the control Pd sample synthesized by the traditional impregnation method, showing the PdO (indexed to PdO (PDF#43-1024)) was reduced to metallic Pd (indexed to Pd (PDF#46-1043)). b, XRD patterns before and after the methane combustion reaction for our 10-MEO-PdO (indexed to ZrO2 (PDF#49-1642)) nanoparticles with excellent thermal stability. c, TEM image and d, elemental mapping of our 10-MEO-PdO nanoparticles after reaction, showing the uniform dispersion and mixing, without particle coarsening or elemental segregation.
Extended Data Fig. 10 The 10-MEO-PdO dispersed on the Al2O3-coated carbon paper for methane combustion performed under wet conditions.
a, b, SEM images of carbon paper without (a) and with (b) Al2O3 coating. Scale bar = 2 μm. c, TGA data (performed in air atmosphere) of carbon paper (CP) without and with the Al2O3 coating. The Al2O3 coating increases the decomposition temperature of the substrate, which shows excellent stability up to 973 K. d, CH4 conversion of 10-MEO-PdO on the Al2O3-coated carbon paper performed under wet conditions at different GHSVs. e, CH4 conversion of 10-MEO-PdO on the Al2O3-coated carbon paper performed under wet conditions at the GHSV of 10,800 L gpd−1 h−1 before and after annealing at 873 K for 5 h under the catalytic reaction condition, demonstrating the excellent stability of the catalyst under wet conditions at high temperatures. f, STEM image and elemental mapping of 10-MEO-PdO nanoparticles on the Al2O3-coated carbon paper after 100 hours of operation carried out under wet conditions, showing the uniform distribution and mixing of the nanoparticles, without coarsening or elemental segregation.
Supplementary information
Supplementary Information
Supplementary Figs. 1–8, Tables 1–8, Methods and references.
Supplementary Data 1
The optimized structures for binary, ternary, quaternary and denary oxides.
Supplementary Data 2
The initial and final configurations for ternary, quaternary and denary oxides.
Rights and permissions
About this article
Cite this article
Li, T., Yao, Y., Huang, Z. et al. Denary oxide nanoparticles as highly stable catalysts for methane combustion. Nat Catal 4, 62–70 (2021). https://doi.org/10.1038/s41929-020-00554-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-020-00554-1
This article is cited by
-
Emerging Atomically Precise Metal Nanoclusters and Ultrasmall Nanoparticles for Efficient Electrochemical Energy Catalysis: Synthesis Strategies and Surface/Interface Engineering
Electrochemical Energy Reviews (2024)
-
The reformation of catalyst: From a trial-and-error synthesis to rational design
Nano Research (2024)
-
Three-dimensional atomic structure and local chemical order of medium- and high-entropy nanoalloys
Nature (2023)
-
Transient and general synthesis of high-density and ultrasmall nanoparticles on two-dimensional porous carbon via coordinated carbothermal shock
Nature Communications (2023)
-
A review on the rational design and fabrication of nanosized high-entropy materials
Nano Research (2023)